Gut Bacterial Communities Vary across Habitats and Their Diversity Increases with Increasing Glucocorticoids in Toad Tadpoles
Abstract
1. Introduction
2. Materials and Methods
2.1. Tadpole Collection and Processing
2.2. Corticosterone Analysis
2.3. Bacterial Communities Analysis
2.4. Statistical Analysis
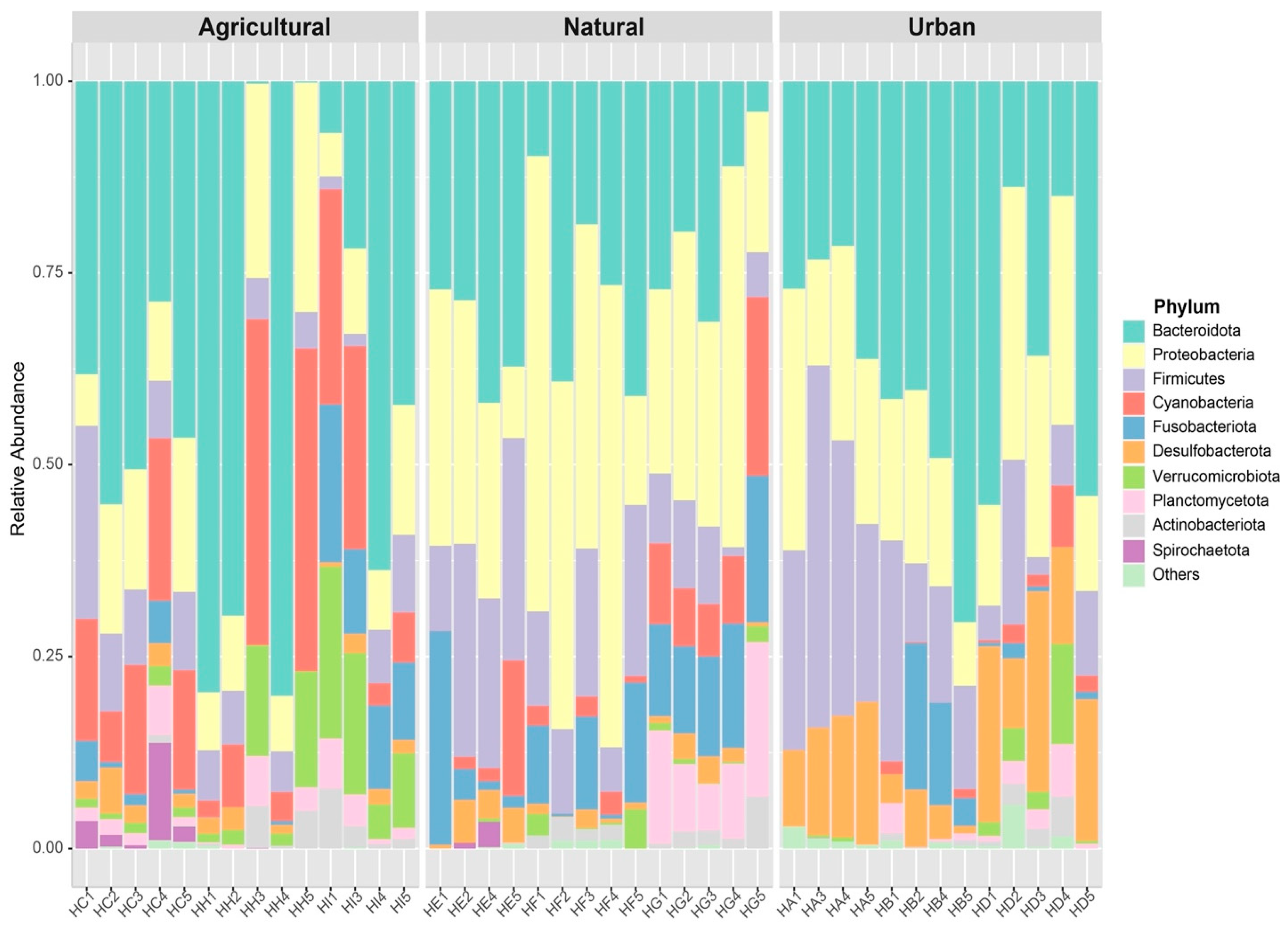
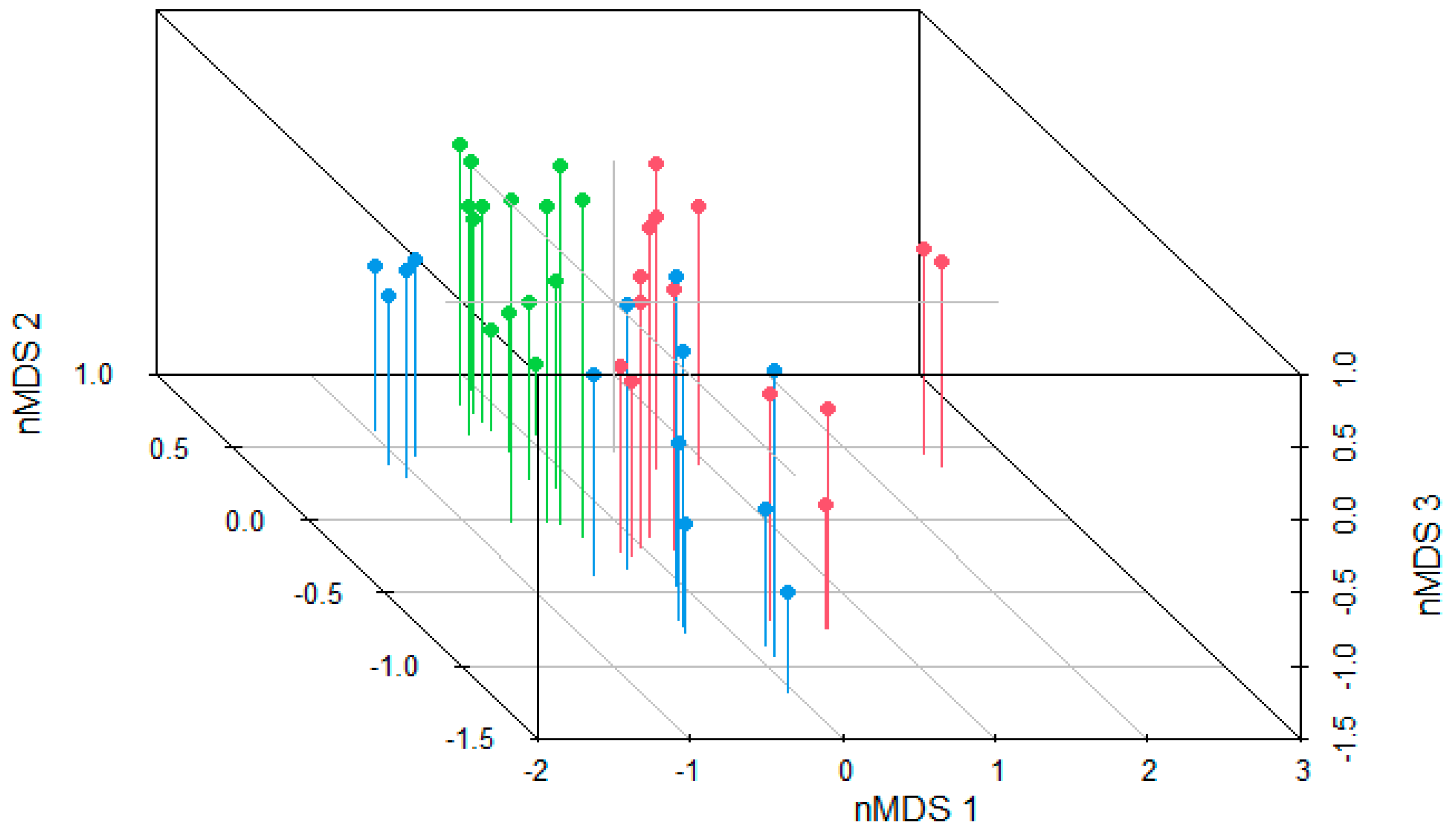
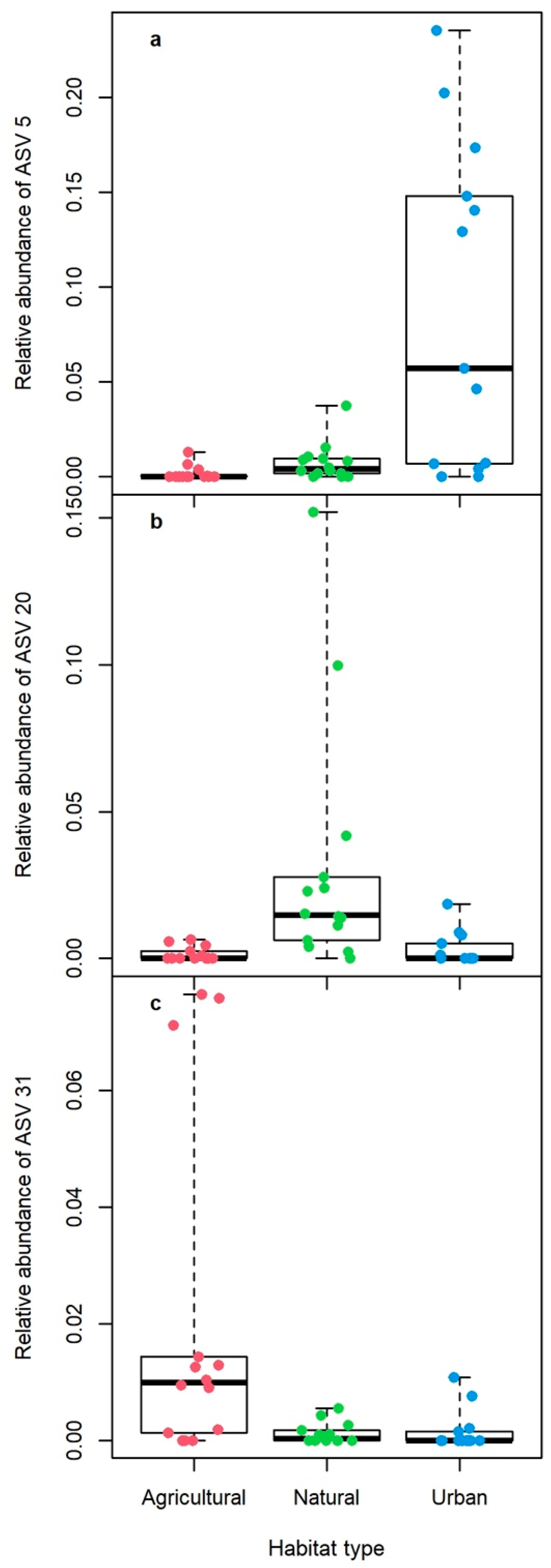
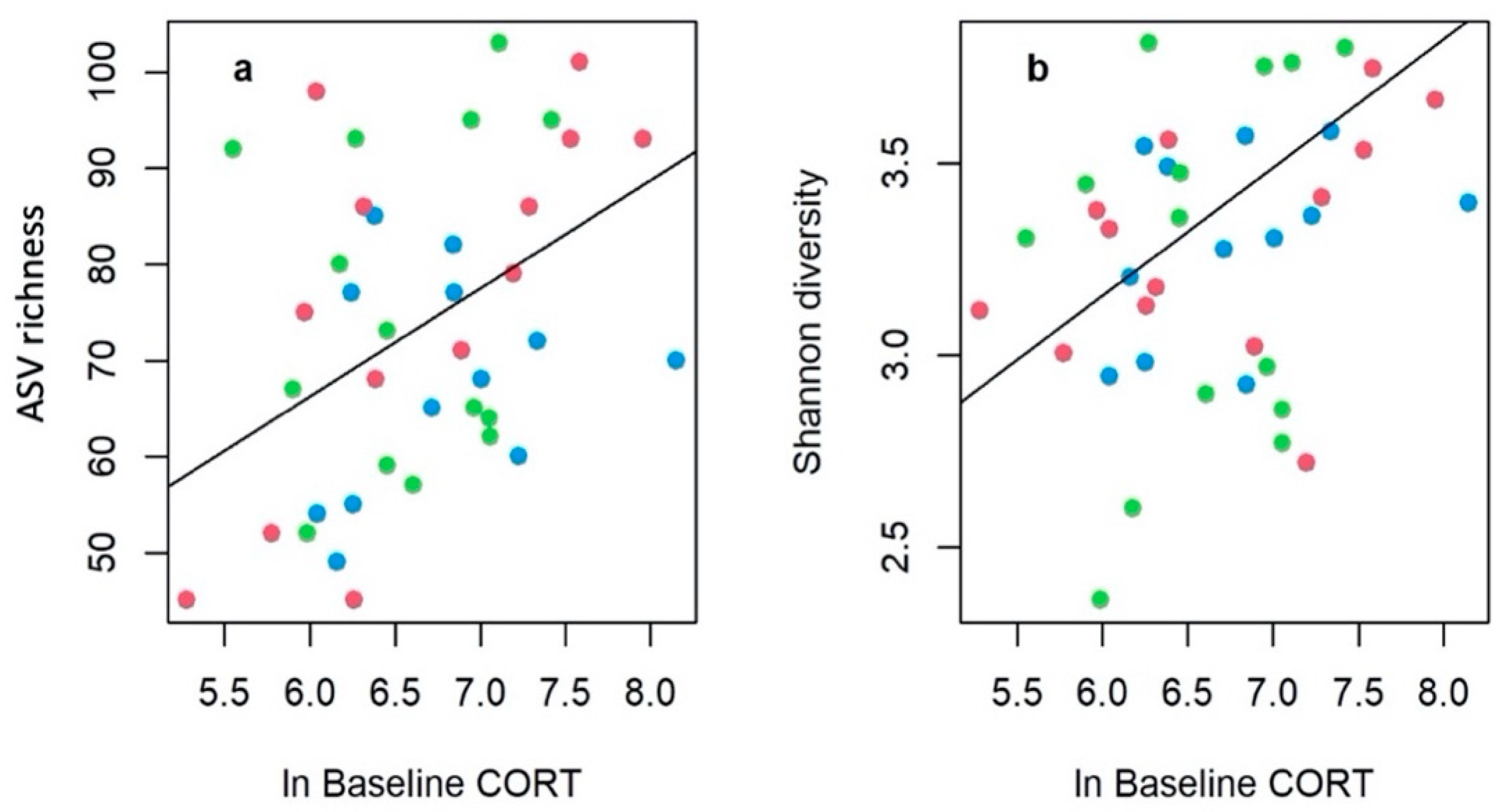
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jandhyala, S.M.; Talukdar, R.; Subramanyam, C.; Vuyyuru, H.; Sasikala, M.; Nageshwar Reddy, D. Role of the normal gut microbiota. World J. Gastroenterol. 2015, 21, 8787–8803. [Google Scholar] [CrossRef] [PubMed]
- McFall-Ngai, M.; Hadfield, M.G.; Bosch, T.C.; Carey, H.V.; Domazet-Lošo, T.; Douglas, A.E.; Dubilier, N.; Eberl, G.; Fukami, T.; Gilbert, S.F.; et al. Animals in a bacterial world, a new imperative for the life sciences. Proc. Natl. Acad. Sci. USA 2013, 110, 3229–3236. [Google Scholar] [CrossRef] [PubMed]
- Hooper, L.V.; Littman, D.R.; Macpherson, A.J. Interactions between the microbiota and the immune system. Science 2012, 336, 1268–1273. [Google Scholar] [CrossRef] [PubMed]
- Rea, K.; Dinan, T.G.; Cryan, J.F. The microbiome: A key regulator of stress and neuroinflammation. Neurobiol. Stress 2016, 4, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Ujszegi, J.; Vajna, B.; Móricz, Á.M.; Krüzselyi, D.; Korponai, K.; Krett, G.; Hettyey, A. Relationships Between Chemical Defenses of Common Toad (Bufo bufo) Tadpoles and Bacterial Community Structure of their Natural Aquatic Habitat. J. Chem. Ecol. 2020, 46, 534–543. [Google Scholar] [CrossRef]
- Teyssier, A.; Matthysen, E.; Hudin, N.S.; de Neve, L.; White, J.; Lens, L. Diet contributes to urban-induced alterations in gut microbiota: Experimental evidence from a wild passerine. Proc. Biol. Sci. 2020, 287, 20192182. [Google Scholar] [CrossRef]
- Barnes, E.M.; Kutos, S.; Naghshineh, N.; Mesko, M.; You, Q.; Lewis, J.D. Assembly of the amphibian microbiome is influenced by the effects of land-use change on environmental reservoirs. Environ. Microbiol. 2021, 23, 4595–4611. [Google Scholar] [CrossRef]
- Stražar, M.; Temba, G.S.; Vlamakis, H.; Kullaya, V.I.; Lyamuya, F.; Mmbaga, B.T.; Joosten, L.A.B.; van der Ven, A.J.A.M.; Netea, M.G.; de Mast, Q.; et al. Gut microbiome-mediated metabolism effects on immunity in rural and urban African populations. Nat. Commun. 2021, 12, 4845. [Google Scholar] [CrossRef]
- Wong, S.; Rawls, J.F. Intestinal microbiota composition in fishes is influenced by host ecology and environment. Mol. Ecol. 2012, 21, 3100–3102. [Google Scholar] [CrossRef]
