Groundwater Amphipods of the Hyporheic Interstitial: A Case Study from Luxembourg and The Greater Region
Abstract
1. Introduction
2. Materials and Methods
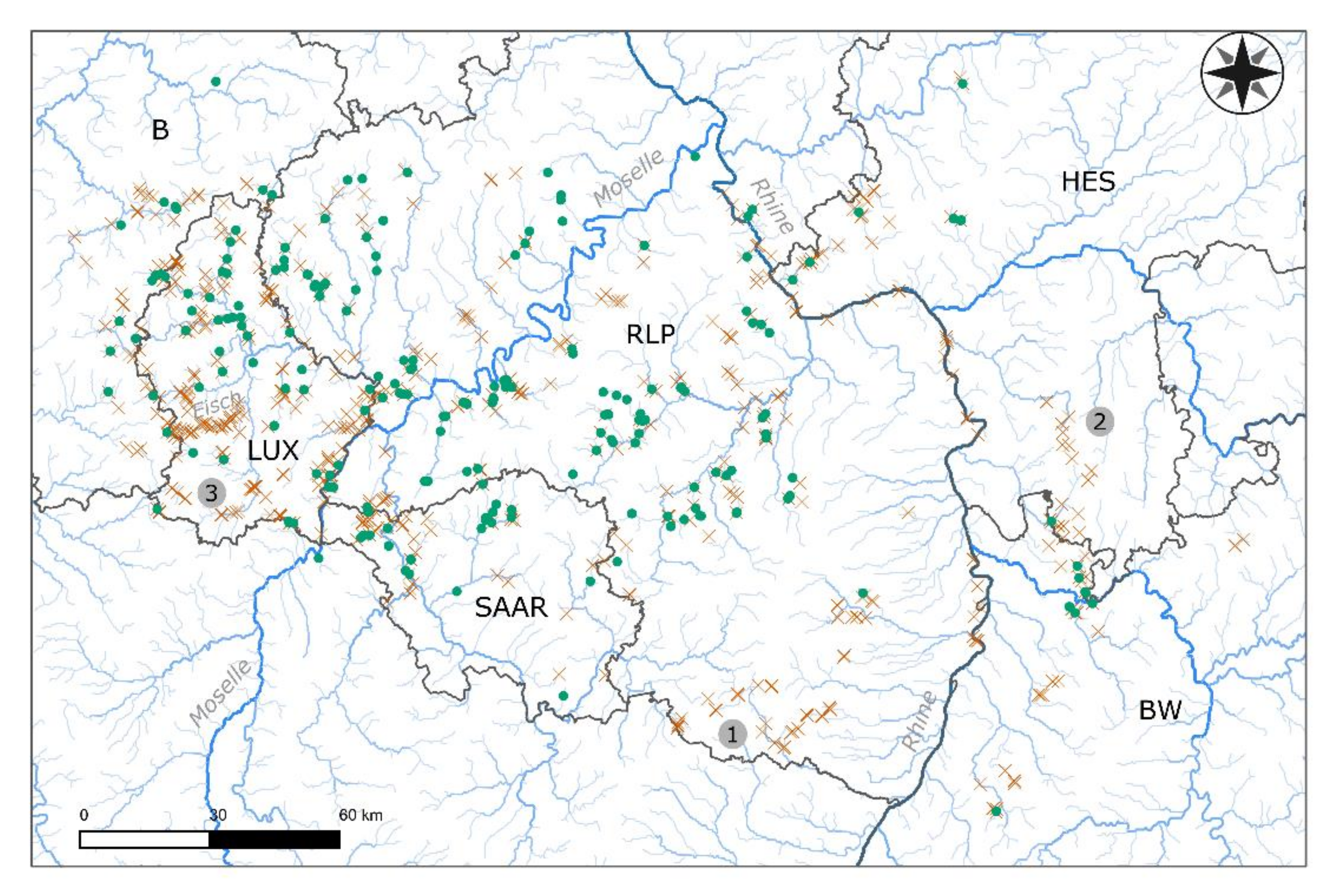
2.1. Sampling Sites and Sampling Method
2.2. Molecular Identification of Specimens
3. Results
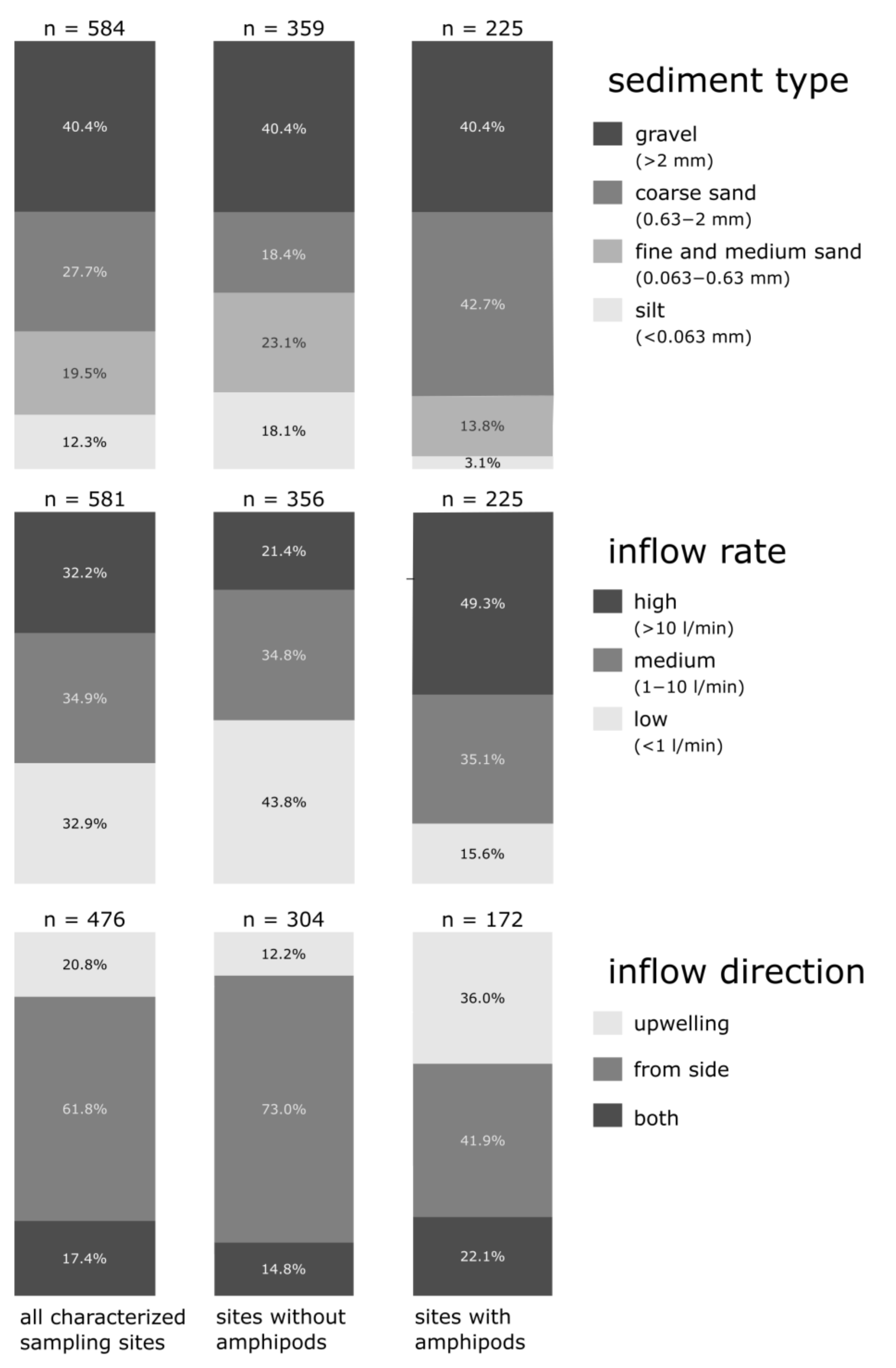
3.1. Characterization of Hyporheic Interstitials and Regional Patterns
3.2. Groundwater Amphipod Species Diversity
4. Discussion
4.1. Overall High Species Diversity in the Hyporheic Interstitial
4.2. Small Individuals but Not Small Species Dominate in the Hyporheic Interstitial
4.3. Regional Absence of Groundwater Amphipods in Hyporheic Interstitials
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Horton, T.; Lowry, J.; De Broyer, C.; Bellan-Santini, D.; Copilaș-Ciocianu, D.; Corbari, L.; Costello, M.J.; Daneliya, M.; Dauvin, J.-C.; Fišer, C.; et al. World Amphipoda Database. Available online: https://www.marinespecies.org/amphipoda/ (accessed on 18 January 2023).
