Millennia-Long Co-Existence of Two Major European Whitefish (Coregonus spp.) Lineages in Switzerland Inferred from Ancient Mitochondrial DNA
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Archaeological Sites
2.3. Neolithic Period
2.3.1. Stansstad-Kehrsiten
2.3.2. Arbon-Bleiche 3
2.4. Roman Era
2.5. Medieval Period
2.6. Methods
2.6.1. DNA Extraction
2.6.2. Primer Design, PCR, and Sequencing
3. Results and Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bernatchez, L.; Renaut, S.; Whiteley, A.R.; Derome, N.; Jeukens, J.; Landry, L.; Lu, G.; Nolte, A.W.; Ostbye, K.; Rogers, S.M.; et al. On the origin of species: Insights from the ecological genomics of lake whitefish. Philos. Trans. R. Soc. B 2010, 365, 1783–1800. [Google Scholar] [CrossRef] [PubMed]
- Kottelat, M.; Freyhof, J. Handbook of European Freshwater Fishes; Publications Kottelat: Cornol, Switzerland, 2007. [Google Scholar]
- Schulz, M.; Freyhof, J.; Saint-Laurent, R.; Østbye, K.; Mehner, T.; Bernatchez, L. Evidence for independent origin of two spring-spawning ciscoes (salmoniformes: Coregonidae) in germany. J. Fish Biol. 2006, 68, 119–135. [Google Scholar] [CrossRef]
- Bernatchez, L. Ecological theory of adaptive radiation: An empirical assessment from coregonine fishes (salmoniformes). In Evolution Illuminated: Salmon and Their Relatives; Hendry, A.P., Stearns, S.C., Eds.; Oxford University Press: Oxford, UK, 2004; pp. 175–207. [Google Scholar]
- Hudson, A.G.; Vonlanthen, P.; Mueller, R.; Seehausen, O. Review: The geography of speciation and adaptive radiation in coregonines. In Biology and Management of Coregonid Fishes—2005; Jankun, M., Brzuzan, P., Hliwa, P., Luczynski, M., Eds.; Schweizerbart Wissenschaftsverlage: Stuttgart, Germany, 2007; Volume 60, pp. 111–146. [Google Scholar]
- Østbye, K.; Amundsen, P.A.; Bernatchez, L.; Klemetsen, A.; Knudsen, R.; Kristoffersen, R.; Næsje, T.F.; Hindar, K. Parallel evolution of ecomorphological traits in the european whitefish Coregonus lavaretus (L.) species complex during postglacial times. Mol. Ecol. 2006, 15, 3983–4001. [Google Scholar] [CrossRef] [PubMed]
- Douglas, M.R.; Brunner, P.C.; Bernatchez, L. Do assemblages of Coregonus (teleostei: Salmoniformes) in the central alpine region of europe represent species flocks? Mol. Ecol. 1999, 8, 589–603. [Google Scholar] [CrossRef]
- Praebel, K.; Knudsen, R.; Siwertsson, A.; Karhunen, M.; Kahilainen, K.K.; Ovaskainen, O.; Ostbye, K.; Peruzzi, S.; Fevolden, S.E.; Amundsen, P.A. Ecological speciation in postglacial european whitefish: Rapid adaptive radiations into the littoral, pelagic, and profundal lake habitats. Ecol. Evol. 2013, 3, 4970–4986. [Google Scholar] [CrossRef] [PubMed]