- Uren Webster, T.M.; Rodriguez-Barreto, D.; Consuegra, S.; Garcia De Leaniz, C. Cortisol-Related Signatures of Stress in the Fish Microbiome. Front. Microbiol. 2020, 11, 01621. [Google Scholar] [CrossRef]
- Galley, J.D.; Nelson, M.C.; Yu, Z.; Dowd, S.E.; Walter, J.; Kumar, P.S.; Lyte, M.; Bailey, M.T. Exposure to a social stressor disrupts the community structure of the colonic mucosa-associated microbiota. BMC Microbiol. 2014, 14, 189. [Google Scholar] [CrossRef] [PubMed]
- Kohl, K.D.; Yahn, J. Effects of environmental temperature on the gut microbial communities of tadpoles. Environ. Microbiol. 2016, 18, 1561–1565. [Google Scholar] [CrossRef] [PubMed]
- Krotman, Y.; Yergaliyev, T.M.; Shani, R.A.; Avrahami, Y.; Szitenberg, A. Dissecting the factors shaping fish skin microbiomes in a heterogeneous inland water system. Microbiome 2020, 8, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Zaneveld, J.R.; McMinds, R.; Vega Thurber, R. Stress and stability: Applying the Anna Karenina principle to animal microbiomes. Nat. Microbiol. 2017, 2, 17121. [Google Scholar] [CrossRef] [PubMed]
- Alberdi, A.; Aizpurua, O.; Bohmann, K.; Zepeda-Mendoza, M.L.; Gilbert, M.T.P. Do Vertebrate Gut Metagenomes Confer Rapid Ecological Adaptation? Trends Ecol. Evol. 2016, 31, 689–699. [Google Scholar] [CrossRef]
- Rennison, D.J.; Rudman, S.M.; Schluter, D. Parallel changes in gut microbiome composition and function during colonization, local adaptation and ecological speciation. Proc. R. Soc. B Biol. Sci. 2019, 286, 20191911. [Google Scholar] [CrossRef]
- Houwenhuyse, S.; Stoks, R.; Mukherjee, S.; Decaestecker, E. Locally adapted gut microbiomes mediate host stress tolerance. ISME J. 2021, 15, 2401–2414. [Google Scholar] [CrossRef]
- Zorriehzahra, M.J.; Delshad, S.T.; Adel, M.; Tiwari, R.; Karthik, K.; Dhama, K.; Lazado, C.C. Probiotics as beneficial microbes in aquaculture: An update on their multiple modes of action: A review. Vet. Q. 2016, 36, 228–241. [Google Scholar] [CrossRef]
- Tetel, M.J.; de Vries, G.J.; Melcangi, R.C.; Panzica, G.; O’Mahony, S.M. Steroids, stress and the gut microbiome-brain axis. J. Neuroendocrinol. 2018, 30, e12548. [Google Scholar] [CrossRef]
- Foster, J.A.; Rinaman, L.; Cryan, J.F. Stress & the gut-brain axis: Regulation by the microbiome. Neurobiol. Stress 2017, 7, 124–136. [Google Scholar] [CrossRef]
- Brans, K.I.; Engelen, J.M.T.; Souffreau, C.; De Meester, L. Urban hot-tubs: Local urbanization has profound effects on average and extreme temperatures in ponds. Landsc. Urban Plann. 2018, 176, 22–29. [Google Scholar] [CrossRef]
