Endemic and Threatened Amazona Parrots of the Atlantic Forest: An Overview of Their Geographic Range and Population Size
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Data Collection
2.2. Data Analysis
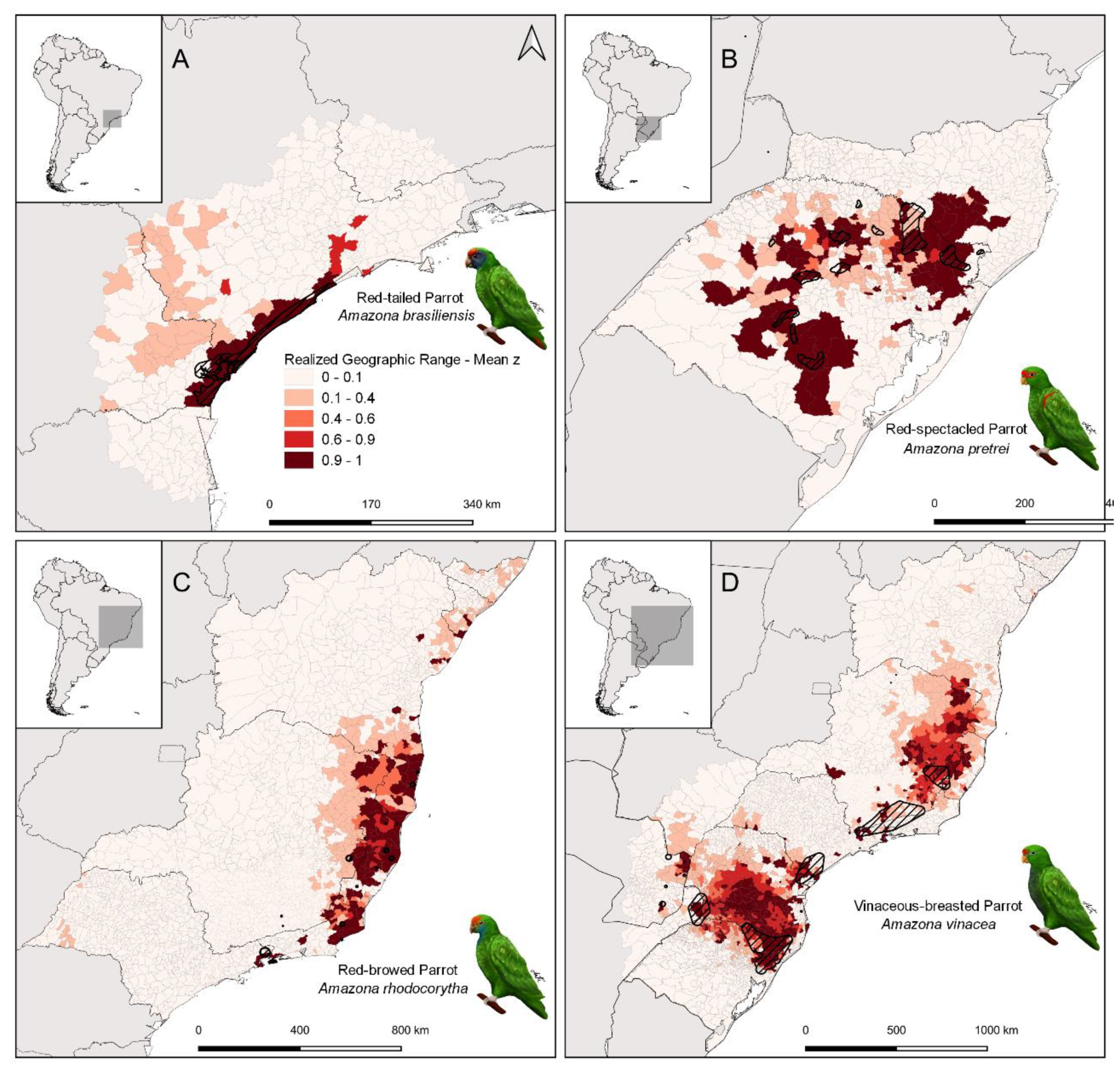
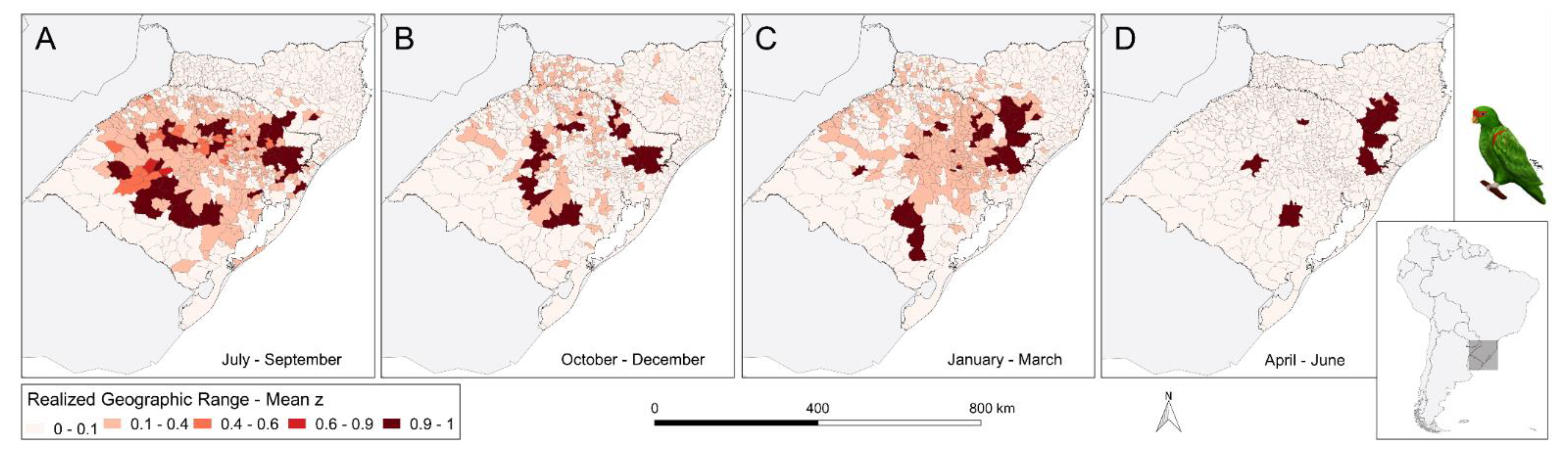
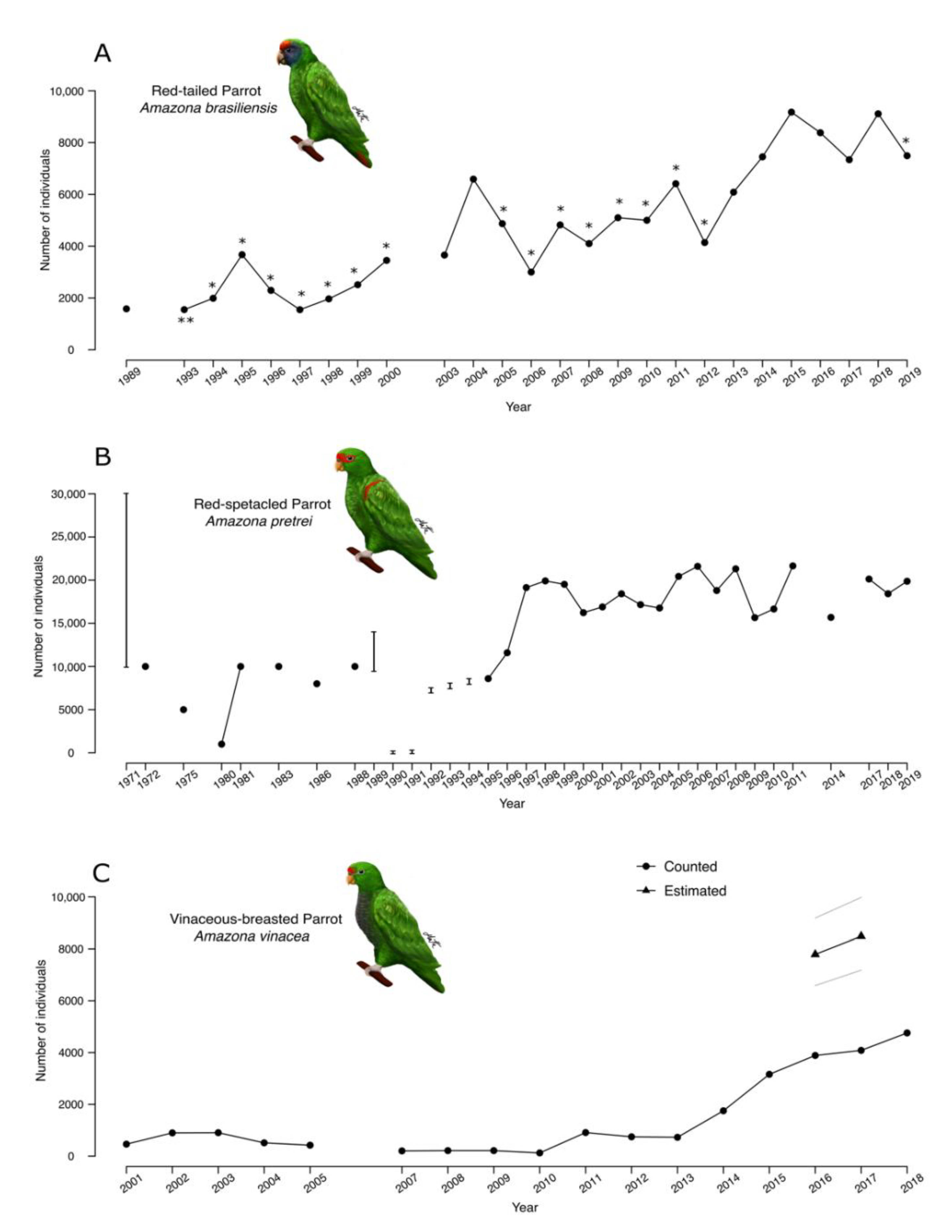
3. Results
4. Discussion
4.1. Geographic Range
4.2. Population Size
4.3. Seasonal Change in Geographic Range
4.4. Long-Term Changes in Geographic Range
4.5. Long-Term Change in Population Size
4.6. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- BirdLife International. The IUCN Red List of Threatened Species. 2021. Available online: https://www.iucnredlist.org/ (accessed on 2 February 2020).
