Polychaetes (Annelida) of Cyprus (Eastern Mediterranean Sea): An Updated and Annotated Checklist including New Distribution Records
Abstract
1. Introduction
2. Materials and Methods
2.1. Development of the Polychaeta Dataset
2.2. Data Analysis
3. Results
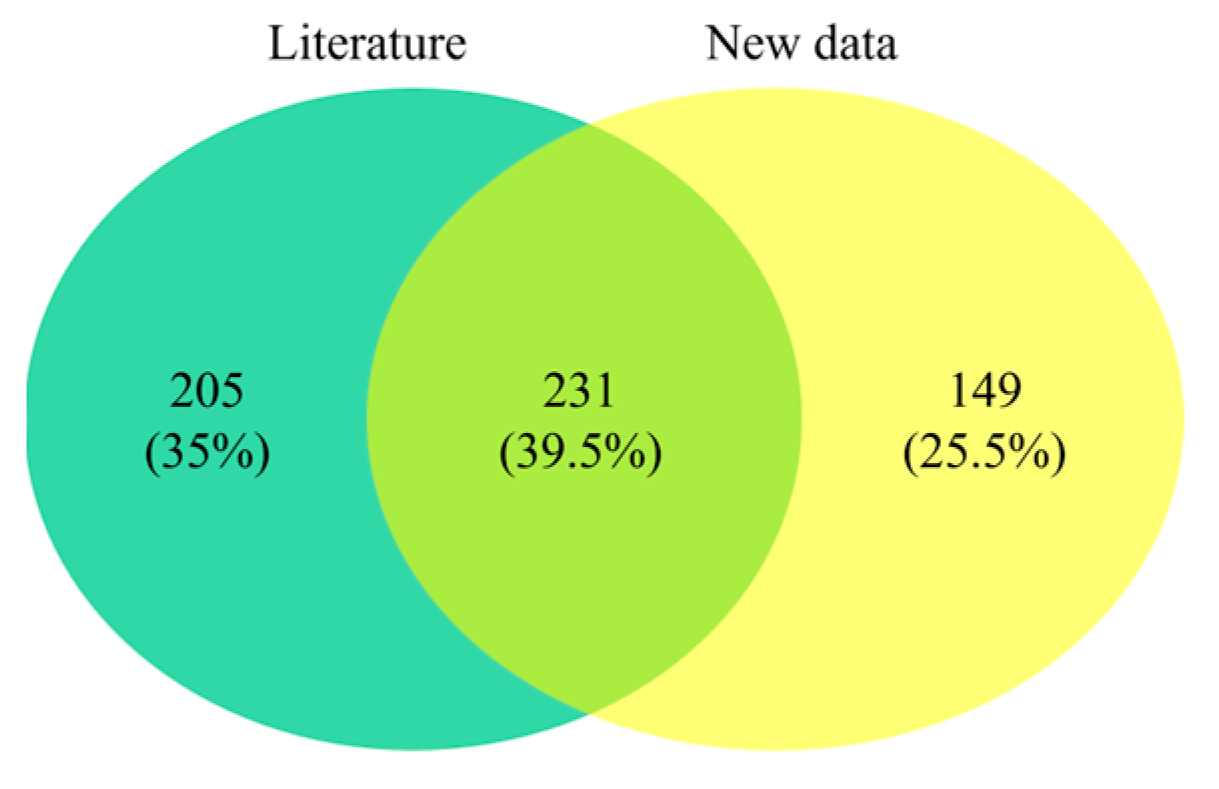
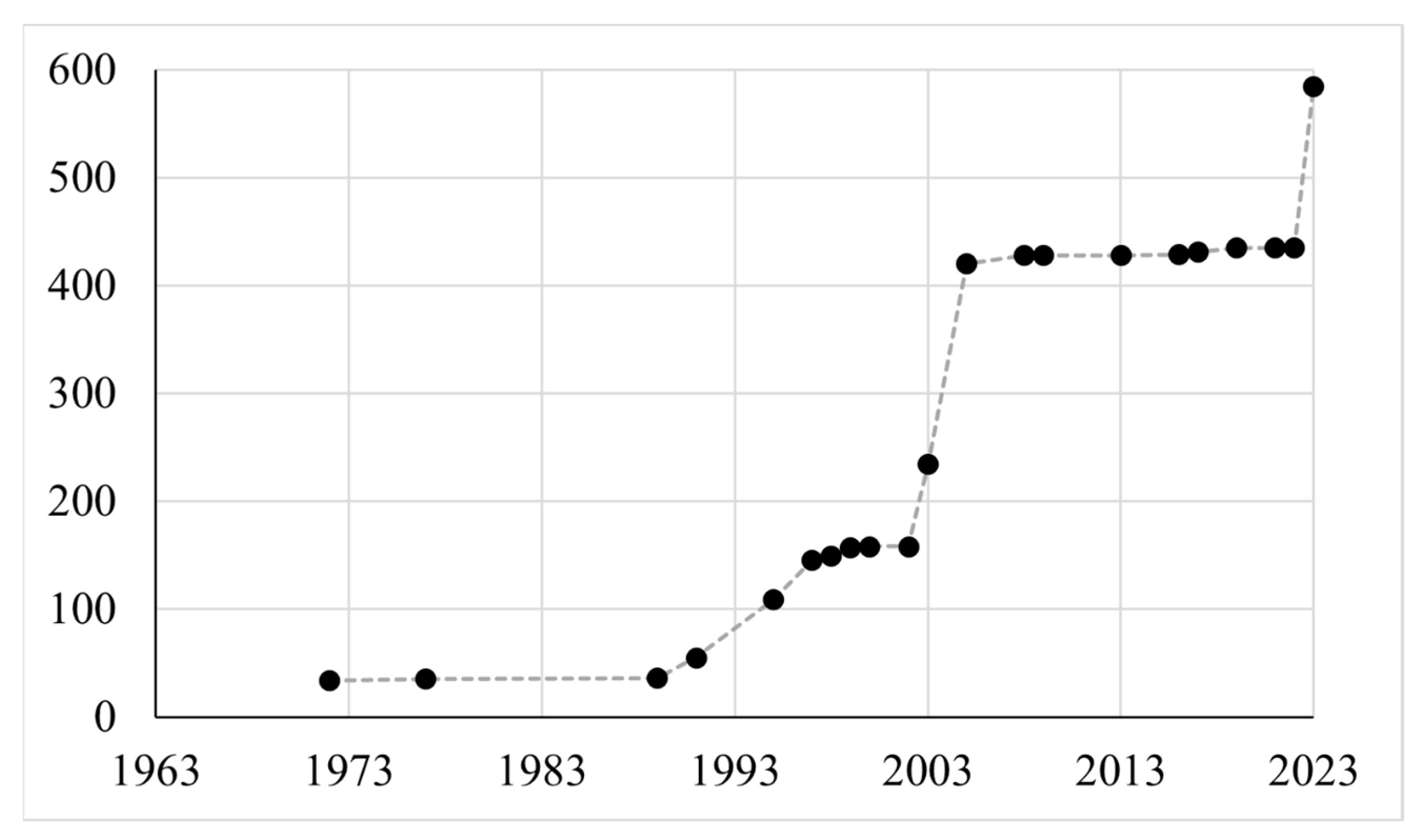
3.1. Bibliographic Sources and New Surveys
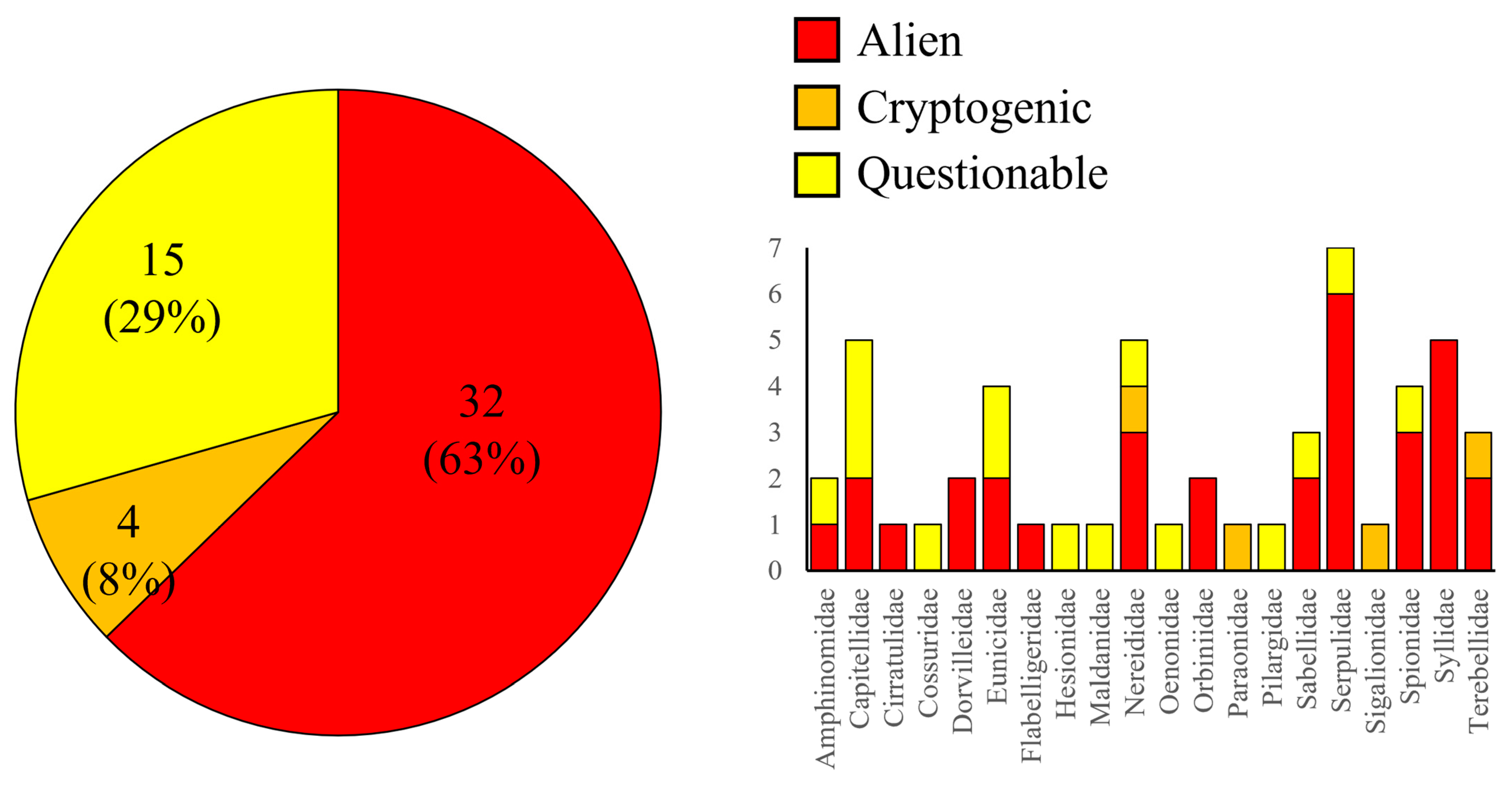
3.2. Polychaete Diversity in Cyprus, including New Records and Non-Indigenous Species
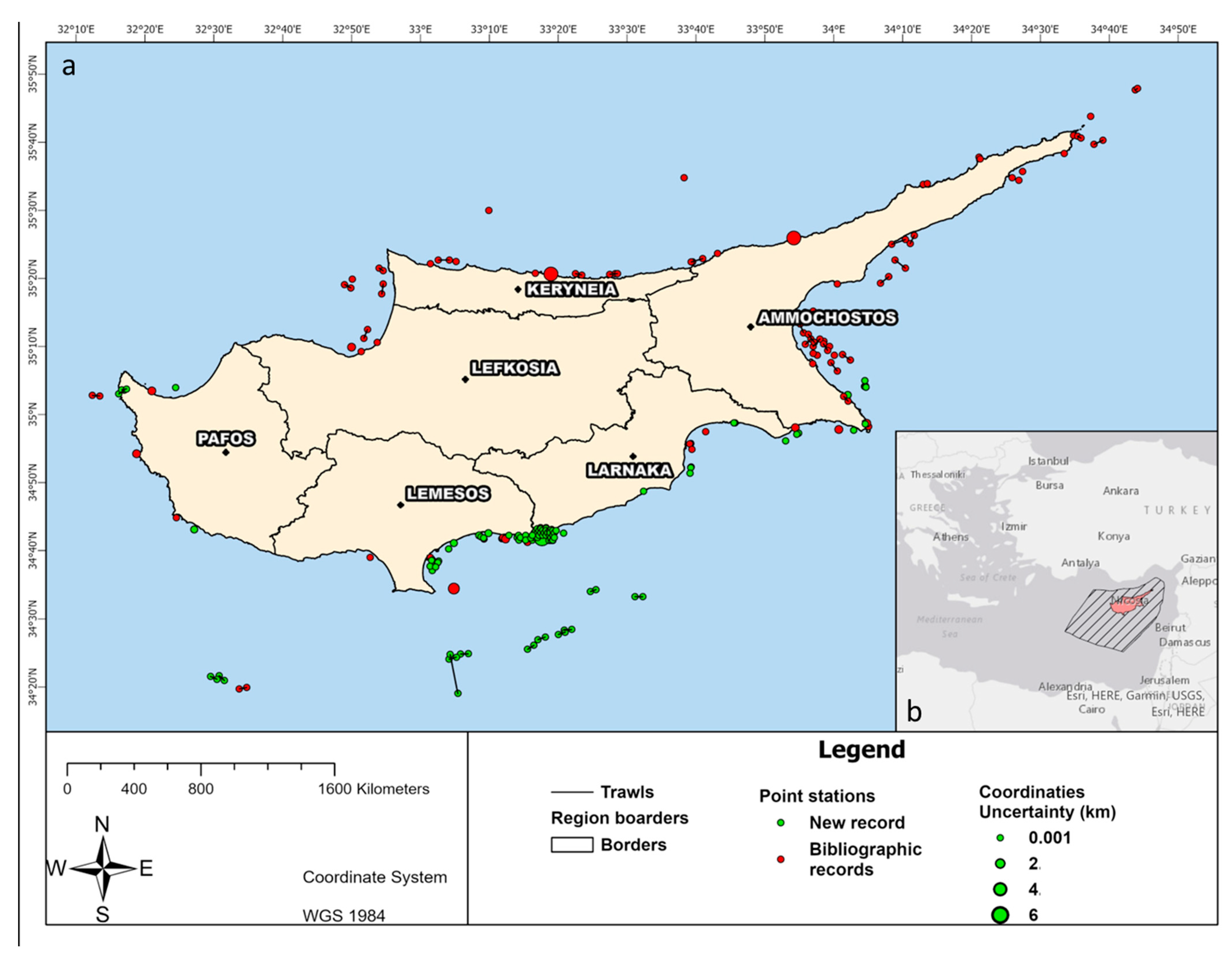
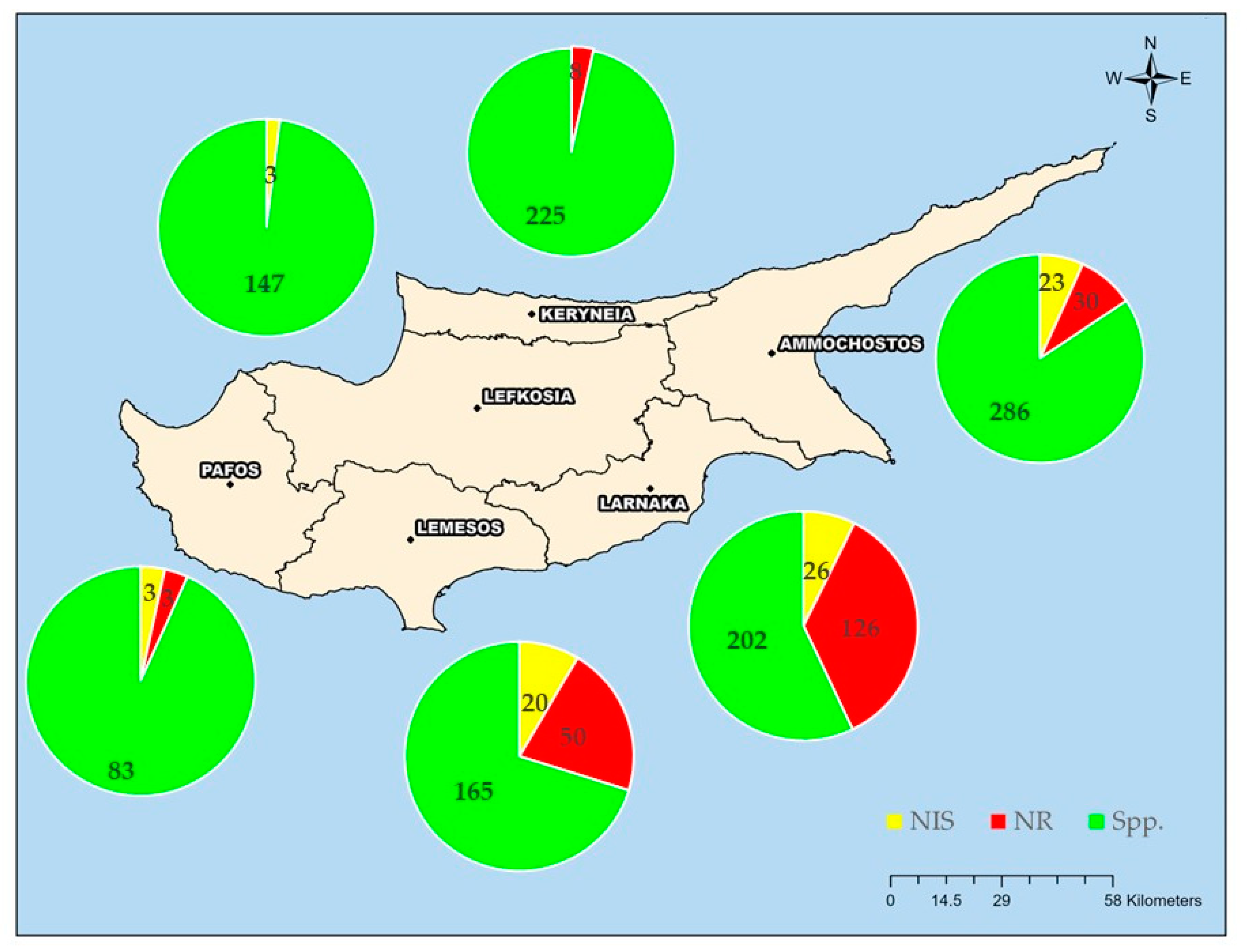
3.3. Spatial Distribution of Polychaeta Species
3.4. Habitat Characteristics and Associations with Flora and Human Activities
4. Discussion
4.1. On the Path to Modern Checklists: What Is Complete and What Is Still Missing
4.2. Non-Indigenous Polychaetes in Cyprus Waters
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AP Marine | AP Marine Environmental Consultancy Ltd. |
| AR | Artificial Reef |
| AUTH | Aristotle University of Thessaloniki |
| DFMR | Department of Fisheries and Marine Research (DFMR) |
| MER | Marine and Environmental (MER) Lab Ltd. |
| MPA | Marine Protected Area |
| IEF | Ichthys EcoFarm Ltd. |
| BI | Blue Island Plc |
| SW | Seawave Fisheries Ltd. |
| TV | Telia Vasiliko Ltd. |
| TL | Telia Liopetri Ltd. |
| KM1 | Kimagro Fishfarming Ltd. (Fishfarm 1) |
| KM2 | Kimagro Fishfarming Ltd. (Fishfarm 2) |
References
- Hutchings, P. Biodiversity and functioning of polychaetes in benthic sediments. Biodiv. Conserv. 1998, 7, 1133–1145. [Google Scholar] [CrossRef]
- Salen-Picard, C.; Arlhac, D. Long-term changes in a Mediterranean benthic community: Relationships between the polychaete assemblages and hydrological variations of the Rhône River. Estuaries 2002, 25, 1121–1130. [Google Scholar] [CrossRef]
- Langeneck, J.; Busoni, G.; Aliani, S.; Lardicci, C.; Castelli, A. Distribution and diversity of polychaetes along a bathyal escarpment in the western Mediterranean Sea. Deep Sea Res. Part I 2019, 144, 85–94. [Google Scholar] [CrossRef]
- Checon, H.H.; Amaral, A.C.Z. Taxonomic sufficiency and the influence of rare species on variation partitioning analysis of a polychaete community. Mar. Ecol. 2017, 38, e12384. [Google Scholar] [CrossRef]
- Golubkov, S.; Tiunov, A.; Golubkov, M. Food-web modification in the eastern Gulf of Finland after invasion of Marenzelleria arctia (Spionidae, Polychaeta). NeoBiota 2021, 66, 75–94. [Google Scholar] [CrossRef]
- Ben-Eliahu, M.N.; Golani, D. Polychaetes (Annelida) in the gut contents of goatfishes (Mullidae), with new polychaete records for the Mediterranean coast of Israel and the Gulf of Elat. Mar. Ecol. 2008, 11, 193–205. [Google Scholar] [CrossRef]
- Beal, B.; Meredith, S.D.; Jourdet, C.B.; Pepperman, K.E. Diet of an underappreciated benthic intertidal fish, Cryptacanthodes maculatus (Cryptacanthodidae), in eastern Maine, USA. AIMS Environ. Sci. 2016, 3, 488–508. [Google Scholar] [CrossRef]
- Eriksen, E.; Benzik, A.N.; Dolgov, A.B.; Skjoldal, H.R.; Vihtakari, M.; Johannesen, E.; Prokhorova, T.A.; Keulder-Stenevik, F.; Prokopchuk, I.; Strand, E. Diet and trophic structure of fishes in the Barents Sea: The Norwegian-Russian program “Year of Stomachs” 2015—Establishing a baseline. Prog. Oceanogr. 2020, 183, 102262. [Google Scholar] [CrossRef]
- Naranjo Ortíz, A.P.; Tobías Jiménez, F.J.; Sardá Borroy, R.; Gil, J. Producción de poliquetos libres de enfermedades para su uso como alimento vivo en la industria camaronera de Ecuador. Aquacultura 2019, 128, 44–48. [Google Scholar]
- Nederlof, M.A.J.; Fang, J.; Dahlgren, T.G.; Rastrick, S.P.S.; Smaal, A.C.; Strand, Ø.; Sveier, H.; Verdegem, M.C.J.; Jansen, H.M. Application of polychaetes in (de)coupled integrated aquaculture: An approach for fish waste bioremediation. Aquacult. Environ. Interact. 2020, 12, 385–399. [Google Scholar] [CrossRef]
- Reish, D. Use of polychaetous annelids as test organisms for marine bioassay experiments. In Aquatic Invertebrate Bioassays; Buikema, A.L., Jr., Cairns, J., Jr., Eds.; American Society for Testing and Materials: Philadelphia, PA, USA, 1980; pp. 140–154. [Google Scholar]
- Durou, C.; Pourier, L.; Amiard, J.C.; Budzinski, H.; Gnassia-Barelli, M.; Lemenach, K.; Peluhet, L.; Mouneyrac, C.; Roméo, M.; Amiard-Triquet, C. Biomonitoring in a clean and multi-contaminated estuary based on biomarkers and chemical analyses in the endobenthic worm Nereis diversicolor. Environ. Pollut. 2007, 148, 445–458. [Google Scholar] [CrossRef] [PubMed]
- Weis, W.A.; Lemes Soares, C.H.; Cunha de Quadros, D.P.; Scheneider, M.; Pagliosa, P.R. Urbanization effects on different biological organization levels of an estuarine polychaete tolerant to pollution. Ecol. Indic. 2017, 73, 698–707. [Google Scholar] [CrossRef]
- Méndez, N.; Flos, J.; Romero, J. Littoral soft-bottom polychaete communities in a pollution gradient in front of Barcelona (Western Mediterranean, Spain). Bull. Mar. Sci. 1998, 63, 167–178. [Google Scholar]
- Belan, T.A. Marine environmental quality assessment using polychaete taxocene characteristics in Vancouver Harbour. Mar. Environ. Res. 2004, 57, 89–101. [Google Scholar] [CrossRef]
- Dean, H.K. The use of polychaetes (Annelida) as indicator species of marine pollution: A review. Rev. Biol. Trop. 2008, 56, 11–38. [Google Scholar]
- Stabili, L.; Schirosi, R.; Licciano, M.; Mola, E.; Giangrande, A. Bioremediation of bacteria in aquaculture waste using the polychaete Sabella spallanzanii. New Biotech. 2010, 27, 774–781. [Google Scholar] [CrossRef]
- Pajand, Z.O.; Soltani, M.; Bahmani, M.; Kamali, A. The role of the polychaete Nereis diversicolor in bioremediation of wastewater and its growth performance and fatty acid composition in an integrated culture system with Huso huso (Linnaeus, 1758). Aquacult. Res. 2017, 48, 5271–5279. [Google Scholar] [CrossRef]
- Mandario, M.A.E.; Alava, V.R.; Añasco, N.C. Evaluation of the bioremediation potential of mud polychaete Marphysa sp. in aquaculture pond sediments. Environ. Sci. Pollut. Res. Int. 2019, 26, 29810–29821. [Google Scholar] [CrossRef]
- Musco, L.; Terlizzi, A.; Licciano, M.; Giangrande, A. Taxonomic structure and the effectiveness of surrogates in environmental monitoring: A lesson from polychaetes. Mar. Ecol. Prog. Ser. 2009, 383, 199–210. [Google Scholar] [CrossRef]
- Hutchings, P.; Lavesque, N. I know who you are, but do others know? Why correct scientific names are so important for the biological sciences. Zoosymposia 2020, 19, 151–163. [Google Scholar] [CrossRef]
- Giangrande, A.; Licciano, M. Factors influencing latitudinal pattern of biodiversity: An example using Sabellidae (Annelida, Polychaeta). Biodiv. Conserv. 2004, 13, 1633–1646. [Google Scholar] [CrossRef]
- Hutchings, P.A. Major issues facing taxonomy—A personal perspective. Megataxa 2020, 1, 46–48. [Google Scholar] [CrossRef]
- Engel, M.S.; Ceriaco, L.M.P.; Daniel, G.M.; Dellapé, P.M.; Löbl, I.; Marinov, M.; Reis, R.E.; Young, M.T.; Dubois, A.; Agarwal, I.; et al. The taxonomic impediment: A shortage of taxonomists, not the lack of technical approaches. Zool. J. Linn. Soc. 2021, 193, 381–387. [Google Scholar] [CrossRef]
- Fauchald, K. Polychaete distribution patterns, or: Can animals with Palaeozoic cousins show large-scale geographical patterns? In Proceedings of the First International Polychaete Conference; Hutchings, P., Ed.; The Linnean Society of New South Wales: Sydney, Australia, 1984; pp. 1–6. [Google Scholar]
- Hutchings, P.; Kupriyanova, E. Cosmopolitan polychaetes—Fact or fiction? Personal and historical perspectives. Invertebr. Syst. 2018, 32, 1–9. [Google Scholar] [CrossRef]
- Fauvel, P. Faune de France 5: Polychètes Errantes; Paul Lechevalier: Paris, France, 1923; 488p. [Google Scholar]
- Fauvel, P. Faune de France 16: Polychètes Sédentaires; Paul Lechevalier: Paris, France, 1927; 494p. [Google Scholar]
- Barroso, R.; Klautau, M.; Solé-Cava, A.M.; Paiva, P.C. Eurythoe complanata (Polychaeta: Amphinomidae), the ‘cosmopolitan’ fireworm, consists of at least three cryptic species. Mar. Biol. 2010, 157, 69–80. [Google Scholar] [CrossRef]
- Nygren, A. Cryptic polychaete diversity: A review. Zool. Scr. 2014, 43, 172–183. [Google Scholar] [CrossRef]
- Nygren, A.; Parapar, J.; Pons, J.; Meißner, K.; Bakken, T.; Kongsrud, J.A.; Oug, E.; Gaeva, D.; Sikorski, A.; Johansen, R.A.; et al. A megacryptic species complex hidden among one of the most common annelids in the NorthEast Atlantic. PLoS ONE 2018, 13, e0198356. [Google Scholar] [CrossRef]