- Wang, H.; Marshall, C.W.; Cheng, M.; Xu, H.; Li, H.; Yang, X.; Zheng, T. Changes in land use driven by urbanization impact nitrogen cycling and the microbial community composition in soils. Sci. Rep. 2017, 7, 44049. [Google Scholar] [CrossRef] [PubMed]
- Colston, T.J.; Jackson, C.R. Microbiome evolution along divergent branches of the vertebrate tree of life: What is known and unknown. Mol. Ecol. 2016, 25, 3776–3800. [Google Scholar] [CrossRef] [PubMed]
- Numberger, D.; Zoccarato, L.; Woodhouse, J.; Ganzert, L.; Sauer, S.; Grossart, H.-P.; Greenwood, A. Urbanization promotes specific bacteria in freshwater microbiomes including potential pathogens. bioRxiv 2020, 845, 157321. [Google Scholar] [CrossRef] [PubMed]
- McLellan, S.L.; Fisher, J.C.; Newton, R.J. The microbiome of urban waters. Int. Microbiol. 2015, 18, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Newton, R.J.; McLellan, S.L.; Dila, D.K.; Vineis, J.H.; Morrison, H.G.; Eren, A.M.; Sogin, M.L. Sewage reflects the microbiomes of human populations. mBio 2015, 6, e02574. [Google Scholar] [CrossRef] [PubMed]
- Vonesh, J.; De la Cruz Cabrera, O. Complex life cycles and density dependence: Assessing the contribution of egg mortality to amphibian declines. Oecologia 2002, 133, 325–333. [Google Scholar] [CrossRef]
- Givens, C.E.; Kolpin, D.W.; Borchardt, M.A.; Duris, J.W.; Moorman, T.B.; Spencer, S.K. Detection of hepatitis E virus and other livestock-related pathogens in Iowa streams. Sci. Total Environ. 2016, 566–567, 1042–1051. [Google Scholar] [CrossRef]
- Walters, E.; Graml, M.; Behle, C.; Müller, E.; Horn, H. Influence of Particle Association and Suspended Solids on UV Inactivation of Fecal Indicator Bacteria in an Urban River. Water Air Soil Pollut. 2013, 225, 1–9. [Google Scholar] [CrossRef]
- Dudgeon, D.; Arthington, A.H.; Gessner, M.O.; Kawabata, Z.-I.; Knowler, D.J.; Lévêque, C.; Naiman, R.J.; Prieur-Richard, A.-H.; Soto, D.; Stiassny, M.L.J.; et al. Freshwater biodiversity: Importance, threats, status and conservation challenges. Biol. Rev. 2006, 81, 163–182. [Google Scholar] [CrossRef]
- Sapolsky, R.M.; Romero, L.M.; Munck, A.U. How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocr. Rev. 2000, 21, 55–89. [Google Scholar] [CrossRef] [PubMed]
- Petrullo, L.; Ren, T.; Wu, M.; Boonstra, R.; Palme, R.; Boutin, S.; McAdam, A.G.; Dantzer, B. Glucocorticoids coordinate changes in gut microbiome composition in wild North American red squirrels. Sci. Rep. 2022, 12, 2605. [Google Scholar] [CrossRef] [PubMed]
- Foster, J.A.; Neufeld, K.-A.M. Gut–brain axis: How the microbiome influences anxiety and depression. Trends Neurosci. 2013, 36, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Sekirov, I.; Russell, S.L.; Antunes, L.C.M.; Finlay, B.B. Gut microbiota in health and disease. Physiol. Rev. 2010, 90, 859–904. [Google Scholar] [CrossRef]