- Collar, N.J.; Juniper, A.T. Dimensions and Causes of the Parrot Conservation Crisis. In New World Parrots in Crisis: Solutions from Conservation Biology; Smithsonian Institution Press: Washington, DC, USA, 1992. [Google Scholar]
- Juniper, T.; Parr, M. Parrots: A Guide to the Parrots of the World; Yale University Press: New Haven, CT, USA; London, UK, 1998. [Google Scholar]
- Cockle, K.L.; Martin, K.; Drever, M.C. Supply of Tree-Holes Limits Nest Density of Cavity-Nesting Birds in Primary and Logged Subtropical Atlantic Forest. Biol. Conserv. 2010, 143, 2851–2857. [Google Scholar] [CrossRef]
- Foley, J.A. Global Consequences of Land Use. Science 2005, 309, 570–574. [Google Scholar] [CrossRef]
- Berkunsky, I.; Quillfeldt, P.; Brightsmith, D.J.; Abbud, M.C.; Aguilar, J.M.R.E.; Alemán-Zelaya, U.; Aramburú, R.M.; Arce Arias, A.; Balas McNab, R.; Balsby, T.J.S.; et al. Current Threats Faced by Neotropical Parrot Populations. Biol. Conserv. 2017, 214, 278–287. [Google Scholar] [CrossRef]
- Tella, J.L.; Hiraldo, F. Illegal and Legal Parrot Trade Shows a Long-Term, Cross-Cultural Preference for the Most Attractive Species Increasing Their Risk of Extinction. PLoS ONE 2014, 9, e107546. [Google Scholar] [CrossRef] [PubMed]
- Wright, T.F.; Toft, C.A.; Enkerlin-Hoeflich, E.; Gonzalez-Elizondo, J.; Albornoz, M.; Rodríguez-Ferraro, A.; Rojas-Suárez, F.; Sanz, V.; Trujillo, A.; Beissinger, S.R.; et al. Nest Poaching in Neotropical Parrots. Conserv. Biol. 2001, 15, 710–720. [Google Scholar] [CrossRef]
- del Hoyo, J.; Elliott, A.; Sargatal, J.; Christie, D.A.; de Juana, E. Handbook of the Birds of the World Alive; Lynx Edicions: Barcelona, Spain, 2017. [Google Scholar]
- Ribeiro, M.C.; Martensen, A.C.; Metzger, J.P.; Tabarelli, M.; Scarano, F.R.; Fortin, M.-J. The Brazilian Atlantic Forest: A Shrinking Biodiversity Hotspot. In Biodiversity Hotspots; Zachos, F.E., Habel, J.C., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; ISBN 978-3-642-20991-8. [Google Scholar]
- Tabarelli, M.; Aguiar, A.V.; Ribeiro, M.C.; Metzger, J.P.; Peres, C.A. Prospects for Biodiversity Conservation in the Atlantic Forest: Lessons from Aging Human-Modified Landscapes. Biol. Conserv. 2010, 143, 2328–2340. [Google Scholar] [CrossRef]
- Ribeiro, M.C.; Metzger, J.P.; Martensen, A.C.; Ponzoni, F.J.; Hirota, M.M. The Brazilian Atlantic Forest: How Much Is Left, and How Is the Remaining Forest Distributed? Implications for Conservation. Biol. Conserv. 2009, 142, 1141–1153. [Google Scholar] [CrossRef]
- Powers, R.P.; Jetz, W. Global Habitat Loss and Extinction Risk of Terrestrial Vertebrates under Future Land-Use-Change Scenarios. Nat. Clim. Chang. 2019, 9, 323–329. [Google Scholar] [CrossRef]
- Vergara-Tabares, D.L.; Cordier, J.M.; Landi, M.A.; Olah, G.; Nori, J. Global Trends of Habitat Destruction and Consequences for Parrot Conservation. Glob. Chang. Biol. 2020, 26, 4251–4262. [Google Scholar] [CrossRef] [PubMed]
- Vale, M.M.; Tourinho, L.; Lorini, M.L.; Rajão, H.; Figueiredo, M.S.L. Endemic Birds of the Atlantic Forest: Traits, Conservation Status, and Patterns of Biodiversity. J. Field Ornithol. 2018, 89, 193–206. [Google Scholar] [CrossRef]
- Mace, G.M.; Collar, N.J.; Gaston, K.J.; Hilton-Taylor, C.; AkçAkaya, H.R.; Leader-Williams, N.; Milner-Gulland, E.J.; Stuart, S.N. Quantification of Extinction Risk: IUCN’s System for Classifying Threatened Species. Conserv. Biol. 2008, 22, 1424–1442. [Google Scholar] [CrossRef] [PubMed]
- Zulian, V.; Miller, D.A.W.; Ferraz, G. Integrating citizen‐science and planned‐survey data improves species distribution estimates. Divers. Distrib. 2021. [Google Scholar] [CrossRef]
- eBird. Available online: https://ebird.org/home (accessed on 2 February 2020).