- Borko, Š.; Trontelj, P.; Seehausen, O.; Moškrič, A.; Fišer, C. A subterranean adaptive radiation of amphipods in Europe. Nat. Commun. 2021, 12, 3688. [Google Scholar] [CrossRef] [PubMed]
- Premate, E.; Borko, Š.; Delić, T.; Malard, F.; Simon, L.; Fišer, C. Cave amphipods reveal co-variation between morphology and trophic niche in a low-productivity environment. Freshw. Biol. 2021, 66, 1876–1888. [Google Scholar] [CrossRef]
- Esmaeili-Rineh, S.; Sari, A.; Delić, T.; Moškrič, A.; Fišer, C. Molecular phylogeny of the subterranean genus Niphargus (Crustacea: Amphipoda) in the Middle East: A comparison with European niphargids. Zool. J. Linn. Soc. 2015, 175, 812–826. [Google Scholar] [CrossRef]
- Fišer, C.; Sket, B.; Trontelj, P. A phylogenetic perspective on 160 years of troubled taxonomy of Niphargus (Crustacea: Amphipoda). Zool. Scr. 2008, 37, 665–680. [Google Scholar] [CrossRef]
- Fišer, C.; Trontelj, P.; Luštrik, R.; Sket, B. Toward a unified taxonomy of Niphargus (Crustacea: Amphipoda): A review of morphological variability. Zootaxa 2009, 2061, 1–22. [Google Scholar] [CrossRef]
- Stoch, F.; Christian, E.; Flot, J.F. Molecular taxonomy, phylogeny and biogeography of the Niphargus tatrensis species complex (Amphipoda, Niphargidae) in Austria. Org. Divers. Evol. 2020, 20, 701–722. [Google Scholar] [CrossRef]
- Fišer, C.; Zagmajster, M. Cryptic species from cryptic space: The case of Niphargus fongi sp. n. (Amphipoda, Niphargidae). Crustaceana 2009, 82, 593–614. [Google Scholar] [CrossRef]
- Delić, T.; Trontelj, P.; Rendoš, M.; Fišer, C. The importance of naming cryptic species and the conservation of endemic subterranean amphipods. Sci. Rep. 2017, 7, 3391. [Google Scholar] [CrossRef]
- Fišer, C.; Robinson, C.T.; Malard, F. Cryptic species as a window into the paradigm shift of the species concept. Mol. Ecol. 2018, 27, 613–635. [Google Scholar] [CrossRef]
- Weber, D.; Stoch, F.; Knight, L.R.F.D.; Chauveau, C.; Flot, J.-F. The genus Microniphargus (Crustacea, Amphipoda): Evidence for three lineages distributed across northwestern Europe and transfer from Niphargidae to Pseudoniphargidae. Belg. J. Zool. 2021, 151, 169–191. [Google Scholar] [CrossRef]
- Weber, D.; Flot, J.-F. Rote Liste und Gesamtartenliste der Grundwasserkrebse (Niphargidae) des Saarlandes. In Rote Liste Gefährdeter Tiere und Pflanzen des Saarlandes; Ministerium für Umwelt und Verbraucherschutz und DELATTINIA: Saarbrücken und Landsweiler-Reden, Germany, 2020; pp. 3–9. [Google Scholar]
- Rütz, N.K.; Marxsen, J.; Wolters, V. Long-term cultivation of the groundwater amphipod Niphargus aquilex (Crustacea). Hydrobiologia 2023, 850, 269–281. [Google Scholar] [CrossRef]
- Husmann, S. Studies on subterranean drift of stygobiont crustaceans (Niphargus, Crangonyx, Graeteriella). Int. J. Speleol. 1976, 8, 81–92. [Google Scholar] [CrossRef]
- Hinz, W.; Danneel, I. Vorkommen von Niphargus a. aquilex Schiödte (Amphipoda) in Oberflächengewässern bei Düsseldorf. Nat. Und Heim. 1974, 34, 7–11. [Google Scholar]
- Spangenberg, H.-J. Beitrag zur Faunistik von Höhlengewässern im Zechstein des Südharzes und Kyffhäusers. Hercynia (Neue Folge) 1973, 10, 143–160. [Google Scholar]