- Grenham, S.; Clarke, G.; Cryan, J.; Dinan, T. Brain-gut-microbe communication in health and disease. Front. Physiol. 2011, 2, 94, External Resources Crossref (DOI)2011. [Google Scholar] [CrossRef]
- Butt, R.L.; Volkoff, H. Gut Microbiota and Energy Homeostasis in Fish. Front. Endocrinol. 2019, 10, 9. [Google Scholar] [CrossRef]
- Kim, P.S.; Shin, N.-R.; Lee, J.-B.; Kim, M.-S.; Whon, T.W.; Hyun, D.-W.; Yun, J.-H.; Jung, M.-J.; Kim, J.Y.; Bae, J.-W. Host habitat is the major determinant of the gut microbiome of fish. Microbiome 2021, 9, 166. [Google Scholar] [CrossRef]
- Stothart, M.R.; Palme, R.; Newman, A.E.M. It’s what’s on the inside that counts: Stress physiology and the bacterial microbiome of a wild urban mammal. Proc. R. Soc. B Biol. Sci. 2019, 286, 20192111. [Google Scholar] [CrossRef]
- Zhang, H.; Sparks, J.B.; Karyala, S.V.; Settlage, R.; Luo, X.M. Host adaptive immunity alters gut microbiota. ISME J. 2015, 9, 770–781. [Google Scholar] [CrossRef]
- Dillon, R.J.; Vennard, C.T.; Buckling, A.; Charnley, A.K. Diversity of locust gut bacteria protects against pathogen invasion. Ecol. Lett. 2005, 8, 1291–1298. [Google Scholar] [CrossRef]
- Bókony, V.; Ujhegyi, N.; Hamow, K.A.; Bosch, J.; Thumsova, B.; Voros, J.; Aspbury, A.S.; Gabor, C.R. Stressed tadpoles mount more efficient glucocorticoid negative feedback in anthropogenic habitats due to phenotypic plasticity. Sci. Total Environ. 2021, 753, 141896. [Google Scholar] [CrossRef] [PubMed]
- Kolonin, A.M.; Bókony, V.; Bonner, T.H.; Zúñiga-Vega, J.J.; Aspbury, A.S.; Guzman, A.; Molina, R.; Calvillo, P.; Gabor, C.R. Coping with urban habitats via glucocorticoid regulation: Physiology, behavior, and life history in stream fishes. Integr. Comp. Biol. 2022, 62, 90–103. [Google Scholar] [CrossRef] [PubMed]
- Blaustein, A.R.; Kiesecker, J.M. Complexity in conservation: Lessons from the global decline of amphibian populations. Ecol. Lett. 2002, 5, 597–608. [Google Scholar] [CrossRef]
- Mann, R.M.; Hyne, R.V.; Choung, C.B.; Wilson, S.P. Amphibians and agricultural chemicals: Review of the risks in a complex environment. Environ. Pollut. 2009, 157, 2903–2927. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.-W.; Huang, B.-H.; Lin, S.-M.; Huang, C.-L.; Liao, P.-C. Changes of diet and dominant intestinal microbes in farmland frogs. BMC Microbiol. 2016, 16, 33. [Google Scholar] [CrossRef] [PubMed]
- Krynak, K.L.; Burke, D.J.; Benard, M.F. Landscape and water characteristics correlate with immune defense traits across Blanchard’s cricket frog (Acris blanchardi) populations. Biol. Conserv. 2016, 193, 153–167. [Google Scholar] [CrossRef]