- WikiAves. WikiAves, a Enciclopédia Das Aves Do Brasil. Available online: http://www.wikiaves.com.br (accessed on 8 January 2020).
- Xeno-Canto. Available online: https://www.xeno-canto.org/ (accessed on 9 January 2019).
- Zulian, V.; Müller, E.S.; Cockle, K.L.; Lesterhuis, A.; Tomasi Júnior, R.; Prestes, N.P.; Martinez, J.; Kéry, M.; Ferraz, G. Addressing Multiple Sources of Uncertainty in the Estimation of Global Parrot Abundance from Roost Counts: A Case Study with the Vinaceous-Breasted Parrot (Amazona vinacea). Biol. Conserv. 2020, 248, 108672. [Google Scholar] [CrossRef]
- MacKenzie, D.I.; Nichols, J.D.; Lachman, G.B.; Droege, S.; Andrew Royle, J.; Langtimm, C.A. Estimating Site Occupancy Rates When Detection Probabilities Are Less than One. Ecology 2002, 83, 2248–2255. [Google Scholar] [CrossRef]
- Collar, N.; Boesman, P.F.D. Red-spectacled Parrot (Amazona pretrei). In Birds of the World; Billerman, S.M., Keeney, B.K., Rodewald, P.G., Schulenberg, T.S., Eds.; Cornell Lab of Ornithology: Ithaca, NY, USA, 2020. [Google Scholar]
- Collar, N.; Boesman, P.F.D.; Sharpe, C. Red-browed Parrot (Amazona rhodocorytha). In Birds of the World; Billerman, S.M., Keeney, B.K., Rodewald, P.G., Schulenberg, T.S., Eds.; Cornell Lab of Ornithology: Ithaca, NY, USA, 2020. [Google Scholar]
- Collar, N.; Boesman, P.F.D.; Sharpe, C. Red-tailed Parrot (Amazona brasiliensis). In Birds of the World; Billerman, S.M., Keeney, B.K., Rodewald, P.G., Schulenberg, T.S., Eds.; Cornell Lab of Ornithology: Ithaca, NY, USA, 2020. [Google Scholar]
- Prestes, N.P.; Martinez, J.; Peres, A.R. Dieta alimentar do papagaio-charão (Amazona pretrei). In Biologia Da Conservação: Estudo de Caso com Papagaio-Charão e Outros Papagaios Brasileiros; Martinez, J., Prestes, N.P., Eds.; UPF Editora: Passo Fundo, RS, Brazil, 2008. [Google Scholar]
- Prestes, N.P.; Martinez, J.; Kilpp, J.C.; Batistela, T.; Turkievicz, A.; Rezende, É.; Gaboardi, V.T.R. Ecologia e Conservação de Amazona vinacea Em Áreas Simpátricas Com Amazona pretrei. Ornithologia 2014, 6, 109–120. [Google Scholar]
- Scherer-Neto, P. Contribuição à Biologia do Papagaio-de-Cara-Roxa Amazona Brasiliensis (Linnaeus, 1758) (Psittacidae, Aves). Master’s Thesis, Universidade Federal do Paraná, Curitiba, PR, Brazil, 1989. [Google Scholar]
- Martuscelli, P. Ecology and Conservation of the Red-Tailed Amazon Amazona brasiliensis in South-Eastern Brazil. Bird Conserv. Int. 1995, 5, 405–420. [Google Scholar] [CrossRef]
- IBGE Mapa de Vegetação. 2012. Available online: https://www.ibge.gov.br (accessed on 30 June 2021).