- Arias, A.; Paxton, H. Hidden diversity within the polychaete Onuphis eremita sensu lato (Annelida: Onuphidae)—Redescription of O. eremita Audouin & Milne-Edwards, 1833 and reinstatement of Onuphis pancerii Claparède, 1868. Zootaxa 2014, 3861, 145–169. [Google Scholar]
- Barroso, M.; Moreira, J.; Parapar, J. Long forgotten: Eunice woodwardi Baird, 1869 (Annelida, Eunicidae) revisited, with an insight on internal anatomy. PeerJ. 2022, 10, e13126. [Google Scholar] [CrossRef]
- Teixeira, M.A.L.; Langeneck, J.; Vieira, P.E.; Hernandes, J.C.; Sampieri, B.R.; Kasapidis, P.; Mucciolo, S.; Bakken, T.; Ravara, A.; Nygren, A.; et al. Reappraisal of the hyperdiverse Platynereis dumerilii (Annelida, Nereididae) species complex in the Northern Atlantic, with the description of two new species. Invertebr. Syst. 2022, 36, 1017–1061. [Google Scholar] [CrossRef]
- Castelli, A.; Bianchi, C.N.; Cantone, G.; Çinar, M.E.; Gambi, M.C.; Giangrande, A.; Iraci Sareri, D.; Lanera, P.; Licciano, M.; Musco, L.; et al. Annelida Polychaeta. Biol. Mar. Medit. 2008, 15, 323–373. [Google Scholar]
- Çinar, M.E.; Dağli, E.; Kurt Şahin, G. Checklist of Annelida from the coast of Turkey. Turkish J. Zool. 2014, 38, 734–764. [Google Scholar] [CrossRef]
- Faulwetter, S.; Simboura, N.; Katsiaras, N.; Chatzigeorgiou, G.; Arvanitidis, C. Polychaetes of Greece: An updated and annotated checklist. Biodiv. Data J. 2017, 5, e20997. [Google Scholar] [CrossRef]
- Bakalem, A.; Gillet, P.; Pezy, J.-P.; Dauvin, J.-C. Inventory and the biogeographical affinities of Annelida Polychaeta in the Algerian coastline (Western Mediterranean). Mediterr. Mar. Sci. 2020, 21, 157–182. [Google Scholar] [CrossRef]
- Zaabi, S.; Gillet, P.; Chambers, S.; Afli, A.; Boumaiza, M. Inventory and new records of polychaete species from the Cap Bon Peninsula, north-east coast of Tunisia, Western Mediterranean Sea. Mediterr. Mar. Sci. 2012, 13, 36–48. [Google Scholar] [CrossRef][Green Version]
- Ayari-Kliti, R.; Bakalem, A.; Fersi, A.; Afli, A.; Dauvin, J.-C. Polychaete diversity in Tunisian waters as of 2021: An update with special emphasis on Non-Indigenous species. Mediterr. Mar. Sci. 2022, 23, 698–724. [Google Scholar] [CrossRef]
- Ben-Eliahu, M.N. A list of Polychaeta along the Levant coast. Haasiana 1995, 1, 78–93. [Google Scholar]
- Mikac, B. A sea of worms: Polychaete checklist of the Adriatic Sea. Zootaxa 2015, 3943, 172p. [Google Scholar] [CrossRef][Green Version]
- Ben-Eliahu, M.N. Littoral Polychaeta from Cyprus. Tethys 1972, 4, 85–94. [Google Scholar]
- Ben-Eliahu, M.N. Red Sea serpulids (Polychaeta) in the eastern Mediterranean. Ophelia 1991, S5, 515–528. [Google Scholar]
- Ben-Eliahu, M.N.; Fiege, D. Polychaeta from the continental shelf and slope of Israel collected by the “Meteor” 5 Expedition (1987). Senckenberg. Maritima 1995, 25, 85–105. [Google Scholar]
- Hadjichristophorou, M.; Argyrou, A.; Demetropoulous, A.; Bianchi, T.S. A species list of the sublittoral soft-bottom macrobenthos of Cyprus. Acta Adriat. 1997, 38, 3–32. [Google Scholar]
- Ben-Eliahu, M.N.; Payiatas, G. Searching for Lessepsian migrant serpulids (Annelida: Polychaeta) on Cyprus—Some results of a recent expedition. Israel J. Zool. 1999, 45, 101–119. [Google Scholar]
- Çinar, M.E.; Ergen, Z. Eusyllinae and Syllinae (Annelida: Polychaeta) form northern Cyprus (eastern Mediterranean Sea) with a checklist of species reported from the Levant Sea. Bull. Mar. Sci. 2003, 72, 769–793. [Google Scholar]
- Çinar, M.E.; Ergen, Z.; Benli, H.A. Autolytinae and Exogoninae (Polychaeta: Syllidae) from northern Cyprus (eastern Mediterranean Sea) with a checklist of species reported from the Levant Sea. Bull. Mar. Sci. 2003, 72, 741–767. [Google Scholar]
- Çinar, M.E. Polychaetes from the coast of northern Cyprus (eastern Mediterranean Sea), with two new records for the Mediterranean Sea. Cahiers Biol. Mar. 2005, 46, 143–159. [Google Scholar]
- Katsanevakis, S.; Tsiamis, K.; Ioannou, G.; Michailidis, N.; Zenetos, A. Inventory of alien marine species of Cyprus (2009). Mediterr. Mar. Sci. 2009, 10, 109–134. [Google Scholar] [CrossRef]
- Russo, A.R. Epifauna living on sublittoral seaweeds around Cyprus. Hydrobiologia 1997, 344, 169–179. [Google Scholar] [CrossRef]
- Argyrou, M.; Demetropoulos, A.; Hadjichristophorou, M. Expansion of the macroalga Caulerpa racemosa and changes in soft-bottom macrofaunal assemblages in Moni Bay, Cyprus. Oceanolog. Acta 1999, 22, 517–528. [Google Scholar] [CrossRef]
- Pavloudi, C.; Christodoulou, M.; Mavidis, M. Macrofaunal assemblages associated with the sponge Sarcotragus foetidus Schmidt, 1862 (Porifera, Demospongiae) at the coasts of Cyprus and Greece. Biodiv. Data J. 2016, 30, e8210. [Google Scholar] [CrossRef]
- Böggemann, M. Revision of the Glyceridae Grube, 1850 (Annelida: Polychaeta). Abhand. Senckenb. Naturfosch. Ges. 2002, 555, 249p. [Google Scholar]
- Barnich, R.; Fiege, D. The Aphroditoidea (Annelida: Polychaeta) of the Mediterranean Sea. Abhand. Senckenb. Naturfosch. Ges. 2003, 559, 1–167. [Google Scholar]
- Böggemann, M. Revision of the Goniadidae. Abhand. Naturwissenschaft. Ver. Hamburg 2005, 39, 1–354. [Google Scholar]
- Guido, A.; Jiménez, C.; Achilleos, K.; Rosso, A.; Sanfilippo, R.; Hadjioannou, L.; Petrou, A.; Russo, F.; Mastandrea, A. Cryptic serpulid-microbialite bioconstructions in the Kakoskali submarine cave (Cyprus, Eastern Mediterranean). Facies 2017, 63, 21. [Google Scholar] [CrossRef]
- Rousou, M. Investigation of Soft Bottom Benthic Macrofauna Communities of Vasiliko Bay (Cyprus, East Mediterranean Sea). Unpublished. Ph.D. Thesis, Aristotle University of Thessaloniki, Thessaloniki, Greece, 2018. (In Greek). [Google Scholar]
- Gerovasileiou, V.; Akel, E.H.K.; Akyol, O.; Alongi, G.; Azevedo, F.; Babali, N.; Bakiu, R.; Bariche, M.; Bennoui, A.; Castriota, L.; et al. New Mediterranean biodiversity records (July; 2017). Mediterr. Mar. Sci. 2017, 18, 355–384. [Google Scholar]
- Myers, A.; Plaiti, W.; Rousou, M. A new species of Microdeutopus, M. periergos sp. nov. (Crustacea, Amphipoda, Senticaudata, Aoridae) from Cyprus (East Mediterranean Sea). Zootaxa 2018, 4378, 144–150. [Google Scholar] [CrossRef]
- Rousou, M.; Plaiti, W.; Lowry, J.; Charalambous, S.; Chintiroglou, C.C. Amphipoda species (Suborders: Amphilochidea and Senticaudata) from Vasiliko Bay, Cyprus: New records, information on their biogeography and an annotated checklist from the coasts of Cyprus. Zootaxa 2020, 4896, 373–408. [Google Scholar] [CrossRef]
- Langeneck, J.; Lezzi, M.; Del Pasqua, M.; Musco, L.; Gambi, M.C.; Castelli, A.; Giangrande, A. Non-indigenous polychaetes along the coasts of Italy: A critical review. Mediterr. Mar. Sci. 2020, 21, 238–275. [Google Scholar] [CrossRef]
- Carlton, J.T. Biological invasions and cryptogenic species. Ecology 1996, 77, 1653–1655. [Google Scholar] [CrossRef]
- Tsiamis, K.; Zenetos, A.; Deriu, I.; Gervasini, E.; Cardoso, A.C. The native distribution range of the European marine non-indigenous species. Aquat. Invasions 2018, 13, 187–198. [Google Scholar] [CrossRef]
- Mayo, S.J.; Allkin, R.; Baker, W.; Blagoderov, V.; Brake, I.; Clark, B.; Govaerts, R.; Godfray, C.; Haigh, A.; Hand, R.; et al. Alpha E-taxonomy: Responses from the systematics community to the biodiversity crisis. Kew Bull. 2008, 63, 1–16. [Google Scholar] [CrossRef]
- Melville, J.; Chapple, D.G.; Keogh, J.S.; Sumner, J.; Amey, A.; Bowles, P.; Brennan, I.G.; Couper, P.; Donnellan, S.C.; Doughty, P.; et al. A return-on-investment approach for prioritization of rigorous taxonomy research needed to inform responses to the biodiversity crisis. PLoS Biol. 2021, 19, e3001210. [Google Scholar] [CrossRef] [PubMed]
- Galil, B.S.; Zibrowius, H. First benthos samples from Eratosthenes Seamount, eastern Mediterranean. Mar. Biodivers. 1998, 28, 111–121. [Google Scholar] [CrossRef]
- World Register of Marine Species (WoRMS). Available online: https://www.marinespecies.org (accessed on 27 June 2023).
- de Jong, Y.; Verbeek, M.; Michelsen, V.; de Place Bjørn, P.; Los, W.; Steeman, F.; Bailly, N.; Basire, C.; Chylarecki, P.; Stloukal, E.; et al. Fauna Europaea—All European animal species on the web. Biodiv. Data J. 2014, 2, e4034. [Google Scholar] [CrossRef] [PubMed]
- Checklist Fauna d’Italia. Available online: https://www.lifewatchitaly.eu/iniziative/checklist-fauna-italia-it/ (accessed on 27 June 2023).