- Walke, J.B.; Becker, M.H.; Loftus, S.C.; House, L.L.; Cormier, G.; Jensen, R.V.; Belden, L.K. Amphibian skin may select for rare environmental microbes. ISME J. 2014, 8, 2207–2217. [Google Scholar] [CrossRef]
- Kohl, K.D.; Cary, T.L.; Karasov, W.H.; Dearing, M.D. Larval exposure to polychlorinated biphenyl 126 (PCB-126) causes persistent alteration of the amphibian gut microbiota. Environ. Toxicol. Chem. 2015, 34, 1113–1118. [Google Scholar] [CrossRef]
- Ujszegi, J.; Ludányi, K.; Móricz, Á.M.; Krüzselyi, D.; Drahos, L.; Drexler, T.; Németh, M.Z.; Vörös, J.; Garner, T.W.J.; Hettyey, A. Exposure to Batrachochytrium dendrobatidis affects chemical defences in two anuran amphibians, Rana dalmatina and Bufo bufo. BMC Ecol. Evol. 2021, 21, 135. [Google Scholar] [CrossRef]
- Jiménez, R.R.; Sommer, S. The amphibian microbiome: Natural range of variation, pathogenic dysbiosis, and role in conservation. Biodivers. Conserv. 2017, 26, 763–786. [Google Scholar] [CrossRef]
- Tong, Q.; Liu, X.-N.; Hu, Z.-F.; Ding, J.-F.; Bie, J.; Wang, H.-B.; Zhang, J.-T. Effects of Captivity and Season on the Gut Microbiota of the Brown Frog (Rana dybowskii). Front. Microbiol. 2019, 10, 01912. [Google Scholar] [CrossRef] [PubMed]
- Taff, C.C.; Vitousek, M.N. Endocrine Flexibility: Optimizing Phenotypes in a Dynamic World? Trends Ecol. Evol. 2016, 31, 476–488. [Google Scholar] [CrossRef] [PubMed]
- Taff, C.C.; Zimmer, C.; Vitousek, M.N. Efficacy of negative feedback in the HPA axis predicts recovery from acute challenges. Biol. Lett. 2018, 14, 20180131. [Google Scholar] [CrossRef]
- Reading, C.J.; Loman, J.; Madsen, T. Breeding pond fidelity in the common toad, Bufo bufo. J. Zool. 1991, 225, 201–211. [Google Scholar] [CrossRef]
- Sinsch, U. Seasonal changes in the migratory behaviour of the toad Bufo bufo: Direction and magnitude of movements. Oecologia 1988, 76, 390–398. [Google Scholar] [CrossRef] [PubMed]
- Kohl, K.D.; Cary, T.L.; Karasov, W.H.; Dearing, M.D. Restructuring of the amphibian gut microbiota through metamorphosis. Environ. Microbiol. Rep. 2013, 5, 899–903. [Google Scholar] [CrossRef] [PubMed]
- Gosner, K. A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 1960, 16, 183–190. [Google Scholar]
- Narayan, E.J.; Forsburg, Z.R.; Davis, D.R.; Gabor, C.R. Non-invasive Methods for Measuring and Monitoring Stress Physiology in Imperiled Amphibians. Front. Ecol. Evol. 2019, 7, 00431. [Google Scholar] [CrossRef]
- Lattin, C.R.; Kelly, T.R. Glucocorticoid negative feedback as a potential mediator of trade-offs between reproduction and survival. Gen. Comp. Endocrinol. 2020, 286, 113301. [Google Scholar] [CrossRef]
