Nationwide Tracing of Two Top Freshwater Fish Invaders in Greece Using Environmental DNA Sampling
Abstract
:1. Introduction
2. Materials and Methods
2.1. Water Sampling and Data Collection
2.2. eDNA Analysis
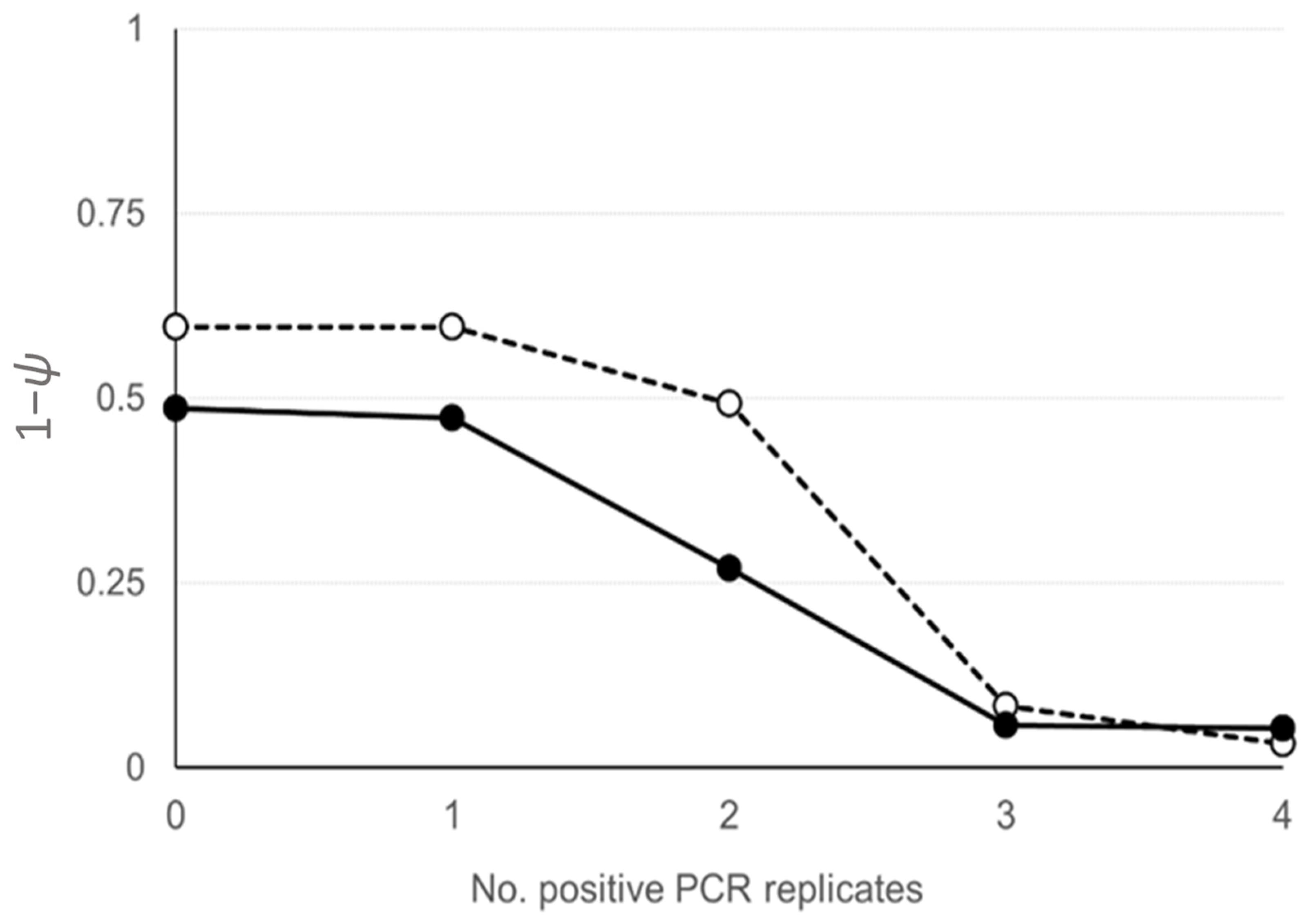
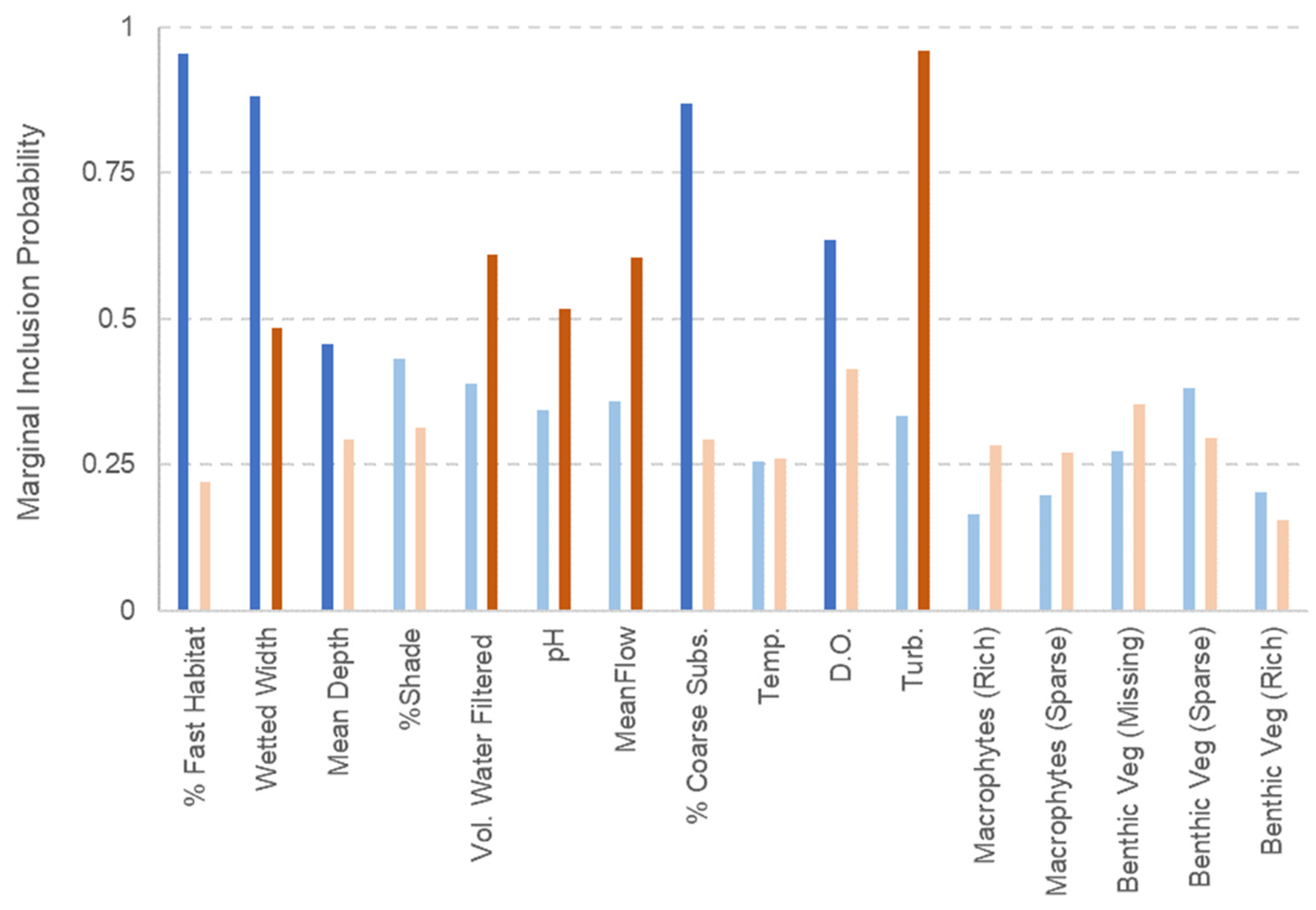
2.3. Statistical Analysis
2.4. Ethical Statement
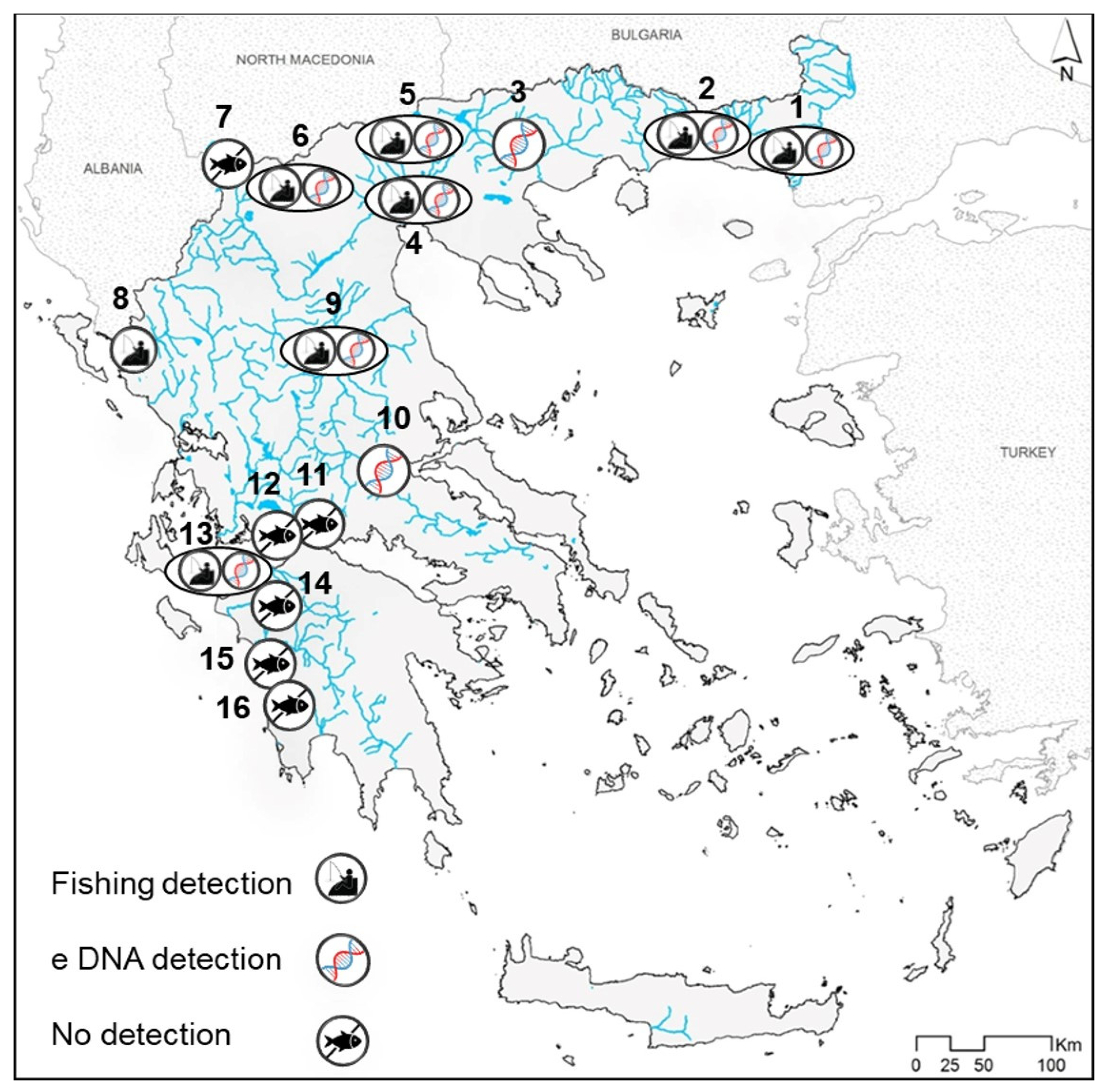
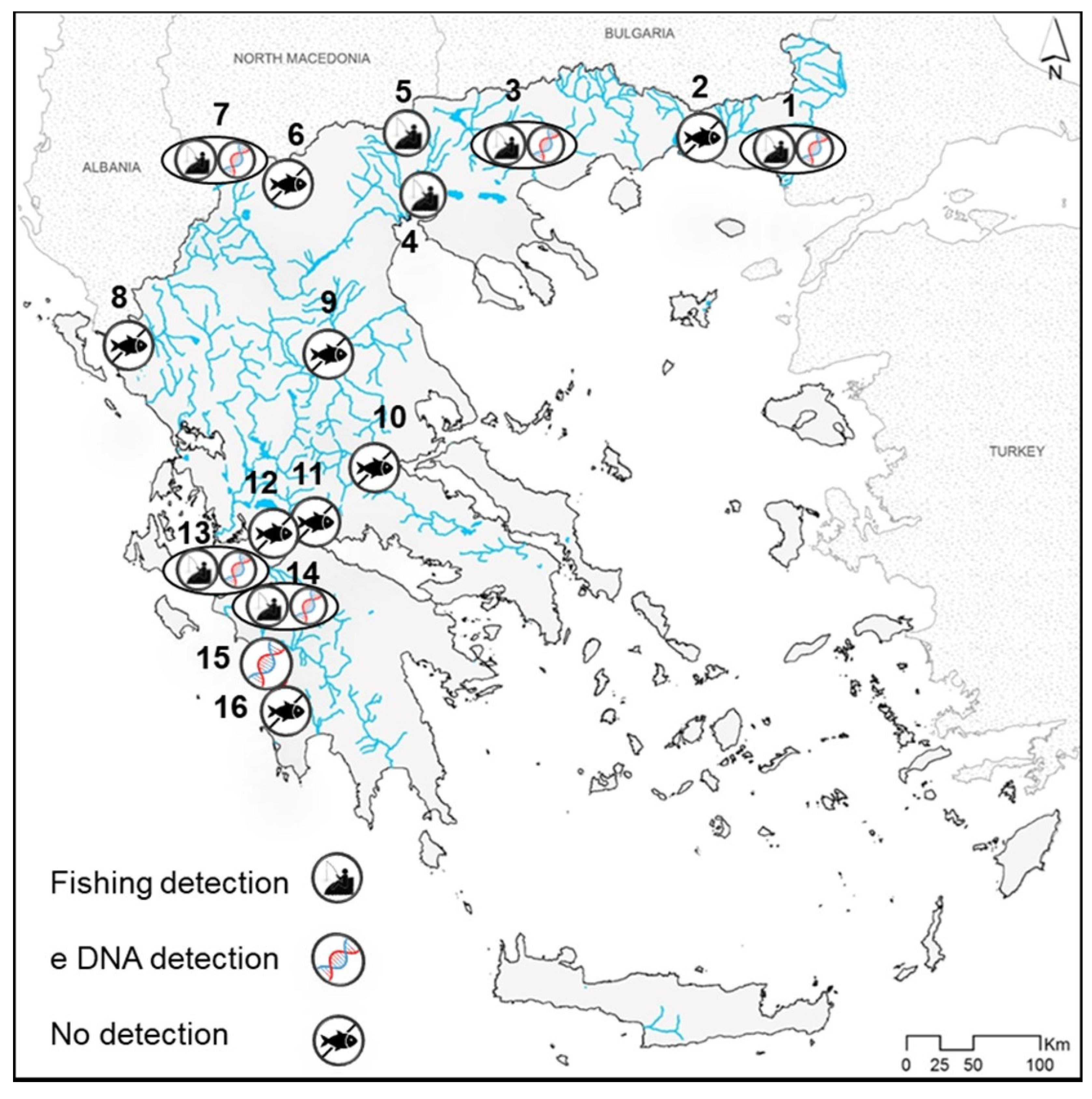
3. Results
4. Discussion
4.1. eDNA—A Technique for Nationwide Invasive Species’ Monitoring
4.2. New Records of the Two Invaders and Insights on Their Dispersal
4.3. Top Invader Pockets, Early Detection and Management Intervention Monitoring with eDNA
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Butchart, S.H.M.; Walpole, M.; Collen, B.; Van Strien, A.; Scharlemann, J.P.W.; Almond, R.E.A.; Baillie, J.E.M.; Bomhard, B.; Brown, C.; Bruno, J.; et al. Global biodiversity: Indicators of recent declines. Science 2010, 328, 1164–1168. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.N.; Balmford, A.; Brook, B.W.; Buettel, J.C.; Galetti, M.; Guangchun, L.; Wilmshurst, J.M. Biodiversity losses and conservation responses in the Anthropocene. Science 2017, 356, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Richman, N.I.; Böhm, M.; Adams, S.B.; Alvarez, F.; Bergey, E.A.; Bunn, J.J.S.; Burnham, Q.; Cordeiro, J.; Coughran, J.; Crandall, K.A.; et al. Multiple drivers of decline in the global status of freshwater crayfish (Decapoda: Astacidea). Philos. Trans. R. Soc Biol. Sci. 2015, 370, 20140060. [Google Scholar] [CrossRef] [PubMed]
- Reid, A.J.; Carlson, A.K.; Creed, I.F.; Eliason, E.J.; Gell, P.A.; Johnson, P.T.J.; Kidd, K.A.; MacCormack, T.J.; Olden, J.D.; Ormerod, S.J.; et al. Emerging threats and persistent conservation challenges for freshwater biodiversity. Biol. Rev. 2019, 94, 849–873. [Google Scholar] [CrossRef]
- Maxwell, S.; Fuller, R.A.; Brooks, T.M.; Watson, J.E.M. The ravages of guns, nets and bulldozers. Nature 2016, 536, 143–145. [Google Scholar] [CrossRef] [PubMed]
- Gherardi, F. Invasive crayfish and freshwater fishes of the world. Rev. Sci. Tech. (Off. Int. Epizoot.) 2010, 29, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Pyšek, P.; Hulme, P.E.; Simberloff, D.; Bacher, S.; Blackburn, T.M.; Carlton, J.T.; Dawson, W.; Essl, F.; Foxcroft, L.C.; Genovesi, P.; et al. Scientists’ warning on invasive alien species. Biol. Rev. 2020, 95, 1511–1534. [Google Scholar] [CrossRef]