- Musco, L. Ecology and diversity of Mediterranean hard-bottom Syllidae (Annelida): A community-level approach. Mar. Ecol. Prog. Ser. 2012, 461, 107–119. [Google Scholar] [CrossRef]
- Tempesti, J.; Langeneck, J.; Romani, L.; Garrido, M.; Lardicci, C.; Maltagliati, F.; Castelli, A. Characterization of fouling structure components and their relationship with associated macrofauna in three Northern Tyrrhenian port systems (Mediterranean Sea). Estuar. Coast. Shelf Sci. 2022, 279, 108156. [Google Scholar] [CrossRef]
- Lobo, J.; Teixeira, M.A.L.; Borges, L.M.S.; Ferreira, M.S.G.; Hollatz, C.; Gomes, P.T.; Sousa, R.; Ravara, A.; Costa, M.H.; Costa, F.O. Starting a DNA barcode reference library for shallow water polychaetes from the Southern European Atlantic coast. Mol. Ecol. Res. 2016, 16, 298–313. [Google Scholar] [CrossRef]
- Gold, Z.; Curd, E.E.; Goodwin, K.D.; Choi, E.S.; Frable, B.W.; Thompson, A.R.; Walker, H.J., Jr.; Burton, R.S.; Kacev, D.; Martz, L.D.; et al. Improving metabarcoding taxonomic assignment: A case study of fishes in a large marine ecosystem. Mol. Ecol. Res. 2021, 21, 2546–2564. [Google Scholar] [CrossRef]
- Pappalardo, P.; Collins, A.G.; Pagenkopp Lohan, K.M.; Hanson, K.M.; Truskey, S.B.; Jaeckle, W.; Lewis Ames, C.; Goodheart, J.A.; Bush, S.L.; Biancani, L.M.; et al. The role of taxonomic expertise in interpretation of metabarcoding studies. ICES J. Mar. Sci. 2021, 78, 3397–3410. [Google Scholar] [CrossRef]
- Nikula, R.; Väinölä, R. Phylogeography of Cerastoderma glaucum (Bivalvia: Cardiidae) across Europe: A major break in the Eastern Mediterranean. Mar. Biol. 2003, 143, 339–350. [Google Scholar] [CrossRef]
- Moussa, M.; Choulak, S.; Rhouma-Chatti, S.; Chatti, N.; Said, K. First insight of genetic diversity, phylogeographic relationships, and population structure of marine sponge Chondrosia reniformis from the eastern and western Mediterranean coasts of Tunisia. Ecol. Evol. 2022, 12, e8494. [Google Scholar] [CrossRef] [PubMed]
- Deli, T.; Kalkan, E.; Karhan, S.Ü.; Uzunova, S.; Keikhosravi, A.; Bilgin, R.; Schubart, C.D. Parapatric genetic divergence among deep evolutionary lineages in the Mediterranean green crab, Carcinus aestuarii (Brachyura, Portunoidea, Carcinidae), accounts for a sharp phylogeographic break in the Eastern Mediterranean. BMC Evol. Biol. 2018, 18, 53. [Google Scholar] [CrossRef] [PubMed]
- Langeneck, J.; Englezou, C.; Di Maggio, M.; Castelli, A.; Maltagliati, F. Phylogeography of Aphanius fasciatus (Osteichthyes: Aphaniidae) in the Mediterranean Sea, with a focus on its conservation in Cyprus. Hydrobiologia 2021, 848, 4093–4114. [Google Scholar] [CrossRef]
- Iannotta, M.A.; Gambi, M.C.; Patti, F.P. Molecular evidence of intraspecific variability in Lysidice ninetta (Polychaeta: Eunicidae) in the Mediterranean Sea. Aquat. Biol. 2009, 6, 121–132. [Google Scholar] [CrossRef]
- Barco, A.; Aissaoui, C.; Houart, R.; Bonomolo, G.; Crocetta, F.; Oliverio, M. Revision of the Ocinebrina aciculata species complex (Mollusca: Gastropoda: Muricidae) in the northeastern Atlantic Ocean and Mediterranean Sea. J. Molluscan Stud. 2018, 84, 19–29. [Google Scholar] [CrossRef]
- Larson, E.R.; Graham, B.M.; Achury, R.; Coon, J.J.; Daniels, M.K.; Gambrell, D.K.; Jonasen, K.L.; King, G.D.; LaRacuente, N.; Perrin-Stowe, T.I.N.; et al. From eDNA to citizen science: Emerging tools for the early detection of invasive species. Front. Ecol. Environ. 2020, 18, 194–202. [Google Scholar] [CrossRef]
- Thomas, A.C.; Tank, S.; Nguyen, P.L.; Ponce, J.; Sinnesael, M.; Goldberg, C.S. A system for rapid eDNA detection of aquatic invasive species. Environ. DNA 2020, 2, 261–270. [Google Scholar] [CrossRef]
- Duarte, S.; Vieira, P.E.; Costa, F.O. Assessment of species gaps in DNA barcode libraries of non-indigenous species (NIS) occurring in European coastal regions. Metabarcoding Metagenom. 2020, 4, 35–46. [Google Scholar] [CrossRef]
- Lavrador, A.S.; Fontes, J.T.; Vieira, P.E.; Costa, F.O.; Duarte, S. Compilation, revision, and annotation of DNA barcodes of marine invertebrate non-indigenous species (NIS) occurring in European coastal regions. Diversity 2023, 15, 174. [Google Scholar] [CrossRef]
- Sikorski, A.V.; Radashevsky, V.I.; Castelli, A.; Pavlova, L.V.; Nygren, A.; Malyar, V.V.; Borisova, P.B.; Mikac, B.; Rousou, M.; Martin, D.; et al. Revision of the Laonice bahusiensis complex (Annelida: Spionidae) with a description of three new species. Zootaxa 2021, 4996, 253–283. [Google Scholar] [CrossRef]
- Langeneck, J.; Fourreau, C.J.L.; Rousou, M.; Barbieri, M.; Maltagliati, F.; Musco, L.; Castelli, A. Environmental features drive lineage diversification in the Aricidea assimilis species complex (Annelida, Paraonidae) in the Mediterranean Sea. Eur. Zool. J. 2022, 89, 1246–1258. [Google Scholar] [CrossRef]
- Zenetos, A.; Albano, P.G.; Lopez Garcia, E.; Stern, N.; Tsiamis, K.; Galanidi, M. Established non-indigenous species increased by 40% in 11 years in the Mediterranean Sea. Mediterr. Mar. Sci. 2022, 23, 196–212. [Google Scholar] [CrossRef]
- Tempesti, J.; Mangano, M.C.; Langeneck, J.; Lardicci, C.; Maltagliati, F.; Castelli, A. Non-indigenous species in Mediterranean ports: A knowledge baseline. Mar. Environ. Res. 2020, 161, 105056. [Google Scholar] [CrossRef] [PubMed]
- Ben-Eliahu, M.N.; ten Hove, H.A. Serpulids (Annelida: Polychaeta) along the Mediterranean coast of Israel—New population build-ups of Lessepsian migrants. Israel J. Zool. 1992, 38, 35–53. [Google Scholar]
- Çinar, M.E.; Dağli, E. Polychaetes (Annelida: Polychaeta) from the Aegean and Levantine coasts of Turkey, with descriptions of two new species. J. Nat. Hist. 2013, 47, 911–947. [Google Scholar] [CrossRef]
- Galil, B.S.; Spanier, E.; Ferguson, W.W. The Scyphomedusae of the Mediterranean coast of Israel, including two Lessepsian migrants new to the Mediterranea. Zool. Meded. 1990, 64, 95–105. [Google Scholar]
- Galil, B.S.; Gershwin, L.-A.; Douek, J.; Rinkevich, B. Marivagia stellata gen. et sp. nov. (Scyphozoa: Rhizostomeae: Cepheidae), another alien jellyfish from the Mediterranean coast of Israel. Aquat. Invasions 2010, 5, 331–340. [Google Scholar] [CrossRef]
- Çinar, M.E.; Bakir, K.; Öztürk, B.; Katağan, T.; Doğan, A.; Açik, S.; Kurt-Şahin, G.; Özcan, T.; Dağli, E.; Bitlis-Bakir, B.; et al. Macrobenthic fauna associated with the invasive alien species Brachidontes pharaonis (Mollusca: Bivalvia) in the Levantine Sea (Turkey). J. Mar. Biol. Assoc. UK 2017, 97, 613–628. [Google Scholar] [CrossRef]
- Mutlu, E.; Çağatay, I.T.; Olguner, M.T.; Yilmaz, H.E. A new sea-nettle from the Eastern Mediterranean Sea: Chrysaora pseudoocellata sp. nov. (Scyphozoa: Pelagiidae). Zootaxa 2020, 4790, 229–244. [Google Scholar] [CrossRef]
- Barnich, R.; Van Haaren, T. Revision of Sthenelais Kinberg, 1856, Fimbriosthenelais Pettibone, 1971 and Eusthenelais McIntosh, 1876 (Polychaeta, Sigalionidae) in the Northeast Atlantic. Eur. J. Taxon. 2021, 740, 138–171. [Google Scholar] [CrossRef]
- Chaibi, M.; Azzouna, A.; Martín, D. First record of Lepidonotus tenuisetosus (Annelida: Polynoidae) from Tunisia with distributional notes. Mediterr. Mar. Sci. 2023, 24, 7–18. [Google Scholar] [CrossRef]
- Cepeda, D.; Lattig, P. New reports and description of a new species of Sabellidae (Annelida) for the Iberian Peninsula and Balearic Archipelago. Mar. Biol. Res. 2017, 13, 832–853. [Google Scholar] [CrossRef]
- Enrichetti, F.; Baldrighi, E.; Bavestrello, G.; Betti, F.; Canese, S.; Costa, A.; del Pasqua, M.; Giangrande, A.; Langeneck, J.; Misic, C.; et al. Ecological role and phylogenetic position of a new habitat-forming species (Canalipalpata, Sabellidae) from the Mediterranean mesophotic soft bottoms. Estuar. Coast. Shelf Sci. 2022, 265, 107737. [Google Scholar] [CrossRef]
- Gogina, M.; Zettler, M.L. Diversity and distribution of benthic macrofauna in the Baltic Sea: Data inventory and its use for species distribution modelling and prediction. J. Sea Res. 2010, 64, 313–321. [Google Scholar] [CrossRef]
- Lee, S.; Park, I.; Koo, B.J.; Ryu, J.-H.; Choi, J.-K.; Woo, H.J. Macrobenthos habitat potential mapping using GIS-based artificial neural network models. Mar. Pollut. Bull. 2013, 67, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Moraitis, M.L.; Tsikopoulou, I.; Geropoulos, A.; Dimitriou, P.D.; Papageorgiou, N.; Giannoulaki, M.; Valavanis, V.D.; Karakassis, I. Molluscan indicator species and their potential use in ecological status assessment using species distribution modeling. Mar. Environ. Res. 2018, 140, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Pettibone, M.H. Revision of the aphroditoid polychaetes of the family Acoetidae Kinberg (= Polyodontidae Augener) and reestablishment of Acoetes Audouin and Milne-Edwards, 1832, and Euarche Ehlers, 1887. Smithsonian Contrib. Zool. 1989, 464, 1–138. [Google Scholar] [CrossRef]
- Holmer, M.; Argyrou, M.; Dalsgaard, T.; Danovaro, R.; Diaz-Almela, E.; Duarte, C.M.; Frederiksen, M.; Grau, A.; Karakassis, I.; Marbà, N.; et al. Effects of fish farm waste on Posidonia oceanica meadows: Synthesis and provision of monitoring and management tools. Mar. Pollut. Bull. 2008, 56, 1618–1629. [Google Scholar] [CrossRef]
- Arias, A.; Barroso, R.; Anadón, N.; Paiva, P.C. On the occurrence of the fireworm Eurythoe complanata complex (Annelida, Amphinomidae) in the Mediterranean Sea with an updated revision of the alien Mediterranean amphinomids. Zookeys 2013, 337, 19–33. [Google Scholar] [CrossRef]
- Heilskov, A.C.; Alperin, M.; Holmer, M. Benthic fauna bio-irrigation effects on nutrient regeneration in fish farm sediments. J. Exp. Mar. Biol. Ecol. 2006, 339, 204–225. [Google Scholar] [CrossRef]
- Aristeidou, K.; Gerovasileiou, V.; Koutsoubas, D. Wrecks as artificial reefs and marine areas of diving interest: The case of Zenobia wreck from Cyprus (E. Mediterranean). In Proceedings of the 9th Symposium on Oceanography & Fisheries, Patra, Greece, 4–8 May 2009. [Google Scholar]
- Jiménez, C.; Hadjioannou, L.; Petrou, A.; Andreou, V.; Georgiou, A. Fouling communities of two accidental artificial reefs (modern shipwrecks) in Cyprus (Levantine Sea). Water 2017, 9, 11. [Google Scholar] [CrossRef]
- Papatheodoulou, M.; Jimenez, C.; Petrou, A.; Thasitis, I. Endobiotic communities of marine sponges in Cyprus (Levantine Sea). Heliyon 2019, 5, e01392. [Google Scholar] [CrossRef]
- Çinar, M.E. Alien polychaete species (Annelida: Polychaeta) on the southern coast of Turkey (Levantine Sea, eastern Mediterranean), with 13 new records for the Mediterranean Sea. J. Nat. Hist. 2009, 42, 1975–1990. [Google Scholar] [CrossRef]
- Grassle, J.P.; Grassle, J.F. Sibling species in the marine pollution indicator Capitella (Polychaeta). Science 1976, 192, 567–569. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Qian, P.-Y.; Zhang, S. Morphology, reproduction, ecology and allozyme electrophoresis of three Capitella sibling species in Qingdao (Polychaeta: Capitellidae). In Systematics, Biology and Morphology of World Polychaeta; Brill: Leiden, The Netherlands, 1991; pp. 391–400. [Google Scholar]
- Blake, J.A.; Grassle, J.P.; Eckelbarger, K.J. Capitella teleta, a new species designation for the opportunistic and experimental Capitella sp. I, with a review of the literature for confirmed records. Zoosymposia 2009, 2, 25–53. [Google Scholar] [CrossRef]
- Silva, C.F.; Seixas, V.C.; Barroso, R.; Di Domenico, M.; Amaral, A.C.Z.; Paiva, P.C. Demystifying the Capitella capitata complex (Annelida, Capitellidae) diversity by morphological and molecular data along the Brazilian coast. PLoS ONE 2017, 12, e0177760. [Google Scholar] [CrossRef]
- Blake, J.A. Redescription of Capitella capitata (Fabricius) from West Greenland and designation of a neotype (Polychaeta, Capitellidae). Zoosymposia 2009, 2, 55–80. [Google Scholar] [CrossRef]
- Kurt-Şahin, G.; Çinar, M.E. A check-list of polychaete species (Annelida: Polychaeta) from the Black Sea. J. Black Sea Mediterr. Environ. 2012, 18, 10–48. [Google Scholar]
- Akoumianaki, I.; Hughes, J.A. The distribution of macroinfauna along a Mediterranean submarine cave with sulphur springs. Cahiers Biol. Mar. 2004, 45, 355–364. [Google Scholar]
- Zenetos, A.; Gofas, S.; Verlaque, M.; Çinar, M.E.; Garcia Raso, J.E.; Bianchi, C.N.; Morri, C.; Azzurro, E.; Bilecenoglu, M.; Froglia, C.; et al. Alien species in the Mediterranean Sea by 2010. A contribution to the application of European Union’s Marine Strategy Framework Directive (MSFD). Part I. spatial distribution. Medit. Mar. Sci. 2010, 11, 381–493. [Google Scholar] [CrossRef]
- Viéitez, J.M.; Alós, C.; Parapar, J.; Besteiro, C.; Moreira, J.; Núñez, J.; Laborda, A.J.; San Martín, G. Annelida Polychaeta I. Fauna Iberica; Museo Nacional de Ciencias Naturales, CSIC: Madrid, Spain, 2004; Volume 25, 530p. [Google Scholar]
- Fauvel, P. Quatrième note préliminaire sur les Polychètes provenant des campagnées de l’Hirondelle et de la Princesse-Alice, ou deposées dans le Musée Océanographique de Monaco. Bull. Inst. Océanogr. Monaco 1913, 270, 1–80. [Google Scholar]
- Lezzi, M. Caulleriella mediterranea, a new species of polychaete (Annelida: Cirratulidae) from the central Mediterranean Sea. Eur. Zool. J. 2017, 84, 380–389. [Google Scholar] [CrossRef]
- Blake, J.A. Bitentaculate Cirratulidae (Annelida, Polychaeta) collected chiefly during cruises of the R/V Anton Bruun, USNS Eltanin, USCG Glacier, R/V Hero, RVIB Nathaniel B. Palmer, and R/V Polarstern from the Southern Ocean, Antarctica, and off Western South America. Zootaxa 2018, 4537, 130p. [Google Scholar] [CrossRef] [PubMed]
- Çinar, M.E.; Ergen, Z. The presence of Chaetozone corona (Polychaeta: Cirratulidae) in the Mediterranean Sea: An alien or a native species? Cahiers Biol. Mar. 2007, 48, 339–346. [Google Scholar]
- Le Garrec, V.; Grall, J.; Chevalier, C.; Guyonnet, B.; Jourde, J.; Lavesque, N.; Bonifacio, P.; Blake, J.A. Chaetozone corona (Polychaeta, Cirratulidae) in the Bay of Biscay: A new alien species for the North-east Atlantic waters? J. Mar. Biol. Assoc. UK 2017, 97, 433–445. [Google Scholar] [CrossRef]
- Grosse, M.; Bakken, T.; Nygren, A.; Kongsrud, J.A.; Capa, M. Species delimitation analysis of NE Atlantic Chaetozone (Annelida, Cirratulidae) reveals hidden diversity among a common and abundant marine annelid. Mol. Phylogenet. Evol. 2020, 149, 106582. [Google Scholar] [CrossRef]
- Grosse, M.; Capa, M.; Bakken, T. Describing the hidden species diversity of Chaetozone (Annelida, Cirratulidae) in the Norwegian Sea using morphological and molecular diagnostics. Zookeys 2021, 1039, 139–176. [Google Scholar] [CrossRef]
- Çinar, M.E.; Dağli, E. Bioeroding (boring) polychaete species (Annelida: Polychaeta) from the Aegean Sea (eastern Mediterranean). J. Mar. Biol. Assoc. UK 2021, 101, 309–318. [Google Scholar] [CrossRef]
- Blake, J.A. Kirkegaardia (Polychaeta, Cirratulidae), new name for Monticellina Laubier, preoccupied in the Rhabdocoela, together with new records and descriptions of eight previously known and sixteen new species from the Atlantic, Pacific, and Southern Oceans. Zootaxa 2016, 4166, 1–93. [Google Scholar] [CrossRef]
- Bogdanos, C.; Fredj, G. Sur la présence de Cossura coasta Kitamori, 1960 (Polychaeta, Cossuridae) dans les eaux côtières grecques (Mer Égée, Mer Ionienne). Thalassographica 1983, 6, 5–15. [Google Scholar]
- Wehe, T.; Fiege, D. Annotated checklist of the polychaete species of the seas surrounding the Arabian Peninsula: Red Sea, Gulf of Aden, Arabian Sea, Gulf of Oman, Arabian Gulf. Fauna Arab. 2002, 19, 7–238. [Google Scholar]
- Arvanitidis, C. Systematic and Bionomic Study of the Macrobenthic Polychaete (Annelida) of the Northern Aegean. Ph.D. Thesis, Aristotle University of Thessaloniki, Thessaloniki, Greece, 1994; 512p. (In Greek). [Google Scholar]
- Grosse, M.; Zhadan, A.; Langeneck, J.; Fiege, D.; Martínez, A. Still digging: Advances and perspectives in the study of the diversity of several sedentarian annelid families. Diversity 2021, 13, 132. [Google Scholar] [CrossRef]
- Corsini-Foka, M.; Zenetos, A.; Crocetta, F.; Çinar, M.E.; Koçak, F.; Golani, D.; Katsanevakis, S.; Tsiamis, K.; Cook, E.; Froglia, C.; et al. Inventory of alien and cryptogenic species of the Dodecanese (Aegean Sea; Greece): Collaboration through COST action training school. Manag. Biol. Invasions 2015, 6, 351–366. [Google Scholar] [CrossRef]
- Tempesti, J.; Langeneck, J.; Romani, L.; Garrido, M.; Lardicci, C.; Maltagliati, F.; Castelli, A. Harbour type and use destination shape fouling community and non-indigenous species assemblage: A study of three Northern Tyrrhenian port systems (Mediterranean Sea). Mar. Pollut. Bull. 2022, 174, 113191. [Google Scholar] [CrossRef] [PubMed]
- Paavo, B.; Bailey-Brock, J.H.; Åkesson, B. Morphology and life history of Ophryotrocha adherens sp. nov. (Polychaeta, Dorvilleidae). Sarsia 2000, 85, 251–264. [Google Scholar] [CrossRef]
- Munari, C.; Ebbe, B. A new species of Protodorvillea (Polychaeta: Dorvilleidae) from the Western Mediterranean Sea. Europ. J. Zool. 2019, 86, 196–209. [Google Scholar] [CrossRef]
- Hutchings, P.A.; Murray, A. Taxonomy of polychaetes from the Hawkesbury River and the southern estuaries of New South Wales, Australia. In Records of the Australian Museum; Australian Museum: Darlinghurst, Australia, 1984; pp. 1–118. [Google Scholar]
- Hartmann-Schröder, G. Die Polychaeten der antiborealen Südküste Australiens (zwischen Port Lincoln im Westen und Port Augusta im Osten). Teil 11. Mitt. Zool. Mus. Hamburg 1985, 82, 61–99. [Google Scholar]
- Haswell, W.A. Observation on some Australian Polychaeta. Part I. I. Syllidae. II. Staurocephalus. III. Eulalia. IV. Psamathe. IV. Siphonostoma. V. Halla. Proc. Linn. Soc. N. S. Wales 1886, 10, 733–756. [Google Scholar] [CrossRef]
- Marenzeller, E.V. Berichte der Commission für Oceanographische Erforschung des Östlichen Mittelmeeres. Zweite Reihe. VI. Zoologische Ergebnisse II. Polychäten des Grundes, gesammelt 1890, 1891, 1892. Denkschr. Akad. Wissensch. Wien 1893, 60, 25–48. [Google Scholar]