- Wasimuddin; Schlaeppi, K.; Ronchi, F.; Leib, S.L.; Erb, M.; Ramette, A. Evaluation of primer pairs for microbiome profiling from soils to humans within the One Health framework. Mol. Ecol. Resour. 2020, 20, 1558–1571. [Google Scholar] [CrossRef]
- Arango, R.A.; Schoville, S.D.; Currie, C.R.; Carlos-Shanley, C. Experimental Warming Reduces Survival, Cold Tolerance, and Gut Prokaryotic Diversity of the Eastern Subterranean Termite, Reticulitermes flavipes (Kollar). Front. Microbiol. 2021, 12, 632715. [Google Scholar] [CrossRef] [PubMed]
- Fadrosh, D.W.; Ma, B.; Gajer, P.; Sengamalay, N.; Ott, S.; Brotman, R.M.; Ravel, J. An improved dual-indexing approach for multiplexed 16S rRNA gene sequencing on the Illumina MiSeq platform. Microbiome 2014, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Kozich, J.J.; Westcott, S.L.; Baxter, N.T.; Highlander, S.K.; Schloss, P.D. Development of a Dual-Index Sequencing Strategy and Curation Pipeline for Analyzing Amplicon Sequence Data on the MiSeq Illumina Sequencing Platform. Appl. Environ. Microbiol. 2013, 79, 5112–5120. [Google Scholar] [CrossRef]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- McMurdie, P.J.; Holmes, S. Waste not, want not: Why rarefying microbiome data is inadmissible. PLoS Comput Biol. 2014, 10, e1003531. [Google Scholar] [CrossRef] [PubMed]
- Weiss, S.; Xu, Z.Z.; Peddada, S.; Amir, A.; Bittinger, K.; Gonzalez, A.; Lozupone, C.; Zaneveld, J.R.; Vázquez-Baeza, Y.; Birmingham, A.; et al. Normalization and microbial differential abundance strategies depend upon data characteristics. Microbiome 2017, 5, 27. [Google Scholar] [CrossRef] [PubMed]
- Lahti, L.; Shetty, S. Microbiome R package. Bioconductor 2017. [Google Scholar] [CrossRef]
- Pike, N. Using false discovery rates for multiple comparisons in ecology and evolution. Methods Ecol. Evol. 2011, 2, 278–282. [Google Scholar] [CrossRef]
- Stojanov, S.; Berlec, A.; Štrukelj, B. The Influence of Probiotics on the Firmicutes/Bacteroidetes Ratio in the Treatment of Obesity and Inflammatory Bowel disease. Microorganisms 2020, 8, 1715. [Google Scholar] [CrossRef]
- Zuur, A.; Ieno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R.; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Fitzpatrick, B.M.; Allison, A.L. Similarity and differentiation between bacteria associated with skin of salamanders (Plethodon jordani) and free-living assemblages. FEMS Microbiol. Ecol. 2014, 88, 482–494. [Google Scholar] [CrossRef] [PubMed]
- Hughey, M.C.; Walke, J.B.; Becker, M.H.; Umile, T.P.; Burzynski, E.A.; Minbiole, K.P.C.; Iannetta, A.A.; Santiago, C.N.; Hopkins, W.A.; Belden, L.K. Short-Term Exposure to Coal Combustion Waste Has Little Impact on the Skin Microbiome of Adult Spring Peepers (Pseudacris crucifer). Appl. Environ. Microbiol. 2016, 82, 3493–3502. [Google Scholar] [CrossRef] [PubMed]