- Hijmans, R.J.; Guarino, L.; Cruz, M.; Rojas, E. Computer Tools for Spatial Analysis of Plant Genetic Resources Data: 1. DIVA-GIS. Plant Genet. Resour. Newsl. 2001, 127, 15–19. [Google Scholar]
- Pacifici, K.; Reich, B.J.; Miller, D.A.W.; Gardner, B.; Stauffer, G.; Singh, S.; McKerrow, A.; Collazo, J.A. Integrating Multiple Data Sources in Species Distribution Modeling: A Framework for Data Fusion. Ecology 2017, 98, 840–850. [Google Scholar] [CrossRef]
- Miller, D.A.W.; Pacifici, K.; Sanderlin, J.S.; Reich, B.J. The Recent Past and Promising Future for Data Integration Methods to Estimate Species’ Distributions. Methods Ecol. Evol. 2019, 10, 22–37. [Google Scholar] [CrossRef]
- Stauffer, G.E.; Miller, D.A.W.; Williams, L.M.; Brown, J. Ruffed Grouse Population Declines after Introduction of West Nile Virus. Jour. Wild. Mgmt. 2018, 82, 165–172. [Google Scholar] [CrossRef]
- Lunn, D.J.; Thomas, A.; Best, N.; Spiegelhalter, D. WinBUGS-a Bayesian Modelling Framework: Concepts, Structure, and Extensibility. Stat. Comput. 2000, 10, 325–337. [Google Scholar] [CrossRef]
- Forshaw, J.M.; Cooper, W.T. Parrots of the World, 2nd ed.; Lansdowne Press: Melbourne, Australia, 1978. [Google Scholar]
- Varty, N.; Bencke, G.A.; Bernardini, L.M.; Cunha, A.S.; Dias, E.V.; Fontana, C.S.; Guadagnin, D.L.; Kindel, A.; Kindel, E.; Raymundo, M.M.; et al. Conservação Do Papagaio-Charão Amazona pretrei No Sul Do Brasil: Um Plano de Ação Preliminar; Divulgações do Museu de Ciências e Tecnologia UBEA/PUCRS: EDIPUCRS: Porto Alegre, RS, Brazil, 1994. [Google Scholar]
- Belton, W. Birds of Rio Grande Do Sul, Brazil. Part 1. Rheidae through Furnariidae; Bulletin of the American Museum of Natural History: New York, NY, USA, 1984; Volume 178. [Google Scholar]
- Martinez, J.; Prestes, N.P. Tamanho populacional, tamanho médio de bando e outros aspectos demográficos do papagaio-charão (Amazona pretrei). In Biologia da Conservação: Estudo de Caso Com Papagaio-Charão e Outros Papagaios Brasileiros; Martinez, J., Prestes, N.P., Eds.; UPF Editora: Passo Fundo, RS, Brazil, 2008. [Google Scholar]
- Tella, J.L.; Dénes, F.V.; Zulian, V.; Prestes, N.P.; Martínez, J.; Blanco, G.; Hiraldo, F. Endangered Plant-Parrot Mutualisms: Seed Tolerance to Predation Makes Parrots Pervasive Dispersers of the Parana Pine. Sci. Rep. 2016, 6, 31709. [Google Scholar] [CrossRef]
- Schunck, F.; Somenzari, M.; Lugarini, C.; Soares, E.S. Plano de Ação Nacional Para a Conservação Dos Papagaios Da Mata Atlântica; Instituto Chico Mendes de Conservação da Biodiversidade-ICMBio: Brasília, DF, Brazil, 2011. [Google Scholar]
- ICMBio. Plano de Ação Nacional Para a Conservação Dos Papagaios—PAN Papagaios: Matriz de Monitoria. 2020. Available online: https://www.icmbio.gov.br/portal/faunabrasileira/plano-de-acao-nacional-lista/837-plano-de-acao-nacional-para-conservacao-dos-papagaios-da-mata-atlantica (accessed on 2 April 2020).
- SPVS Relatório Anual 2019; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, Brazil, 2019; p. 52.
- Scherer-Neto, P.; Toledo, M.C. Avaliação Populacional Do Papagaio-de-Cara-Roxa (Amazona brasiliensis) (Psittacidae) No Estado Do Paraná, Brasil. Ornitol. Neotrop. 2007, 18, 379–393. [Google Scholar]
- Galetti, M.; Schunck, F.; Ribeiro, M.; Paiva, A.A.; Toledo, R.; Fonseca, L. Distribuição e Tamanho Populacional Do Papagaio-de-Cara-Roxa Amazona brasiliensis No Estado de São Paulo. Rev. Bras. Ornitol. 2006, 14, 239–247. [Google Scholar]
- SPVS Relatório Anual 2003; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2003; p. 20.
