1. Introduction
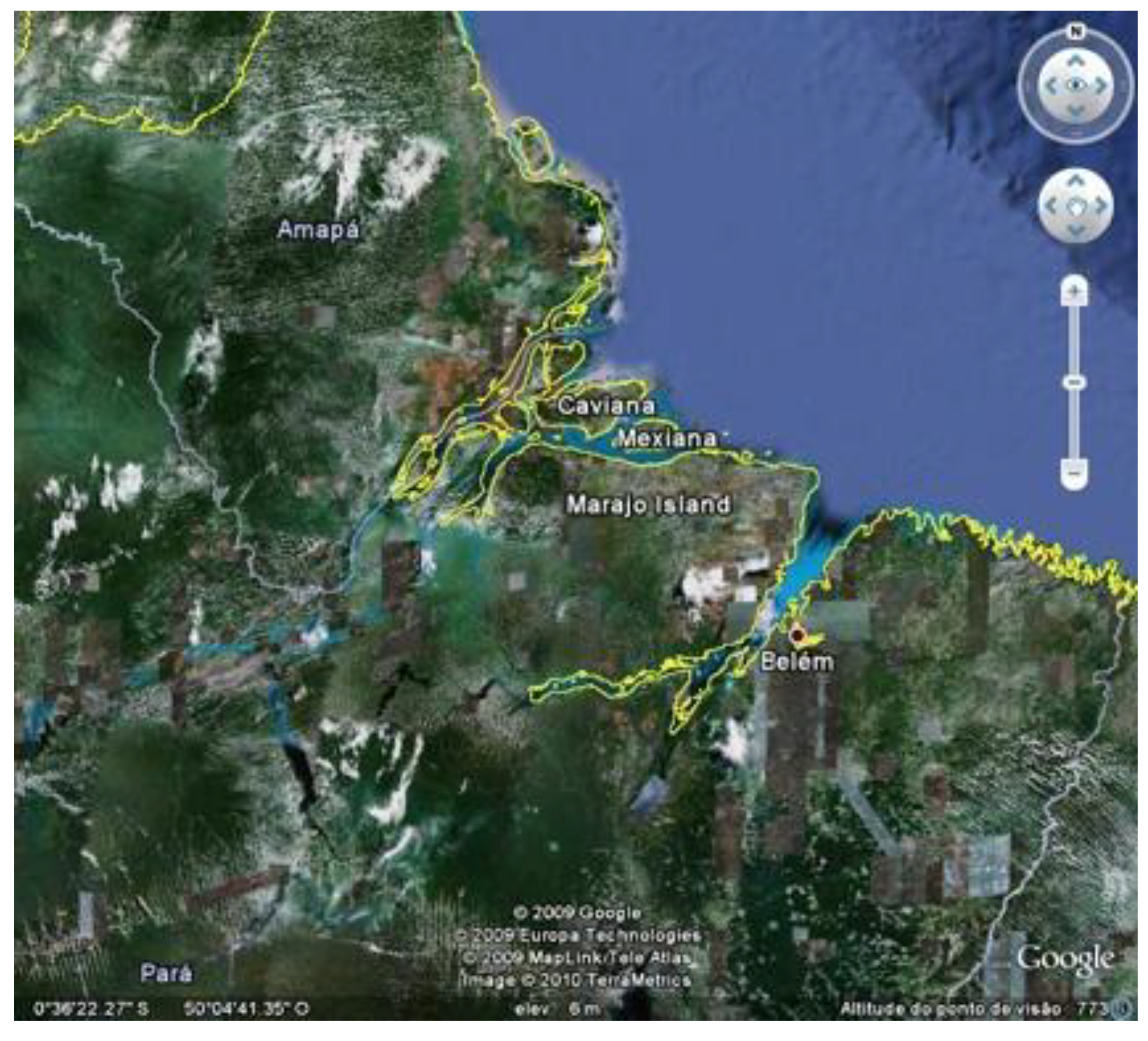
Marajó Island provides a rich laboratory for the study of long-term interaction between human societies and the environment (
Figure 1). The history of human occupation of the island dates from about 5,000 years BP, showing a long history, up to the present, especially of aquatic resource management. The seasonally flooded savannas, which occupy about 40% of the island, are a natural fish nursery, and these were, from time to time, successfully managed in order to feed native and subsequently, colonial populations. The peak of the fishing economy occurred during an archaeological period known as the Marajoara Phase (400−1350 AD), which featured archaeologically-known societies that built mounds and dams as part of efficient hydraulic systems, with the underlying purpose of controlling aquatic fauna and water supplies. Such systems, transformed during centuries of manipulation, have persisted to the present, even if they have been affected negatively by the interference of cattle ranching. It is believed that conflicts over the best fishing areas gave rise to complex societies before European contact. Today this situation still affects social relations and exacerbates social conflicts on the island.
Marajó Island has been studied by ecologists, geographers, and bioscientists interested in its enormous diversity of organisms; these studies underscore the threat of ecological disruption due to infelicitous and unwise management of natural resources [
1,
2,
3]. After enduring for almost a century on an economy strongly based on cattle—mostly water buffaloes—in the last couple of decades, the island’s large ranches have been sold to entrepreneurs with an eye to ecological tourism. The main attraction for such economic activity is a rustic landscape located a mere one to three hour voyage from Belém, the state’s capital of 1.8 million people. The island’s small towns and farm-like scenery supply the visitor with a scent of an untouched Eden.
Twentieth-century literature [
4,
5,
6,
7] has described life on the island as being harsh for cowboys and their families, who took upon their shoulders most of the burdens of administering large ranches with little support and investment from landowners, who preferred to spend their wealth in Belém, Rio de Janeiro, and the cities of Europe, a lifestyle which incidentally also ultimately drove many of them into bankruptcy. The typical cowboy family would have had no formal education, nor access to health and market systems. Trapped on ranches, they had to hunt, fish, and plant their own food. This native work force, of course, would have been favored by an immense knowledge of Marajó Island’s ecology, where perceptions of time and space are measured in terms of seasonal weather, river tidal systems, and reproductive behaviors of flora and fauna.