- Pruvot, G.; Racovitza, E.G. Matériaux pour la faune des annélides de Banyuls. Arch. Zool. Exp Gen. Sér. 3 1895, 3, 339–492. [Google Scholar]
- Gravina, M.F.; Pierri, C.; Mercurio, M.; Nonnis Marzano, C.; Giangrande, A. Polychaete diversity related to different mesophotic bioconstructions along the Southeastern Italian coast. Diversity 2021, 13, 239. [Google Scholar] [CrossRef]
- Zanol, J.; Halanych, K.M.; Fauchald, K. Reconciling taxonomy and phylogeny in the bristleworm family Eunicidae (polychaete, Annelida). Zool. Scr. 2014, 43, 79–100. [Google Scholar] [CrossRef]
- Martín, D. Anélidos poliquetos y moluscos asociados a algas calcáreas del litoral catalán. Miscellània Zool. 1987, 11, 61–75. [Google Scholar]
- Kurt-Şahin, G.; Çinar, M.E. Eunicidae (Polychaeta) species in and around Iskenderun Bay (Levantine Sea, Eastern Mediterranean) with a new alien species for the Mediterranean Sea and a re-description of Lysidice collaris. Turk. J. Zool. 2009, 33, 331–347. [Google Scholar]
- Lavesque, N.; Daffe, G.; Bonifácio, P.; Hutchings, P. A new species of the Marphysa sanguinea complex from French waters (Bay of Biscay, NE Atlantic) (Annelida, Eunicidae). Zookeys 2017, 716, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Martín, D.; Gil, J.; Zanol, J.; Meca, M.A.; Pérez Portela, R. Digging the diversity of Iberian bait worms Marphysa (Annelida, Eunicidae). PLoS ONE 2020, 15, e0226749. [Google Scholar] [CrossRef]
- Elgetany, A.H.; El-Ghobashy, A.E.; Ghoneim, A.; Struck, T.H. Description of a new species of the genus Marphysa (Eunicidae), Marphysa aegypti sp. n., based on molecular and morphological evidence. Invertebr. Zool. 2018, 15, 71–84. [Google Scholar] [CrossRef]
- Lavesque, N.; Hutchings, P.; Abe, H.; Daffe, G.; Gunton, L.M.; Glasby, C.J. Confirming the exotic status of Marphysa victori Lavesque, Daffe, Bonifácio & Hutchings, 2017 (Annelida) in French waters and synonymy of Marphysa bulla Liu, Hutchings & Kupriyanova, 2018. Aquat. Invasions 2020, 15, 355–366. [Google Scholar]
- Katsiaras, N.; Simboura, N.; Koutsoubas, D. The rare subgroup C1 of Marphysa (Polychaeta, Eunicidae): Re-description of species and first records in the Mediterranean Sea. Zootaxa 2014, 3873, 201–217. [Google Scholar] [CrossRef]
- Molina-Acevedo, I.C. Morphological revision of the Subgroup 1 Fauchald, 1970 of Marphysa de Quatrefages, 1865 (Eunicidae: Polychaeta). Zootaxa 2018, 4480, 125p. [Google Scholar] [CrossRef]
- Salazar-Vallejo, S.I. Revision of Stylarioides Delle Chiaje, 1831 (Annelida: Flabelligeridae). Ital. J. Zool. 2011, 78, 163–200. [Google Scholar] [CrossRef]
- Grube, A.E. Beschreibungen einiger von Georg Ritter von Frauenfeld gesammelter Anneliden und Gephyreen des rothen Meeres. Verhandlungen Kais. Königlichen Zool. Bot. Ges. Wien 1868, 18, 629–650. [Google Scholar]
- Borghese, J.; Musco, L.; Arduini, D.; Tamburello, L.; Del Pasqua, M.; Giangrande, A. A comparative approach to detect macrobenthic response to the conversion of an inshore mariculture plant into an IMTA system in the Mar Grande of Taranto (Mediterranean Sea, Italy). Water 2023, 15, 68. [Google Scholar] [CrossRef]
- Parapar, J.; Adarraga, I.; Aguado, M.T.; Aguirrezabalaga, F.; Arias, A.; Besteiro, C.; Bleidorn, C.; Capa, M.; Capaccioni-Azzati, R.; El-Haddad, M.; et al. Fauna Iberica 45. Annelida; Polychaeta V.; Museo Nacional de Ciencias Naturales, CSIC: Madrid, Spain, 2018; 631p. [Google Scholar]
- Moraitis, M.; Papageorgiou, N.; Dimitriou, P.D.; Petrou, A.; Karakassis, I. Effects of offshore tuna farming on benthic assemblages in the Eastern Mediterranean. Aquacult. Environ. Interact. 2013, 4, 41–51. [Google Scholar] [CrossRef]
- Salazar-Vallejo, S.I. Revision of Hesione Savigny in Lamarck, 1818 (Annelida, Errantia, Hesionidae). Zoosystema 2018, 40, 227–325. [Google Scholar] [CrossRef]
- Kurt-Şahin, G.; Çinar, M.E.; Gönülal, O. A new species of Augeneria (Polychaeta: Lumbrineridae) from deep waters of the Aegean Sea (eastern Mediterranean). Mediterr. Mar. Sci. 2016, 17, 708–713. [Google Scholar] [CrossRef]
- Katsiaras, N.; Rousou, M.; Carrera-Parra, L.; Garcia-Gomez, S.; Simboura, N.; Louizidou, P.; Chintiroglou, C.C.; Martins, R. Taxonomy; ecology and geographic distribution of Gallardoneris iberica (Polychaeta; Lumbrineridae) in southern Europe. J. Mar. Biol. Assoc. UK 2018, 98, 1609–1618. [Google Scholar] [CrossRef]
- Martin, D.; Estefa, J.; Gil, J. Taxonomic review of Gallardoneris nonatoi (Ramos, 1976) comb. nov. (Annelida, Lumbrineridae), and description of a new species of Lumbrineris from the Gulf of Mexico. Zookeys 2022, 1114, 35–57. [Google Scholar] [CrossRef]
- Carrera-Parra, L.F. Lumbrineridae (Annelida: Polychaeta) from the Gran Caribbean region with the description of six new species. J. Mar. Biol. Assoc. UK 2001, 81, 599–621. [Google Scholar] [CrossRef]
- Ramos, J.M. Lumbrineridae (Polychètes errantes) de Méditerranée. Ann. Inst. Océanogr. 1976, 52, 103–137. [Google Scholar]
- Grube, A.E. Beschreibung neuer oder wenig bekannter Anneliden. Sechster Beitrag. Arch. Für Naturgeschichte 1863, 29, 37–69. [Google Scholar] [CrossRef]
- Claparède, É. Les annélides chétopodes du Golfe de Naples. Mém. Soc. Phys. Hist. Nat. Genève 1868, 19, 313–584. [Google Scholar]
- Ehlers, E.H. Die Borstenwürmer (Annelida Chaetopoda) Nach Systematischen und Anatomischen Untersuchungen Dargestellt; Wilhelm Enelmann: Leipzig, Germany, 1868; 748p. [Google Scholar]
- Hartmann-Schröder, G. Annelida, Borstenwürmer, Polychaeta. 2nd Revised ed. The Fauna of Germany and Adjacent Seas with Their Characteristics and Ecology, 58; Gustav Fischer: Jena, Germany, 1996; 648p. [Google Scholar]
- Oug, E. Guide to Identification of Lumbrineridae (Polychaeta) in North East Atlantic Waters v.3.2; NMBAQC 2010 Taxonomic Workshop; Dove Marine Laboratory: North Shields, UK, 2012; 31p. [Google Scholar]
- Quatrefages, A. Histoire Naturelle des Annelés Marins et D’Eau Douce. Annélides et Gephyriens. Volume 2; Librarie Encyclopédique de Roret: Paris, France, 1866; 794p. [Google Scholar]
- Çinar, M.E.; Bilecenoglu, M.; Öztürk, B.; Katağan, T.; Aysel, V. Alien species on the coasts of Turkey. Mediterr. Mar. Sci. 2005, 6, 119–146. [Google Scholar] [CrossRef]
- Zenetos, A.; Çinar, M.E.; Pancucci-Papadopoulou, M.A.; Harmelin, J.-G.; Furnari, G.; Andaloro, F.; Bellou, N.; Streftaris, N.; Zibrowius, H. Annotated list of marine alien species in the Mediterranean with records of the worst invasive species. Mediterr. Mar. Sci. 2005, 6, 63–118. [Google Scholar] [CrossRef]
- Kuş, S.; Kurt, G.; Çinar, M.E. Nephtyidae (Annelida: Polychaeta) from the Sea of Marmara and Black Sea, with descriptions of two new species. Zootaxa 2021, 5060, 33–64. [Google Scholar] [CrossRef] [PubMed]
- Christomanos, A.; Giannitsis, D. Dredging results from the oceanographic cruise “Tethys” during August-September 1961. Mar. Sci. Pap. 1962, 1, 7–11. [Google Scholar]
- Occhipinti-Ambrogi, A.; Andaloro, F.; Azzurro, E.; Cardeccia, A.; Carnevali, L.; Castelli, A.; Cecere, E.; Cerri, J.; Donnarumma, L.; Ferrario, J.; et al. Prioritizzazione delle specie aliene marine italiane per l’implementazione di una lista di specie aliene invasive di rilevanza nazionale ai sensi del regolamento EU 1143/2014 e del decreto legislativo 230/2017. Biol. Mar. Medit. 2019, 26, 219–222. [Google Scholar]
- Teixeira, M.A.L.; Bakken, T.; Vieira, P.E.; Langeneck, J.; Sampieri, B.R.; Kasapidis, P.; Ravara, A.; Nygren, A.; Costa, F.O. The curious and intricate case of the European Hediste diversicolor (Annelida, Nereididae) species complex, with description of two new species. Syst. Biodivers. 2022, 20, 2116124. [Google Scholar] [CrossRef]
- Muir, A.I.; Smith, B.D.; Garcia-Alonso, J. Neanthes nubila (Annelida: Polychaeta)—A review of its biogeography and breeding habits. Cah. Biol. Mar. 2014, 55, 275–280. [Google Scholar]
- Villalobos-Guerrero, T.F.; Kara, J.; Simon, C.; Idris, I. Systematic review of Neanthes Kinberg, 1865 (Annelida: Errantia: Nereididae) from southern Africa, including a preliminary molecular phylogeny of the genus. Mar. Biodivers. 2022, 52, 21. [Google Scholar] [CrossRef]
- López, E.; Richter, A. Non-indigenous species (NIS) of polychaetes (Annelida: Polychaeta) from the Atlantic and Mediterranean coasts of the Iberian Peninsula: An annotated checklist. Helgol. Mar. Res. 2017, 71, 19. [Google Scholar] [CrossRef]
- Gravina, M.F.; Lezzi, M.; Bonifazi, A.; Giangrande, A. The genus Nereis L., 1758 (Polychaeta, Nereididae): State of the art for identification of Mediterranean species. Atti Soc. Toscana Sci. Nat. Mem. Ser. B 2015, 122, 147–164. [Google Scholar]
- Malmgren, A.J. Annulata Polychaeta Spetsbergiæ, Grœnlandiæ, Islandiæ et Scandinaviæ Hactenus Cognita; Ex Officina Frenckelliana: Helsinki, Finland, 1867; 127p. [Google Scholar]
- Maltagliati, F.; Camilli, L.; Lardicci, C.; Castelli, A. Evidence for morphological and genetic divergence in Perinereis cultrifera (Polychaeta: Nereididae) from two habitat types at Elba Island. J. Mar. Biol. Assoc. UK 2001, 81, 411–414. [Google Scholar] [CrossRef]
- Rouabah, A.; Scaps, P. Two-dimensional electrophoresis analysis of proteins from epitokous forms of the polychaete Perineris cultrifera from the English Channel and the Mediterranean Sea. Cah. Biol. Mar. 2003, 44, 227–236. [Google Scholar]
- Özpolat, B.D.; Randel, N.; Williams, E.A.; Bezares-Calderón, L.A.; Andreatta, G.; Balavoine, G.; Bertucci, P.Y.; Ferrier, D.E.K.; Gambi, M.C.; Gazave, E.; et al. The Nereid on the rise: Platynereis as a model system. EvoDevo 2021, 12, 10. [Google Scholar] [CrossRef] [PubMed]
- Wäge, J.; Valvassori, G.; Hardege, J.D.; Schulze, A.; Gambi, M.C. The sibling polychaetes Platynereis dumerilii and Platynereis massiliensis in the Mediterranean Sea: Are phylogeographic patterns related to exposure to ocean acidification? Mar. Biol. 2017, 164, 199. [Google Scholar] [CrossRef]
- Cantone, G. Primo rinvenimento in Mediterraneo di Rullierinereis Pettibone, 1971 (Policheti Nereidi) con descrizione di una nuova specie. Animalia 1982, 9, 103–107. [Google Scholar]
- Tena, J.; Capaccioni-Azzati, R.; Porras, R.; Torres-Gavilá, F.J. Cuatro especies de poliquetos nuevas para las costas mediterráneas españolas en los sedimentos del antepuerto de Valencia. Miscellània Zool. 1991, 15, 29–41. [Google Scholar]
- Borisova, P.B.; Schepetov, D.M.; Budaeva, N.E. Aponuphis Kucheruk, 1978 (Annelida: Onuphidae) from western African waters. Invertebr. Zool. 2018, 15, 19–41. [Google Scholar] [CrossRef]
- Paxton, H. A new species of Diopatra (Annelida: Onuphidae) from Namibia, southwestern Africa. Mar. Biodiv. 2016, 46, 889–895. [Google Scholar] [CrossRef]
- Elgetany, A.H.; van Rensburg, H.; Hektoen, M.; Matthee, C.; Budaeva, N.; Simon, C.A.; Struck, T.H. Species delimitation in the speciation grey zone: The case of Diopatra (Annelida, Onuphidae). Zool. Scripta 2020, 49, 516–534. [Google Scholar] [CrossRef]
- European Nature Information System (EUNIS). Available online: https://eunis.eea.europa.eu/ (accessed on 28 June 2023).
- SeaLifeBase. Available online: https://www.sealifebase.ca/ (accessed on 28 June 2023).
- De Filippi, F. Armandia nuovo genere di Annelidi nel Mediterraneo. Arch. Zool. L’anatomia Fisiol. 1861, 1, 215–219. [Google Scholar]
- Harmelin, J.-G. Contribution à l’étude de l’endofaune des prairies d’Halophila stipulacea de Méditerranée orientale. Recl. Trav. Stn. Mar. Endoume 1969, 45, 305–320. [Google Scholar]
- Blake, J.A. Polychaeta Orbiniidae from Antarctica, the Southern Ocean, the Abyssal Pacific Ocean, and off South America. Zootaxa 2018, 4218, 145p. [Google Scholar] [CrossRef]
- Day, J.H. Some Polychaeta from the Israel South Red Sea Expedition, 1962. Bull. Sea Fisher. Res. Stat. Haifa 1965, 38, 15–27. [Google Scholar]
- Blake, J.A.; Giangrande, A. Naineris setosa (Verrill) (Polychaeta, Orbiniidae), an American subtropical–tropical polychaete collected from an aquaculture facility in Brindisi (Adriatic Sea, Italy): A possible alien species. Ital. J. Zool. 2011, 78, 20–26. [Google Scholar] [CrossRef]
- Khedhri, I.; Lavesque, N.; Bonifácio, P.; Djabou, H.; Afli, A. First record of Naineris setosa (Verrill, 1900) (Annelida: Polychaeta: Orbiniidae) in the Western Mediterranean Sea. BioInvasions Rec. 2014, 3, 83–88. [Google Scholar] [CrossRef]
- Atzori, G.; López, E.; Addis, P.; Sabatini, A.; Cabiddu, S. First record of the alien polychaete Naineris setosa (Scolecida; Orbiniidae) in Tyrrhenian Sea (Western Mediterranean). Mar. Biodivers. Rec. 2016, 9, e05. [Google Scholar] [CrossRef]
- Tempesti, J.; Langeneck, J.; Maltagliati, F.; Castelli, A. Macrobenthic fouling assemblages and NIS success in a Mediterranean port: The role of use destination. Mar. Pollut. Bull. 2020, 150, 110768. [Google Scholar] [CrossRef]
- Bleidorn, C.; Kruse, I.; Albrecht, S.; Bartolomaeus, T. Mitochondrial sequence data expose the putative cosmopolitan polychaete Scoloplos armiger (Annelida, Orbiniidae) as a species complex. BMC Evol. Biol. 2006, 6, 47. [Google Scholar] [CrossRef]
- Luttikhuizen, P.C.; Bol, A.; Cardoso, J.F.M.F.; Dekker, R. Overlapping distributions of cryptic Scoloplos cf. armiger species in the western Wadden Sea. J. Sea Res. 2011, 66, 231–237. [Google Scholar]
- Koh, B.S.; Bhaud, M. Description of Owenia gomsoni n. sp. (Oweniidae, Annelida Polychaeta) from the Yellow Sea and evidence that Owenia fusiformis is not a cosmopolitan species. Vie Milieu 2001, 51, 77–86. [Google Scholar]
- Koh, B.S.; Bhaud, M. Identification of differentiating criteria between populations of Owenia fusiformis (Annelida Polychaeta) from different origins. Vie Milieu 2003, 53, 64–95. [Google Scholar]
- Langeneck, J.; Barbieri, M.; Maltagliati, F.; Castelli, A. Molecular phylogeny of Paraonidae (Annelida). Mol. Phylogenet. Evol. 2019, 136, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Laubier, L. Sur quelques Aricidea (Polychètes, Paraonidae) de Banyuls-sur-Mer. Vie Milieu 1967, 18, 99–132. [Google Scholar]
- Langeneck, J.; Mazziotti, C.; Mikac, B.; Scirocco, T.; Castelli, A. Aricidea fragilis (Annelida: Paraonidae) in the Mediterranean Sea: Overlooked native or alien species? Eur. Zool. J. 2018, 85, 267–272. [Google Scholar] [CrossRef]
- Vatova, A. La fauna bentonica dell’alto e medio Adriatico. Nova Thalass. 1949, 1, 110p. [Google Scholar]
- Fauvel, P. Annélides Polychètes de la Haute-Adriatique. Thalass. Jena 1940, 4, 24p. [Google Scholar]
- Strelzov, V.E. Polychaete Worms of the Family Paraonidae Cerruti, 1909 (Polychaeta, Sedentaria); Akademia Nauk: Moscow, Russia, 1973; 170p. (In Russian) [Google Scholar]
- Çinar, M.E.; Dağli, E.; Erdoğan-Dereli, D. The diversity of polychaetes (Annelida: Polychaeta) in a long-term pollution monitoring study from the Levantine coast of Turkey (Eastern Mediterranean), with the descriptions of four species new to science and two species new to the Mediterranean fauna. J. Nat. Hist. 2022, 56, 1383–1426. [Google Scholar] [CrossRef]
- Laubier, L.; Ramos, J. Paraonidae (Polychètes Sédentaires) de Méditerranée. Bull. Mus. Natn. Hist. Nat. Paris 1974, 113, 1097–1148. [Google Scholar]
- Hartley, J.P. The family Paraonidae (Polychaeta) in British waters: A new species and new records with a key to species. J. Mar. Biol. Assoc. UK 1981, 61, 133–149. [Google Scholar] [CrossRef]
- Erdoğan-Dereli, D.; Çinar, M.E. The genus Paradoneis (Annelida: Paraonidae) from the Sea of Marmara, with descriptions of two new species. Zootaxa 2019, 4686, 465–496. [Google Scholar] [CrossRef] [PubMed]
- Erdoğan-Dereli, D.; Çinar, M.E. Levinsenia species (Annelida: Polychaeta: Paraonidae) from the Sea of Marmara with descriptions of two new species. Zootaxa 2021, 4908, 151–180. [Google Scholar] [CrossRef]
- Langeneck, J.; Barbieri, M.; Maltagliati, F.; Castelli, A. A new species of Cirrophorus (Annelida: Paraonidae) from Mediterranean organically enriched coastal environments, with taxonomic notes on the family. J. Mar. Biol. Assoc. UK 2017, 97, 871–880. [Google Scholar] [CrossRef]
- Langeneck, J.; Diez, M.E.; Nygren, A.; Salazar-Vallejo, S.; Carrera-Parra, L.F.; Vega Fernández, T.; Badalamenti, F.; Castelli, A.; Musco, L. Worming its way into Patagonia: An integrative approach reveals the cryptic invasion by Eulalia clavigera (Annelida: Phyllodocidae). Mar. Biodivers. 2019, 49, 851–861. [Google Scholar] [CrossRef]
- Teixeira, M.A.L.; Vieira, P.E.; Fenwick, D.; Langeneck, J.; Pleijel, F.; Sampieri, B.R.; Hernandez, J.C.; Ravara, A.; Costa, F.O.; Nygren, A. Revealing the diversity of the green Eulalia (Annelida, Phyllodocidae) species complex along the European coast, with description of three new species. Org. Divers. Evol. 2023, 1–27. [Google Scholar] [CrossRef]
- Nygren, A.; Pleijel, F. From one to ten in a single stroke—Resolving the European Eumida sanguinea (Phyllodocidae, Annelida) species complex. Mol. Phylogenet. Evol. 2011, 58, 132–141. [Google Scholar] [CrossRef]
- Teixeira, M.A.L.; Vieira, P.E.; Ravara, A.; Costa, F.O.; Nygren, A. From 13 to 22 in a second stroke: Revisiting the European Eumida sanguinea (Phyllodocidae: Annelida) species complex. Zool. J. Linn. Soc. 2022, 196, 169–197. [Google Scholar] [CrossRef]
- Pleijel, F.; Dales, R.P. Polychaetes: British Phyllodocoideans, Typhloscolecoideans and Tomopteroideans. Synop. Br. Fauna (NS) 1991, 45, 202. [Google Scholar]
- Pleijel, F. Polychaeta, Phyllodocidae. Marine Invertebrates of Scandinavia 8; Scandinavian University Press: Oslo, Norway, 1993; 159p. [Google Scholar]
- Nygren, A.; Eklöf, J.; Pleijel, F. Cryptic species of Notophyllum (Polychaeta: Phyllodocidae) in Scandinavian waters. Organisms Divers. Evol. 2010, 10, 193–204. [Google Scholar] [CrossRef]
- Giangrande, A.; Licciano, M. Revision of the species of Megalomma (Polychaeta: Sabellidae) from the Mediterranean Sea, with the description of M. messapicum n. sp. Ital. J. Zool. 2008, 75, 207–217. [Google Scholar] [CrossRef]