- SPVS Relatório Anual 2004; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2004; p. 16.
- SPVS Relatório Anual 2005; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2005; p. 8.
- SPVS Relatório Anual 2006; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2006; p. 12.
- SPVS Relatório Anual 2007; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2007; p. 16.
- SPVS Relatório Anual 2008; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2008; p. 12.
- SPVS Relatório Anual 2009; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2009; p. 6.
- SPVS Relatório Anual 2010; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2010; p. 4.
- SPVS Relatório Anual 2011; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2011; p. 13.
- SPVS Relatório Anual 2012; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2012; p. 38.
- SPVS Relatório Anual 2013; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2013; p. 27.
- SPVS Relatório Anual 2014; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2014; p. 27.
- SPVS Relatório Anual 2015; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2015; p. 35.
- SPVS Relatório Anual 2016; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2016; p. 52.
- SPVS Relatório Anual 2017; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2017; p. 48.
- SPVS Relatório Anual 2018; Sociedade de Pesquisa em Vida Selvagem e Educação Ambiental: Curitiba, PR, Brazil, 2018; p. 44.
- Abe, L.M. Caracterização do Hábitat do Papagaio-de-Peito-Roxo Amazona vinacea (Kuhl, 1820) No Município de Tunas do Paraná, Região Metropolitana de Curitiba, Paraná. Master’s Thesis, Universidade Federal do Paraná, Curitiba, PR, Brazil, 2004. [Google Scholar]
- Cockle, K.; Capuzzi, G.; Bodrati, A.; Clay, R.; del Castillo, H.; Velázquez, M.; Areta, J.I.; Fariña, N.; Fariña, R. Distribution, Abundance, and Conservation of Vinaceous Amazons (Amazona vinacea) in Argentina and Paraguay. J. Field Ornithol. 2007, 78, 21–39. [Google Scholar] [CrossRef]
- Segovia, J.M.; Cockle, K.L. Conservación del Loro Vinoso (Amazona vinacea) En Argentina. El Hornero 2012, 27, 27–37. [Google Scholar]
- Marsden, S.J.; Whiffin, M.; Sadgrove, L.; Guimarães, P. Parrot Populations and Habitat Use in and around Two Lowland Atlantic Forest Reserves, Brazil. Biol. Conserv. 2000, 96, 209–217. [Google Scholar] [CrossRef]
- Klemann-Júnior, L.; Neto, P.S.; Monteiro, T.V.; Ramos, M.; de Almeida, R. Mapeamento da distribuição e conservação do chauá (Amazona rhodocorytha) no estado do Espírito Santo, Brasil. Ornitol. Neotrop. 2008, 19, 14. [Google Scholar]
- Silva, F. Contribuição Ao Conhecimento Da Biologia Do Papagaio Charão, Amazona pretrei (TEMMINCK, 1830) (Psittacidae, Aves). Iheringia Sér. Zool. 1981, 58, 79–85. [Google Scholar]
- Gaston, K.J.; Fuller, R.A. The Sizes of Species’ Geographic Ranges. J. Appl. Ecol. 2009, 46, 1–9. [Google Scholar] [CrossRef]
- Prestes, N.P.; Martinez, J.; Meyrer, P.A.; Hansen, L.H.; de Negri Xavier, M. Nest Characteristics of the Red-Spectacled Amazon Amazona pretrei Temminck, 1830 (Psittacidae). Ararajuba 1997, 5, 151–158. [Google Scholar]
- Prestes, N.P.; Martinez, J.; Rezende, É. Biologia reprodutiva do papagaio-charão (Amazona pretrei). In Biologia Da Conservação: Estudo de Caso com Papagaio-Charão e Outros Papagaios Brasileiros; Martinez, J., Prestes, N.P., Eds.; Editora UPF: Passo Fundo, RS, Brazil, 2008; Volume 1. [Google Scholar]
- Forshaw, J.M. Parrots of the World; Princeton Field Guides; Princeton University Press: Princeton, NJ, USA, 2010; ISBN 978-0-691-14285-2. [Google Scholar]
- BirdLife International IUCN Red List of Threatened Species: Amazona Vinacea. 2017. Available online: http://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T22686374A93109194.en (accessed on 18 March 2019).
- BirdLife International IUCN Red List of Threatened Species: Amazona Rhodocorytha. 2017. Available online: https://dx.doi.org/10.2305/IUCN.UK.2017-3.RLTS.T22686288A118968809.en (accessed on 18 March 2019).
- ICMBio. Livro Vermelho da Fauna Brasileira Ameaçada de Extinção: Volume III—Aves. In Livro Vermelho da Fauna Brasileira Ameaçada de Extinção; Instituto Chico Mendes de Conservação da Biodiversidade. (Org.): Brasília, DF, Brazil, 2018; Volume 3, p. 712. [Google Scholar]
- BirdLife International IUCN Red List of Threatened Species: Amazona Pretrei. 2016. Available online: https://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T22686251A93104759.en (accessed on 18 March 2019).