- Steinmann, P. Monographie der schweizerischen Koregonen. Beitrag zum Problem der Entstehung neuer Arten. Spezieller Teil. Schweiz Z. Hydrobiol. 1950, 12, 340–491. [Google Scholar]
- Vonlanthen, P.; Bittner, D.; Hudson, A.G.; Young, K.A.; Muller, R.; Lundsgaard-Hansen, B.; Roy, D.; Di Piazza, S.; Largiader, C.R.; Seehausen, O. Eutrophication causes speciation reversal in whitefish adaptive radiations. Nature 2012, 482, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Douglas, M.R.; Brunner, P.C. Biodiversity of central alpine Coregonus (salmoniformes): Impact of one-hundred years of management. Ecol. Appl. 2002, 12, 154–172. [Google Scholar] [CrossRef]
- Hudson, A.G.; Vonlanthen, P.; Seehausen, O. Rapid parallel adaptive radiations from a single hybridogenic ancestral population. Proc. R. Soc. B 2011, 278, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Hudson, A.G.; Lundsgaard-Hansen, B.; Lucek, K.; Vonlanthen, P.; Seehausen, O. Managing cryptic biodiversity: Fine-scale intralacustrine speciation along a benthic gradient in alpine whitefish (Coregonus spp.). Evol. Appl. 2017, 10, 251–266. [Google Scholar] [CrossRef] [PubMed]
- Østbye, K.; Bernatchez, L.; Næsje, T.F.; Himberg, K.J.M.; Hindar, K. Evolutionary history of the european whitefish Coregonus lavaretus (L.) species complex as inferred from mtdna phylogeography and gill-raker numbers. Mol. Ecol. 2005, 14, 4371–4387. [Google Scholar] [CrossRef] [PubMed]
- Alexander, T.J.; Vonlanthen, P.; Seehausen, O. Does eutrophication-driven evolution change aquatic ecosystems? Philos. Trans. R. Soc. B 2017, 372, 20160041. [Google Scholar] [CrossRef] [PubMed]
- Ruhlé, C.; Kindle, T. Morphological comparison of river-spawning whitefish of the alpine rhine with the whitefish of lake constance. Pol. Arch. Hydrobiol. 1992, 39, 403–408. [Google Scholar]
- Rick, T.C.; Lockwood, R. Integrating paleobiology, archeology, and history to inform biological conservation. Conserv. Biol. 2013, 27, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Hüster Plogmann, H. Fischfang und Kleintierbeute. Ergebnisse der Untersuchung aus den Schlämmproben. In Die Jungsteinzeitliche Seeufersiedlung Arbon—Bleiche 3. Umwelt und Wirtschaft. Archäologie im Thurgau 12; Jacomet, S., Leuzinger, U., Schibler, J., Eds.; Amt für Archäologie des Kantons Thurgau: Frauenfeld, Switzerland, 2004; pp. 253–276. [Google Scholar]
- Michel, C.; Bleicher, N.; Brombacher, C.; Hüster Plogmann, H.; Ismail-Meyer, K.; Rehazek, A. Pfahlbauten am Vierwaldstättersee-der steinzeitliche Siedlungsplatz in Kehrsiten. Archäol. Schweiz 2012, 35, 56–71. [Google Scholar]
- Hüster Plogmann, H. Befunde und Fundkomplexe-der steingebaute Gutshof: Knochenreste aus Schlämmproben. In Der Römische Gutshof in Neftenbach. Monographien der Kantonsarchäologie Zürich 31, Band 1; Kantonsarchäologie Zürich, Ed.; Fotorotar AG: Egg/Zürich, Switzerland, 1999; pp. 413–414. [Google Scholar]