- Doherty, T.S.; Dickman, C.R.; Nimmo, D.G.; Ritchie, E.G. Multiple threats, or multiplying the threats? Interactions between invasive predators AC S. Kaminskas Pacific Conservation Biology and other ecological disturbances. Biol. Conserv. 2015, 190, 60–68. [Google Scholar] [CrossRef]
- Clavero, M.; García-Berthou, M. Invasive species are a leading cause of animal extinctions. Trends Ecol. Evol. 2005, 20, 110. [Google Scholar] [CrossRef]
- Kaminskas, S.; Lymbery, A. Alien fish ascendancy and native fish extinction: Ecological history and observations on the Lower Goodradigbee River, Australia. Pac. Conserv. Biol. 2022, 29, 38–73. [Google Scholar] [CrossRef]
- Piria, M.; Simonović, P.; Kalogianni, E.; Vardakas, L.; Koutsikos, N.; Zanella, D.; Ristovska, M.; Apostolou, A.; Adrović, A.; Mrdak, D.; et al. Alien freshwater fish species in the Balkans—Vectors and pathways of introduction. Fish. Fish. 2018, 19, 138–169. [Google Scholar] [CrossRef]
- Yao, M.; Zhang, S.; Lu, Q.; Chen, X.; Zhang, S.Y.; Kong, Y.; Zhao, J. Fishing for fish environmental DNA: Ecological applications, methodological considerations, surveying designs, and ways forward. Mol. Ecol. 2022, 31, 5132–5164. [Google Scholar] [CrossRef] [PubMed]
- Harper, L.R.; Handley, L.L.; Hahn, C.; Boonham, N.; Rees, H.C.; Gough, K.C.; Lewis, E.; Adams, I.P.; Brotherton, P.; Phillips, S.; et al. Needle in a haystack? A comparison of eDNA metabarcoding and targeted qPCR for detection of the great crested newt (Triturus cristatus). Ecol. Evol. 2018, 8, 6330–6341. [Google Scholar] [CrossRef] [PubMed]
- Barnes, M.A.; Turner, C.R.; Jerde, C.L.; Renshaw, M.A.; Chadderton, W.L.; Lodge, D.M. Environmental conditions influence eDNA persistence in aquatic systems. Environ. Sci. Technol. 2014, 48, 1819–1827. [Google Scholar] [CrossRef] [PubMed]
- McKee, A.M.; Spear, S.F.; Pierson, T.W. The effect of dilution and the use of a post-extraction nucleic acid purification column on the accuracy, precision, and inhibition of environmental DNA samples. Biol. Conserv. 2015, 183, 70–76. [Google Scholar] [CrossRef]
- Mauvisseau, Q.; Kalogianni, E.; Zimmerman, B.; Bulling, M.; Brys, R.; Sweet, M. eDNA-based monitoring: Advancement in management and conservation of critically endangered killifish species. Environ. DNA 2020, 2, 601–613. [Google Scholar] [CrossRef]
- Francis, R.A. Handbook of Global Freshwater Invasive Species, 2nd ed.; Earthscan: New York, NY, USA, 2012; 455p. [Google Scholar]
- Pyke, G.H. Plague minnow or mosquito fish? A review of the biology and impacts of introduced Gambusia species. Annu. Rev. Ecol. Evol. 2008, 39, 171–191. [Google Scholar] [CrossRef]