- Calosi, P.; Rastrick, S.P.S.; Lombardi, C.; de Guzman, H.J.; Davidson, L.; Jahnke, M.; Giangrande, A.; Hardege, J.D.; Schulze, A.; Spicer, J.I.; et al. Adaptation and acclimatization to ocean acidification in marine ectotherms: An in situ transplant experiment with polychaetes at a shallow CO2 vent system. Phil. Trans. Royal Soc. Ser. B 2013, 368, 20120444. [Google Scholar] [CrossRef] [PubMed]
- Tilic, E.; Feerst, K.G.; Rouse, G.W. Two new species of Amphiglena (Sabellidae, Anellida), with an assessment of hidden diversity in the Mediterranean. Zootaxa 2019, 4648, 337–353. [Google Scholar] [CrossRef] [PubMed]
- Giangrande, A.; Putignano, M.; Licciano, M.; Gambi, M.C. The Pandora’s box: Morphological diversity within the genus Amphiglena Claparède, 1864 (Sabellidae, Annelida) in the Mediterranean Sea, with description of nine new species. Zootaxa 2021, 4949, 201–239. [Google Scholar] [CrossRef] [PubMed]
- Del Pasqua, M.; Schulze, A.; Tovar-Hernández, M.A.; Keppel, E.; Lezzi, M.; Gambi, M.C.; Giangrande, A. Clarifying the taxonomic status of the alien species Branchiomma bairdi and Branchiomma boholense (Annelida: Sabellidae) using molecular and morphological evidence. PLoS ONE 2018, 13, e0197104. [Google Scholar] [CrossRef] [PubMed]
- Tovar-Hernández, M.A.; Licciano, M.; Giangrande, A. Revision of Chone Krøyer, 1856 (Polychaeta: Sabellidae) from the eastern central Atlantic and Mediterranean Sea with descriptions of two new species. Sci. Mar. 2007, 71, 315–338. [Google Scholar] [CrossRef]
- Fitzhugh, K. Fan worm polychaetes (Sabellidae: Sabellinae) collected during the Thai-Danish BIOSHELF Project. Phuket Mar. Biol. Center Spec. Publ. 2002, 24, 353–424. [Google Scholar]
- Selim, S.A.; Rzhavsky, A.V.; Britayev, T.A. Dialychone and Paradialychone (Polychaeta: Sabellidae) from the Mediterranean Coast of Egypt with description of Dialychone egyptica sp. n. Invertebr. Zool. 2012, 9, 105–114. [Google Scholar] [CrossRef]
- Giangrande, A.; Licciano, M. The genus Euchone (Polychaeta, Sabellidae) in the Mediterranean Sea, addition of two new species and discussion on some closely related taxa. J. Nat. Hist. 2006, 40, 1301–1330. [Google Scholar] [CrossRef]
- Giangrande, A. Censimento dei policheti dei mari italiani: Sabellidae Malmgren, 1867. Atti Soc. Toscana Sci. Nat. Mem. Ser. B 1989, 96, 153–189. [Google Scholar]
- Putignano, M.; Gravili, C.; Giangrande, A. The peculiar case of Myxicola infundibulum (Polychaeta: Sabellidae): Echo from a science 200 years old and description of four new taxa in the Mediterranean Sea. Eur. Zool. J. 2023, 90, 506–546. [Google Scholar] [CrossRef]
- Iroso, I. Revisione dei Serpulidi e Sabellidi del Golfo di Napoli. Pubbl. Stn. Zool. Napoli 1921, 3, 47–91. [Google Scholar]
- Sun, Y.; Wong, E.; Keppel, E.; Williamson, J.E.; Kupriyanova, E.K. A global invader or a complex of regionally distributed species? Clarifying the status of an invasive calcareous tubeworm Hydroides dianthus (Verrill, 1873) (Polychaeta: Serpulidae) using DNA barcoding. Mar. Biol. 2017, 164, 28. [Google Scholar] [CrossRef]
- Grosse, M.; Pérez, R.; Juan-Amengual, M.; Pons, J.; Capa, M. The elephant in the room: First record of invasive gregarious species of serpulids (calcareous tube annelids) in Majorca (western Mediterranean). Sci. Mar. 2021, 85, 15–28. [Google Scholar] [CrossRef]
- Ulman, A.; Ferrario, J.; Occhipinti-Ambrogi, A.; Arvanitidis, C.; Bandi, A.; Bertolino, M.; Bogi, C.; Chatzigeorgiou, G.; Çiçek, B.A.; Deidun, A.; et al. A massive update of non-indigenous species records in Mediterranean marinas. PeerJ. 2017, 5, e3954. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, C.N. Guide per il Riconoscimento Delle Specie Animali Delle Acque Lagunari e Costiere Italiane. 5. Policheti Serpuloidei; Consiglio Nazionale delle Ricerche: Roma, Italy, 1981; 190p. [Google Scholar]
- Mastrototaro, F.; D’Onghia, G.; Corriero, G.; Matarrese, A.; Maiorano, P.; Panetta, P.; Gherardi, M.; Longo, C.; Rosso, A.; Sciuto, F.; et al. Biodiversity of the white coral bank off Cape Santa Maria di Leuca (Mediterranean Sea): An update. Deep Sea Res. Part II 2010, 57, 412–430. [Google Scholar] [CrossRef]
- Zibrowius, H. Étude morphologique, systématique et écologique des Serpulidae (Annelida Polychaeta) de la region de Marseille. Rec. Trav. Stn. Mar. Endoume 1968, 43, 81–252. [Google Scholar]
- Scacchi, A. Catalogus Conchyliorum Regni Neapolitani Quae Usque Adhuc Reperit A. Scacchi; Francisci Xaverii Tornese: Napoli, Italy, 1836; 18p. [Google Scholar]
- Ben-Eliahu, M.N.; ten Hove, H.A. Redescription of Rhodopsis pusilla Bush, a little known but widely distributed species of Serpulidae (Polychaeta). Zool. Scr. 1989, 18, 381–395. [Google Scholar] [CrossRef]
- Ben-Eliahu, M.N. Polychaeta errantia of the Suez Canal. Israel J. Zool. 1972, 21, 189–237. [Google Scholar]
- Palero, F.; Torrado, H.; Perry, O.; Kupriyanova, E.; Ulman, A.; Genis-Armero, R.; ten Hove, H.A.; Capaccioni-Azzati, R. Following the Phoenician example: Western Mediterranean colonization by Spirobranchus cf. tetraceros (Annelida: Serpulidae). Sci. Mar. 2020, 84, 83–92. [Google Scholar] [CrossRef]
- Pettibone, M.H. Partial revision of the genus Sthenelais Kinberg (Polychaeta: Sigalionidae) with diagnoses of two new genera. Smithsonian Contrib. Zool. 1971, 109, 40p. [Google Scholar] [CrossRef]
- Grube, A.E. Beschreibungen neuer oder weniger bekannter von Hrn. Ehrenberg gesammelter Anneliden des rothen Meeres. Monatsberichte K. Preuss. Akad. Wiss. Berl. 1869, 484–521. [Google Scholar]
- Meißner, K.; Götting, M.; Nygren, A. Do we know who they are? On the identity of Pholoe (Annelida: Sigalionidae: Pholoinae) species from northern Europe. Zool. J. Linn. Soc. 2020, 189, 178–206. [Google Scholar] [CrossRef]
- Capa, M.; Bakken, T.; Meißner, K.; Nygren, A. Three, two, one! Revision of the long-bodied sphaerodorids (Sphaerodoridae, Annelida) and synonymization of Ephesiella, Ephesiopsis and Sphaerodorum. PeerJ 2018, 6, e5783. [Google Scholar] [CrossRef]
- Dağli, E.; Çinar, M.E. Species of the subgenera Aquilaspio and Prionospio (Polychaeta: Spionidae: Prionospio) from the southern coast of Turkey (Levantine Sea, eastern Mediterranean), with description of a new species and two new reports for the Mediterranean fauna. Zootaxa 2009, 2275, 20p. [Google Scholar] [CrossRef]
- Katsanevakis, S.; Poursanidis, D.; Hoffman, R.; Rizgalla, J.; Rothman, S.B.S.; Levitt-Barmats, Y.; Hadjioannou, L.; Trkov, D.; Garmendia, J.M.; Rizzo, M.; et al. Unpublished Mediterranean records of marine alien and cryptogenic species. BioInvasions Rec. 2020, 9, 165–182. [Google Scholar] [CrossRef]
- Maciolek, N.J. A revision of the genus Prionospio Malmgren, with special emphasis on species from the Atlantic Ocean, and new records of species belonging to the genera Apoprionospio Foster and Paraprionospio Caullery (Polychaeta, Annelida, Spionidae). Zool. J. Linn. Soc. 1985, 84, 325–383. [Google Scholar] [CrossRef]
- Dağli, E.; Cinar, M.E. Species of the subgenus Minuspio (Polychaeta: Spionidae: Prionospio) from the southern coast of Turkey (Levantine Sea, eastern Mediterranean), with the description of two new species. Zootaxa 2011, 3043, 35–53. [Google Scholar] [CrossRef]
- Radashevsky, V. I Pseudopolydora (Annelida: Spionidae) from European and adjacent waters with a key to identification and description of a new species. Mar. Biodivers. 2021, 51, 31. [Google Scholar] [CrossRef]
- Meißner, K. Revision of the genus Spiophanes (Polychaeta, Spionidae) with new synonymies, new records and descriptions of new species. Zoosyst. Evol. 2005, 81, 3–65. [Google Scholar] [CrossRef]
- Laubier, L. Un Spionidien des vases bathyales de Banyuls-sur-Mer, Spiophanes kroyeri reyssi ssp. n. Bull. Soc. Zool. Fr. 1964, 89, 562–577. [Google Scholar]
- Abd-Elnaby, F.A.; San Martín, G. Eusyllinae, Anoplosyllinae, and Exogoninae (Polychaeta: Syllidae) for the Mediterranean coasts of Egypt, together the description of one new species. Life Sci. J. 2010, 7, 132–139. [Google Scholar]
- Çinar, M.E.; Dağli, E. New records of alien polychaete species for the coasts of Turkey. Mediterr. Mar. Sci. 2012, 13, 103–107. [Google Scholar] [CrossRef]
- Aguado, M.T.; San Martín, G. Syllidae (Polychaeta) from Lebanon with two new reports for the Mediterranean Sea. Cah. Biol. Mar. 2007, 48, 207–224. [Google Scholar]
- Nygren, A. Revision of Autolytinae (Syllidae: Polychaeta). Zootaxa 2004, 680, 314p. [Google Scholar] [CrossRef]
- Álvarez-Campos, P.; Giribet, G.; San Martín, G.; Rouse, G.W.; Riesgo, A. Straightening the striped chaos: Systematics and evolution of Trypanosyllis and the case of its pseudocryptic type species Trypanosyllis krohnii (Annelida, Syllidae). Zool. J. Linn. Soc. 2017, 179, 492–540. [Google Scholar] [CrossRef]
- Kurt-Şahin, G.; Çinar, M.E.; Dağli, E. New records of polychaetes (Annelida) from the Black Sea. Cah. Biol. Mar. 2019, 60, 153–165. [Google Scholar]
- San Martín, G. Fauna Iberica 21. Annelida, Polychaeta II: Syllidae; Museo Nacional de Ciencias Naturales, CSIC: Madrid, Spain, 2003; 554p. [Google Scholar]
- Musco, L.; Çinar, M.E.; Giangrande, A. A new species of Sphaerosyllis (Polychaeta: Syllidae: Exogoninae) from the coasts of Italy and Cyprus (Eastern Mediterranean Sea). Ital. J. Zool. 2005, 72, 161–166. [Google Scholar] [CrossRef][Green Version]
- Del-Pilar-Ruso, Y.; San Martín, G. Description of a new species of Sphaerosyllis Claparède, 1863 (Polychaeta: Syllidae: Exogoninae) from the Alicante coast (W Mediterranean) and first reports of two other species of Syllidae for the Mediterranean Sea and the Iberian Peninsula. Mediterr. Mar. Sci. 2012, 13, 187–197. [Google Scholar] [CrossRef][Green Version]
- Licher, F. Revision der Gattung Typosyllis Langerhans, 1879 (Polychaeta: Syllidae). Morphologie, Taxonomie und Phylogenie. Abh. Senckenb. Naturfosch. Ges. 1999, 551, 1–336. [Google Scholar]
- Langeneck, J.; Musco, L.; Busoni, G.; Conese, I.; Aliani, S.; Castelli, A. Syllidae (Annelida: Phyllodocida) from the deep Mediterranean Sea, with the description of three new species. Zootaxa 2018, 4369, 197–220. [Google Scholar] [CrossRef]
- San Martín, G. Estudio biogeografico, faunistico y sistematico de los poliquetos de la familia silidos (Stlidae: Polychaeta) en Baleares. Publicaciones de la Universidad Complutense de Madrid. Colecc. Tesis Dr. 1984, 187/84, xxii+529. [Google Scholar]
- Çinar, M.E. Syllis ergeni: A new species of Syllidae (Annelida: Polychaeta) from Izmir Bay (Aegean Sea, eastern Mediterranean Sea). Zootaxa 2005, 1036, 43–54. [Google Scholar] [CrossRef]
- Abd-Elnaby, F.A.; San Martín, G. Syllinae (Syllidae: Polychaeta) from the Mediterranean coast of Egypt with the description of two new species. Mediterr. Mar. Sci. 2011, 12, 43–52. [Google Scholar] [CrossRef]
- Langeneck, J.; Scarpa, F.; Maltagliati, F.; Sanna, D.; Barbieri, M.; Cossu, P.; Mikac, B.; Curini Galletti, M.; Castelli, A.; Casu, M. A complex species complex: The controversial role of ecology and biogeography in the evolutionary history of Syllis gracilis Grube; 1840 (Annelida; Syllidae). J. Zool. Syst. Evol. Res. 2020, 58, 66–78. [Google Scholar] [CrossRef]
- Ben-Eliahu, M.N. Polychaete cryptofauna from rims of similar intertidal vermetid reefs on the Mediterranean coast of Israel and in the Gulf of Elat: Syllinae and Eusyllinae (Polychaeta Errantia: Syllidae). Israel J. Zool. 1977, 26, 1–58. [Google Scholar]
- Cosentino, A. Microhabitat selection in a local syllid assemblage with the first record of Syllis hyllebergi (Syllinae) in the central Mediterranean. Ital. J. Zool. 2011, 78, 267–279. [Google Scholar] [CrossRef]
- Katzmann, W. Polychaeten von Sedimentboden der mittleren Adria (50–525 m). Zool. Jahrbücher Abt. Für Syst. Okol. Geogr. Tiere 1973, 100, 436–450. [Google Scholar]
- Ba-Akdah, M.A.; Satheesh, S.; Al-Sofyani, A.M.A.; Lucas, Y.; Álvarez-Campos, P.; San Martín, G. Taxonomy of some species of the genus Syllis (Annelida: Syllidae: Syllinae) from the Red Sea found among the first colonizers of an artificial substrate. Mar. Biol. Res. 2018, 14, 790–805. [Google Scholar] [CrossRef]
- Hutchings, P.; Glasby, C.J. Redescription of Loimia medusa and Amphitrite rubra (Polychaeta: Terebellidae). Mitteilungen Hambg. Zool. Mus. Inst. 1995, 92, 149–154. [Google Scholar]
- Lavesque, N.; Daffe, G.; Londoño-Mesa, M.H.; Hutchings, P. Revision of the French Terebellidae sensu stricto (Annelida: Terebelliformia), with descriptions of nine new species. Zootaxa 2021, 5038, 63p. [Google Scholar] [CrossRef] [PubMed]
- Lavesque, N.; Hutchings, P.; Daffe, G.; Londoño-Mesa, M.H. Revision of the French Polycirridae (Annelida, Terebelliformia), with descriptions of eight new species. Zootaxa 2020, 4869, 151–186. [Google Scholar] [CrossRef]
- Lezzi, M.; Giangrande, A. New species of Streblosoma (Thelepodidae, Annelida) from the Mediterranean Sea: S. pseudocomatus sp. nov., S. nogueirai sp. nov. and S. hutchingsae sp. nov. J. Nat. Hist. 2018, 52, 2857–2873. [Google Scholar] [CrossRef]
- Lavesque, N.; Hutchings, P.; Daffe, G.; Nygren, A.; Londoño-Mesa, M.H. A revision of the French Trichobranchidae (Polychaeta), with descriptions of nine new species. Zootaxa 2019, 4664, 151–190. [Google Scholar] [CrossRef] [PubMed]
- Parapar, J.; Capa, M.; Nygren, A.; Moreira, J. To name but a few: Descriptions of five new species of Terebellides (Annelida, Trichobranchidae) from the North East Atlantic. Zookeys 2020, 992, 58p. [Google Scholar] [CrossRef]
- Barroso, M.; Moreira, J.; Capa, M.; Nygren, A.; Parapar, J. A further step towards the characterisation of Terebellides (Annelida, Trichobranchidae) diversity in the Northeast Atlantic, with the description of a new species. Zookeys 2022, 1132, 85–126. [Google Scholar] [CrossRef]
| Family/Species | Taxonomic Remarks | Depth Range (m) | Habitat Type | Source | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Hard (Including Rocks) | Soft/Hard | Soft | Artificial Construction | Sea Caves | In Sponges | Combination of Habitats | In Association with Flora | ||||
| Acoetidae | |||||||||||
| Eupanthalis kinbergi McIntosh, 1876 | 69–338 | x | x | Literature; This study | |||||||
| Panthalis oerstedii Kinberg, 1856 | NR | 359 | x | This study | |||||||
| Acrocirridae | |||||||||||
| Acrocirrus frontifilis (Grube, 1860) | 0–150 | x | x | x | Literature | ||||||
| Macrochaeta clavicornis (Sars, 1835) | 0–92 | x | x | x | x | Literature; This study | |||||
| Ampharetidae | |||||||||||
| Amage adspersa (Grube, 1863) | 12–210 | x | x | Literature; This study | |||||||
| Amage gallasii Marion, 1875 | 62–210 | x | x | Literature | |||||||
| Ampharete acutifrons (Grube, 1860) | 20–210 | x | x | Literature; This study | |||||||
| Ampharete octocirrata (Sars, 1835) | NR | 31–58 | x | x | This study | ||||||
| Amphicteis gunneri (Sars, 1835) | 20–59 | x | x | Literature | |||||||
| Amphicteis midas (Gosse, 1855) | NR | 30–37 | x | This study | |||||||
| Auchenoplax worsfoldi Jirkov & Leontovich, 2013 | NR | 141 | x | This study | |||||||
| Amphinomidae | |||||||||||
| Chloeia venusta Quatrefages, 1866 | 100–145 | x | Literature | ||||||||
| Euphrosine armadillo Sars, 1851 | 100–145 | x | Literature | ||||||||
| Euphrosine foliosa Audouin & Milne-Edwards, 1833 | 0–70 | x | x | x | x | Literature; This study | |||||
| Eurythoe complanata (Pallas, 1766) | SC; *NIS | - | Literature | ||||||||
| Hermodice carunculata (Pallas, 1766) | 0–69 | x | x | x | x | x | x | Literature; This study | |||
| Linopherus canariensis Langerhans, 1881 | NIS | 0–337 | x | x | x | x | Literature | ||||
| Aphroditidae | |||||||||||
| Aphrodita aculeata Linnaeus, 1758 | 20 | x | Literature | ||||||||
| Aphrodita perarmata Roule, 1898 | NR | 19 | x | x | This study | ||||||
| Laetmonice filicornis Kinberg, 1866 | 155–187 | x | Literature | ||||||||
| Laetmonice hystrix (Savigny in Lamarck, 1818) | 10–150 | x | x | x | Literature; This study | ||||||
| Pontogenia chrysocoma (Baird, 1865) | 0–47 | x | x | x | x | x | Literature; This study | ||||
| Arenicolidae | x | ||||||||||
| Branchiomaldane vincentii Langerhans, 1881 | NR | 32 | x | x | This study | ||||||
| Capitellidae | |||||||||||
| Capitella capitata (Fabricius, 1780) | SC | 0–600 | x | x | x | x | Literature; This study | ||||
| Capitella minima Langerhans, 1880 | 3–14 | x | Literature; This study | ||||||||
| Capitellethus dispar (Ehlers, 1907) | NR; *NIS | 25–33 | x | x | This study | ||||||
| Dasybranchus caducus (Grube, 1846) | 0–42 | x | x | x | Literature; This study | ||||||
| Dasybranchus gajolae Eisig, 1887 | 0–15 | x | x | Literature | |||||||
| Heteromastus filiformis (Claparède, 1864) | NR | 12–45 | x | This study | |||||||
| Leiocapitella dollfusi (Fauvel, 1936) | NR | 25–27 | x | x | This study | ||||||
| Mastobranchus trinchesii Eisig, 1887 | NR | 12 | x | This study | |||||||
| Mediomastus capensis Day, 1961 | *NIS | 0–42 | x | x | x | Literature; This study | |||||
| Mediomastus fragilis Rasmussen, 1973 | NR | 37–42 | x | This study | |||||||
| Neopseudocapitella brasiliensis Rullier & Amoureux, 1979 | *NIS | 15–42 | x | x | Literature; This study | ||||||
| Notomastus aberans Day, 1957 | NIS | 0–150 | x | x | x | x | Literature; This study | ||||
| Notomastus formianus Eisig, 1887 | NR | 5–42 | x | x | This study | ||||||
| Notomastus latericeus Sars, 1851 | 0–300 | x | x | x | x | Literature; This study | |||||
| Notomastus lineatus Claparède, 1869 | 0–120 | x | x | x | Literature; This study | ||||||
| Notomastus mossambicus (Thomassin, 1970) | NIS | 38–70 | x | x | x | Literature; This study | |||||
| Notomastus profundus Eisig, 1887 | 37–600 | x | Literature; This study | ||||||||
| Peresiella clymenoides Harmelin, 1968 | NR | 25–40 | x | This study | |||||||
| Pseudocapitella incerta Fauvel, 1913 | 20 | x | Literature | ||||||||
| Pseudoleiocapitella fauveli Harmelin, 1964 | 8–150 | x | x | Literature; This study | |||||||
| Chrysopetalidae | |||||||||||
| Arichlidon reyssi (Katzmann, Laubier & Ramos, 1974) | 0–337 | x | x | x | x | Literature; This study | |||||