- Copilaş-Ciocianu, D.; Fišer, C.; Borza, P.; Petrusek, A. Is subterranean lifestyle reversible? Independent and recent large-scale dispersal into surface waters by two species of the groundwater amphipod genus Niphargus. Mol. Phylogenetics Evol. 2018, 119, 37–49. [Google Scholar] [CrossRef]
- Weber, D.; Flot, J.-F.; Weigand, H.; Weigand, A.M. Demographic history, range size and habitat preferences of the groundwater amphipod Niphargus puteanus (C.L. Koch in Panzer, 1836). Limnol. Ecol. Manag. Inland Waters 2020, 82, 125765. [Google Scholar] [CrossRef]
- Boulton, A.J.; Findlay, S.; Marmonier, P.; Stanley, E.H.; Valett, H.M. The functional significance of the hyporheic zone in streams and rivers. Annu. Rev. Ecol. Syst. 1998, 29, 59–81. [Google Scholar] [CrossRef]
- Gómez-Gener, L.; Siebers, A.R.; Arce, M.I.; Arnon, S.; Bernal, S.; Bolpagni, R.; Datry, T.; Gionchetta, G.; Grossart, H.-P.; Mendoza-Lera, C.; et al. Towards an improved understanding of biogeochemical processes across surface-groundwater interactions in intermittent rivers and ephemeral streams. Earth-Sci. Rev. 2021, 220, 103724. [Google Scholar] [CrossRef]
- Mathers, K.L.; Hill, M.J.; Wood, C.D.; Wood, P.J. The role of fine sediment characteristics and body size on the vertical movement of a freshwater amphipod. Freshw. Biol. 2019, 64, 152–163. [Google Scholar] [CrossRef]
- Lawrence, J.E.; Skold, M.E.; Hussain, F.A.; Silverman, D.R.; Resh, V.H.; Sedlak, D.L.; Luthy, R.G.; McCray, J.E. Hyporheic zone in urban streams: A review and opportunities for enhancing water quality and improving aquatic habitat by active management. Environ. Eng. Sci. 2013, 30, 480–501. [Google Scholar] [CrossRef]
- Stubbington, R.; Dole-Olivier, M.-J.; Galassi, D.M.P.; Hogan, J.-P.; Wood, P.J. Characterization of macroinvertebrate communities in the hyporheic zone of river ecosystems reflects the pump-sampling technique used. PLoS ONE 2016, 11, e0164372. [Google Scholar] [CrossRef] [PubMed]
- Datry, T.; Singer, G.; Sauquet, E.; Jorda-Capdevila, D.; Von Schiller, D.; Stubbington, R.; Magand, C.; Pařil, P.; Miliša, M.; Acuña, V.; et al. Science and management of intermittent rivers and ephemeral streams (SMIRES). Res. Ideas Outcomes 2017, 3, e21774. [Google Scholar] [CrossRef]
- Weigand, A.M.; Macher, J.-N. A DNA metabarcoding protocol for hyporheic freshwater meiofauna: Evaluating highly degenerate COI primers and replication strategy. Metabarcoding Metagenom. 2018, 2, e26869. [Google Scholar] [CrossRef]
- Fišer, C.; Zagmajster, M.; Dethier, M. Overview of Niphargidae (Crustacea: Amphipoda) in Belgium: Distribution, taxonomic notes and conservation issues. Zootaxa 2018, 4387, 47. [Google Scholar] [CrossRef]
- Gerecke, R.; Stoch, F.; Meisch, C.; Schrankel, I. Die Fauna der Quellen und des hyporheischen Interstitials in Luxembourg. Unter Besonderer Berücksichtigung der Milben (Acari), Muschelkrebse (Ostracoda) und Ruderfusskrebse (Copepoda). Ferrantia 2005, 41, 1–140. [Google Scholar]