- Hüster Plogmann, H. Von Leckerbissen und Schädlingen-die Untersuchung der Kleintierreste. In Zur Frühzeit von Vindonissa. Auswertung der Holzbauten der Grabung Windisch-Breite 1996–1998. Veröffentlichungen der Gesellschaft pro Vindonissa XVIII; Kantonsarchäologie Aargau, Ed. Kantonsarchäologie Aargau: Brugg, Switzerland, 2003; pp. 231–243. [Google Scholar]
- Zaugg, B.; Stucki, P.; Pedroli, J.-C.; Kirchhofer, A. Pisces—Atlas Fauna Helvetica 7; CSCF: Chaumont, France, 2003. [Google Scholar]
- Häberle, S.; Fuller, B.T.; Nehlich, O.; Van Neer, W.; Schibler, J.; Hüster Plogmann, H. Inter- and intraspecies variability in stable isotope ratio values of archaeological freshwater fish remains from switzerland (11th–19th centuries ad). Environ. Archaeol. 2016, 21, 119–132. [Google Scholar] [CrossRef]
- Hüster Plogmann, H. (Ed.) Fisch und Fischer aus zwei Jahrtausenden. Eine Fischereiwirtschaftliche Zeitreise durch die Nordwestschweiz. Forschungen in Augst 39; Römermuseum Augst: Augst, Switzerland, 2006. [Google Scholar]
- Müller, L. Die Fischerei im Spätmittelalterlichen Basel. Lizentiazsarbeit; Universität Basel: Basel, Switzerland, 1989. [Google Scholar]
- Chassaing, O.; Desse-Berset, N.; Hanni, C.; Hughes, S.; Berrebi, P. Phylogeography of the european sturgeon (acipenser sturio): A critically endangered species. Mol. Phylogenet. Evol. 2016, 94, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Grealy, A.; Douglass, K.; Haile, J.; Bruwer, C.; Gough, C.; Bunce, M. Tropical ancient DNA from bulk archaeological fish bone reveals the subsistence practices of a historic coastal community in southwest madagascar. J. Archaeol. Sci. 2016, 75, 82–88. [Google Scholar] [CrossRef]
- Grier, C.; Flanigan, K.; Winters, M.; Jordan, L.G.; Lukowski, S.; Kemp, B.M. Using ancient DNA identification and osteometric measures of archaeological pacific salmon vertebrae for reconstructing salmon fisheries and site seasonality at dionisio point, british columbia. J. Archaeol. Sci. 2013, 40, 544–555. [Google Scholar] [CrossRef]
- Ólafsdóttir, G.Á.; Westfall, K.M.; Edvardsson, R.; Pálsson, S. Historical DNA reveals the demographic history of atlantic cod (gadus morhua) in medieval and early modern iceland. Proc. R. Soc. B 2014, 281. [Google Scholar] [CrossRef]
- Speller, C.F.; Hauser, L.; Lepofsky, D.; Moore, J.; Rodrigues, A.T.; Moss, M.L.; McKechnie, I.; Yang, D.Y. High potential for using DNA from ancient herring bones to inform modern fisheries management and conservation. PLoS ONE 2012, 7, e51122. [Google Scholar] [CrossRef] [PubMed]