- Koutsikos, N.; Zogaris, S.; Vardakas, L.; Kalantzi, O.I.; Dimitriou, E.; Economou, A.N. Tracking non-indigenous fishes in lotic ecosystems: Invasive patterns at different spatial scales in Greece. Sci. Total Environ. 2019, 659, 384–400. [Google Scholar] [CrossRef]
- Vardakas, L.; Koutsikos, N.; Perdikaris, C.; Petriki, O.; Bobori, D.; Zogaris, S.; Giakoumi, S.; Fitoka, E.; Tompoulidou, M.; Tsiaoussi, V.; et al. The fish fauna in lentic ecosystems of Greece. Mediterr. Mar. Sci. 2022, 23, 223–265. [Google Scholar] [CrossRef]
- CEN. EN 14011; Water Quality—Sampling of Fish with Electricity. European Committee for Standardization: Brussels, Belgium, 2003; 18p.
- Ye, J.; Coulouris, G.; Zaretskaya, L.; Cutcutache, L.; Rozen, S.; Madden, T.L. Primer-BLAST: A tool to design target-specific primers for polymerase chain reaction. BMC Bioinform. 2012, 13, 134. [Google Scholar] [CrossRef]
- Spens, J.; Evans, A.R.; Halfmaerten, D.; Knudsen, S.W.; Sengupta, M.E.; Mak, S.S.T.; Sigsgaard, E.E.; Hellström, M. Comparison of capture and storage methods for aqueous macrobial eDNA using an optimized extraction protocol: Advantage of enclosed filter. Methods Ecol. Evol. 2017, 8, 635–645. [Google Scholar] [CrossRef]
- Weldon, L.; O’Leary, C.; Steer, M.; Newton, L.; Macdonald, H.; Sargeant, S.L. A comparison of European eel Anguilla anguilla eDNA concentrations to fyke net catches in five Irish lakes. Environ. DNA 2020, 2, 587–600. [Google Scholar] [CrossRef]
- Mauvisseau, Q.; Burian, A.; Gibson, C.; Brys, R.; Ramsey, A.; Sweet, M. Influence of accuracy, repeatability and detection probability in the reliability of species-specific eDNA based approaches. Sci. Rep. 2019, 9, 580. [Google Scholar] [CrossRef] [PubMed]
- Mauvisseau, Q.; Davy-Bowker, J.; Bulling, M.; Brys, R.; Neyrinck, S.; Troth, C.; Sweet, M. Combining ddPCR and environmental DNA to improve detection capabilities of a critically endangered freshwater invertebrate. Sci. Rep. 2019, 9, 14064. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, D.I.; Nichols, J.D.; Lachman, G.B.; Droege, S.; Andrew Royle, J.; Langtimm, C.A. Estimating site occupancy rates when detection probabilities are less than one. Ecology 2002, 83, 2248–2255. [Google Scholar] [CrossRef]
- MacKenzie, D.I.; Nichols, J.D.; Royle, J.A.; Pollock, K.H.; Bailey, L.L.; Hines., J.E. Occupancy Estimation and Modeling: Inferring Patterns and Dynamics of Species Occurrence, 1st ed.; Academic Press: New York, NY, USA, 2006; 643p. [Google Scholar]