- Weber, D.; Flot, J.-F.; Frantz, A.; Weigand, A.M. Molecular Analyses of Groundwater Amphipods (Crustacea: Niphargidae) from Luxembourg: New species reveal limitations of morphology-based checklists. Zootaxa 2022, 5222, 501–533. [Google Scholar] [CrossRef]
- Flot, J.-F.; Weber, D. Amphipods from caves of the Grand Duchy of Luxembourg. Ferrantia 2013, 69, 186–190. [Google Scholar]
- McInerney, C.E.; Maurice, L.; Robertson, A.L.; Knight, L.R.F.D.; Arnscheidt, J.; Venditti, C.; Dooley, J.S.G.; Mathers, T.; Matthijs, S.; Erikkson, K.; et al. The ancient Britons: Groundwater fauna survived extreme climate changes over tens of millions of years across NW Europe. Mol. Ecol. 2014, 23, 1153–1166. [Google Scholar] [CrossRef]
- Alther, R.; Bongni, N.; Borko, Š.; Fišer, C.; Altermatt, F. Citizen science approach reveals groundwater fauna in Switzerland and a new species of Niphargus (Amphipoda, Niphargidae). Subterr. Biol. 2021, 39, 1–31. [Google Scholar] [CrossRef]
- Rendoš, M.; Delić, T.; Copilaș-Ciocianu, D.; Fišer, C. First insight into cryptic diversity of a Caucasian subterranean amphipod of the genus Niphargus (Crustacea: Amphipoda: Niphargidae). Zool. Anz. 2021, 290, 1–11. [Google Scholar] [CrossRef]
- Husmann, S. Eine neue Methode zur Entnahme von Interstitialwasser aus subaquatischen Lockergesteinen. Arch. Für Hydrobiol. 1971, 68, 519–527. [Google Scholar]
- Chappuis, P.-A. Eine neue Methode zur Untersuchung der Grundwasserfauna. Acta Scientiarum Mathematicarum. Nat. Univ. Fr. Josephinae Kolozsvar 1942, 6, 3–7. [Google Scholar]
- Husmann, S. Eine Rammpumpe zur Untersuchung des subaquatischen Stygorheals. Gewässer Abwässer 1971, 50–51, 115–119. [Google Scholar]
- Pearson, K.X. On the criterion that a given system of deviations from the probable in the case of a correlated system of variables is such that it can be reasonably supposed to have arisen from random sampling. Lond. Edinb. Dublin Philos. Mag. J. Sci. 1900, 50, 157–175. [Google Scholar] [CrossRef]
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; Buxton, S.; Cooper, A.; Markowitz, S.; Duran, C.; et al. Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Grabner, D.; Weber, D.; Weigand, A.M. Updates to the sporadic knowledge on microsporidian infections in groundwater amphipods (Crustacea, Amphipoda, Niphargidae). Subterr. Biol. 2020, 33, 71–85. [Google Scholar] [CrossRef]
- Weber, D. Molecular Phylogeny and Systematics of Central and Western European Niphargids (Crustacea: Amphipoda); Université liebre de Bruxelles: Bruxelles, Belgium, 2022. [Google Scholar]
- Hoffmann, J. Faune des Amphipodes du Grand-Duché de Luxembourg. Crustacea, Malacostraca, Amphipoda. Arch. de la Sect. Des. Sci. de L’institut Grand-Ducal Nouv. Série 1963, 29, 77–128. [Google Scholar]
- Weber, D.; Brad, T.; Stoch, F.; Flot, J.-F. Rediscovery and redescription of Niphargus enslini Karaman, 1932 (Amphipoda, Niphargidae) in southern Germany. Subterr. Biol. 2021, 40, 65–89. [Google Scholar] [CrossRef]
- Knight, L.R.F.D.; Gledhill, T. The discovery of Microniphargus leruthi Schellenberg, 1934 (Crustacea: Amphipoda: Niphargidae) in Britain and its distribution in the British Isles. Zootaxa 2010, 2655, 52–56. [Google Scholar] [CrossRef]