| Chrysopetalum debile (Grube, 1855) | 0–131 | x | x | x | x | x | Literature; This study | ||||
| Paleanotus chrysolepis Schmarda, 1861 | NR | 17–33 | x | x | This study | ||||||
| Cirratulidae | |||||||||||
| Aphelochaeta filiformis (Keferstein, 1862) | NR | 4–59 | x | x | This study | ||||||
| Aphelochaeta marioni (Saint-Joseph, 1894) | NR | 17–40 | x | x | This study | ||||||
| Caulleriella bioculata Keferstein, 1862 | 0–45 | x | x | x | x | Literature; This study | |||||
| Caulleriella mediterranea Lezzi, 2017 | 0–210 | x | x | x | x | Literature; This study | |||||
| Chaetozone caputesocis (Saint-Joseph, 1894) | NR | 25–30 | x | This study | |||||||
| Chaetozone carpenteri McIntosh, 1911 | NR | 25–42 | x | x | This study | ||||||
| Chaetozone corona Berkeley & Berkeley, 1941 | NR; NIS | 8–42 | x | This study | |||||||
| Chaetozone gibber Woodham & Chambers, 1994 | NR | 16–42 | x | x | This study | ||||||
| Chaetozone setosa Malmgren, 1867 | SC | 20–44 | x | x | Literature; This study | ||||||
| Chaetozone zetlandica McIntosh, 1911 | NR | 38–47 | x | This study | |||||||
| Cirriformia tentaculata (Montagu, 1808) | 0–50 | x | x | x | Literature; This study | ||||||
| Dodecaceria concharum Örsted, 1843 | NR | 16–30 | x | This study | |||||||
| Fauvelicirratulus dollfusi (Fauvel, 1928) | NR | 30 | x | This study | |||||||
| Kirkegaardia dorsobranchialis (Kirkegaard, 1959) | 22–300 | x | Literature; This study | ||||||||
| Kirkegaardia heterochaeta (Laubier, 1961) | 9–300 | x | x | Literature; This study | |||||||
| Kirkegaardia marypetersenae (Lezzi, Çinar & Giangrande, 2016) | NR | 25–30 | x | This study | |||||||
| Protocirrineris chrysoderma (Claparède, 1868) | NR | 25 | x | This study | |||||||
| Protocirrineris purgamentorum Lezzi, Çinar & Giangrande, 2016 | NR | 37–42 | x | This study | |||||||
| Timarete filigera (Delle Chiaje, 1828) | 37–70 | x | Literature; This study | ||||||||
| Cossuridae | |||||||||||
| Cossura coasta Kitamori, 1960 | *NIS | 20 | x | Literature | |||||||
| Cossura soyeri Laubier, 1964 | 32–69 | x | x | Literature; This study | |||||||
| Dorvilleidae | |||||||||||
| Dorvillea rubrovittata (Grube, 1855) | 0–300 | x | x | x | x | x | Literature; This study | ||||
| Dorvillea similis (Crossland, 1924) | NR; NIS | 27–49 | x | This study | |||||||
| Ophryotrocha adherens Paavo, Bailey-Brock & Åkesson, 2000 | - | Literature | |||||||||
| Parougia caeca (Webster & Benedict, 1884) | NR | 6–59 | x | x | This study | ||||||
| Pettiboneia urciensis Campoy & San Martín, 1980 | 0–33 | x | x | x | x | Literature; This study | |||||
| Protodorvillea artemidis Munari & Ebbe, 2019 | NR | 42–45 | x | This study | |||||||
| Protodorvillea kefersteini (McIntosh, 1869) | 0–210 | x | x | x | x | Literature; This study | |||||
| Schistomeringos loveni (Kinberg, 1865) | NR; NIS | 37–42 | x | This study | |||||||
| Schistomeringos neglecta (Fauvel, 1923) | 5–145 | x | x | x | Literature; This study | ||||||
| Schistomeringos rudolphi (Delle Chiaje, 1828) | 0–59 | x | x | x | x | Literature; This study | |||||
| Eunicidae | |||||||||||
| Eunice floridana (Pourtalés, 1867) | *NIS | 20–50 | x | Literature | |||||||
| Eunice pennata (O. F. Müller, 1776) | 5–114 | x | x | Literature; This study | |||||||
| Eunice vittata (Delle Chiaje, 1828) | SC | 0–300 | x | x | x | x | Literature; This study | ||||
| Leodice torquata (Quatrefages, 1866) | 32 | x | x | Literature | |||||||
| Lysidice collaris Grube, 1868 | NIS | 0–46 | x | x | x | x | x | Literature; This study | |||
| Lysidice margaritacea Claparède, 1868 | NR | 17–40 | x | x | This study | ||||||
| Lysidice ninetta Audouin & Milne Edwards, 1833 | SC | 0–300 | x | x | x | x | x | Literature; This study | |||
| Lysidice unicornis (Grube, 1840) | 0–300 | x | x | x | x | x | Literature | ||||
| Marphysasanguinea (Montagu, 1813) | SC | 5–100 | x | x | x | Literature; This study | |||||
| Palola siciliensis (Grube, 1840) | 0–46 | x | x | x | x | Literature | |||||
| Palola valida (Gravier, 1900) | NR; NIS | 32–47 | x | This study | |||||||
| Paucibranchia adenensis (Gravier, 1900) | NR; *NIS | 25–141 | x | This study | |||||||
| Paucibranchia bellii (Audouin & Milne Edwards, 1833) | 2–210 | x | x | Literature; This study | |||||||
| Paucibranchia fallax (Marion & Bobretzky, 1875) | 0–300 | x | x | x | x | Literature; This study | |||||
| Fabriciidae | |||||||||||
| Fabricia stellaris (O. F. Müller. 1774) | NR | 30–57 | x | This study | |||||||
| Novafabricia posidoniae Licciano & Giangrande, 2006 | NR | 6–48 | x | x | This study | ||||||
| Pseudofabricia aberrans Cantone, 1972 | 0–30 | x | x | x | x | Literature; This study | |||||
| Fauveliopsidae | |||||||||||
| Fauveliopsis adriatica Katzmann & Laubier, 1974 | NR | 37 | x | This study | |||||||
| Fauveliopsis fauchaldi Katzmann & Laubier, 1974 | 25–140 | x | x | Literature | |||||||
| Flabelligeridae | |||||||||||
| Bradabyssa villosa (Rathke, 1843) | NR | 17–25 | x | x | This study | ||||||
| Diplocirrus glaucus (Malmgren, 1867) | 11–70 | x | x | Literature; This study | |||||||
| Flabelligera affinis Sars, 1829 | 32–145 | x | x | x | x | Literature; This study | |||||
| Flabelligera diplochaitus (Otto, 1820) | - | Literature | |||||||||
| Pherusa plumosa (O. F. Müller, 1776) | 5–50 | x | x | Literature; This study | |||||||
| Piromis eruca (Claparède, 1869) | 17–70 | x | x | Literature; This study | |||||||
| Stylarioides grubei Salazar-Vallejo, 2011 | NR; NIS | 45–59 | x | x | This study | ||||||
| Stylarioides moniliferus Delle Chiaje, 1831 | NR | 2–42 | x | x | This study | ||||||
| Therochaeta flabellata (Sars in Sars, 1872) | 37–210 | x | x | Literature; This study | |||||||
| Glyceridae | |||||||||||
| Glycera alba (O. F. Müller, 1776) | 10–69 | x | x | Literature; This study | |||||||
| Glycera celtica O’Connor, 1987 | NR | 22–27 | x | This study | |||||||
| Glycera fallax Quatrefages, 1850 | 17–324 | x | x | x | Literature; This study | ||||||
| Glycera lapidum Quatrefages, 1866 | 20–300 | x | x | Literature; This study | |||||||
| Glycera oxycephala Ehlers, 1887 | NR | 24–47 | x | This study | |||||||
| Glycera rouxii Audouin & Milne Edwards, 1833 | 5–50 | x | Literature; This study | ||||||||
| Glycera tesselata Grube, 1863 | 0–210 | x | x | x | x | x | Literature; This study | ||||
| Glycera tridactyla Schmarda, 1861 | 2–150 | x | x | Literature; This study | |||||||
| Glycera unicornis Lamarck, 1818 | 5–59 | x | x | Literature; This study | |||||||
| Goniadidae | |||||||||||
| Goniada emerita Audouin & Milne-Edwards, 1833 | 20–120 | x | x | Literature; This study | |||||||
| Goniada maculata Örsted, 1843 | 6–300 | x | x | Literature; This study | |||||||
| Goniada norvegica Örsted, 1845 | 20–60 | x | x | Literature; This study | |||||||
| Goniada vorax (Kinberg, 1866) | NR | 8–38 | x | This study | |||||||
| Hesionidae | |||||||||||
| Gyptis propinqua Marion & Bobretzky, 1875 | 6–32 | x | x | Literature; This study | |||||||
| Hesione pantherina Risso, 1826 | 0–20 | x | x | Literature | |||||||
| Hesiospina aurantiaca (Sars, 1842) | 12–210 | x | x | Literature; This study | |||||||
| Leocrates claparedii (Costa in Claparède, 1868) | NR | 33 | x | This study | |||||||
| Oxydromus flexuosus (Delle Chiaje, 1827) | 0–55 | x | x | x | Literature; This study | ||||||
| Oxydromus pallidus Claparède, 1864 | 11–48 | x | x | Literature; This study | |||||||
| Podarkeopsis capensis (Day, 1963) | NR; *NIS | 17–45 | x | x | This study | ||||||
| Psamathe fusca Johnston, 1836 | 0–210 | x | x | x | x | x | Literature; This study | ||||
| Syllidia armata Quatrefages, 1866 | 0–131 | x | x | x | x | Literature; This study | |||||
| Lacydoniidae | |||||||||||
| Lacydonia miranda Marion, 1874 | 19–45 | x | x | Literature; This study | |||||||
| Lumbrineridae | |||||||||||
| Abyssoninoe bidentata D’Alessandro, Cosentino, Giacobbe, Andaloro & Romeo, 2014 | NR | 6–59 | x | x | This study | ||||||
| Abyssoninoe hibernica (McIntosh, 1903) | NR | 6–58 | x | x | This study | ||||||
| Augeneria profundicola Kurt-Şahin, Çinar & Gönülal, 2016 | NR | 310–313 | x | This study | |||||||
| Gallardoneris nonatoi (Ramos, 1976) | 3–59 | x | x | x | Literature; This study | ||||||
| Lumbricalus adriatica (Fauvel, 1940) | NR | 12–58 | x | x | This study | ||||||
| Lumbrinerides amoureuxi Miura, 1981 | 3–120 | x | x | Literature; This study | |||||||
| Lumbrineriopsis paradoxa (Saint-Joseph, 1888) | 16–58 | x | x | x | Literature; This study | ||||||
| Lumbrineris coccinea (Renier, 1804) | 0–120 | x | x | x | x | Literature; This study | |||||
| Lumbrineris geldiayi Carrera-Parra, Çinar & Dağli, 2011 | NR | 25–49 | x | x | This study | ||||||
| Lumbrineris gracilis (Ehlers, 1868) | 0–210 | x | x | x | x | Literature; This study | |||||
| Lumbrineris latreilli Audouin & Milne-Edwards, 1833 | 0–210 | x | x | x | x | x | Literature; This study | ||||
| Lumbrineris luciliae Martins, Carrera-Parra, Quintino & Rodrigues, 2012 | NR | 19–39 | x | This study | |||||||
| Lumbrineris lusitanica Martins, Carrera-Parra, Quintino & Rodrigues, 2012 | NR | 5–59 | x | x | This study | ||||||
| Lumbrineris pinaster Martins, Carrera-Parra, Quintino & Rodrigues, 2012 | NR | 5–59 | x | x | This study | ||||||
| Scoletoma funchalensis (Kinberg, 1865) | - | Literature | |||||||||
| Scoletoma laurentiana (Grube, 1863) | 0–150 | x | x | x | x | Literature; This study | |||||
| Magelonidae | |||||||||||
| Magelona alleni Wilson, 1958 | 8–50 | x | Literature; This study | ||||||||
| Magelona equilamellae Harmelin, 1964 | 14–20 | x | Literature; This study | ||||||||
| Magelona filiformis Wilson, 1959 | NR | 5–55 | x | x | This study | ||||||
| Magelona minuta Eliason, 1962 | 10–120 | x | x | Literature; This study | |||||||
| Magelona mirabilis (Johnston, 1865) | NR | 1 | x | This study | |||||||
| Magelona wilsoni Glémarec, 1966 | NR | 4–55 | x | This study | |||||||
| Maldanidae | |||||||||||
| Axiothella constricta (Claparède, 1868) | NR | 12 | x | This study | |||||||
| Chirimia biceps biceps (Sars, 1861) | 35–69 | x | x | Literature; This study | |||||||
| Euclymene collaris (Claparède, 1869) | 17–40 | x | x | Literature; This study | |||||||
| Euclymene lombricoides (Quatrefages, 1866) | 10–150 | x | x | Literature; This study | |||||||
| Euclymene oerstedii (Claparède, 1863) | 13–50 | x | x | Literature | |||||||
| Euclymene palermitana (Grube, 1840) | NR | 25 | x | This study | |||||||
| Leiochone leiopygos (Grube, 1860) | 2–42 | x | x | Literature; This study | |||||||
| Leiochone tricirrata (Bellan & Reyss, 1967) | 33–120 | x | x | Literature; This study | |||||||
| Macroclymene santanderensis (Rioja, 1917) | 35–38 | x | x | Literature | |||||||
| Maldane glebifex Grube, 1860 | NR | 25–40 | x | x | This study | ||||||
| Maldane sarsi Malmgren, 1865 | 25–120 | x | x | Literature; This study | |||||||
| Metasychis gotoi (Izuka, 1902) | *NIS | 92–120 | x | Literature | |||||||
| Micromaldane ornithochaeta Mesnil, 1897 | NR | 15–32 | x | x | This study | ||||||
| Nicomache lumbricalis (Fabricius, 1780) | 0–69 | x | x | x | x | Literature | |||||
| Petaloproctus terricola Quatrefages, 1866 | 0–120 | x | x | x | Literature; This study | ||||||
| Praxillella affinis (Sars in Sars, 1872) | NR | 30–37 | x | x | This study | ||||||
| Praxillella gracilis (Sars, 1861) | 8–69 | x | x | Literature; This study | |||||||
| Praxillella lophoseta (Orlandi, 1898) | 37–70 | x | x | Literature; This study | |||||||
| Praxillella praetermissa (Malmgren, 1865) | 25–150 | x | x | x | x | Literature; This study | |||||
| Rhodine gracilior Tauber, 1879 | NR | 37 | x | This study | |||||||
| Rhodine loveni Malmgren, 1866 | 10–70 | x | x | x | Literature; This study | ||||||
| Melinnidae | |||||||||||
| Melinna monoceroides Fauvel, 1936 | NR | 22–55 | x | x | This study | ||||||
| Melinna palmata (Grube, 1860) | 5–120 | x | x | x | x | Literature; This study | |||||
| Microphthalmidae | |||||||||||
| Microphthalmus similis Bobretzky, 1870 | 0–15 | x | x | x | Literature; This study | ||||||
| Nephtyidae | |||||||||||
| Aglaophamus agilis (Langerhans, 1880) | 47–120 | x | Literature; This study | ||||||||
| Inermonephtys inermis (Ehlers, 1887) | 20–92 | x | x | Literature | |||||||
| Micronephthys longicornis (Perejaslavtseva, 1891) | 6–120 | x | x | x | x | Literature; This study | |||||
| Micronephthys sphaerocirrata Wesenberg-Lund, 1949 | NR | 1 | x | This study | |||||||
| Nephtys assimilis Örsted, 1843 | NR | 7–12 | x | x | This study | ||||||
| Nephtys caeca (Fabricius, 1780) | 5–50 | x | Literature | ||||||||
| Nephtys ciliata (O. F. Müller, 1788) | 100 | Literature | |||||||||
| Nephtys hombergii (Savigny in Lamarck, 1818) | 2–150 | x | x | Literature; This study | |||||||
| Nephtys hystricis McIntosh, 1900 | 20 | x | Literature | ||||||||
| Nephtys incisa Malmgren, 1865 | 13–120 | x | x | Literature; This study | |||||||
| Nephtys paradoxa Malm, 1874 | 140 | x | Literature | ||||||||
| Nereididae | |||||||||||
| Alitta virens (Sars, 1835) | NR; NIS | 28–58 | x | x | This study | ||||||
| Ceratonereis mirabilis Kinberg, 1865 | NIS | 20–210 | x | x | x | Literature; This study | |||||
| Composetia costae (Grube, 1840) | 0–145 | x | x | x | x | x | Literature; This study | ||||
| Composetia hircinicola (Eisig, 1869) | 27–210 | x | x | x | x | Literature | |||||
| Eunereis longissima (Johnston, 1840) | NR | 19–42 | x | x | This study | ||||||
| Hediste diversicolor (O. F. Müller, 1776) | SC | 45–116 | x | Literature | |||||||
| Leonnates aylaoberi Çinar & Dağli, 2013 | NR; NIS | 25–42 | x | x | This study | ||||||
| Namanereis littoralis (Grube, 1872) | NR | 25 | x | This study | |||||||
| Neanthes acuminata (Ehlers, 1868) | 0–150 | x | x | x | x | Literature; This study | |||||
| Neanthes fucata (Savigny, 1822) | 10 | Literature | |||||||||
| Neanthes kerguelensis (McIntosh, 1885) | 31–337 | x | x | Literature; This study | |||||||
| Neanthes nubila (Savigny, 1822) | 19–92 | x | x | x | x | Literature; This study | |||||
| Neanthes rubicunda (Ehlers, 1868) | NR | 5–50 | x | x | This study | ||||||
| Nereis agulhana Day, 1963 | NR; *NIS | 8–47 | x | x | This study | ||||||
| Nereis pelagica Linnaeus, 1758 | SC | 0–150 | x | x | x | x | x | Literature | |||
| Nereis rava Ehlers, 1868 | 0–210 | x | x | x | x | Literature; This study | |||||
| Nereis zonata Malmgren, 1867 | SC | 0–100 | x | x | x | x | Literature | ||||
| Perinereis cultrifera (Grube, 1840) | SC | 0–35 | x | x | x | x | Literature; This study | ||||
| Platynereis coccinea (Delle Chiaje, 1822) | 0–15 | x | x | Literature | |||||||
| Platynereis dumerilii (Audouin & Milne Edwards, 1833) | SC | 0–92 | x | x | x | x | x | Literature; This study | |||
| Platynereis nadiae Abbiati & Castelli, 1992 | NR | 12 | x | This study | |||||||
| Pseudonereis anomala Gravier, 1900 | NIS | 0–15 | x | x | Literature; This study | ||||||
| Rullierinereis anoculata Cantone, 1982 | 92 | x | x | Literature | |||||||
| Websterinereis glauca (Claparède, 1870) | 27–45 | x | x | Literature; This study | |||||||
| Oenonidae | |||||||||||
| Arabella geniculata (Claparède, 1868) | 20–70 | x | x | Literature | |||||||
| Arabella iricolor (Montagu, 1804) | 0–120 | x | x | x | x | x | Literature; This study | ||||
| Drilonereis filum (Claparède, 1868) | 0–120 | x | x | x | Literature; This study | ||||||
| Notocirrus scoticus McIntosh, 1879 | 12–42 | x | x | Literature; This study | |||||||
| Oenone fulgida (Lamarck, 1818) | *NIS | 62–70 | x | x | Literature | ||||||
| Onuphidae | |||||||||||
| Aponuphis bilineata (Baird, 1870) | SC | 6–300 | x | x | Literature; This study | ||||||
| Aponuphis brementi (Fauvel, 1916) | SC | 7–120 | x | x | Literature; This study | ||||||
| Aponuphis ornata (Fauvel, 1928) | NR | 5–22 | x | x | This study | ||||||
| Aponuphis rigida (Claparède, 1868) | NR | 38 | x | This study | |||||||
| Diopatra neapolitana Delle Chiaje, 1841 | SC | 2–22 | x | x | Literature; This study | ||||||
| Hyalinoecia tubicola (O. F. Müller, 1776) | 10–150 | x | Literature | ||||||||
| Nothria conchylega (Sars, 1835) | 210 | x | Literature | ||||||||
| Onuphis eremita Audouin & Milne-Edwards, 1833 | SC | 2–150 | x | x | Literature; This study | ||||||
| Paradiopatra calliopae Arvanitidis & Koukouras, 1997 | 25–600 | x | x | Literature; This study | |||||||
| Paradiopatra quadricuspis (Sars in Sars, 1872) | 0–15 | x | x | Literature | |||||||
| Opheliidae | |||||||||||
| Armandia cirrhosa De Filippi, 1861 | 0–51 | x | x | x | Literature; This study | ||||||
| Armandia polyophthalma Kükenthal, 1887 | NR | 3–47 | x | This study | |||||||
| Ophelina abranchiata Støp-Bowitz, 1948 | NR | 141 | x | This study | |||||||
| Ophelina cylindricaudata (Hansen, 1879) | 0–210 | x | x | x | Literature | ||||||
| Polyophthalmus pictus (Dujardin, 1839) | 0–55 | x | x | x | x | Literature; This study | |||||
| Tachytrypane jeffreysi McIntosh in Jeffreys, 1876 | 25–210 | x | x | Literature; This study | |||||||
| Orbiniidae | |||||||||||
| Leodamas chevalieri candiensis (Harmelin, 1969) | 27–45 | x | x | Literature; This study | |||||||
| Naineris laevigata (Grube, 1855) | 0–210 | x | x | x | x | Literature; This study | |||||
| Naineris quadraticeps Day, 1965 | NR; NIS | 38 | x | This study | |||||||
| Naineris setosa Verrill, 1900 | NR; NIS | 27–50 | x | This study | |||||||
| Orbinia sertulata (Savigny, 1822) | 2–10 | x | Literature; This study | ||||||||
| Phylo foetida (Claparède, 1868) | 4–46 | x | Literature; This study | ||||||||
| Protoaricia oerstedii (Claparède, 1864) | NR | 4–39 | x | x | This study | ||||||
| Scoloplos armiger (O. F. Müller, 1776) | 0–210 | x | x | x | Literature; This study | ||||||
| Scoloplos haasi (Monro, 1937) | NR | 0.5–50 | x | x | This study | ||||||
| Scoloplos typicus (Eisig, 1914) | 2–51 | x | x | Literature; This study | |||||||
| Oweniidae | |||||||||||
| Galathowenia oculata (Zachs, 1923) | NR | 37–40 | x | This study | |||||||