Figure 1.
Google Earth satellite image of the mouth of the Amazon River, showing Marajó, Caviana and Mexiana Islands.
Figure 1.
Google Earth satellite image of the mouth of the Amazon River, showing Marajó, Caviana and Mexiana Islands.
Major ecological problems in the island were reported during the 20
th century by journalists, geographers, geologists, ecologists, and even archaeologists, based on both scientific literature and oral tradition, which mentioned river obstructions, disappearance of lakes, longer floods, harsher droughts, lack of water, saltiness of water courses, decrease in fauna species (caimans, jaguars, piranhas, large fish), among others [
3,
8,
9,
10,
11]. Many of these problems are due to ecologically irrational water management (ranchers dam rivers to retain water for their livestock), overfishing, overhunting, and sedimentation of rivers by aquatic buffaloes (carrying heavy loads of sediments into the water courses).
In this article, I intend to show that traditional knowledge of the environment and strategies to gather resources are based on a long term history of landscape management, which started about 2,000 years BP, when indigenous populations were organized into small chiefdoms, articulated within a larger context of complex regional systems. This continuity is visible not only in landscape management but also in mythical narratives that continue to the present day. I also aim to discuss the importance of restoring ancient land management systems in order to restore the landscape and its biotic wealth of resources, for this has been greatly affected to date by decidedly irrational economic decisions.
2. The Savanna’s Landscape
The largest extent of savannas in the estuary is located on Marajó Island, where they comprise 23,046 square kilometers (
Figure 2). Extensive areas of savannas are also found in Caviana and Mexiana, the northern islands. These are poorly-drained, heavily mottled clay soils, covered with grasses and eventually dotted by some forest islands and shrubs. These flat, grassy areas are locally called
campos.
Figure 2.
Aerial view of the savannas of southeastern Marajó Island. Photograph by Denise Schaan, 2002.
Figure 2.
Aerial view of the savannas of southeastern Marajó Island. Photograph by Denise Schaan, 2002.
Although the term savanna is frequently associated with certain types of vegetation (mostly grasses and occasionally shrubs and scattered small trees) [
12], some scientists understand that savannas can be defined by their climate (as indexed by things such as annual average temperature and length of the dry season) [
13]. In both systems of classification, the Marajó
campos are a typical example of low, grassy, and seasonally inundated savannas.
Both the predominance of grasses (Poaceae) and sedges (Cyperaceae), together with the half-year long submersion of these, are points of similarity between the lowland savannas and the lower Amazon floodplain, or
várzea. But the similarities, for the most part, end there. The
campos of eastern Marajó are flooded by rain water, while the Amazon lower floodplain is invaded by the river overflow [
12]. As Moran [
14] has pointed out, the differential influence of oceanic tides (twice daily on Marajó and once a year in the lower Amazon more generally) also produce a very different ecology in each area as well as requires diverse adaptive responses. Marajó, as part of the Amazon River Estuary, is characterized by major dominance of a small number of species, in contrast to vast numbers of species with few individual representatives, as one finds in much of the rest of the Amazon Basin. The predominance of a few species of palms, in fact, which were used by indigenous populations for multitudinous products, from food to craft production, indicates long-term manipulation of the environment by people who were likely attracted to that area for its well known abundance of aquatic resources (
Figure 3) [
3,
14].
Figure 3.
A miriti (Mauritia flexuosa) forest, aerial view, southeastern Marajó Island. Photograph by Denise Schaan, 2002.
Figure 3.
A miriti (Mauritia flexuosa) forest, aerial view, southeastern Marajó Island. Photograph by Denise Schaan, 2002.
The climate of Marajó Island is characterized by two well-defined seasons: a rainy and hot winter, and a dry summer. Although this is true for most of the Amazon region, rainfall on Marajó is far more intense (from 2,800 to 3,400 mm per year) and the rainshowers are concentrated in a period of 5 to 6 months, from January to June, reflective of its location in the Intertropical Convergence Zone (ITCZ). The summer, on the other hand, is excessively dry, with only a few localized showers falling from August to December. Average temperature is 27 ºC year round and can reach 32 ºC during the day, especially during the rainy season. The relative lack of rainfall during the summer brings tradewinds and cooler, pleasant temperatures of around 26 ºC during the day and 22 ºC during the night.
Differences in precipitation between the rainy winters and the dry summers translate into dramatic landscape dynamics in the savannas. The intense rainfall causes river and lake levels to rise up to four meters. The impermeable clay soils of the campos are covered by rainwater and remain so for most of the season. Conversely, during the summer months, the lack of precipitation and the temporary availability of most water sources cause severe desiccation of soils and vegetation. This situation is particularly critical at the rivers’ headwaters, which depend on occasional rainshowers to maintain a minimal flow. Because the rivers are regulated by tides, twice in a 24-hour period the waters move back towards the headwaters, but the currents and volume of water in general are not enough to replenish water throughout the system. Life along the main rivers, on the other hand, is less affected, because water is available year-round.
The impact of the fluctuation of water resources on the lowland savannas cannot be overemphasized. Local people often refer to the seasons—
cheia (flood) and
seca (drought)—to explain their way of life, because the environmental conditions influence availability of resources and different strategies for their exploitation (
Figure 4).
Figure 4.
Landscape change due to climate and human action: (a) Teso dos Bichos archaeological mound in January and (b) in May. (c) Fishing in temporary lakes. (d) contemporary dam built in the savannas. Photographs by Denise Schaan, 2005.