- Bou, C.; Rouch, R. Un nouveau champ de recherches sur la faune aquatique souterraine. Comptes Rendus De L’académie Des Sci. Série D 1967, 265, 369–370. [Google Scholar]
- Schellenberg, A. Eine neue Amphipoden-Gattung aus einer belgischen Höhle, nebst Bemerkungen über die Gattung Crangonyx. Zool. Anz. 1934, 106, 215–218. [Google Scholar]
- Weber, D.; Knight, L.R.F.D.; Salussolia, A. Microniphargus, first results of phylogenetic analysis. In Proceedings of the 18th UIS Congress—Volume I. Karstologia Mémoires, Savoie Technolac, France, 24–31 July 2022; Volume 21, pp. 357–360. [Google Scholar]
- Spence Bate, C. On the genus Niphargus (Schiödte). In Proceedings of the Dublin University Zoological and Botanical Association; Dublin University: Dublin, Ireland, 1859; Volume 1, pp. 237–240. [Google Scholar]
- Schminke, H.K.; Gad, G. Grundwasserfauna Deutschlands. Ein Bestimmungswerk; Deutsche Vereinigung für Wasserwirtschaft, Abwasser und Abfall e.V.: Hennef, Germany, 2007. [Google Scholar]
- Schellenberg, A. Krebstiere oder Crustacea. IV: Flohkrebse oder Amphipoda. Die Tierwelt Deutschlands und der angrenzenden Meeresteile nach ihren Merkmalen und nach ihrer Lebensweise; Fischer: Jena, Germany, 1942. [Google Scholar]
- Stock, J.H.; Gledhill, T. The Niphargus kochianus-group in north-western Europe. Crustaceana Suppl. 1977, 4, 212–243. [Google Scholar]
- Schellenberg, A. Höhlenflohkrebse des Adelsberger Grottensystems nebst Bemerkung über Niphargus kochianus. Mitt. über Höhlen- und Karstforschung 1933, 2, 32–36. [Google Scholar]
- Weigand, A.M.; Kremers, J.; Grabner, D.S. Shared microsporidian profiles between an obligate (Niphargus) and facultative subterranean amphipod population (Gammarus) at sympatry provide indications for underground transmission pathways. Limnol. Ecol. Manag. Inland Waters 2016, 58, 7–10. [Google Scholar] [CrossRef]
- van de Kerkhof, S. Die Industrialisierung der lothringisch-luxemburgischen Minette-Region. In Die Industrialisierung Europäischer Montanregionen im 19. Jahrhundert (=Regionale Industrialisierung); Pierenkemper, A., Ed.; F. Steiner: Stuttgart, Germany, 2002; pp. 225–276. [Google Scholar]
- Konen, J.; Scheffen, J.-L. Bergwerke: Luxemburgs Vergangene Industrie unter Tage; Jérôme Konen Productions: Luxembourg, 2015. [Google Scholar]
- Weigand, H.; Weiss, M.; Cai, H.; Li, Y.; Yu, L.; Zhang, C.; Leese, F. Fishing in troubled waters: Revealing genomic signatures of local adaptation in response to freshwater pollutants in two macroinvertebrates. Sci. Total Environ. 2018, 633, 875–891. [Google Scholar] [CrossRef]
- Iniguez-Armijos, C.; Tapia-Armijos, M.F.; Wilhelm, F.; Breuer, L. Urbanisation process generates more independently-acting stressors and ecosystem functioning impairment in tropical Andean streams. J. Environ. Manag. 2022, 304, 114211. [Google Scholar] [CrossRef]
- United Nations. Globally Harmonized System of Classification and Labelling of Chemicals (GHS), 9th ed.; United Nations: New York, NY, USA; Geneva, Switzerland, 2021. [Google Scholar]
- Neyens, N. Das Steinforter Phenolwerk und die Umweltprobleme an der Eisch. Nos Cah. Kanton Capellen 2003, 3–4, 75–88. [Google Scholar]