- Splendiani, A.; Fioravanti, T.; Giovannotti, M.; Negri, A.; Ruggeri, P.; Olivieri, L.; Nisi Cerioni, P.; Lorenzoni, M.; Caputo Barucchi, V. The effects of paleoclimatic events on mediterranean trout: Preliminary evidences from ancient DNA. PLoS ONE 2016, 11, e0157975. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, A.; Arndt, U.; Lippold, S.; Benecke, N.; Debus, L.; King, T.; Matsumura, S. Tracing the first steps of american sturgeon pioneers in europe. BMC Evol. Biol. 2008, 8, 221. [Google Scholar] [CrossRef] [PubMed]
- Oleksiak, M.F. Genomic approaches with natural fish populations. J. Fish Biol. 2010, 76, 1067–1093. [Google Scholar] [CrossRef] [PubMed]
- Macqueen, D.J.; Primmer, C.R.; Houston, R.D.; Nowak, B.F.; Bernatchez, L.; Bergseth, S.; Davidson, W.S.; Gallardo-Escarate, C.; Goldammer, T.; Guiguen, Y.; et al. Functional annotation of all salmonid genomes (faasg): An international initiative supporting future salmonid research, conservation and aquaculture. BMC Genom. 2017, 18, 484. [Google Scholar] [CrossRef] [PubMed]
- Zohar, I.; Belmaker, M. Size does matter: Methodological comments on sieve size and species richness in fishbone assemblages. J. Archaeol. Sci. 2005, 32, 635–641. [Google Scholar] [CrossRef]
- Leuzinger, U. Die Jungsteinzeitliche Seeufersiedlung Arbon—Bleiche 3. Befunde. Archäologie im Thurgau. Band 9; Amt für Archäologie des Kantons Thurgau: Frauenfeld, Switzerland, 2000; Volume 9. [Google Scholar]
- Hagendorn, A. Zur Frühzeit von Vindonissa. Auswertung der Holzbauten der Grabung Windisch-Breite 1996–1998. Veröffentlichungen der Gesellschaft pro Vindonissa XVIII; Kantonsarchäologie Aargau: Brugg, Switzerland, 2003. [Google Scholar]
- Hüster Plogmann, H. Befunde und Fundkomplexe-der steingebaute Gutshof: Knochen aus Schlämmproben. In Der römische Gutshof in Neftenbach. Monographien der Kantonsarchäologie Zürich 31, Band 1; Kantonsarchäologie Zürich, Ed.; Fotorotar AG: Egg/Zürich, Switzerland, 1999; pp. 264–265. [Google Scholar]
- Rychener, J. Der Römische Gutshof in Neftenbach. Monographien der Kantonsarchäologie Zürich 31; Fotorotar AG: Egg/Zürich, Switzerland, 1999. [Google Scholar]
- Caduff, B.; Hüster Plogmann, H.; Diaz Tabernero, J.; Durst, M. Zum Frühmittelalterlichen Speisezettel in Tumegl/Tomils, Sogn Murez. In Jahresberichte des Archäologischen Dienstes Graubünden und der Denkmalpflege Graubünden 2002; Archäologischer Dienst Graubünden, Ed.; Archäologischer Dienst Graubünden: Haldenstein/Chur, Switzerland, 2003; pp. 96–115. [Google Scholar]
- Hüster Plogmann, H. Die frühmittelalterlichen Speisereste. In Zum frühmittelalterlichen Speisezettel in Tumegl/Tomils, Sogn Murezi. Jahresberichte des Archäologischen Dienstes Graubünden und der Denkmalpflege Graubünden 2002; Archäologischer Dienst Graubünden, Ed.; Archäologischer Dienst Graubünden: Haldenstein/Chur, Switzerland, 2003; pp. 100–113. [Google Scholar]