- Chai, L.; Dong, Z.; Chen, A.; Wang, H. Changes in intestinal microbiota of Bufo gargarizans and its association with body weight during metamorphosis. Arch. Microbiol. 2018, 200, 1087–1099. [Google Scholar] [CrossRef] [PubMed]
- Scalvenzi, T.; Clavereau, I.; Bourge, M.; Pollet, N. Gut microbial ecology of Xenopus tadpoles across life stages. Peer Community J. 2021, 1, e41. [Google Scholar] [CrossRef]
- Bókony, V.; Üveges, B.; Ujhegyi, N.; Verebélyi, V.; Nemesházi, E.; Csíkvári, O.; Hettyey, A. Endocrine disruptors in breeding ponds and reproductive health of toads in agricultural, urban and natural landscapes. Sci. Total Environ. 2018, 634, 1335–1345. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Guo, R.; Yang, Y.; Ding, J.; Zhang, Y. Long-term effect of heavy-metal pollution on diversity of gastrointestinal microbial community of Bufo raddei. Toxicol. Lett. 2016, 258, 192–197. [Google Scholar] [CrossRef]
- Lin, X.; Zhao, J.; Zhang, W.; He, L.; Wang, L.; Chang, D.; Cui, L.; Gao, Y.; Li, B.; Chen, C.; et al. Acute oral methylmercury exposure perturbs the gut microbiome and alters gut-brain axis related metabolites in rats. Ecotoxicol. Environ. Saf. 2020, 190, 110130. [Google Scholar] [CrossRef]
- Ellison, S.; Knapp, R.; Vredenburg, V. Longitudinal patterns in the skin microbiome of wild, individually marked frogs from the Sierra Nevada, California. ISME Commun. 2021, 1, 45. [Google Scholar] [CrossRef]
- Sullam, K.E.; Essinger, S.D.; Lozupone, C.A.; O’Connor, M.P.; Rosen, G.L.; Knight, R.; Kilham, S.S.; Russell, J.A. Environmental and ecological factors that shape the gut bacterial communities of fish: A meta-analysis. Mol. Ecol. 2012, 21, 3363–3378. [Google Scholar] [CrossRef]
- Huang, B.-H.; Chang, C.-W.; Huang, C.-W.; Gao, J.; Liao, P.-C. Composition and Functional Specialists of the Gut Microbiota of Frogs Reflect Habitat Differences and Agricultural Activity. Front. Microbiol. 2018, 8, 02670. [Google Scholar] [CrossRef]
- Goff, C.B.; Walls, S.C.; Rodriguez, D.; Gabor, C.R. Changes in physiology and microbial diversity in larval ornate chorus frogs are associated with habitat quality. Conserv. Physiol. 2020, 8, coaa047. [Google Scholar] [CrossRef] [PubMed]
- Worsley, S.F.; Davies, C.S.; Mannarelli, M.-E.; Hutchings, M.I.; Komdeur, J.; Burke, T.; Dugdale, H.L.; Richardson, D.S. Gut microbiome composition, not alpha diversity, is associated with survival in a natural vertebrate population. Anim. Microbiome 2021, 3, 84. [Google Scholar] [CrossRef] [PubMed]