- Royle, J.A.; Dorazio, R.M. Hierarchical Modeling and Inference in Ecology: The Analysis of Data from Populations, Metapopulations and Communities, 1st ed.; Academic Press: London, UK, 2008; 443p. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2023. [Google Scholar]
- Burnham, K.P.; Anderson, D.R. Multimodel inference: Understanding AIC and BIC in model selection. Sociol. Methods Res. 2004, 33, 261–304. [Google Scholar] [CrossRef]
- Dorazio, R.M.; Erickson, R.A. ednaoccupancy: An r package for multiscale occupancy modelling of environmental DNA data. Mol. Ecol. Resour. 2018, 18, 368–380. [Google Scholar] [CrossRef]
- Ghosh, S.K.; Gelfand, A.E. Latent risk approach for modeling individual level data consisting of multiple event times. J. Statist. Res. 1998, 32, 23–38. [Google Scholar]
- Watanabe, S.; Opper, M. Asymptotic equivalence of Bayes cross validation and widely applicable information criterion in singular learning theory. J. Mach. Learn. Res. 2010, 11, 3571–3594. [Google Scholar]
- Watanabe, S. A widely applicable Bayesian information criterion. J. Mach. Learn. Res. 2013, 14, 867–897. [Google Scholar]
- Diana, A.; Matechou, E.; Griffin, J.E.; Buxton, A.S.; Griffiths, R.A. An RShiny app for modelling environmental DNA data: Accounting for false positive and false negative observation error. Ecography 2021, 44, 1838–1844. [Google Scholar] [CrossRef]
- Griffin, J.E.; Matechou, E.; Buxton, A.S.; Bormpoudakis, D.; Griffiths, R.A. Modelling environmental DNA data; Bayesian variable selection accounting for false positive and false negative errors. J. R. Stat. Soc. Ser. C Appl. Stat. 2020, 69, 377–392. [Google Scholar] [CrossRef]
- Kalogianni, E.; Koutsikos, N.; Karaouzas, I.; Smeti, E.; Kapakos, Y.; Laschou, S.; Dimitriou, E.; Vardakas, L. REVIVE: A feasibility assessment tool for freshwater fish conservation translocations in Mediterranean rivers. Sci. Total Environ. 2023, 862, 160595. [Google Scholar] [CrossRef] [PubMed]
- Jerde, C.L.; Chadderton, W.L.; Mahon, A.R.; Renshaw, M.A.; Corush, J.; Budny, M.L.; Mysorekar, S.; Lodge, D.M. Detection of Asian carp DNA as part of a Great Lakes basin-wide surveillance program. Can. J. Fish. Aquat. Sci. 2013, 70, 522–526. [Google Scholar] [CrossRef]
- Evans, N.T.; Shirey, P.D.; Wieringa, J.G.; Mahon, A.R.; Lamberti, G.A. Comparative cost and effort of fish distribution detection via environmental DNA analysis and electrofishing. Fisheries 2017, 42, 90–99. [Google Scholar] [CrossRef]