- Giger, W. The Rhine red, the fish dead—The 1986 Schweizerhalle disaster, a retrospect and long-term impact assessment. Environ. Sci. Pollut. Res. 2009, 16, 98–111. [Google Scholar] [CrossRef]
- Van Dijk, G.M.; Marteijn, E.C.L.; Schulte-Wülwer-Leidig, A. Ecological rehabilitation of the River Rhine: Plans, progress and perspectives. Regul. Rivers: Res. Manag. 1995, 11, 377–388. [Google Scholar] [CrossRef]
| Species | n | LUX | B-Wal | D-RLP | D-Saar | D-Hes | D-BW |
|---|---|---|---|---|---|---|---|
| Crangonyx subterraneus Spence Bate, 1859 | 4 | x | x | x | |||
| Microniphargus leruthi Schellenberg, 1934 | 1 | x | |||||
| Niphargellus nolli Schellenberg, 1938 | 6 | x | x | x | x | ||
| Niphargus aquilex A | 7 | x | x | ||||
| Niphargus aquilex B (sensu stricto) | 28 | x | x | x | x | x | |
| Niphargus aquilex EF (sublineage F-1) | 114 | x | x | x | x | x | |
| Niphargus aquilex EF (sublineage F-2) | 11 | x | x | x | |||
| Niphargus aquilex G (sublineage G-1) | 12 | x | x | x | x | ||
| Niphargus aquilex G (sublineage G-2) | 1 | x | |||||
| Undescribed species of the N. aquilex-complex | 1 | x | |||||
| Niphargus fontanus Spence Bate, 1859 | 1 | x | |||||
| Niphargus puteanus (C.L. Koch in Panzer, 1836) | 5 | x | x | ||||
| Niphargus schellenbergi S. Karaman, 1932 | 101 | x | x | x | x | x | x |
| Species | Luxembourg Interstitial (This Study) | Luxembourg Interstitial [27] | Luxembourg All [28] | Saarland Interstitial (This Study) | Saarland All [12] |
|---|---|---|---|---|---|
| Microniphargus leruthi Schellenberg, 1934 | x | ||||
| Niphargellus nolli Schellenberg, 1938 | x | x | (x) | ||
| Niphargus aquilex-complex sensu Schiødte, 1855 | x | ||||
| Niphargus aquilex A | x | x | |||
| Niphargus aquilex B (sensu stricto) | x | x | x | ||
| Niphargus aquilex EF (sublineage F-1) | x | x | x | x | |
| Niphargus aquilex EF (sublineage F-2) | x | x | |||
| Niphargus aquilex G (sublineage G-1) | x | x | x | ||
| Niphargus aquilex I | x | ||||
| Niphargus fontanus Spence Bate, 1859 | x | x | |||
| Niphargus kochianus-complex sensu Spence Bate, 1859 | x | (x) | |||
| Niphargus puteanus (C.L. Koch in Panzer, 1836) | x | ||||
| Niphargus schellenbergi S. Karaman, 1932 | x | x | x | x | x |
| Niphargus virei Chevreux, 1896 | x |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weber, D.; Weigand, A.M. Groundwater Amphipods of the Hyporheic Interstitial: A Case Study from Luxembourg and The Greater Region. Diversity 2023, 15, 411. https://doi.org/10.3390/d15030411
Weber D, Weigand AM. Groundwater Amphipods of the Hyporheic Interstitial: A Case Study from Luxembourg and The Greater Region. Diversity. 2023; 15(3):411. https://doi.org/10.3390/d15030411
Chicago/Turabian StyleWeber, Dieter, and Alexander M. Weigand. 2023. "Groundwater Amphipods of the Hyporheic Interstitial: A Case Study from Luxembourg and The Greater Region" Diversity 15, no. 3: 411. https://doi.org/10.3390/d15030411
APA StyleWeber, D., & Weigand, A. M. (2023). Groundwater Amphipods of the Hyporheic Interstitial: A Case Study from Luxembourg and The Greater Region. Diversity, 15(3), 411. https://doi.org/10.3390/d15030411