Figure 4.
Landscape change due to climate and human action: (a) Teso dos Bichos archaeological mound in January and (b) in May. (c) Fishing in temporary lakes. (d) contemporary dam built in the savannas. Photographs by Denise Schaan, 2005.
The enormous impact on local economies of the timing and intensity of the seasons also would have impacted prehistoric aboriginal societies. Although palynological data are limited, and restricted to peripheral areas of Amazonia, changes in vegetation can be correlated with severely dry periods that may have greatly affected ancient subsistence systems, and perhaps destabilized political economies. The only available data for Marajó comes from a pollen profile from Lake Arari that indicates an expansion of savannas around 2,600 BP, probably correlated with an arid episode [
15]. This date is associated with the pre-Marajoara period.
Savanna soils are in general infertile, due to the combination of poor mineral content and intense leaching during the rainy season, removing nutrients from the upper horizon [
16]. As with most savanna environments, the Marajó
campos are characterized by a fine-textured, clayey soil, with limited infiltration capacity, which has the tendency to become waterlogged during the rainy season and to dry out excessively during the period without precipitation. In areas where the soil is exposed all day long to the equatorial heat, the earth cracks deeply, sometimes forming rounded, grassy berms. This desiccated soil is locally called “
terroada”.
Agricultural productivity in Amazonia is known to be greatly affected by rainfall and extension of the dry season [
12]. Both lack of water and flooding, conditions that exist in most tropical savannas, can create problems for long-term cultivation [
13]. It seems like a no-win situation, but indigenous societies of the past took an active hand in defying these seemingly inexorable conditions, though not for agricultural purposes
per se.
These anti-agricultural conditions are reflected in earlier research and reports. A study undertaken on Marajó Island by the Organization of American States had long ago characterized the “Type III” soils of Marajó coast as “restricted” for agriculture. The savanna soils were characterized as “Type IV”, or “unsuitable” for cultivation. The study concluded that under present conditions (poor soils and inadequate drainage systems), the area was not appropriate for cultivation [
17].
Another study, carried out by Chomitz and Thomas [
18], compared several areas of Amazonia regarding crop production. Areas in which precipitation is higher than 2,800 mm have yielded the production of manioc, banana, and pineapple, while maize has been produced only in areas where local rainfall is between 1,400 and 1,600 mm. The study has also demonstrated that the proportion of agricultural land tends to decrease with increased precipitation.
Even among the savanna soils, however, there are some differences between lowland and upland savannas. The different topography of these two habitats translates into variable rates of flooding, which, in turn, influence observed soil conditions. Overall soil chemical composition, in part caused by continuous submergence and leaching, results in distinctive vegetation patterns.
In Marajó, the soils are good for palms. Palms require humid soils; for this reason, they are well-adapted to flooded areas. The moriche palm (
Mauritia flexuosa), known locally as
buriti or
miriti, is one of the most useful palms in the Amazon (
Figure 3). The leaves can be used in roof thatching and weaving for hammocks, baskets, and the
tipiti (a tube used to press squeeze water out of bitter manioc dough) [
10]. The sap itself can be used to prepare brown sugar, alcoholic beverages, and yeast. The starch can be transformed into flour, starch pearls (similar to tapioca), and a kind of bread. The kernel of the nut yields oil similar to food oils. The larvae that grow in felled trunks are collected and eaten [
19]. The vitamin A and C-rich fruits are used to produce soft drinks and alcohol. Miriti palms are widely present on Marajó Island [
19].
In several areas of the Amazon, including the estuary, ethnographers, ethnobotanists, and geographers have identified domesticated and semi-domesticated plants that are believed to have been the result of centuries or even millennia of intentional human management [
3,
20,
21,
22,
23,
24,
25]. On Marajó, the dominance of certain plant species such as palms (especially miriti and
açaí [
Euterpe oleracea]), nondomesticated cotton, and several edible fruits, including a nondomesticated bromeliad, may indicate that prehistoric populations cultivated these plants for their consumption [
26].
It has been argued that intentional management, such as construction of raised fields, drainage systems, and soil mulching, might have overcome the adverse conditions to cultivation noted in environments such as poorly drained savannas [
27,
28]. To date, however, no evidence shows that such management ever had taken place on Marajó Island. The archaeological mounds display comparatively small areas, and are otherwise heavily stocked with cultural remains, which indicate they were primarily used as habitation and cemetery sites, not as agricultural fields. In addition, the anthropogenic dark earth (
terra preta) soils found in some archaeological sites on Marajó [
29] have varied horizontal and vertical distributions associated with abundant cultural materials, indicating they result from anthropogenic activities typically associated with the formation of middens and the like, not from intentional mulching for cultivation of domesticates.
3. Human Occupation of Marajó Island Savannas before the Conquest
The first inhabitants of those equinoctial lands were riverine populations dedicated to small-scale fishing and gathering of riverine shellfish. The archaeological record accounts for the presence of shell middens in the south of Marajó Island, along secondary small rivers, were shell-tempered pottery and stone ornaments have been reported [
30]. Because such sites have not yet been investigated, their chronology can only be guessed by relating them to shell midden populations known to have inhabited both the Atlantic northern coast and the lower Amazon, which have been dated, respectively, to 5,000 and 7,000 BP [
31,
32,
33].
Subsequently, small villages of pottery producers—probably incipient farming communities—appeared 3,500 years ago in the north, center, and southeast [
15]. They probably lived by hunting, fishing and gathering, as well as by small-scale cultivation. Such occupations were superficially studied by neo-evolutionist scholars in the late 1940s, who characterized them as “tropical forest tribes” [
34].