- Parrots: Status Survey and Conservation Action Plan, 2000–2004; Snyder, N.F.R., McGowan, P., Gilardi, J., Grajal, A., Eds.; IUCN/SSC Action Plans for the Conservation of Biological Diversity; IUCN: Gland, Switzerland, 2000; ISBN 978-2-8317-0504-0. [Google Scholar]
- BirdLife International IUCN Red List of Threatened Species: Amazona Brasiliensis. 2017. Available online: https://dx.doi.org/10.2305/IUCN.UK.2017-3.RLTS.T22686296A118478685.en (accessed on 27 April 2021).
- Holt, A.R.; Gaston, K.J.; He, F. Occupancy-Abundance Relationships and Spatial Distribution: A Review. Basic Appl. Ecol. 2002, 3, 1–13. [Google Scholar] [CrossRef]
- Sipinski, E.A.B.; Abbud, M.C.; Sezerban, R.M.; Serafini, P.P.; Boçon, R.; Manica, L.T.; de Camargo Guaraldo, A. Tendência Populacional Do Papagaio-de-Cara-Roxa (Amazona brasiliensis) No Litoral Do Estado Do Paraná. Ornithologia 2014, 6, 136–143. [Google Scholar]
- Marini, M.Â.; Barbet-Massin, M.; Martinez, J.; Prestes, N.P.; Jiguet, F. Applying Ecological Niche Modelling to Plan Conservation Actions for the Red-Spectacled Amazon (Amazona pretrei). Biol. Conserv. 2010, 143, 102–112. [Google Scholar] [CrossRef]
- Martinez, J.; Prestes, N.P. Um pouco da história do papagaio-charão (Amazona pretrei). In Biologia da Conservação: Estudo de Caso Com Papagaio-Charão E Outros Papagaios Brasileiros; Martinez, J., Prestes, N.P., Eds.; UPF Editora: Passo Fundo, RS, Brazil, 2008. [Google Scholar]
- Martinez, J.; Prestes, N.P.; Rezende, É. As ameaças enfrentadas pelo papagaio-charão (Amazona pretrei). In Biologia Da Conservação: Estudo de Caso com Papagaio-Charão e Outros Papagaios Brasileiros.; Martinez, J., Prestes, N.P., Eds.; Editora UPF: Passo Fundo, RS, Brazil, 2008. [Google Scholar]
- Ministerio de Ambiente e Desarollo Sustentable. Aves Argentinas. In Categorizacion de Las Aves de La Argentina Según Su Estado de Conservación; Informe del Ministerio de Ambiente y Desarrollo Sustentable de la Nación y de Aves Argentinas: Buenos Aires, Argentina, 2015. [Google Scholar]
- BirdLife International Handbook of the Birds of the World Amazona Pretrei. 2016. Available online: https://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T22686251A93104759.en (accessed on 18 March 2019).
- Bodrati, A.; Cockle, K. New Records of Rare and Threatened Birds from the Atlantic Forest of Misiones, Argentina. Cotinga 2006, 26, 20–24. [Google Scholar]
- Brooks, M.; Clay, R.P.; Lowen, C.; Butchart, H.M.; Barnes, R.; Esquivel, E.Z.; Etcheverry, N.I.; Vincent, J.P. New Information on Nine Birds from Paraguay. Ornitol. Neotrop. 1995, 6, 129–134. [Google Scholar]
- Ministerio del Ambiente y Desarrollo Sostenible. Resolución n 254/19 Por La Cual Se Actualiza El Listado de Las Especies Protegidas de La Vida Silvestre de La Classe Aves; Gobierno Nacional: Asunción, Paraguay, 2019. [Google Scholar]
- Alerstam, T. Ecological Causes and Consequences of Bird Orientation. Experientia 1990, 46, 405–415. [Google Scholar] [CrossRef]
- Roda, S.A. Distribuição de aves Endêmicas e Ameaçadas em Usinas de Açúcar e Unidades de Conservação do Centro Pernambuco; Centro de Pesquisas Ambientais do Nordeste: Recife, PE, Brazil, 2005; p. 42. [Google Scholar]
- Katayama, M.V.; Zima, P.V.Q.; Perrella, D.F.; Francisco, M.R. Successional Stage Effect on the Availability of Tree Cavities for Cavity-Nesting Birds in an Atlantic Forest Park from the State of São Paulo, Brazil. Biota Neotrop. 2017, 17. [Google Scholar] [CrossRef]