- Rees, H.C.; Maddison, B.C.; Middleditch, D.J.; Patmore, J.R.; Gough, K.C. The detection of aquatic animal species using environmental DNA–a review of eDNA as a survey tool in ecology. J. Applied Ecol. 2014, 51, 1450–1459. [Google Scholar] [CrossRef]
- Taberlet, P.; Bonin, A.; Zinger, L.; Coissac, E. Environmental DNA for Biodiversity Research and Monitoring, 1st ed.; Oxford University Press: Oxford, UK, 2018; 252p. [Google Scholar]
- Troth, C.R.; Sweet, M.J.; Nightingale, J.; Burian, A. Seasonality, DNA degradation and spatial heterogeneity as drivers of eDNA detection dynamics. Sci. Total Environ. 2021, 768, 144466. [Google Scholar] [CrossRef]
- Burian, A.; Mauvisseau, Q.; Bulling, M.; Domisch, S.; Qian, S.; Sweet, M. Improving the reliability of eDNA data interpretation. Mol. Ecol. Resour. 2021, 21, 1422–1433. [Google Scholar] [CrossRef]
- Brönmark, C.; Herrmann, J.; Malmqvist, B.; Otto, C.; Sjöström, P. Animal community structure as a function of stream size. Hydrobiologia 1984, 112, 73–79. [Google Scholar] [CrossRef]
- Oberdoff, T.; Guégan, J.F.; Hugueny, B. Global scale patterns of fish species richness in rivers. Ecography 1995, 18, 345–352. [Google Scholar] [CrossRef]
- Guégan, J.F.; Lek, S.; Oberdorff, T. Energy availability and habitat heterogeneity predict global riverine fish diversity. Nature 1998, 391, 382–384. [Google Scholar] [CrossRef]
- Shogren, A.J.; Tank, J.L.; Andruszkiewicz, E.; Olds, B.; Mahon, A.R.; Jerde, C.L.; Bolster, D. Controls on eDNA movement in streams: Transport, retention, and resuspension. Sci. Rep. 2017, 7, 5065. [Google Scholar] [CrossRef] [PubMed]
- Zenetos, A.; Pancucci-Papadopoulou, M.; Zogaris, S.; Papastergiadou, E.; Vardakas, L.; Aligizaki, K.; Economou, A.N. Aquatic alien species in Greece (2009): Tracking sources, patterns and effects on the ecosystem. J. Biol. Res. Thessalon. 2009, 12, 135–172. [Google Scholar]
- Livadas, G.A.; Sphangos, J.C. Malaria in Greece (1930–1940), Research Control, 1st ed.; Pyrsos Press: Athens, Greece, 1941; 238p. [Google Scholar]
- Bartlett, B.R. Introduced Parasites and Predators of Arthropod Pests and Weeds: A World Review. No. 480 Agricultural Research Service; US Department of Agriculture: Washington, DC, USA, 1978; 545p. [Google Scholar]
- Kalogianni, E.; Giakoumi, S.; Andriopoulou, A.; Chatzinikolaou, Y. Prey utilisation and trophic overlap between the non native mosquitofish and a native fish in two Mediterranean rivers. Mediterr. Mar. Sci. 2014, 15, 287–301. [Google Scholar] [CrossRef]