In the neighborhood of 1,600 years ago, regional, administratively autonomous societies, which can be referred to as “simple chiefdoms”, arguably on the basis of significant evidence, emerged and flourished on the center of the island [
35,
36]. Sociopolitical complexity is suggested by regional aggregation of mound sites, site size hierarchy, elaborate ceremonial ceramics, ancestor worship, differential treatment of the dead, and the presence of long distance exchange items [
34,
37] (
Figure 5). Landscape management would have led to forms of economic, symbolic, and political control of food resources and people, institutionalizing social hierarchy and inequality, even in the absence of documented intensive or semi-intensive cultivation. In a few decades, Marajoara mounds spread over the seasonally inundated savannas, occupying even places where the tropical forest peoples once lived. Organized around regional centers, the mound builders also constructed dams and fish ponds, arranged in very efficient hydraulic systems. Archaeological research has demonstrated that in choosing river headwaters to live along, the Marajoarans were taking advantage of extremely bountiful areas, such as where fish seasonally migrated to spawn [
35].
Figure 5.
Excavation of a Marajoara mound; Camutins site. Photograph by Carlos Mora, 2002.
Figure 5.
Excavation of a Marajoara mound; Camutins site. Photograph by Carlos Mora, 2002.
Figure 6.
Marajoara phase ceramics and green stone trade item. Plate, tanga, and greenstone from the Banco Santos Colletion, Museu de Arqueologia e Etnologia da Universidade de São Paulo. Photographs by Denise Andrade. Funerary urn from an anonymous collector. Anonymous photographer.
Figure 6.
Marajoara phase ceramics and green stone trade item. Plate, tanga, and greenstone from the Banco Santos Colletion, Museu de Arqueologia e Etnologia da Universidade de São Paulo. Photographs by Denise Andrade. Funerary urn from an anonymous collector. Anonymous photographer.
The flooded savannas are known to be generous in aquatic resources. At the beginning of the rainy season, after the first rains, fish move to the rivers headwaters, looking for a place to reproduce away from their predators. From there, they spread out over the flooded savannas, feeding off the riparian forest and floating meadows [
10,
38]. At the end of the rainy season, seasonal and permanent lakes shrink, whereas fish density increases, becoming more available to humans. It is during the period of transition between the two seasons that fish harvesting can be very productive, if managed correctly. Similar ecological conditions were observed in the region of Baurés, in the Bolivian Llanos de Mojos, where Erickson [
39,
40] identified a complex system of hydraulic earthworks designed for massive fish capture.
According to Roosevelt, analysis of faunal remains recovered from
Teso dos Bichos soil flotation samples show an almost complete absence of terrestrial fauna, but abundance of small fish. Large fish, such as the osteoglossids pirarucu (
Arapaina gigas) and aruana (
Osteoglossum bicirrhosum), were recovered only from pits and caches. Mammals and birds were rare and present only in one of the excavations. Small aquatic fauna, including small shells of mussuã turtle (
Kinosternon scorpioides), teeth of piranha (
Serrasalmus sp.), bones of traíra (
Hoplias malabaricus) and tamoatá (an armored catfish,
Hoplosternum littorale), comprised the bulk of the assemblage recovered from garbage and hearths. The specialists concluded that the assemblage patterns were consistent with massive capture in fishponds [
37].
Having their protein intake mainly from fish, Marajoara populations also relied on other diverse wild resources. Data on diet during Marajoara phase, however, is very limited and come mainly from the preliminary results of Roosevelt’s research in
Teso dos Bichos mound, as well as skeletal and dental analysis [
35,
37]. Carbonized plant remains recovered by Roosevelt in Teso dos Bichos site were comprised mainly of seeds, wood, and palm fronds. Açaí (
Euterpe olearacea), tucumã (
Astrocaryum vulgare), ingá (
Inga sp.), muruci (
Byrsonima crassifola) and taperebá (
Spondias lutea) are among the species that were identified [
37]. Taperebá and tucumã, besides being consumed by humans, are also an important part of the diet for several fish species. Fishermen mix manioc flour with the orange flesh of tucumã to produce bait balls [
10,
41]. Açaí seeds and bark were identified during the excavations at the Belém mound (M-17), indicating it was consumed at the site since at least A.D. 700 [
35]. The widespread consumption of açaí in Amazonia was reported by early travelers, as well as was by colonial documents [
10].
“As a staff of life, few palms around the world rival açaí” [
10]. It is difficult to exaggerate the economic, nutritive, and cultural importance of this palm in Amazonia, which has today become a fashion food in the Western world [
42]. Together with manioc flour and fish, the açaí (
Euterpe oleracea) juice has constituted the staple meal of thousands of riverine people. The current exploitation of açaí palm constitutes one of the most important economic activities on Marajó Island.
Every açaí palm yields several fruit stalks, containing about a thousand small fruits each [
10]. These blueberry-sized fruits consist of a hard seed surrounded by a thin (1-2 mm) layer of purple pulp. The pulp represents from 5 to 15% of the fruit’s volume [
43]. After released from the stalk, the fruits are washed and soaked in water for a while in order to soften the pulp. The fruits are then kneaded by hand over a sieve, while water is added. This is done several times until all the pulp has been extracted and diluted in water, producing a thick purple juice of high caloric value and exquisite taste. Five kilos of fruit are necessary to prepare, on average, three liters of juice [
43]. One liter of prepared açaí juice has 2.6 grams of protein, representing 25 to 30% of the daily protein need [
43]. A comparative study on the aminoacid content of açaí, indicated a profile similar to the chicken egg [
43]. The same study also compared açaí to
castanha do pará (Brazil nut,
Bertholletia excelsa) and
palmito (heart of palm) indicating that castanha do pará and açaí can complement each other, although both
castanha and
palmito show some deficiencies in several necessary amino acids [
43].