| Owenia fusiformis Delle Chiaje, 1844 | 4–50 | x | x | Literature; This study | |||||||
| Paralacydoniidae | |||||||||||
| Paralacydonia paradoxa Fauvel, 1913 | 30–35 | x | x | Literature; This study | |||||||
| Paraonidae | |||||||||||
| Aricidea aberrans Laubier & Ramos, 1974 | 600 | x | Literature | ||||||||
| Aricidea annae Laubier, 1967 | 92 | x | Literature | ||||||||
| Aricidea assimilis Tebble, 1959 | SC | 0–141 | x | x | x | Literature; This study | |||||
| Aricidea bansei Laubier & Ramos, 1974 | NR | 5–58 | x | x | This study | ||||||
| Aricidea catherinae Laubier, 1967 | SC | 0–58 | x | x | x | Literature; This study | |||||
| Aricidea cerrutii Laubier, 1966 | 0–58 | x | x | x | Literature; This study | ||||||
| Aricidea claudiae Laubier, 1967 | 2–69 | x | x | x | Literature; This study | ||||||
| Aricidea fragilis Webster, 1879 | NR; NIS | 22–56 | x | This study | |||||||
| Aricidea jeaneteae Langeneck, Busoni, Aliani & Castelli, 2017 | 600 | x | Literature | ||||||||
| Aricidea katzmanni Erdoğan-Dereli & Çinar, 2020 | NR | 17 | x | x | This study | ||||||
| Aricidea monicae Laubier, 1967 | 92–141 | x | Literature; This study | ||||||||
| Aricidea pseudoarticulata Hobson, 1972 | 4–600 | x | x | Literature; This study | |||||||
| Aricidea simonae Laubier & Ramos, 1974 | 69–210 | x | Literature | ||||||||
| Cirrophorus branchiatus Ehlers, 1908 | 0.5–300 | x | x | Literature; This study | |||||||
| Cirrophorus nikebianchii Langeneck, Barbieri, Maltagliati & Castelli, 2017 | 17–57 | x | x | Literature; This study | |||||||
| Cirrophorus turcicus Erdoğan-Dereli, Çinar & Dağli, 2017 | NR | 25–40 | x | This study | |||||||
| Levinsenia demiri Çinar, Dağli & Açik, 2011 | NR | 30–141 | x | This study | |||||||
| Levinsenia gracilis (Tauber, 1879) | 7–600 | x | x | x | Literature; This study | ||||||
| Levinsenia kosswigi Çinar, Dağli & Açik, 2011 | NR | 33 | x | This study | |||||||
| Levinsenia materi Çinar & Dağli, 2013 | NR | 25–42 | x | x | This study | ||||||
| Levinsenia vulgaris Erdoğan-Dereli & Çinar, 2021 | NR | 42 | x | This study | |||||||
| Paradoneis armata Glémarec, 1966 | 0–59 | x | x | x | Literature; This study | ||||||
| Paradoneis heterochaeta Erdoğan-Dereli & Çinar, 2019 | NR | 25 | x | This study | |||||||
| Paradoneis ilvana Castelli, 1985 | NR | 6–31 | x | x | This study | ||||||
| Paradoneis lyra (Southern, 1914) | 0–600 | x | x | x | Literature; This study | ||||||
| Paraonella myriamae (Katzmann & Laubier, 1975) | 210 | x | Literature | ||||||||
| Paraonides neapolitana Cerruti, 1909 | 69–210 | x | Literature | ||||||||
| Paraonis paucibranchiata Cerruti, 1909 | 300 | x | Literature | ||||||||
| Pectinariidae | |||||||||||
| Amphictene auricoma (O. F. Müller, 1776) | 10–60 | x | x | x | Literature; This study | ||||||
| Lagis koreni Malmgren, 1866 | NR | 12–50 | x | x | This study | ||||||
| Pectinaria belgica (Pallas, 1766) | - | Literature | |||||||||
| Petta pusilla Malmgren, 1866 | NR | 38–40 | x | This study | |||||||
| Phyllodocidae | |||||||||||
| Eteone flava (Fabricius, 1780) | NR | 38 | x | This study | |||||||
| Eteone longa (Fabricius, 1780) | 5 | Literature | |||||||||
| Eulalia clavigera (Audouin & Milne Edwards, 1833) | 0–48 | x | x | x | x | Literature; This study | |||||
| Eulalia expusilla Pleijel, 1987 | - | Literature | |||||||||
| Eulalia mustela Pleijel, 1987 | 35–92 | x | x | x | Literature | ||||||
| Eulalia tripunctata McIntosh, 1874 | 0–37 | x | x | x | Literature; This study | ||||||
| Eumida punctifera (Grube, 1860) | NR | 40 | x | This study | |||||||
| Eumida sanguinea (Örsted, 1843) | 0–92 | x | x | x | Literature; This study | ||||||
| Hesionura coineaui (Laubier, 1962) | 0–15 | x | x | x | x | Literature | |||||
| Hesionura elongata (Southern, 1914) | 3–35 | x | x | Literature; This study | |||||||
| Hypereteone foliosa (Quatrefages, 1866) | NR | 13–40 | x | This study | |||||||
| Mysta barbata Malmgren, 1865 | NR | 14–50 | x | This study | |||||||
| Mysta picta (Quatrefages, 1866) | 0–210 | x | x | x | Literature; This study | ||||||
| Mysta siphodonta (Delle Chiaje, 1830) | 5 | Literature | |||||||||
| Mystides caeca (Langerhans, 1880) | 30–32 | x | x | Literature; This study | |||||||
| Nereiphylla paretti Blainville, 1828 | 0–32 | x | x | x | Literature | ||||||
| Nereiphylla rubiginosa (Saint-Joseph, 1888) | 0–70 | x | x | x | x | Literature | |||||
| Notophyllum foliosum (Sars, 1835) | 0–100 | x | x | x | x | Literature | |||||
| Paranaitis kosteriensis (Malmgren, 1867) | 19–40 | x | x | Literature; This study | |||||||
| Phyllodoce maculata (Linnaeus, 1767) | 38–45 | x | x | Literature | |||||||
| Phyllodoce mucosa Örsted, 1843 | 0–30 | x | x | x | Literature; This study | ||||||
| Protomystides bidentata (Langerhans, 1880) | NR | 6–58 | x | x | This study | ||||||
| Pseudomystides limbata limbata (Saint-Joseph, 1888) | 0–210 | x | x | x | Literature; This study | ||||||
| Pseudomystides limbata nigrolineata (Rioja, 1925) | NR | 19 | x | This study | |||||||
| Pterocirrus macroceros (Grube, 1860) | 0–15 | x | x | x | Literature | ||||||
| Sige fusigera Malmgren, 1865 | 50–100 | x | Literature | ||||||||
| Pilargidae | |||||||||||
| Ancistrosyllis groenlandica McIntosh, 1879 | 600 | x | Literature | ||||||||
| Pilargis verrucosa Saint-Joseph, 1899 | 7–57 | x | x | Literature; This study | |||||||
| Sigambra parva (Day, 1963) | NR; *NIS | 40 | x | This study | |||||||
| Poecilochaetidae | |||||||||||
| Poecilochaetus serpens Allen, 1904 | 210 | x | Literature | ||||||||
| Polynoidae | |||||||||||
| Bylgides groenlandicus (Malmgren, 1867) | 48–60 | x | Literature | ||||||||
| Harmothoe antilopes McIntosh, 1876 | 27–140 | x | Literature; This study | ||||||||
| Harmothoe imbricata (Linnaeus, 1767) | NR | 37 | x | This study | |||||||
| Harmothoe impar (Johnston, 1839) | 0–15 | x | x | x | Literature | ||||||
| Harmothoe spinifera (Ehlers, 1864) | 0–145 | x | x | x | x | Literature; This study | |||||
| Lepidasthenia elegans (Grube, 1840) | 32–100 | x | x | x | x | Literature | |||||
| Lepidonotus clava (Montagu, 1808) | 0–100 | x | x | x | x | Literature; This study | |||||
| Malmgrenia lilianae (Pettibone, 1993) | 35 | x | x | Literature | |||||||
| Malmgrenia ljungmani (Malmgren, 1867) | 35 | x | x | Literature | |||||||
| Malmgrenia lunulata (Delle Chiaje, 1830) | 31–85 | x | x | x | Literature; This study | ||||||
| Subadyte pellucida (Ehlers, 1864) | 17–300 | x | x | x | Literature; This study | ||||||
| Sabellidae | |||||||||||
| Acromegalomma adriaticum (Giangrande, Caruso, Mikac & Licciano, 2015) | NR | 30 | x | This study | |||||||
| Acromegalomma lanigerum (Grube, 1846) | 0–145 | x | x | x | x | Literature; This study | |||||
| Acromegalomma messapicum (Giangrande & Licciano, 2008) | NR | 25 | x | This study | |||||||
| Acromegalomma pseudogesae (Mikac, Giangrande & Licciano, 2013) | NR | 8–33 | x | This study | |||||||
| Amphicorina armandi (Claparède, 1864) | 0–15 | x | x | Literature | |||||||
| Amphiglena mediterranea (Leydig, 1851) | 0–38 | x | x | x | x | Literature | |||||
| Bispira mariae Lo Bianco, 1893 | 35–131 | x | x | Literature | |||||||
| Bispira melanostigma (Schmarda, 1861) | *NIS | - | Literature | ||||||||
| Branchiomma boholense (Grube, 1878) | NIS | 0–15 | x | x | x | Literature | |||||
| Branchiomma bombyx (Dalyell, 1853) | 0–145 | x | x | x | x | x | Literature; This study | ||||
| Branchiomma luctuosum (Grube, 1870) | NIS | 0–30 | x | x | Literature | ||||||
| Branchiomma lucullanum (Delle Chiaje, 1828) | 0–38 | x | x | x | Literature | ||||||
| Branchiomma moebii Knight-Jones, 1994 | 0–15 | x | x | Literature | |||||||
| Chone duneri Malmgren, 1867 | 20–145 | x | x | x | Literature | ||||||
| Claviramus candela (Grube, 1863) | 37–100 | x | x | x | Literature | ||||||
| Dialychone acustica Claparède, 1868 | NR | 12–58 | x | x | This study | ||||||
| Dialychone arenicola (Langerhans, 1880) | NR | 25–40 | x | x | This study | ||||||
| Dialychone collaris (Langerhans, 1880) | 0–92 | x | x | x | x | Literature; This study | |||||
| Dialychone dunerificta (Tovar-Hernández, Licciano & Giangrande, 2007) | NR | 12–45 | x | This study | |||||||
| Dialychone egyptica Selim, Rzhavsky & Britayev, 2012 | NR | 16 | x | This study | |||||||
| Euchone pararosea Giangrande & Licciano, 2006 | NR | 22–51 | x | x | This study | ||||||
| Euchone pseudolimnicola Giangrande & Licciano, 2006 | NR | 12–55 | x | x | This study | ||||||
| Euchone rosea (Langerhans, 1884) | 19–300 | x | x | Literature; This study | |||||||
| Euchone rubrocincta Sars, 1835 | 35 | x | x | Literature | |||||||
| Euchone southerni Banse, 1970 | 27–145 | x | x | x | x | Literature | |||||
| Euratella salmacidis (Claparède, 1869) | 0–42 | x | x | x | Literature; This study | ||||||
| Hypsicomus stichophthalmos (Grube, 1863) | - | Literature | |||||||||
| Jasmineira caudata Langerhans, 1880 | 24–804 | x | Literature; This study | ||||||||
| Jasmineira elegans Saint-Joseph, 1894 | 3–38 | x | x | Literature; This study | |||||||
| Myxicola infundibulum (Montagu, 1808) | SC | 38–70 | x | x | Literature; This study | ||||||
| Paradialychone filicaudata (Southern, 1914) | 3–300 | x | x | x | x | Literature; This study | |||||
| Parasabella langerhansi (Knight-Jones, 1993) | 28–227 | x | x | Literature | |||||||
| Parasabella saxicola (Grube, 1861) | 37–227 | x | x | x | Literature | ||||||
| Parasabella tenuicollaris (Grube, 1861) | 37–92 | x | x | x | Literature | ||||||
| Perkinsiana rubra (Langerhans, 1880) | 0–145 | x | x | x | Literature | ||||||
| Pseudopotamilla reniformis (Bruguière, 1789) | 0–131 | x | x | x | x | Literature; This study | |||||
| Sabella discifera Grube, 1874 | 62–70 | x | x | Literature | |||||||
| Sabella pavonina Savigny, 1822 | 21–227 | x | x | x | x | x | x | Literature; This study | |||
| Sabella spallanzanii (Gmelin, 1791) | 17–92 | x | x | x | Literature; This study | ||||||
| Saccocirridae | |||||||||||
| Saccocirrus papillocercus Bobretzky, 1872 | NR | 3 | x | This study | |||||||
| Scalibregmatidae | |||||||||||
| Asclerocheilus intermedius (Saint-Joseph, 1894) | 22–48 | x | x | Literature; This study | |||||||
| Polyphysia crassa (Örsted, 1843) | NR | 19–40 | x | x | This study | ||||||
| Scalibregma celticum Mackie, 1991 | 16–50 | x | x | Literature; This study | |||||||
| Scalibregma inflatum Rathke, 1843 | 12–50 | x | x | Literature; This study | |||||||
| Sclerocheilus minutus Grube, 1863 | 0–145 | x | x | x | x | Literature; This study | |||||
| Serpulidae | |||||||||||
| Apomatus similis Marion & Bobretzky, 1875 | 0–180 | x | x | x | x | x | x | Literature | |||
| Ditrupa arietina (O. F. Müller, 1776) | 32–180 | x | x | x | Literature; This study | ||||||
| Filograna implexa Berkeley, 1835 | NR | - | x | This study | |||||||
| Filogranula stellata (Southward, 1963) | 779–804 | x | Literature | ||||||||
| Hyalopomatus variorugosus Ben-Eliahu & Fiege, 1996 | 779–804 | x | Literature | ||||||||
| Hydroides dianthus (Verrill, 1873) | SC; *NIS | - | x | Literature | |||||||
| Hydroides dirampha (Mörch, 1863) | SC | - | x | Literature | |||||||
| Hydroides elegans (Haswell, 1883) | SC; NIS | 0–180 | x | Literature | |||||||
| Hydroides helmata (Iroso, 1921) | 0–35 | x | x | x | x | Literature | |||||
| Hydroides heterocera (Grube, 1868) | NIS | - | x | Literature | |||||||
| Hydroides homoceros Pixell, 1913 | NIS | - | x | Literature | |||||||
| Hydroides nigra Zibrowius, 1971 | 0–100 | x | x | x | x | x | Literature | ||||
| Hydroides norvegica Gunnerus, 1768 | 20–227 | x | x | x | x | Literature | |||||
| Hydroides pseudouncinata Zibrowius, 1968 | 0–180 | x | x | x | Literature | ||||||
| Hydroides stoichadon Zibrowius, 1971 | 0–180 | x | x | x | x | Literature; This study | |||||
| Janita fimbriata (Delle Chiaje, 1822) | 9–600 | x | x | x | x | Literature | |||||
| Janua heterostropha (Montagu, 1803) | 0–15 | x | x | x | Literature | ||||||
| Josephella marenzelleri Caullery & Mesnil, 1896 | 0–180 | x | x | x | x | Literature | |||||
| Metavermilia multicristata (Philippi, 1844) | 9–804 | x | x | x | x | Literature | |||||
| Neodexiospira pseudocorrugata (Bush, 1905) | 0–70 | x | x | x | x | Literature | |||||
| Nidificaria clavus (Harris, 1968) | 27–45 | x | x | Literature | |||||||
| Pileolaria heteropoma (Zibrowius, 1968) | 9 | x | Literature | ||||||||
| Pileolaria militaris Claparède, 1870 | 0–15 | x | x | x | Literature | ||||||
| Placostegus crystallinus (Scacchi, 1836) | 0–180 | x | x | x | Literature | ||||||
| Placostegus tridentatus (Fabricius, 1779) | 55–337 | x | x | x | Literature | ||||||
| Protula intestinum (Lamarck, 1818) | 0–100 | x | x | x | x | x | Literature; This study | ||||
| Protula tubularia (Montagu, 1803) | 20–187 | x | Literature | ||||||||
| Rhodopsis pusilla Bush, 1905 | NIS | 0–100 | x | x | Literature | ||||||
| Semivermilia agglutinata (Marenzeller, 1893) | 779–804 | x | Literature | ||||||||
| Semivermilia crenata (O.G. Costa, 1861) | 0–180 | x | x | x | x | Literature | |||||
| Semivermilia cribrata (O.G. Costa, 1861) | 0–100 | x | x | Literature | |||||||
| Semivermilia pomatostegoides (Zibrowius, 1969) | - | Literature | |||||||||
| Serpula cavernicola Fassari & Mollica, 1991 | 0–100 | x | Literature | ||||||||
| Serpula concharum Langerhans, 1880 | 0–328 | x | x | x | x | x | x | Literature; This study | |||
| Serpula vermicularis Linnaeus, 1767 | 0–328 | x | x | x | x | x | x | x | Literature | ||
| Spiraserpula massiliensis (Zibrowius, 1968) | 0–100 | x | x | x | Literature | ||||||
| Spirobranchus lamarcki (Quatrefages, 1866) | 0–100 | x | x | x | Literature; This study | ||||||
| Spirobranchus polytrema (Philippi, 1844) | 0–180 | x | x | x | x | x | x | Literature | |||
| Spirobranchus tetraceros (Schmarda, 1861) | NIS | - | Literature; This study | ||||||||
| Spirobranchus triqueter (Linnaeus, 1758) | 0–100 | x | x | x | x | Literature | |||||
| Vermiliopsis infundibulum (Philippi, 1844) | 0–328 | x | x | x | x | x | x | x | Literature; This study | ||
| Vermiliopsis labiata (O. G. Costa, 1861) | 0–145 | x | x | x | x | x | Literature | ||||
| Vermiliopsis striaticeps (Grube, 1862) | 0–180 | x | x | x | x | x | x | Literature; This study | |||
| Vinearia koehleri (Caullery & Mesnil, 1897) | 9–145 | x | x | Literature | |||||||
| Sigalionidae | |||||||||||
| Claparedepelogenia inclusa (Claparède, 1868) | 32–92 | x | x | x | Literature; This study | ||||||
| Euthalenessa oculata (Peters, 1854) | 38–45 | x | x | Literature | |||||||
| Fimbriosthenelais longipinnis (Grube, 1870) | NIS | 30–141 | x | Literature; This study | |||||||
| Fimbriosthenelais minor (Pruvot & Racovitza, 1895) | 37–70 | x | x | Literature | |||||||
| Fimbriosthenelais zetlandica (McIntosh, 1879) | 35 | x | x | Literature | |||||||
| Pelogenia arenosa (Delle Chiaje, 1830) | 0–15 | x | x | Literature | |||||||
| Pholoe inornata Johnston, 1839 | 0–46 | x | x | x | x | x | Literature; This study | ||||
| Pholoe minuta (Fabricius, 1780) | 2–20 | x | x | x | Literature | ||||||
| Pholoides dorsipapillatus (Marenzeller, 1893) | 155–337 | x | x | Literature | |||||||
| Pisione remota (Southern, 1914) | NR | 40 | x | This study | |||||||
| Sigalion mathildae Audouin & Milne-Edwards, 1832 | 0–300 | x | x | x | Literature; This study | ||||||
| Sthenelais boa (Johnston, 1833) | 0–70 | x | x | x | Literature; This study | ||||||
| Sthenelais limicola (Ehlers, 1864) | NR | 32 | x | x | This study | ||||||
| Sphaerodoridae | |||||||||||
| Sphaerodoridium claparedii (Greeff, 1866) | NR | 46–47 | x | This study | |||||||
| Sphaerodoridium minutum (Webster & Benedict, 1887) | 19–100 | x | x | x | Literature; This study | ||||||
| Sphaerodorum abyssorum Hansen, 1878 | NR | 30 | x | This study | |||||||
| Sphaerodorum gracile (Rathke, 1843) | NR | 44 | x | This study | |||||||
| Spionidae | |||||||||||
| Aonidella dayi López-Jamar, 1989 | 210 | x | Literature | ||||||||
| Aonides oxycephala (Sars, 1862) | 0–40 | x | x | x | Literature; This study | ||||||
| Aonides paucibranchiata Southern, 1914 | NR | 19 | x | This study | |||||||
| Aurospio banyulensis (Laubier, 1966) | 210 | x | Literature | ||||||||
| Dipolydora armata (Langerhans, 1880) | 5–10 | x | x | Literature | |||||||
| Dispio uncinata Hartman, 1951 | NR | 4–5 | x | This study | |||||||
| Laonice mediterranea Sikorski, Rousou & Nygren, 2021 | 12–337 | x | x | x | x | Literature; This study | |||||
| Laubieriellus salzi (Laubier, 1970) | 27–92 | x | x | x | x | Literature | |||||
| Malacoceros fuliginosus (Claparède, 1868) | 2–20 | x | x | Literature; This study | |||||||
| Paraprionospio pinnata (Ehlers, 1901) | 140 | x | Literature | ||||||||
| Polydora ciliata (Johnston, 1838) | NR | 38–50 | x | This study | |||||||
| Prionospio caspersi Laubier, 1962 | NR | 16–30 | x | x | This study | ||||||
| Prionospio cirrifera Wirén, 1883 | 17–40 | x | x | Literature; This study | |||||||
| Prionospio depauperata Imajima, 1990 | NR; NIS | 14–37 | x | x | This study | ||||||
| Prionospio dubia Day, 1961 | NIS | 25–600 | x | x | Literature | ||||||
| Prionospio ehlersi Fauvel, 1928 | 22–140 | x | x | Literature | |||||||
| Prionospio maciolekae Dağli & Çinar, 2013 | NR | 12–33 | x | This study | |||||||
| Prionospio malmgreni Claparède, 1869 | 20 | x | x | Literature | |||||||
| Prionospio multibranchiata Berkeley, 1927 | *NIS | 92 | x | Literature | |||||||
| Prionospio paucipinnulata Blake & Kudenov, 1978 | NR; NIS | 22–50 | x | This study | |||||||
| Prionospio steenstrupi Malmgren, 1867 | 0–37 | x | x | x | Literature; This study | ||||||
| Pseudopolydora pulchra (Carazzi, 1893) | NR | 13–40 | x | This study | |||||||
| Scolelepis (Parascolelepis) tridentata (Southern, 1914) | 24–69 | x | x | Literature; This study | |||||||
| Scolelepis (Scolelepis) bonnieri (Mesnil, 1896) | 69 | x | Literature | ||||||||
| Scolelepis (Scolelepis) cantabra (Rioja, 1918) | 0–300 | x | x | x | x | Literature | |||||