- Jecklin-Tischhauser, U. Die Kirchenanlage Sogn Murezi in Tomils (GR). Kirchliches und herrschaftliches Zentrum im frühmittelalterlichen Churrätien. Ph.D. Thesis, University of Zürich, Zürich, Switzerland, July 2017. [Google Scholar]
- Ohnsorg, P. Zwischen Limmat und Fraumünster. Neue Untersuchungen zur Uferzone am Zürcher Stadthausquai und zur Fraumünster-Abtei. In Stadt Zürich Archäologie und Denkmalpflege 2008–2010; Stadt Zürich: Zürich, Switzerland, 2011; pp. 1–65. [Google Scholar]
- Brombacher, C.; Helmig, G.; Hüster Plogmann, H.; Klee, M.; Rentzel, P.; Rodel, S.; Veszeli, M. Und was davon übrig bleibt—Untersuchungen an einem mittelalterlichen Latrinenschacht an der Bäumleingasse 14 (1992/20). In Archäologische Bodenforschung des Kantons Basel-Stadt. Jahresbericht 1998; Archäologische Bodenforschung des Kantons Basel-Stadt, Ed.; Archäologische Bodenforschung des Kantons Basel-Stadt: Basel, Switzerland, 1999; pp. 93–132. [Google Scholar]
- Häberle, S.; Schibler, J.; Van Neer, W.; Hüster Plogmann, H. Fischknochen als Indikatoren für Gewässerzustand und menschliche Fischselektion. Eine zusammenfassende Auswertung mittelalterlicher und neuzeitlicher Fischreste aus dem Rheineinzugsgebiet der Schweiz. Archäol. Korresp. 2015, 45, 417–437. [Google Scholar]
- Homberger, V. Ein neu entdecktes spätrömisches Kastell bei Weesen SG. Jahrb. Archäol. Schweiz 2008, 91, 141–149. [Google Scholar]
- Straumann, S. 2008/3, Münsterplatz 19, Museum der Kulturen: Ausgrabungen und Funde im Jahr 2009: Münsterhügel. In Archäologische Bodenforschung des Kantons Basel-Stadt. Jahresbericht 2009; Archäologische Bodenforschung des Kantons Basel-Stadt, Ed.; Archäologische Bodenforschung des Kantons Basel-Stadt: Basel, Switzerland, 2010; pp. 32–34. [Google Scholar]
- Jacomet, S.; Ebersbach, R.; Akeret, Ö.; Antolín, F.; Baum, T.; Bogaard, A.; Brombacher, C.; Bleicher, N.K.; Heitz-Weniger, A.; Hüster-Plogmann, H.; et al. On-site data cast doubts on the hypothesis of shifting cultivation in the late neolithic (c. 4300–2400 cal. BC): Landscape management as an alternative paradigm. Holocene 2016, 26, 1858–1874. [Google Scholar] [CrossRef]
- Menotti, F. Wetland Archaeology and Beyond: Theory and Practice; Oxford Universtity Press: Oxford, UK, 2012. [Google Scholar]
- Schibler, J. Zooarchaeological results from neolithic and bronze age wetland and dryland sites in the central alpine foreland: Economic, ecologic and taphonomic relevance. In Oxford Handbook of Archaeozoology; Albarella, U., Rizzetto, M., Russ, H., Vickers, K., Viner-Daniels, S., Eds.; Oxford University Press: Oxford, UK, 2017; pp. 83–98. [Google Scholar]
- Elsner, J.; Hofreiter, M.; Schibler, J.; Schlumbaum, A. Ancient mtdna diversity reveals specific population development of wild horses in switzerland after the last glacial maximum. PLoS ONE 2017, 12, e0177458. [Google Scholar] [CrossRef] [PubMed]