Research indicates that açaí palms are adapted to soils with poor levels of P, N, Ca and Mg, due to two main mechanisms. One is the presence of bacteria that fix the N on the aerial surface of their roots. In addition, due to the large amount of roots, the palm is able to extract nutrients from a large volume of soil [
43]. Açaí trees in general occur together with other trees, especially because young trees need protection from the sun. For this reason, açaí trees are usually found associated with
buriti, another palm.
The probable consumption of huge quantities of açaí in pre-Columbian Marajó Island is also supported by other lines of evidence. Studies of bone chemistry and dental analysis in specimens in museum collections indicated staple consumption of plant protein, particularly seeds [
37]. In several samples, specialists have identified dental wear consistent with the consumption of gritty food, which had caused heavy dental abrasion, even in youngsters [
34,
35,
37]. Given the rarity of maize in bone chemistry, Roosevelt considered it an accessory crop, at most, perhaps used for producing beverages.
Three possible sources of starch (palm, manioc, and wild rice) for Marajoara populations have been suggested in the specialized literature. Based on the available data, it is neither possible to evaluate which one of these options was in fact in use nor to assess their relative importance in the diet. The ceramic assemblages, however, seem to indicate that a major source of starch was used. The presence of griddles may indicate both baking of
beiju (flat manioc bread) and roasting of the sieved dough to produce crunchy flour. On the other hand, the abundance of mortars is consistent with grinding of shredded palm pith or manioc [
35].
Considering the unsuitability of the low savannas for agriculture and the need to envision a staple food resource for Marajoara populations, Meggers [
44] proposed that prehistoric populations may have used palms for starch. The use of palms for starch has been recorded mainly among foragers, probably because sedentary populations would have had to travel constantly in search of palms. In spite of the availability of palms in South American tropical forests, it is unclear why palm starch is not widely used among ethnographic societies. Piperno and Pearsall [
45] point out that it may well be related to historical factors or to the availability of yet other forms of starch.
The Marajoara political economy, based on massive fish capture and the symbolic control of such resources, through the occupation of the best fishing areas by the elite, would weaken after 1,350 AD. The 14th century is taken as the epoch of the Marajoara social organization’s downfall, for still poorly known reasons. It is possible that environmental changes caused by excessive rains or periods of prolonged droughts affected aboriginal subsistence strategies on Marajó, as Meggers [
46,
47,
48] has suggested, causing economic crises that could have distressed already unstable sociopolitical systems. This period is also marked by the arrival on the island of Arawakan populations (Aruã Phase) coming from the northern islands and the Amapá coast [
34]. Thermoluminescence dates indicate that Aruã sites were established on Marajó’s northern coast around A.D. 1300 [
15]. It is not totally impossible that the arrival of the Aruã had played some role in the breakdown of Marajoara political economy. Climate change, causing subsistence stress, might also have been a cause for cultural change, as scholars have suggested. Sufficient data seem incomplete; nevertheless, at present to retrodict conclusively the proximate causes of the Marajoara collapse.
4. Impacts of the Conquest and Colonial Economies
The first Europeans to contact the Marajó natives were Dutch and French trade companies’ employees who arrived in the Amazon Estuary at the beginning of the 16th century. There they met Arawakan speakers, who since the 14th century had been migrating along the Guianas’ northern shores, entering Marajó Island through the northern coast [
34,
49]. Since the natives already had well-established trade networks, the European companies exploited them in order to acquire desired raw materials, such as hides, dyes, and spices, for European markets. Modifications in indigenous local markets and exchange systems by the introduction of European goods are thought to have contributed to severe political imbalances, spread of diseases, demographic decrease, and resettling of indigenous populations [
50,
51]. This process was well on its way when the Portuguese arrived in the delta of the Amazon River in 1616, founding what is now the city of Belém, just across Marajó Bay.
Ethnohistorical information about the populations that inhabited Marajó Island on the eve of European contact is consistent with the archaeological record for the terminal Marajoara period or Cacoal phase [
52]. Colonial documents indicate that several "unrelated" Indian “Nations” generically known as
Nheengaíbas (meaning non-Tupian speaking people)
, inhabited the "islands" [mounds] located on the savannas. Those nations were reportedly extremely bellicose, and organized in up to 29 distinct social groups, each one led by a warrior chief [
53]. The existence of several distinct indigenous societies would have been consistent with several chiefdoms, which later would have become decentralized but probably still could have aggregated people around a leader in times of war. When the Portuguese launched offensive expeditions for the enslavement and “pacification” of the
Nheengaíbas, the Indians reportedly abandoned their settlements and spread over the territory [
55]. Their resistance was so efficient that they were never defeated in their own land, which at least partly explains the total absence of colonial descriptions of their villages and way of life. A peace treaty in 1659 put an end to the conflict. By the end of the 18th century, the remaining population had been relocated to missions in the lower Amazon [
34]. Available evidence suggests that the
Nheengaíbas were the ethnohistorical counterpart of the Marajoara societies in their last period [
52].
In the first years, the relationship between the native chiefs and the Portuguese was not friendly both on the mainland and the islands. If for the Dutch and the French the Amerindians were seen as consumers and producers, as far as the Portuguese were concerned, Indians were a cheap labor force for their colonial enterprises. The indigenous response to labor exploitation and involuntary servitude was war and rebellion. In the archipelago, which terrain the natives knew better than the Portuguese, they often employed guerrilla tactics against the intruders, abandoning their villages and hiding in the forest along the rivers, surprising their would-be capturers in bloody encounters [
52,
55]. In this context, the role of missionaries would become crucial in establishing a sort of armistice that would in effect guarantee Portuguese sovereignty over the land, putting an end to the Indians’ autonomy [
56].
By the middle of the 17th century, missionaries were able to first settle in the island, counting on the Indians’ labor. At that time, fish was the main resource they could exploit there. During the colonial period, fishing companies (
pesqueiros reais) employing
caboclos (non-Indian natives, or indigenous peasantry), militia, and Indians, were established by the crown in a few places of the lower Amazon. The objective was to exploit highly productive riverine and coastal areas [
57].