| Spio decorata Bobretzky, 1870 | 0–35 | x | x | x | x | Literature; This study | |||||
| Spio filicornis (O. F. Müller, 1776) | SC | 38–40 | x | Literature; This study | |||||||
| Spiophanes bombyx (Claparède, 1870) | 49–300 | x | Literature; This study | ||||||||
| Spiophanes kroyeri Grube, 1860 | 20 | x | x | Literature | |||||||
| Spiophanes mediterraneus Meißner, 2005 | NR | 141 | x | This study | |||||||
| Spiophanes reyssi Laubier, 1964 | 210 | x | Literature | ||||||||
| Sternaspidae | |||||||||||
| Sternaspis scutata (Ranzani, 1817) | 30–100 | x | Literature; This study | ||||||||
| Syllidae | |||||||||||
| Amblyosyllis spectabilis (Johnston in Baird, 1861) | 0–15 | x | x | Literature | |||||||
| Anoplosyllis edentula Claparède, 1868 | - | Literature | |||||||||
| Branchiosyllis exilis (Gravier, 1900) | 0–15 | x | x | Literature | |||||||
| Brania arminii (Langerhans, 1881) | 32 | x | x | Literature | |||||||
| Brevicirrosyllis weismanni (Langerhans, 1879) | 12–300 | x | x | x | Literature; This study | ||||||
| Dioplosyllis cirrosa Gidholm, 1962 | 35 | x | x | Literature | |||||||
| Epigamia macrophthalma (Marenzeller, 1875) | 33–145 | x | x | x | x | Literature | |||||
| Erinaceusyllis cryptica (Ben-Eliahu, 1977) | 89–90 | x | Literature | ||||||||
| Eurysyllis tuberculata Ehlers, 1864 | 0–150 | x | x | x | x | Literature; This study | |||||
| Eusyllis assimilis Marenzeller, 1875 | 27–150 | x | x | x | x | x | Literature | ||||
| Eusyllis blomstrandi Malmgren, 1867 | 66–150 | x | x | Literature | |||||||
| Eusyllis kupfferi Langerhans, 1879 | NIS | 0–15 | x | x | x | Literature | |||||
| Eusyllis lamelligera Marion & Bobretzky, 1875 | 0–45 | x | x | x | x | Literature | |||||
| Exogone africana Hartmann-Schröder, 1974 | NR; NIS | 25–42 | x | This study | |||||||
| Exogone dispar (Webster, 1879) | 0–40 | x | x | x | x | Literature; This study | |||||
| Exogone naidina Örsted, 1845 | 0–140 | x | x | x | x | Literature; This study | |||||
| Exogone rostrata Naville, 1933 | 0–150 | x | x | x | x | Literature; This study | |||||
| Exogone verugera (Claparède, 1868) | 0–140 | x | x | x | x | Literature; This study | |||||
| Haplosyllis spongicola (Grube, 1855) | 0–300 | x | x | x | x | Literature; This study | |||||
| Myrianida brachycephala (Marenzeller, 1875) | 0–145 | x | x | x | x | Literature | |||||
| Myrianida convoluta (Cognetti, 1953) | 89–145 | x | x | Literature | |||||||
| Myrianida dentalia (Imajima, 1966) | 100–145 | x | Literature | ||||||||
| Myrianida edwarsi (Saint-Joseph, 1887) | 0–70 | x | x | x | x | Literature | |||||
| Myrianida prolifera (O. F. Müller, 1788) | 0–100 | x | x | x | Literature | ||||||
| Myrianida quindecimdentata (Langerhans, 1884) | 0–15 | x | x | Literature | |||||||
| Odontosyllis ctenostoma Claparède, 1868 | 0–42 | x | x | x | x | Literature; This study | |||||
| Odontosyllis fulgurans (Audouin & Milne-Edwards, 1833) | 0–38 | x | x | x | Literature | ||||||
| Odontosyllis gibba Claparède, 1863 | 30–150 | x | x | x | x | Literature; This study | |||||
| Opisthodonta morena Langerhans, 1879 | 28–38 | x | x | Literature | |||||||
| Opisthodonta serratisetosa (López, San Martín & Jiménez, 1997) | 0–300 | x | x | x | Literature | ||||||
| Opisthosyllis brunnea Langerhans, 1889 | NR | 0.5 | x | x | This study | ||||||
| Paraehlersia dionisi (Núñez & San Martín, 1991) | NR | 10–11 | x | This study | |||||||
| Paraehlersia ferrugina (Langerhans, 1881) | 0–210 | x | x | x | Literature; This study | ||||||
| Parapionosyllis brevicirra Day, 1954 | 0–48 | x | x | Literature; This study | |||||||
| Parapionosyllis elegans (Pierantoni, 1903) | 0–35 | x | x | x | Literature | ||||||
| Parapionosyllis minuta (Pierantoni, 1903) | 0–20 | x | x | x | Literature | ||||||
| Parexogone cognettii (Castelli, Badalamenti & Lardicci, 1987) | NR | 30 | x | This study | |||||||
| Parexogone gambiae (Lanera, Sordino & San Martín, 1994) | 12–70 | x | x | Literature; This study | |||||||
| Parexogone hebes (Webster & Benedict, 1884) | NR | 6–10 | x | x | This study | ||||||
| Parexogone meridionalis (Cognetti, 1955) | 35 | x | x | Literature | |||||||
| Parexogone wolfi (San Martín, 1991) | 70 | x | Literature | ||||||||
| Perkinsyllis anophthalma (Capaccioni & San Martín, 1990) | 0–15 | x | x | Literature | |||||||
| Plakosyllis brevipes Hartmann-Schröder, 1956 | 30–92 | x | x | Literature; This study | |||||||
| Proceraea aurantiaca Claparède, 1868 | 0–70 | x | x | x | Literature | ||||||
| Proceraea picta Ehlers, 1864 | 0–35 | x | x | x | Literature; This study | ||||||
| Prosphaerosyllis campoyi (San Martín, Acero, Contonente & Gomez, 1982) | 69 | x | Literature | ||||||||
| Prosphaerosyllis longipapillata (Hartmann-Schröder, 1979) | NIS | 92 | x | Literature | |||||||
| Prosphaerosyllis xarifae (Hartmann-Schröder, 1960) | 0–92 | x | x | x | Literature; This study | ||||||
| Pseudosyllis brevipennis Grube, 1863 | 0–337 | x | x | x | x | Literature | |||||
| Salvatoria clavata (Claparède, 1863) | SC | 0–92 | x | x | x | x | Literature | ||||
| Salvatoria dolichopoda (Marenzeller, 1874) | 33–35 | x | Literature | ||||||||
| Salvatoria euritmica (Sardá, 1984) | 0–92 | x | x | x | Literature | ||||||
| Salvatoria limbata (Claparède, 1868) | 0–15 | x | x | x | Literature | ||||||
| Salvatoria vieitezi (San Martín, 1984) | 0–45 | x | x | x | Literature | ||||||
| Sphaerosyllis austriaca Banse, 1959 | 0–35 | x | x | x | x | Literature | |||||
| Sphaerosyllis boeroi Musco, Çinar & Giangrande, 2005 | 27–150 | x | x | x | x | Literature | |||||
| Sphaerosyllis bulbosa Southern, 1914 | 35–59 | x | x | Literature; This study | |||||||
| Sphaerosyllis climenti Del-Pilar-Ruso & San Martín, 2012 | NR | 45 | x | This study | |||||||
| Sphaerosyllis glandulata Perkins, 1981 | NR | 27–40 | x | This study | |||||||
| Sphaerosyllis hystrix Claparède, 1863 | SC | 0–210 | x | x | x | x | Literature | ||||
| Sphaerosyllis parabulbosa San Martín & López, 2002 | NR | 47 | x | This study | |||||||
| Sphaerosyllis pirifera Claparède, 1868 | 0–150 | x | x | x | x | Literature; This study | |||||
| Sphaerosyllis taylori Perkins, 1981 | 22–35 | x | x | Literature; This study | |||||||
| Sphaerosyllis thomasi San Martín, 1984 | NR | 32 | x | x | This study | ||||||
| Syllides bansei Perkins, 1981 | NR | 19–40 | x | x | This study | ||||||
| Syllides fulvus (Marion & Bobretzky, 1875) | 0–92 | x | x | x | x | Literature | |||||
| Syllis alternata Moore, 1908 | 0–90 | x | x | x | x | Literature; This study | |||||
| Syllis amica Quatrefages, 1866 | 0–25 | x | x | x | Literature; This study | ||||||
| Syllis armillaris (O. F. Müller, 1776) | 0–150 | x | x | x | x | x | Literature | ||||
| Syllis beneliahuae (Campoy, 1982) | 30–36 | x | x | Literature; This study | |||||||
| Syllis bouvieri Gravier, 1900 | 0–131 | x | x | x | Literature | ||||||
| Syllis columbretensis (Campoy, 1982) | 0–45 | x | x | x | x | Literature | |||||
| Syllis corallicola Verrill, 1900 | 0–50 | x | x | x | x | Literature; This study | |||||
| Syllis cruzi Núñez & San Martín, 1991 | 300 | x | Literature | ||||||||
| Syllis ergeni Çinar, 2005 | NR; NIS | 40 | x | This study | |||||||
| Syllis ferrani Alós & San Martín, 1987 | 0–46 | x | x | Literature; This study | |||||||
| Syllis garciai (Campoy, 1982) | 0–150 | x | x | x | x | x | Literature; This study | ||||
| Syllis gerundensis (Alós & Campoy, 1981) | 27–45 | x | x | Literature | |||||||
| Syllis gracilis Grube, 1840 | SC | 0–20 | x | x | x | x | Literature; This study | ||||
| Syllis hyalina Grube, 1863 | 0–145 | x | x | x | x | Literature; This study | |||||
| Syllis hyllebergi (Licher, 1999) | NIS | - | Literature | ||||||||
| Syllis jorgei San Martín & López, 2000 | 0–145 | x | x | x | x | Literature | |||||
| Syllis kabilica Ben-Eliahu, 1977 | NR | 25–59 | x | x | This study | ||||||
| Syllis katzmanni Arvanitidis, 2017 | 600 | x | Literature | ||||||||
| Syllis krohnii Ehlers, 1864 | 1–58 | x | x | x | Literature; This study | ||||||
| Syllis licheri Ravara, San Martín & Moreira, 2004 | NR | 34–50 | x | x | This study | ||||||
| Syllis parapari San Martín & López, 2000 | 7–69 | x | x | Literature; This study | |||||||
| Syllis pontxioi San Martín & López, 2000 | 0–35 | x | x | x | Literature | ||||||
| Syllis prolifera Krohn, 1852 | 0–51 | x | x | x | x | Literature; This study | |||||
| Syllis pulvinata (Langerhans, 1881) | 0–15 | x | x | Literature | |||||||
| Syllis rosea (Langerhans, 1879) | 0–15 | x | x | x | Literature | ||||||
| Syllis torquata Marion & Bobretzky, 1875 | 0–48 | x | x | x | Literature; This study | ||||||
| Syllis truncata cryptica Ben-Eliahu, 1977 | 0–150 | x | x | x | x | x | Literature; This study | ||||
| Syllis tyrrhena (Licher & Kuper, 1998) | NR | 24–47 | x | This study | |||||||
| Syllis variegata Grube, 1860 | 0–48 | x | x | x | x | Literature; This study | |||||
| Syllis westheidei San Martín, 1984 | NR | 5–17 | x | This study | |||||||
| Synmerosyllis lamelligera (Saint-Joseph, 1887) | 0–70 | x | x | x | x | Literature; This study | |||||
| Trypanosyllis aeolis Langerhans, 1879 | 0–50 | x | x | Literature | |||||||
| Trypanosyllis zebra (Grube, 1860) | SC | 0–70 | x | x | x | x | Literature | ||||
| Xenosyllis scabra (Ehlers, 1864) | 40–328 | x | x | x | x | Literature; This study | |||||
| Terebellidae | |||||||||||
| Amaeana trilobata (Sars, 1863) | 35 | x | x | Literature | |||||||
| Amphitrite cirrata (O. F. Müller, 1776) | 16–131 | x | Literature; This study | ||||||||
| Amphitrite edwardsii (Quatrefages, 1866) | 0–50 | x | x | Literature | |||||||
| Amphitrite figulus (Dalyell, 1853) | 10 | Literature | |||||||||
| Amphitrite gracilis (Grube, 1860) | 5 | Literature | |||||||||
| Amphitrite rubra (Risso, 1826) | SC | 32–35 | x | x | Literature | ||||||
| Amphitrite variabilis (Risso, 1826) | 0–42 | x | x | x | x | Literature; This study | |||||
| Eupolymnia nebulosa (Montagu, 1819) | 0–145 | x | x | x | x | Literature; This study | |||||
| Eupolymnia nesidensis (Delle Chiaje, 1828) | 0–70 | x | x | x | x | x | Literature; This study | ||||
| Lanice conchilega (Pallas, 1766) | 0–49 | x | x | x | Literature; This study | ||||||
| Nicolea venustula (Montagu, 1819) | 0–100 | x | x | x | x | Literature; This study | |||||
| Nicolea zostericola Örsted, 1844 | 45–96 | x | Literature | ||||||||
| Parathelepus collaris (Southern, 1914) | 27–45 | x | x | Literature | |||||||
| Pista cretacea (Grube, 1860) | NR | 5–40 | x | x | This study | ||||||
| Pista cristata (O. F. Müller, 1776) | 0–70 | x | x | x | x | Literature; This study | |||||
| Pista labruneae Lavesque, Daffe, Londoño-Mesa & Hutchings, 2021 | 8–92 | x | x | x | Literature; This study | ||||||
| Pista maculata (Dalyell, 1853) | 0–32 | x | x | x | Literature | ||||||
| Pistella rovignensis Mikac & Hutchings, 2017 | NR | 8–42 | x | x | This study | ||||||
| Polycirrus aurantiacus Grube, 1860 | 0–15 | x | x | Literature | |||||||
| Polycirrus haematodes (Claparède, 1864) | 0–15 | x | x | Literature | |||||||
| Polycirrus medusa Grube, 1850 | NR | 30–40 | x | x | This study | ||||||
| Polycirrus nogueirai Lavesque, Hutchings, Daffe & Londoño-Mesa, 2020 | NR | 40 | x | This study | |||||||
| Polycirrus twisti Potts, 1828 | NR; NIS | 26–38 | x | x | This study | ||||||
| Streblosoma pseudocomatus Lezzi & Giangrande, 2018 | NR; NIS | 33 | x | This study | |||||||
| Terebella ehrenbergi Grube, 1869 | NIS | 144–150 | Literature | ||||||||
| Terebella lapidaria Linnaeus, 1767 | - | Literature | |||||||||
| Thelepus cincinnatus (Fabricius, 1780) | 11–145 | x | x | x | x | Literature; This study | |||||
| Thelepus setosus (Quatrefages, 1866) | NR | 23–48 | x | x | This study | ||||||
| Trichobranchidae | |||||||||||
| Octobranchus lingulatus (Grube, 1863) | NR | 40 | x | This study | |||||||
| Terebellides stroemii Sars, 1835 | SC | 10–59 | x | x | Literature; This study | ||||||
| Trichobranchus glacialis Malmgren, 1866 | 19–48 | x | x | Literature; This study | |||||||
| Family | TN | NRs | NIS | Family | TN | NRs | NIS |
|---|---|---|---|---|---|---|---|
| Acoetidae | 2 | 1 | Nereididae | 24 | 7 | 5 | |
| Acrocirridae | 2 | Oenonidae | 5 | 1 | |||
| Ampharetidae | 7 | 3 | Onuphidae | 10 | 2 | ||
| Amphinomidae | 6 | 2 | Opheliidae | 6 | 2 | ||
| Aphroditidae | 5 | 1 | Orbiniidae | 10 | 4 | 2 | |
| Arenicolidae | 1 | 1 | Oweniidae | 2 | 1 | ||
| Capitellidae | 20 | 7 | 5 | Paralacydoniidae | 1 | ||
| Chrysopetalidae | 3 | 1 | Paraonidae | 28 | 10 | 1 | |
| Cirratulidae | 19 | 12 | 1 | Pectinariidae | 4 | 2 | |
| Cossuridae | 2 | 1 | Phyllodocidae | 26 | 6 | ||
| Dorvilleidae | 10 | 4 | 2 | Pilargidae | 3 | 1 | 1 |
| Eunicidae | 14 | 3 | 4 | Poecilochaetidae | 1 | ||
| Fabriciidae | 3 | 2 | Polynoidae | 11 | 1 | ||
| Fauveliopsidae | 2 | 1 | Sabellidae | 39 | 9 | 3 | |
| Flabelligeridae | 9 | 3 | 1 | Saccocirridae | 1 | 1 | |
| Glyceridae | 9 | 2 | Scalibregmatidae | 5 | 1 | ||
| Goniadidae | 4 | 1 | Serpulidae | 44 | 1 | 7 | |
| Hesionidae | 9 | 2 | 1 | Sigalionidae | 13 | 2 | 1 |
| Lacydoniidae | 1 | Sphaerodoridae | 4 | 3 | |||
| Lumbrineridae | 16 | 8 | Spionidae | 31 | 9 | 4 | |
| Magelonidae | 6 | 3 | Sternaspidae | 1 | |||
| Maldanidae | 21 | 6 | 1 | Syllidae | 100 | 15 | 5 |
| Melinnidae | 2 | 1 | Terebellidae | 28 | 7 | 3 | |
| Microphthalmidae | 1 | Trichobranchidae | 3 | 1 | |||
| Nephtyidae | 11 | 2 |
| Category | Nb of Species | NIS | NRs | % Nb Overall Species | % NIS Overall | % NR Overall | %NIS per Category | %NR per Category | |
|---|---|---|---|---|---|---|---|---|---|
| Habitat Category | Artificial construction | 13 | 3 | 0 | 2.23 | 5.88 | 0 | 23.07 | |
| Combination of habitats | 16 | 0 | 0 | 2.74 | 0 | 0 | 0 | 0 | |
| H8330 Submerged or partially submerged sea caves | 7 | 0 | 0 | 1.20 | 0 | 0 | 0 | 0 | |
| Hard/rock | 186 | 11 | 2 | 31.85 | 21.56 | 1.34 | 5.91 | 1.08 | |
| Soft | 500 | 35 | 147 | 85.62 | 68.62 | 98.66 | 7.00 | 29.40 | |
| Soft/hard | 179 | 7 | 0 | 30.65 | 13.73 | 0 | 3.91 | 0 | |
| Sponge | 51 | 1 | 0 | 8.73 | 2.04 | 0 | 1.96 | 0 | |
| Flora association | Caulerpa cylindracea | 137 | 4 | 33 | 23.46 | 8.16 | 22.15 | 2.92 | 24.09 |
| Caulerpa prolifera | 192 | 7 | 52 | 32.88 | 14.29 | 34.90 | 3.65 | 27.08 | |
| Caulerpa racemosa | 164 | 7 | 18 | 28.08 | 14.29 | 12.08 | 4.27 | 10.98 | |
| Coralline algae | 11 | 1 | 1 | 1.88 | 2.04 | 0.67 | 9.09 | 9.09 | |
| Cymodocea nodosa | 86 | 0 | 15 | 14.73 | 0 | 10.07 | 0 | 17.44 | |
| Cystoseira crinita | 149 | 7 | 0 | 25.51 | 14.29 | 0 | 4.70 | 0 | |
| Cystoseira sp. | 68 | 4 | 0 | 11.64 | 8.16 | 0 | 5.88 | 0 | |
| Cystosiera barbata | 6 | 1 | 0 | 1.03 | 2.04 | 0 | 16.67 | 0 | |
| Dasycladus clavaeformis | 2 | 0 | 0 | 0.34 | 0 | 0 | 0 | 0 | |
| decaying Posidonia matte | 48 | 4 | 12 | 8.22 | 8.16 | 8.05 | 8.33 | 25.00 | |
| Dictyota sp. | 48 | 1 | 11 | 8.22 | 2.04 | 7.38 | 2.08 | 22.92 | |
| Green algae | 1 | 1 | 0 | 0.17 | 2.04 | 0 | 100.00 | 0 | |
| Halimeda tuna | 6 | 1 | 0 | 1.03 | 2.04 | 0 | 16.67 | 0 | |
| Halophila stipulacea | 84 | 1 | 15 | 14.38 | 2.04 | 10.07 | 1.19 | 17.86 | |
| Jania rubens | 1 | 0 | 0 | 0.17 | 0.00 | 0 | 0 | 0 | |
| Laurencia obtusa | 6 | 1 | 0 | 1.03 | 2.04 | 0 | 16.67 | 0 | |
| Laurencia papillosa | 6 | 1 | 0 | 1.03 | 2.04 | 0 | 16.67 | 0 | |
| Posidonia oceanica | 280 | 11 | 0 | 47.95 | 22.45 | 0 | 3.93 | 0 | |
| Padina pavonica | 39 | 5 | 0 | 6.68 | 10.20 | 0 | 12.82 | 0 | |
| Palisada perforata | 18 | 1 | 0 | 3.08 | 2.04 | 0 | 5.56 | 0 | |
| Sargassum vulgare | 32 | 1 | 0 | 5.48 | 2.04 | 0 | 3.13 | 0 | |
| Udotea petiolata | 68 | 1 | 0 | 11.64 | 2.04 | 0 | 1.47 | 0 | |
| Human Activity | Artificial reefs | 12 | 1 | 1 | 2.05 | 2.04 | 0.67 | 8.33 | 8.33 |
| Desalination plant | 68 | 3 | 18 | 11.64 | 6.12 | 12.08 | 4.41 | 26.47 | |
| Discarge point of treated fresh-water from a sewage factory | 23 | 0 | 6 | 3.94 | 0 | 4.03 | 0 | 26.09 | |
| Fishfarm (stations max 500m from cages) | 254 | 19 | 70 | 43.49 | 38.78 | 46.98 | 7.48 | 27.56 | |
| Touristic facilities (near hotels) | 14 | 1 | 0 | 2.40 | 2.04 | 7.14 | |||
| Ports/marinas | 56 | 5 | 8 | 9.42 | 9.8 | 5.37 | 7.27 | 14.55 | |
| Single buoy mooring (for transferring fuels to the power plant) | 41 | 2 | 10 | 7.02 | 4.08 | 6.71 | 4.88 | 24.39 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rousou, M.; Langeneck, J.; Apserou, C.; Arvanitidis, C.; Charalambous, S.; Chrysanthou, K.; Constantinides, G.; Dimitriou, P.D.; García Gómez, S.C.; Hadjieftychiou, S.I.; et al. Polychaetes (Annelida) of Cyprus (Eastern Mediterranean Sea): An Updated and Annotated Checklist including New Distribution Records. Diversity 2023, 15, 941. https://doi.org/10.3390/d15080941
Rousou M, Langeneck J, Apserou C, Arvanitidis C, Charalambous S, Chrysanthou K, Constantinides G, Dimitriou PD, García Gómez SC, Hadjieftychiou SI, et al. Polychaetes (Annelida) of Cyprus (Eastern Mediterranean Sea): An Updated and Annotated Checklist including New Distribution Records. Diversity. 2023; 15(8):941. https://doi.org/10.3390/d15080941
Chicago/Turabian StyleRousou, Maria, Joachim Langeneck, Chara Apserou, Christos Arvanitidis, Stephanos Charalambous, Kyproula Chrysanthou, George Constantinides, Panagiotis D. Dimitriou, Sergio Carlos García Gómez, Soteria Irene Hadjieftychiou, and et al. 2023. "Polychaetes (Annelida) of Cyprus (Eastern Mediterranean Sea): An Updated and Annotated Checklist including New Distribution Records" Diversity 15, no. 8: 941. https://doi.org/10.3390/d15080941
APA StyleRousou, M., Langeneck, J., Apserou, C., Arvanitidis, C., Charalambous, S., Chrysanthou, K., Constantinides, G., Dimitriou, P. D., García Gómez, S. C., Hadjieftychiou, S. I., Katsiaras, N., Kleitou, P., Kletou, D., Küpper, F. C., Louizidou, P., Martins, R., Moraitis, M. L., Papageorgiou, N., Papatheodoulou, M., ... Castelli, A. (2023). Polychaetes (Annelida) of Cyprus (Eastern Mediterranean Sea): An Updated and Annotated Checklist including New Distribution Records. Diversity, 15(8), 941. https://doi.org/10.3390/d15080941