- Bókony, V.; Üveges, B.; Verebélyi, V.; Ujhegyi, N.; Móricz, Á.M. Toads phenotypically adjust their chemical defences to anthropogenic habitat change. Sci. Rep. 2019, 9, 3163. [Google Scholar] [CrossRef] [PubMed]
- Stothart, M.R.; Bobbie, C.B.; Schulte-Hostedde, A.I.; Boonstra, R.; Palme, R.; Mykytczuk, N.C.; Newman, A.E. Stress and the microbiome: Linking glucocorticoids to bacterial community dynamics in wild red squirrels. Biol. Lett. 2016, 12, 20150875. [Google Scholar] [CrossRef]
- Vlčková, K.; Shutt-Phillips, K.; Heistermann, M.; Pafčo, B.; Petrželková, K.J.; Todd, A.; Modrý, D.; Nelson, K.E.; Wilson, B.A.; Stumpf, R.M. Impact of stress on the gut microbiome of free-ranging western lowland gorillas. Microbiology 2018, 164, 40–44. [Google Scholar] [CrossRef]
- Glennemeier, K.A.; Denver, R.J. Small changes in whole-body corticosterone content affect larval Rana pipiens fitness components. Gen. Comp. Endocrinol. 2002, 127, 16–25. [Google Scholar] [CrossRef]
- Romero, L.M.; Wikelski, M. Corticosterone levels predict survival probabilities of Galapagos marine iguanas during El Nino events. Proc. Natl. Acad. Sci. USA 2001, 98, 7366–7370. [Google Scholar] [CrossRef]
- Suzuki, T.A. Links between Natural Variation in the Microbiome and Host Fitness in Wild Mammals. Integr. Comp. Biol. 2017, 57, 756–769. [Google Scholar] [CrossRef]
- Chong , J.; Liu, P.; Zhou, G.; Xia, J. Using MicrobiomeAnalyst for comprehensive statistical, functional, and meta-analysis of microbiome data. Nat. Protoc. 2020, 15, 799–821. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gabor, C.R.; Villatoro-Castañeda, M.; Carlos-Shanley, C.; Ujhegyi, N.; Bókony, V. Gut Bacterial Communities Vary across Habitats and Their Diversity Increases with Increasing Glucocorticoids in Toad Tadpoles. Diversity 2023, 15, 23. https://doi.org/10.3390/d15010023
Gabor CR, Villatoro-Castañeda M, Carlos-Shanley C, Ujhegyi N, Bókony V. Gut Bacterial Communities Vary across Habitats and Their Diversity Increases with Increasing Glucocorticoids in Toad Tadpoles. Diversity. 2023; 15(1):23. https://doi.org/10.3390/d15010023
Chicago/Turabian StyleGabor, Caitlin R., Melissa Villatoro-Castañeda, Camila Carlos-Shanley, Nikolett Ujhegyi, and Veronika Bókony. 2023. "Gut Bacterial Communities Vary across Habitats and Their Diversity Increases with Increasing Glucocorticoids in Toad Tadpoles" Diversity 15, no. 1: 23. https://doi.org/10.3390/d15010023
APA StyleGabor, C. R., Villatoro-Castañeda, M., Carlos-Shanley, C., Ujhegyi, N., & Bókony, V. (2023). Gut Bacterial Communities Vary across Habitats and Their Diversity Increases with Increasing Glucocorticoids in Toad Tadpoles. Diversity, 15(1), 23. https://doi.org/10.3390/d15010023