- Baptista, S.R.; Rudel, T.K. A Re-Emerging Atlantic Forest? Urbanization, Industrialization and the Forest Transition in Santa Catarina, Southern Brazil. Environ. Conserv. 2006, 33, 195. [Google Scholar] [CrossRef]
- Bonfanti, T.; Meurer, C.; Martinez, J.; Prestes, N.P. A captura de papagaios: Espécies encontradas em cativeiro no norte e nordeste do Rio Grande do Sul. In Biologia Da Conservação: Estudo de Caso com Papagaio-Charão e Outros Papagaios Brasileiros; Martinez, J., Prestes, N.P., Eds.; Editora UPF: Passo Fundo, RS, Brazil, 2008. [Google Scholar]
- Martuscelli, P. A Parrot with a Tiny Distribution and a Big Problem: Will Illegal Trade Wipe out the Red-Tailed Amazon? PsittaScene 1994, 6, 3–7. [Google Scholar]
- de Cavalheiro, M.L. Qualidade do Ambiente e Características Fisiológicas do Papagaio-de-Cara-Roxa (Amazona brasiliensis) Ilha Comprida-São Paulo. Master’s Thesis, Universidade Federal do Paraná UFPR, Curitiba, PR, Brazil, 1999. [Google Scholar]
- Carrillo, A.C.; Batista, D.B. A conservação do papagaio-da-cara-roxa (Amazona brasiliensis) no estado do Paraná: Uma experiência de Educação Ambiental no ensino formal. Rev. Árvore 2007, 31, 113–122. [Google Scholar] [CrossRef]
- Cordeiro, P.H.C. A fragmentação da Mata Atlântica no sul da Bahia e suas implicaçãoes na conservação dos psitacídeos. In Ecologia e Conservação de Psitacídeos no Brasil; Melopsittacus Publicações Científicas: Belo Horizonte, MG, Brazil, 2002; p. 236. [Google Scholar]
| Species | Sample Size | Coverage % | ndet | nmuni | IUCN Extant Area (km2) | Estimated Geographic Range (km2) |
|---|---|---|---|---|---|---|
| Amazona brasiliensis (Red-tailed Parrot) | 16,705 | 99.7 | 192 | 15 | 4750 | 15,627 ± 8843 |
| (3127–31,414) | ||||||
| Amazona pretrei (Red-spectacled Parrot) | 5477 | 92.7 | 187 | 73 | 10,430 | 66,203 ± 11,425 |
| (45,727–90,367) | ||||||
| Amazona rhodocorytha (Red-browed Parrot) | 30,867 | 94.2 | 346 | 86 | 2672 | 134,355 ± 13,922 |
| (109,288–162,828) | ||||||
| Amazona vinacea (Vinaceous-breasted Parrot) | 47,240 | 91.9 | 1007 | 339 | 145,700 | 434,670 ± 28,911 (382,887–496,550) |
| Species | Atlantic Forest | Dense Forest | Araucaria Forest | Altitude |
|---|---|---|---|---|
| Amazona brasiliensis | −0.63 ± 2.23 (−5.39–3.48) | −1.33 ± 3.14 (−7.71–4.77) | — | 0.23 ± 0.44 (−0.72–0.94) |
| Amazona pretrei | −0.55 ± 1.08 (−2.70–1.56) | — | 0.47 ± 1.05 (−1.55–2.53) | 0.15 ± 0.18 (−0.21–0.52) |
| Amazona rhodocorytha | 0.84 ± 0.91 (−1.70–1.84) | — | — | −0.14 ± 0.20 (−0.51–0.25) |
| Amazona vinacea | 2.11 ± 0.86 (0.37–3.79) | — | 2.13 ± 0.98 (0.29–4.10) | 0.85 ± 0.12 (0.58–1.05) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zulian, V.; Miller, D.A.W.; Ferraz, G. Endemic and Threatened Amazona Parrots of the Atlantic Forest: An Overview of Their Geographic Range and Population Size. Diversity 2021, 13, 416. https://doi.org/10.3390/d13090416
Zulian V, Miller DAW, Ferraz G. Endemic and Threatened Amazona Parrots of the Atlantic Forest: An Overview of Their Geographic Range and Population Size. Diversity. 2021; 13(9):416. https://doi.org/10.3390/d13090416
Chicago/Turabian StyleZulian, Viviane, David A. W. Miller, and Gonçalo Ferraz. 2021. "Endemic and Threatened Amazona Parrots of the Atlantic Forest: An Overview of Their Geographic Range and Population Size" Diversity 13, no. 9: 416. https://doi.org/10.3390/d13090416
APA StyleZulian, V., Miller, D. A. W., & Ferraz, G. (2021). Endemic and Threatened Amazona Parrots of the Atlantic Forest: An Overview of Their Geographic Range and Population Size. Diversity, 13(9), 416. https://doi.org/10.3390/d13090416