- Kalogianni, E.; Koutsikos, N.; Vardakas, L.; Giakoumi, S.; Chatzinikolaou, Y.; Economou, A.N. Impacts of the alien mosquitofish on the abundance and condition of two Mediterranean native fish. Mediterr. Mar. Sci. 2019, 20, 727–735. [Google Scholar] [CrossRef]
- Rosecchi, E.; Crivelli, A.J.; Catsadorakis, G. The establishment and impact of Pseudorasbora parva, an exotic fish species introduced into Lake Mikri Prespa (north-western Greece). Aquat. Conserv: Mar. Freshw. Ecosyst. 1993, 3, 223–231. [Google Scholar] [CrossRef]
- Holčík, J. Fish introductions in Europe with particular reference to its central and eastern part. Can. J. Fish. Aquat. Sci. 1991, 48, 13–23. [Google Scholar] [CrossRef]
- Koutsikos, N.; Vardakas, L.; Vavalidis, T.; Kalogianni, E.; Dimitriou, E.; Kalantzi, O.I.; Zogaris, S. Defining non-indigenous fish assemblage types in Mediterranean rivers: Network analysis and management implications. J. Environ. Manag. 2021, 278, 111551. [Google Scholar] [CrossRef]
- Simon, A.; Gozlan, R.E.; Robert Britton, J.; Van Oosterhout, C.; Hänfling, B. Human induced stepping-stone colonisation of an admixed founder population: The spread of topmouth gudgeon (Pseudorasbora parva) in Europe. Aquat. Sci. 2015, 77, 17–25. [Google Scholar] [CrossRef]
- Gozlan, R.E.; Andreou, D.; Asaeda, T.; Beyer, K.; Bouhadad, R.; Burnard, D.; Caiola, N.; Cakic, P.; Djikanovic, V.; Esmaeili, H.R.; et al. Pan-continental invasion of Pseudorasbora parva: Towards a better understanding of freshwater fish invasions. Fish. Fish. 2010, 11, 315–340. [Google Scholar] [CrossRef]
- Pinder, A.C.; Davies, G.D.; Brazier, M.; Britton, J.R. A case study of the population ecology of a topmouth gudgeon (Pseudorasbora parva) population in the UK and the implication for native fish communities. Aquat. Conserv: Mar. Freshw. Ecosyst. 2006, 17, 749–759. [Google Scholar] [CrossRef]
- Spikmans, F.; Lemmers, P.; op den Camp, H.J.; van Haren, E.; Kappen, F.; Blaakmeer, A.; van Der Velde, G.; van Langevelde, F.; Leuven, R.S.; van Alen, T.A. Impact of the invasive alien topmouth gudgeon (Pseudorasbora parva) and its associated parasite Sphaerothecum destruens on native fish species. Biol. Invasions 2020, 22, 587–601. [Google Scholar] [CrossRef]
- Hulme, P.E. Beyond control: Wider implications for the management of biological invasions. J. Appl. Ecol. 2006, 43, 835–847. [Google Scholar] [CrossRef]
- Koutsikos, N.; Vardakas, L.; Kalantzi, O.I.; Zogaris, S. Patterns of spatial overlap between non-indigenous and critically endangered freshwater fishes from a Mediterranean biodiversity hotspot. Diversity 2021, 13, 233. [Google Scholar] [CrossRef]