In 1692 one of these companies was located in the town of Joanes, on the Marajó eastern coast, in order to fish
tainha (
Mugil brasiliensis) (a kind of white mullet) and
gurijuba (
Tachysurus luniscutis) [
57,
58,
59]. Although the written sources do not give many details,
tainhas were likely caught in poisoned fenced streams, where they went to spawn in early August. Colonial accounting documents dated to the second half of the 18th century indicate that thousands of dried fish (especially tainhas), turtles, manatees, crabs, as well as turtle butter, among other goods, were being sent from Joanes (Vila de Monforte), Salvaterra, and Monsarás periodically to Belém. For example, between September 18 and October 12, 1767, the towns of Monsarás and Monforte provided 48,500
tainhas and 21 baskets of fish [
57]. Considering that the
tainhas would weigh 4 kg in average, it means a production of 194,000 kg of dried
tainhas in 24 days, or 8,083 kg a day.
At the beginning of the 18th century, however, that economy was already facing problems. Due to predatory fishing, fish productivity enormously decreased. At the same time, the Indians were forbidden to cultivate their gardens and their families were often set apart according to missionary needs of work force in their villages. Also, the lack of fish and salt affected the Indians; thus many of them decided to return to their previous way of life, thus abandoning work for the fish company. Arguments between missionaries and government employees became more frequent. Missions from diverse orders also disagreed on the treatment to be dispensed to the Indians.
Given such a situation, the Mercedarians decided to abandon fishing and start raising cattle. It is not clear when cattle were introduced in the island, but there are reports that the first animals arrived in 1644 together with African slaves brought from the Cape Verde Islands [
60]. In the beginning, the cattle destroyed the few Indian gardens known to exist, so the Portuguese crown stipulated norms and conditions for cattle ranching, which then started to proliferate. Once fisherman, Indians were now becoming cowboys.
The pressure to change economic lifeways was so intense that many Indians escaped to the island’s vast interior. As if economic transformations were not sufficient to bring about instability in lifeways, in 1750 a measles epidemic had a devastating effect on the island, killing many Indians. As the work force was thus dramatically reduced, in the second half of the 18th century, ranchers introduced African slaves. These slaves were set to work on ranches, sugar cane mills, and in the production of manioc flour and extrativism related to forest and savanna products. Eventually fleeing from ranches, Indian and blacks would join forces in the mocambos (Maroon settlements). The fact that African males were more numerous led to frequent marriages with Indian females, originating the ethnic miscegenation that characterizes Marajoara people today. Some elders tell that their Indian grand-grandmothers were captured (laced up) by their black grandfathers in the savannas.
5. Conflicts over Land and Water
During the 20th century, and up to today, fishing continues to be rewarding. Current fishing practices on Marajó are comprised of very simple, cost-effective strategies, similar to those in pre-Colonial times. Along small streams,
caboclos (descendants of native Indians, African slaves, and Portuguese colonists) place removable fences or build temporary earth and wood dams at high tide. At low tide, fish that had entered the streams are trapped behind the fences and can be easily harvested with nets or poison [
10]. This practice has yielded greater returns at the headwaters of rivers on the savanna, causing seasonal movement of fisherman to work in these areas during the peak of the fishing season, beginning in July. Since these areas are often owned by ranchers, serious conflicts have been reported among
caboclos and landowners, both aiming at selling the fish to the
geleiras (refrigerated boats that transport the fish to the Belém market) [
38]. Competition over the best fishing areas and eagerness for fast cash return has led
caboclos to practice predatory fishing, using inappropriate net size, and consequently discarding small fish, which has virtually no market value. The decrease of both fish species diversity and relative size has been noticed since the 1950s [
9], and references to decrease in fishing productivity are increasingly abundant in the literature (e.g., [
61]). For instance, [
38] pointed out that the São Pedro festival, which was normally celebrated in November during the 1970s, was transferred to October and then to September, because of fish scarcity. In Salvaterra, a town located on the eastern coast, community fisheries formerly yielded abundant fish resources for the entire town, but the
pesca de arrastão (seine nets) have apparently jeopardized fish reproduction [
62].
In Pre-Columbian times, better management strategies would stretch the availability of aquatic resources throughout the dry season and well into the first months of the rainy season. Ability to store dry fish and retain live turtles would account evidently for the provision of protein during the remainder of the year.
Serious conflicts also have been reported between descendants of African slaves (
quilombolas) and ranchers over fishing territories. Organized into small communities (
quilombos), the
quilombolas have argued for the ownership of land that has been traditionally exploited by their groups for decades. In the Gurupá River headwaters, located in southeastern Marajó Island, 154 families struggle to survive since they were pushed off their lands, 36 years ago, and prohibited to have access to the resource areas that had up to that time guaranteed their sustenance. They had lived from fish caught in seasonal lakes and streams, small garden cultivation, and wild resources [
63], but their economic strategies changed due to resettling, so did the environment.
Teles and Acevedo-Marin [
63] recount that in the 1980s the African-descendants lived along the small streams that make up the Arari River headwaters, where they exploited fish. As they were relocated to the Gurupá River, they had to adapt to a number of different niches and turn to the cultivation of manioc (but also maize, yam, sesame, and banana) and the gathering of açaí, which has market value. In the 1990s, as the market value for açaí increased, riparian vegetation was substituted by açaí palms. This has led to different forms of property, either exploited by domestic units or by independent entrepreneurs. Together with manioc flour and fish, the açaí juice constitutes a staple meal for most riverine populations. The current exploitation of açaí palm constitutes one of the most important economic activities of Marajó Island society. Although the açaí fruit was historically important in the diet, as the archaeological data also suggest, the large scale exploitation of it has happened at the expense of the environment.