- Schibler, J.; Elsner, J.; Schlumbaum, A. Incorporation of aurochs into a cattle herd in neolithic europe: Single event or breeding? Sci. Rep. 2014, 4, 5798. [Google Scholar] [CrossRef] [PubMed]
- Binladen, J.; Gilbert, M.T.; Campos, P.F.; Willerslev, E. 5′-tailed sequencing primers improve sequencing quality of pcr products. BioTechniques 2007, 42, 174, 176. [Google Scholar] [CrossRef] [PubMed]
- Bandelt, H.-J.; Macaulay, V.; Richards, M. Median networks: Speedy construction and greedy reduction, one simulation, and two case studies from human mtdna. Mol. Phylogenet. Evol. 2000, 16, 8–28. [Google Scholar] [CrossRef] [PubMed]
- Popović, D.; Panagiotopoulou, H.; Baca, M.; Stefaniak, K.; Mackiewicz, P.; Makowiecki, D.; King, T.L.; Gruchota, J.; Weglenski, P.; Stankovic, A. The history of sturgeon in the baltic sea. J. Biogeogr. 2014, 41, 1590–1602. [Google Scholar] [CrossRef]
- Schlumbaum, A.; Edwards, C.J. Ancient DNA reasearch on wetland archaeological evidence. In The Oxford Handbook of Wetland Archaeology; Menotti, F., O′Sullivan, A., Eds.; Oxford University Press: Oxford, UK, 2013; pp. 569–583. [Google Scholar]
- Elsner, J.; Schibler, J.; Hofreiter, M.; Schlumbaum, A. Burial condition is the most important factor for mtdna pcr amplification success in palaeolithic equid remains from the alpine foreland. Archaeol. Anthropol. Sci. 2015, 7, 505–515. [Google Scholar] [CrossRef]
- Pruvost, M.; Schwarz, R.; Correia, V.B.; Champlot, S.; Braguier, S.; Morel, N.; Fernandez-Jalvo, Y.; Grange, T.; Geigl, E.M. Freshly excavated fossil bones are best for amplification of ancient DNA. Proc. Natl. Acad. Sci. USA 2007, 104, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Giguet-Covex, C.; Pansu, J.; Arnaud, F.; Rey, P.J.; Griggo, C.; Gielly, L.; Domaizon, I.; Coissac, E.; David, F.; Choler, P.; et al. Long livestock farming history and human landscape shaping revealed by lake sediment DNA. Nat. Commun. 2014, 5, 3211. [Google Scholar] [CrossRef] [PubMed]
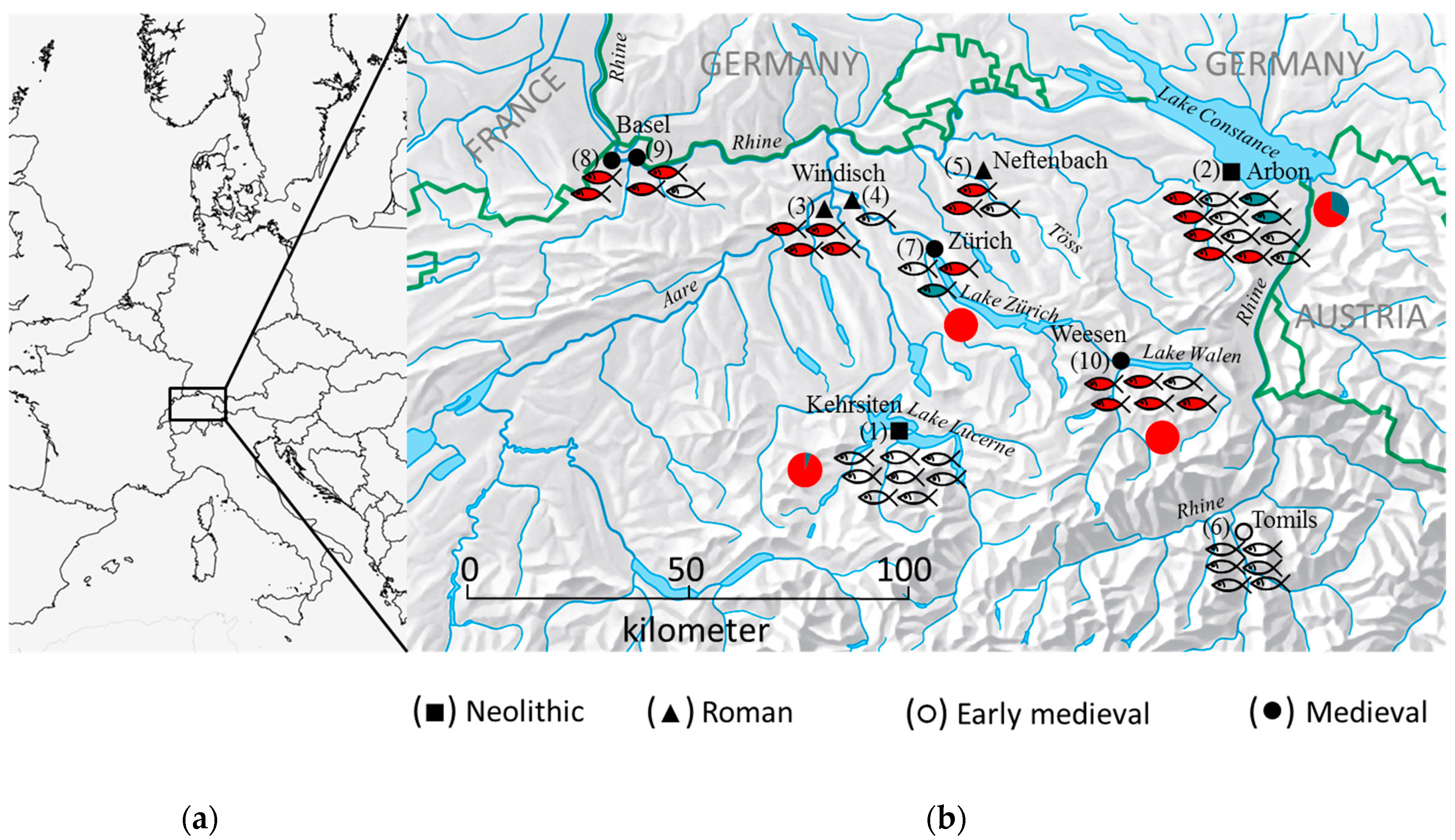

| Archaeological Site | City/Canton | Dating | Code Figure 1 | Elements | Site Type | Depositional Context | Reference |
|---|---|---|---|---|---|---|---|
| Stansstad-Kehrsiten | Kehrsiten/NW | 3500–3400 BC | 1 | 4 scales 4 vertebrae | Lake-shore settlement | Layer samples | [19] |
| Arbon Bleiche 3 | Arbon/TG | 3384–3370 BC | 2 | 4 vertebrae 4 scales 1 4 vertebrae 1 | Lake-shore settlement | Cultural layers | [36] |
| Breite | Windisch/AG | 1st century AD | 3 | 2 scales 2 vertebrae | dryland | Barrel pits, pits | [21,37] |
| Römerblick | Windisch/AG | 1st century AD | 4 | 1 vertebra | dryland | Kitchen floor of a peristyle house | [21,37] |