- Aschonitis, V.G.; Gavioli, A.; Lanzoni, M.; Fano, E.A.; Feld, C.; Castaldelli, G. Proposing priorities of intervention for the recovery of native fish populations using hierarchical ranking of environmental and exotic species impact. J. Environ. Manag. 2018, 210, 36–50. [Google Scholar] [CrossRef]
- Brys, R.; Haegeman, A.; Halfmaerten, D.; Neyrinck, S.; Staelens, A.; Auwerx, J.; Ruttink, T. Monitoring of spatiotemporal occupancy patterns of fish and amphibian species in a lentic aquatic system using environmental DNA. Mol. Ecol. 2021, 30, 3097–3110. [Google Scholar] [CrossRef]
- Trebitz, A.S.; Hoffman, J.C.; Darling, J.A.; Pilgrim, E.M.; Kelly, J.R.; Brown, E.A.; Chadderton, W.L.; Egan, S.P.; Grey, E.K.; Hashsham, S.A.; et al. Early detection monitoring for aquatic non-indigenous species: Optimizing surveillance, incorporating advanced technologies, and identifying research needs. J. Environ. Manag. 2017, 202, 299–310. [Google Scholar] [CrossRef]
- Davies, S.J.; Jordaan, M.S.; Karsten, M.; Terblanche, J.S.; Turner, A.A.; van Wilgen, N.J.; Veldtman, R.; Zengeya, T.A.; Measey, J. Experience and Lessons from Alien and Invasive Animal Control Projects in South Africa. In Biological Invasions in South Africa, 1st ed.; van Wilgen, N.J., Measey, J., Richardson, D.M., Wilson, J., Zengeya, T.A., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 629–663. [Google Scholar] [CrossRef]
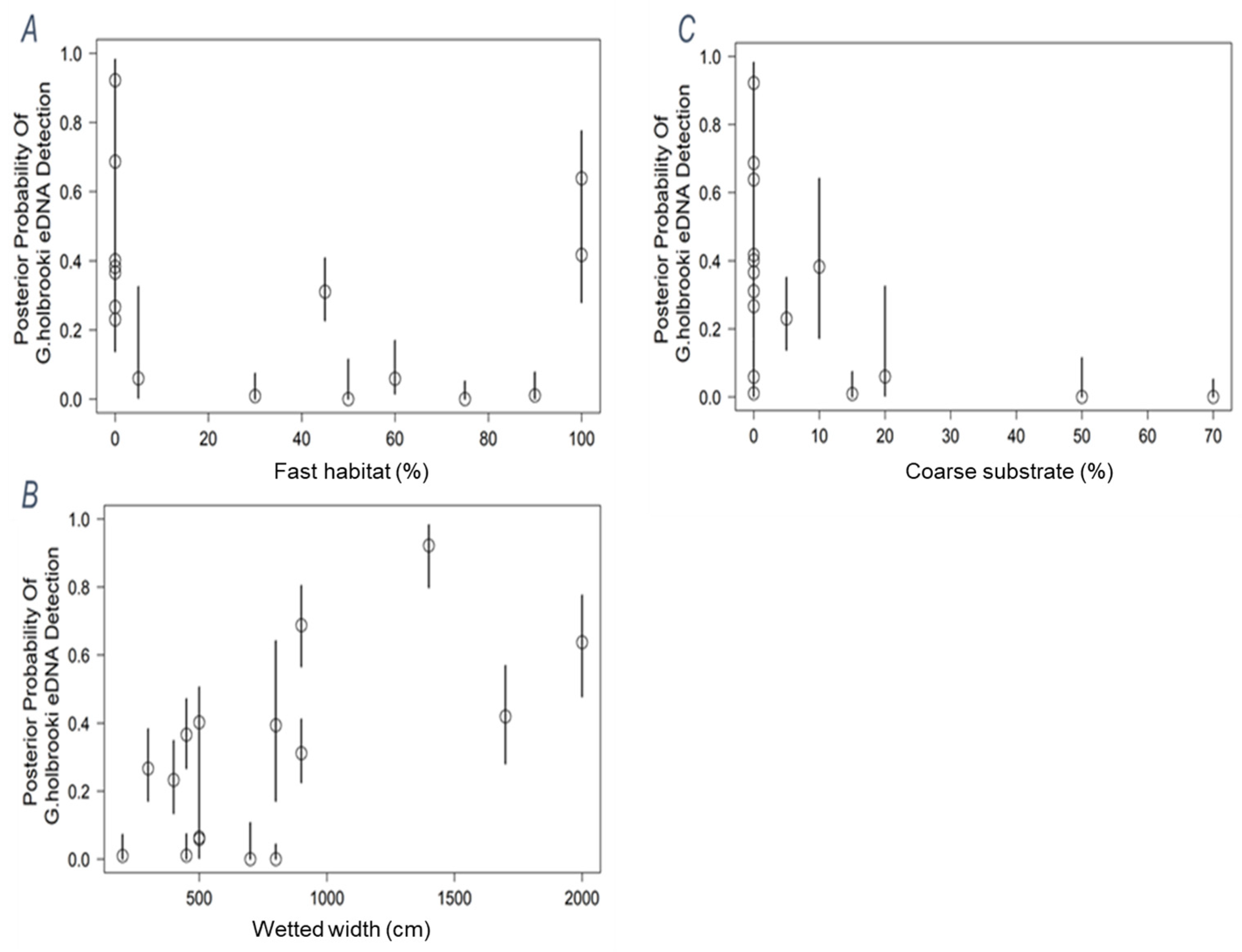
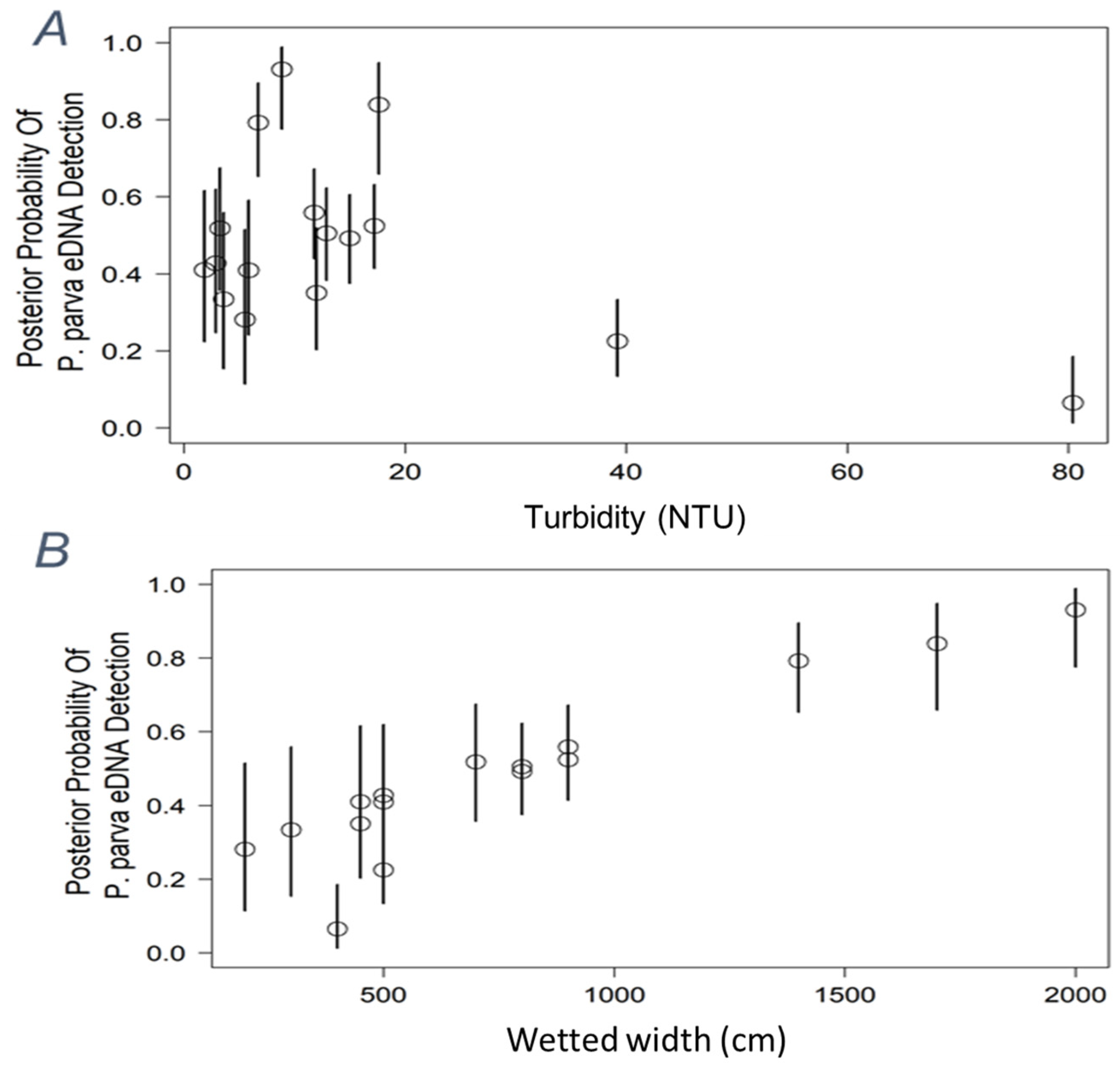
| Site | River Basin | G. holbrooki eDNA Score | G. holbrooki Density | P. parva eDNA Score | P. parva Density | Volume Filtered (X3) | Mean Flow (m/s) | Temperature (°C) | pH |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Evros | 18/18 | 0.94 | 11/12 | 0.31 | 1000 | 0.00 | 19.44 | 7.66 |
| 2 | Bospos | 1/18 | 0.30 | 0/12 | 0.00 | 180 | 0.35 | 20.97 | 7.57 |
| 3 | Strymonas | 17/18 | 0.00 | 11/12 | 0.02 | 300 | 1.33 | 18.25 | 7.9 |
| 4 | Gallikos | 12/18 | 0.27 | 0/12 | 0.76 | 540 | 0.11 | 19 | 8.23 |
| 5 | Axios | 4/18 | 0.42 | 0/12 | 0.09 | 720 | 0.05 | 17.9 | 7.9 |
| 6 | Aliakmonas | 7/18 | 0.01 | 0/12 | 0.00 | 420 | 0.26 | 11 | 8.13 |
| 7 | Agios Germanos (Prespa L.) | 0/18 | 0.00 | 8/12 | 1.53 | 1000 | 0.24 | 11.06 | 6.59 |
| 8 | Kalamas | 0/18 | 0.17 | 0/12 | 0.00 | 780 | 0.14 | 16.04 | 7.14 |
| 9 | Pinios Thessaly | 13/18 | 0.60 | 0/12 | 0.00 | 180 | 0.01 | 16.52 | 7.88 |
| 10 | Spercheios | 2/18 | 0.00 | 0/12 | 0.00 | 300 | 0.35 | 17.13 | 7.98 |
| 11 | Evinos | 0/18 | 0.00 | 0/12 | 0.00 | 1000 | 0.03 | 23.24 | 7.66 |