Both the concentration of domestic households along small stretches of the Gurupá River, and the need for land to substitute the more extensive fishing and gathering economy of the past, have cased important landscape transformations and environmental problems. At the river margins, the removal of riparian vegetation for gardens, cattle raising, house building, and logging have caused environmental distress. The transportation of açaí by motor boats has led to the contamination of the river with oil and plastic. Pigs and cattle have caused pollution and sedimentation. Altogether these factors have caused a decrease in fish and shrimp, besides an increasing lack of water for domestic use [
63].
While landowners have prohibited the population to fish in the headwaters located inside their ranches, cattle owners have also practiced hydraulic management unrestrictedly. In order to maintain the availability of water for cattle during the dry months, they have deviated water courses, excavated “rampas” (rectangular artificial ponds), and built dams. In some places, such actions have created several ecological problems, including shortages of fish and water. Lakes that once were permanent now dry up soon after the rains have ceased in July or August. The savannas have become more and more desiccated; the fires are more frequent, and the population have suffered with the diminution of fish and water. The headwaters of rivers, the once bountiful areas were Marajoara culture emerged, have become the worst places to live. Those who do not abandon their houses during the summer tend to endure lack of water and suffer the spread of tropical diseases.
In recent decades, cattle ranchers have seen their gains decline because of the competition with ranchers from the south of Pará state, whose cattle have more credibility in the market. Several of them have sold their properties, which have been also divided between multiple heirs. Some have transformed their ranches into country hotels for ecological tourism, which is, as noted above, an increasingly attractive economic option for some investors. If this trend is to continue, it might lead to the decrease of environmental degradation, but a regional ecological policy would be necessary in order to avoid the micro-management of rivers inside the large landowners’ ranches without the proper care for the whole landscape.
6. The Snake People
Before undertaking archaeological research in Marajó, I had studied a Museum collection of Marajoara phase archaeological ceramics. Museum work is something many researchers do before going to the field, as a first approach to the culture in question. I was then impressed by the ubiquitous depiction of snakes over the ceramics, and realized that snakes were powerful beings that once populated Marajoara myths (
Figure 7).
Not surprisingly, the myth of the anaconda or
cobra-grande, in its many variations, is a recurrent theme in Amazonian mythology [
64,
65,
66]. According to the Desana, a Tucanoan group living in the Northwest Amazon, snakes are the progenitors offish, thus snake tales are frequently related to fish abundance [
65]. One of the variations of the myth tells that the Sun created everything on Earth except people and fish, which were created later. All the tribes were transported to the Earth inside of the live snake-canoe, which was surrounded by fish. The tribes settled at the headwaters, and the position of every tribe along the river ultimately corresponds to their position inside the mystical snake-canoe (63: 26). The presence of snake skin patterns on funerary urns shows the importance of snakes in Marajoara religious symbolism. If Marajoara mythology was similar to that of the northwestern Amazonian populations, according to which an ancestral anaconda (the snake-canoe) brought the elite to the Earth, (justifying their presence in areas of resource concentration and their rights to the land of their ancestors), the snake symbolism was not only a cultural marker, but it also would have played a critical role in the ideological justification of Marajoara polities’ political economy, landscape management, and effects on diversity [
67].
Figure 7.
Depiction of snakes over Marajoara phase vessels. Banco Santos collection, at Museu de Arqueologia e Etnologia at the University of São Paulo. Photographs by Denise Andrade.
Figure 7.
Depiction of snakes over Marajoara phase vessels. Banco Santos collection, at Museu de Arqueologia e Etnologia at the University of São Paulo. Photographs by Denise Andrade.
What is even more interesting is the fact that the relation between snakes, fish, and rivers has survived throughout the colonial period to these days in collective memories. Perhaps there is synergy in this. In a tale recorded by Alexandre Rodrigues Ferreira, an explorer and early naturalist, who navigated across the center of the island in 1783, in the beginning the island did not have its current labyrinth of rivers. He was told that the center of the island was primordially inhabited by uncountable snakes. Those snakes were suffering because of the drought, so they escaped from the center to the coast, in search of water. In their path, given the weight and magnitude of their bodies, they left imprinted in the land their sinuous figures. Such imprints turned into rivers filled with water when rain water came, and together they filled up the large, forming large rivers [
54].
In a Dalcídio Jurandir novel, the theme of snakes appears in the voice of an Afro-Brazilian woman. She describes the time of drought, when all life goes away with the waters. In her account, the mother snake was sleeping in the bottom of the river, but awakes as the waters recede, and then leaves for the great waters. In doing so, she takes the fishes and all the life with her, provoking much suffering [
41,
68].
Snakes appear in cowboy tales as well. In a novel by Tocantins [
41,
69], a cow used to defy the most skilled cowboy, running to the river when lassoed. As the rope got cut and left behind, the next day they would find the rest of it rolled up in the river margin, transformed into an enormous snake. Cows and snakes, like all life, have something in common, in this contemporary Marajoara view of reality.
A more recent account comes from the work of Teles and Acevedo-Marin in the Gurupá River community of African descendants. Their subjects recall that there were two snakes in the river; their breathing marked the pace of tidal waters. They inhaled at the high tide, and exhaled during the low tide. People explained that the lakes are getting drier and drier every year, and the fishes were getting scarcer, because one of the snakes had left. They believe that the snake was frightened away by the cultivation of açaí palms and logging [
63], both recent economic and landscape transformations.
The myths and tales of Marajoara populations have survived throughout the centuries in different forms, yet have retained similar content. This literature, both written and oral, constitutes part of the cultural and literary diversity of the area. Their connection with the waters, the ultimate substance of diversity and life itself, is always acknowledged in their relation to fishes, as well as to the snakes, the fishes’ protectors, which are clearly ancient ideas traceable to pre-Columbian societies of Amazonia.