| Neftenbach | Neftenbach/ZH | 3th/4th century AD | 5 | 3 vertebrae | dryland | Cesspits | [38,39] |
| Tomils | Tomils/GR | 7th century AD | 6 | 6 vertebrae | dryland | Floor insulation structure | [40,41,42] |
| Fraumünsterstrasse | Zürich/ZH | 1010–1160 AD | 7 | 3 vertebrae | dryland | Occupation layer, filling of a fireplace | [43] |
| Bäumleingasse 14 | Basel/BS | 13th century AD | 8 | 2 vertebrae | dryland | Cesspits | [44] |
| Weesen Rosengärten | Weesen/SG | 14th century AD | 10 | 6 vertebrae | dryland | Waste trench | [45,46] |
| Museum der Kulturen, Im Schürhof | Basel/BS | 15th/16th century AD | 9 | 3 vertebrae | dryland | Cesspits | [45,47] |
| Primer Pairs | Coordinates Reference Sequence NC002646 | Nucleotide Sequence (5′–3′) | Annealing Temperature (°C) | Primer Length (bps) | Amplicon Length (bps) | Target Region Contains |
|---|---|---|---|---|---|---|
| CORb1F | 15,794–15,818 | TCAACATAAGTGATTTTAAGCCCTC | 54–55 | 25 | 90–91 | SNP1 |
| CORb1R | 15,864–15,883 | AGAACGGTTCCGTTGGTGAT | 20 | |||
| CORb1F | 15,794–15,818 | TCAACATAAGTGATTTTAAGCCCTC | 54 | 25 | 134–135 | SNP1 and SNP2 |
| COR1R | 15,908–15,927 | GCCCGTGTTAGTTGGAGGTT | 20 | |||
| CORc1F | 15,853–15,874 | AGACTCGGATAATCACCAACGG | 54–55 | 22 | 80 | SNP2 |
| CORc1R | 15,914–15,932 | ACGGAGCCCGTGTTAGTTG | 19 | |||
| CORa2F | 16,447–16,466 | TGTCAAACCCCAAAACCAGG | 54–55 | 20 | 78 | SNP3 |
| CORa2R | 16,502–16,524 | TGTCGGTGCCAAAGTTTGTTAAT | 23 | |||
| COR3F | 16,690–16,709 | TTGGCACCGACAACCCTATC | 54–55 | 20 | 86 | SNP4 |
| COR3R | 15–38 | ACAGCTTCAGTGTTATGCTTTAGT | 24 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alonso, J.D.G.; Häberle, S.; Plogmann, H.H.; Schibler, J.; Schlumbaum, A. Millennia-Long Co-Existence of Two Major European Whitefish (Coregonus spp.) Lineages in Switzerland Inferred from Ancient Mitochondrial DNA. Diversity 2017, 9, 34. https://doi.org/10.3390/d9030034
Alonso JDG, Häberle S, Plogmann HH, Schibler J, Schlumbaum A. Millennia-Long Co-Existence of Two Major European Whitefish (Coregonus spp.) Lineages in Switzerland Inferred from Ancient Mitochondrial DNA. Diversity. 2017; 9(3):34. https://doi.org/10.3390/d9030034
Chicago/Turabian StyleAlonso, José David Granado, Simone Häberle, Heidemarie Hüster Plogmann, Jörg Schibler, and Angela Schlumbaum. 2017. "Millennia-Long Co-Existence of Two Major European Whitefish (Coregonus spp.) Lineages in Switzerland Inferred from Ancient Mitochondrial DNA" Diversity 9, no. 3: 34. https://doi.org/10.3390/d9030034
APA StyleAlonso, J. D. G., Häberle, S., Plogmann, H. H., Schibler, J., & Schlumbaum, A. (2017). Millennia-Long Co-Existence of Two Major European Whitefish (Coregonus spp.) Lineages in Switzerland Inferred from Ancient Mitochondrial DNA. Diversity, 9(3), 34. https://doi.org/10.3390/d9030034