| 12 | Mornos | 0/18 | 0.00 | 0/12 | 0.00 | 600 | 0.10 | 17.04 | 7.61 |
| 13 | Peirou-Verga-Pineiou | 3/18 | 0.13 | 2/12 | 0.01 | 480 | 0.15 | 19.36 | 7.69 |
| 14 | Peirou-Verga-Pineiou | 0/18 | 0.00 | 1/12 | 0.11 | 480 | 0.19 | 23.27 | 7.91 |
| 15 | Neda | 0/18 | 0.00 | 7/12 | 0.00 | 1000 | 0.47 | 19.46 | 7.42 |
| 16 | Peristera | 0/18 | 0.00 | 0/12 | 0.00 | 1000 | 0.13 | 19.77 | 7.69 |
| Mean | 2.50% | 97.50% | |
|---|---|---|---|
| beta. (intercept) | 1.154 (0.0108) | 0.133 (0.0120) | 2.484 (0.0163) |
| alpha. (intercept) | 1.137 (0.0158) | 0.131 (0.0104) | 2.481 (0.0919) |
| delta. (intercept) | −1.115 (0.0071) | −1.782 (0.0154) | −0.516 (0.0172) |
| delta.fasthabpr | −0.870 (0.0036) | −1.261 (0.0092) | −0.508 (0.0087) |
| delta.wetwidcm | 0.955 (0.0035) | 0.604 (0.0091) | 1.333 (0.0089) |
| delta.coarsubpr | −1.215 (0.0140) | −2.505 (0.0292) | −0.068 (0.0414) |
| Mean | 2.5% | 97.5% | |
|---|---|---|---|
| beta. (Intercept) | 0.363 (0.0172) | −0.686 (0.0096) | 1.973 (0.0281) |
| alpha. (Intercept) | 0.576 (0.0990) | −0.644 (0.0084) | 6.553 (0.0000) |
| delta. (Intercept) | −0.049 (0.0011) | −0.345 (0.0027) | 0.243 (0.0023) |
| delta.Turb | −0.310 (0.0010) | −0.556 (0.0030) | −0.072 (0.0021) |
| delta.WetWidcm | 0.603 (0.0014) | 0.273 (0.0028) | 0.951 (0.0040) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kalogianni, E.; Kalaitzakis, N.; Vardakas, L.; Koutsikos, N.; Zimmerman, B.; Meek, S.; Weldon, L.; Sargeant, S.; Steer, M.D. Nationwide Tracing of Two Top Freshwater Fish Invaders in Greece Using Environmental DNA Sampling. Diversity 2024, 16, 28. https://doi.org/10.3390/d16010028
Kalogianni E, Kalaitzakis N, Vardakas L, Koutsikos N, Zimmerman B, Meek S, Weldon L, Sargeant S, Steer MD. Nationwide Tracing of Two Top Freshwater Fish Invaders in Greece Using Environmental DNA Sampling. Diversity. 2024; 16(1):28. https://doi.org/10.3390/d16010028
Chicago/Turabian StyleKalogianni, Eleni, Nektarios Kalaitzakis, Leonidas Vardakas, Nicholas Koutsikos, Brian Zimmerman, Sarah Meek, Laura Weldon, Stephanie Sargeant, and Mark D. Steer. 2024. "Nationwide Tracing of Two Top Freshwater Fish Invaders in Greece Using Environmental DNA Sampling" Diversity 16, no. 1: 28. https://doi.org/10.3390/d16010028
APA StyleKalogianni, E., Kalaitzakis, N., Vardakas, L., Koutsikos, N., Zimmerman, B., Meek, S., Weldon, L., Sargeant, S., & Steer, M. D. (2024). Nationwide Tracing of Two Top Freshwater Fish Invaders in Greece Using Environmental DNA Sampling. Diversity, 16(1), 28. https://doi.org/10.3390/d16010028