7. Conclusions
In order to model and study aboriginal subsistence strategies in Amazonia, scholars have historically relied on ethnographic studies of surviving indigenous groups, which despite the population losses and displacement suffered in the last five hundred years, still represent a living testimony of human strategies on Amazonian landscapes. Research among
caboclo populations are also regarded as important in providing information about strategies of food production, since their household economies are organized around traditional practices such as hunting, fishing, foraging, and slash-and-burn cultivation. Although the
caboclo communities tend to rely both on conventional forest products and industrialized goods obtained from the city markets, they maintain some traditional knowledge that is in fact instructive about the relations between human societies and the tropical forest environment [
14,
63,
70,
71,
72].
Aquatic resources are considered to have been a major protein source for aboriginal Amazonians, favoring sedentary settlements, population growth, and the development of complex social systems. Carneiro [
73] points out that the availability of aquatic resources encouraged sedentary life, agricultural development, and population growth.
Although scholars are ready to accept that the exploitation of aquatic resources favored sedentary life and population growth, they are reluctant in considering those resources as a staple food source that could also generate surplus. Seasonal fluctuations in water levels in the
várzea and flooded savannas, for example, have been regarded as a major limitation on such a subsistence system [
37,
76]. Meggers [
79,
80] has pointed out, however, that seasonality could have been mitigated by exchange between neighboring groups, who occupied environments with different productivity levels. Beckerman [
81], in contrast, pointed out that although water levels and fish migratory patterns may be factors that affect the seasonal availability of aquatic resources, fishing is still a very reliable source of protein in Amazonia when compared to other foods. In fact, aquatic mammals can yield as much protein as large terrestrial animals, and turtles and reptiles can be kept alive in pens for future consumption.
Fish availability can be predicted on the basis of spawning and feeding behavior, allowing for mass capture in optimal areas where they are isolated in shallow water and ponds. Aquatic resources allow for management and consumption patterns that could be more accurately described as husbandry or farming, instead of the traditional comparison with hunting. Ethnohistorical and ethnographic evidence suggests that aboriginal population developed an array of different techniques for the capture and storage of aquatic resources in the face of diverse geographic and seasonal conditions. Reported methods include spearing, angling, netting, trapping, and poisoning. Use of traps, dams, and weirs are also reported and may have included more permanent facilities. In shallow waters and small streams, the use of poison obtained from either wild or cultivated plants is widely disseminated in Amazonia [
76,
81,
82,
83,
84,
85,
86,
87,
88]. High productivity in fishing is obtained when there is territorial control, especially since some permanent devices, such as traps and dams, require periodical maintenance [
14].
In the upper Uaupés, for example, access and control of the most productive fisheries have established patterns of hierarchy, mirrored in the kinship system [
88]. Techniques for storage of aquatic resources have also been observed. The Siona and Secoya, a Western Tukanoan group living at the Aguarico River, a tributary of the Rio Napo in Ecuador, smoked turtle eggs in order to keep them edible for months [
88]. Fish and meat could also be preserved in manatee fat or turtle oil [
76]. Early chronicles reported that fish and turtles were kept in corrals and pens to be eaten when needed, while dried fish was also used for trade [
50,
51,
54,
90,
91]. Aboriginal populations were also concerned with the preservation of fisheries. The Tukano, for example, have restricted rules regarding the preservation of the riparian forest, which is an essential source of food for aquatic animals [
83]. The literature on Amazonian environments [
92,
93] emphasizes the abundance of aquatic life, even today, when logging, cattle ranching and fires have greatly reduced the flooded forests necessary for the reproduction and sustainability of fisheries.
In order to maintain large, dense, permanent settlements, and also generate surplus, aboriginal subsistence strategies must have involved intensification of production of foodstuffs that could be stored, and in doing so, they changed environments into landscapes. Marajoara mounds are located in areas highly affected by seasonal variation in water levels. These areas are known by islanders to furnish abundant aquatic resources when the waters subside and natural ponds and pools retain aquatic animals that cannot return to the rivers. This natural reflux of water and biological behavior of aquatic species is in some measure managed today, through simple techniques such as damming small streams and poisoning ponds with locally occurring plants that are rich in bioactive compounds. Although poorly managed, these resources are still highly productive. Practices of water-management at ranches located on open savannas, such as the construction of rampas (ponds for retaining rain water) and the damming of streams, indicate that timing of flood and drought requires administration in order to guarantee the survival of people and livestock in those locations.
It is possible that the present day savanna environments in eastern Marajó have been maintained through seasonal fires. Researchers have pointed out that fire has been a fact of landscape change in prehistoric times [
94,
95]. Both wild and controlled fires might have helped to define the savanna environment which has also been subjected to fire during the 20th century for terrestrial turtle hunting, as well as lizard egg collecting [
42].
Today researchers agree that pre-Columbian Amazonian societies profoundly impacted their immediate surroundings, through dynamic manipulation of crops, animals, and landscape [
22,
96,
97,
98,
99]. However, population losses, and the replacement of indigenous population by European descendants have contributed to the loss of knowledge of landscape, crops, and aquatic fauna management practices [
35,
97].
We have much to learn with pre-Columbian cultural systems on Marajó Island, since they made it possible for a complex society to flourish based on local resources and simple, but ingenious, techniques. Today the environment is dramatically affected by poor management of ecological resources, leading to predictable degradation, and huge social inequality. The ancient Marajoaras once built their own ecology, exploiting natural resources in sustainable ways, and leaving us lessons which are still imprinted in the landscape and reflected in much of the island’s diversity. Such lessons need to be included in the array of scientific research data that is being produced on Amazonian landscapes; all of them to be heard by those interested in a better future for Amazonia, and consequently, for the planet.