Dittrichia viscosa: Native-Non Native Invader
Abstract
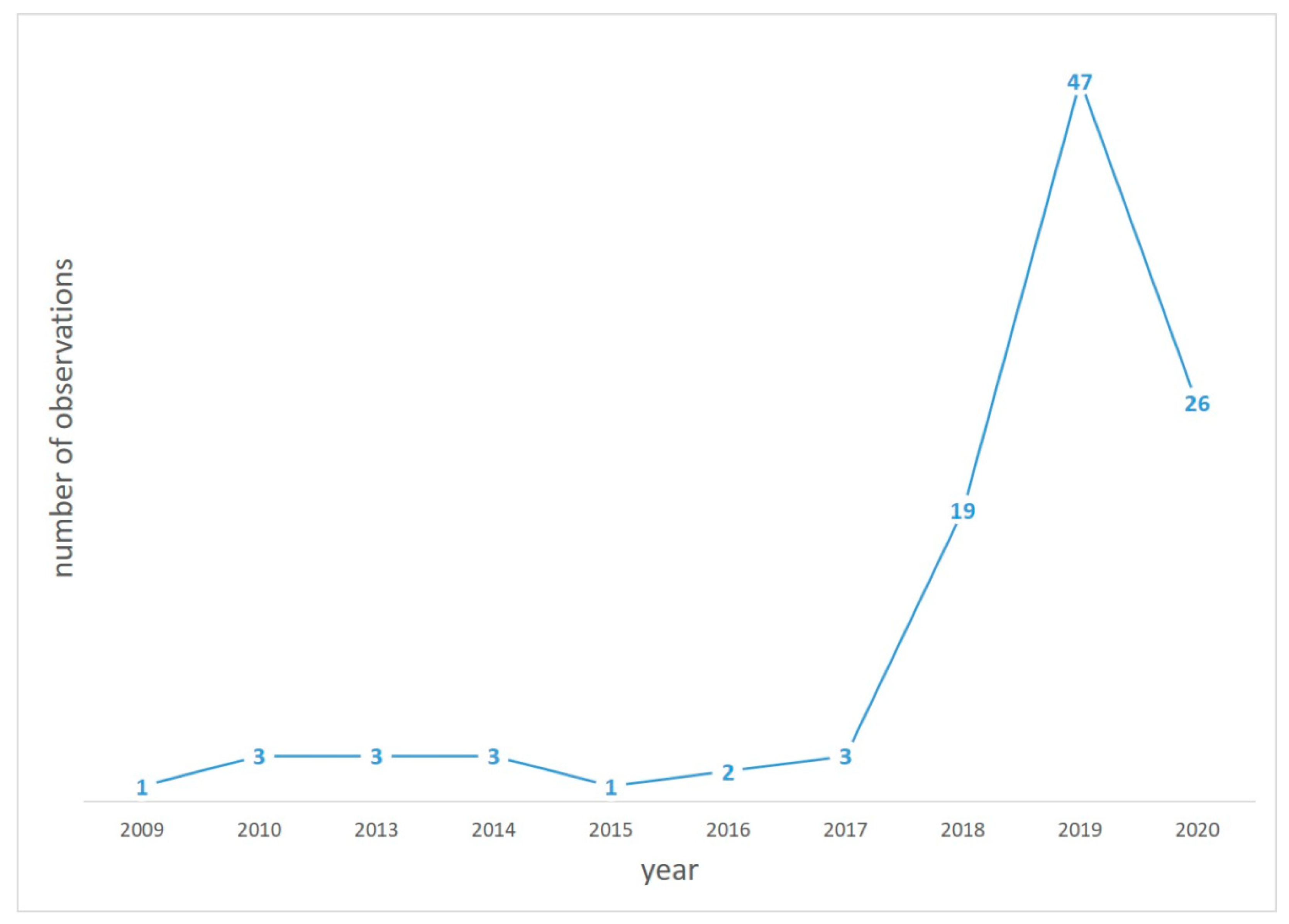
:1. Introduction
2. Dittrichia viscosa Invasive Properties
2.1. D. viscosa in Native Areas
2.1.1. Biology
2.1.2. Reproduction, Regeneration, and Dispersal
2.1.3. Allelopathy
2.1.4. Tolerance of a Broad Range of Environmental Conditions
2.1.5. Lack of Predators
2.2. D. viscosa in New Areas
3. Dittrichia viscosa Ecosystem Services
4. Plant Chemistry as a Competitive Advantage and Potential Ecosystem Service
5. Invasive Native-Non Native D. viscosa
6. Species Management
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Parolin, P.; Scotta, I.; Bresch, C. Biology of Dittrichia viscosa, a Mediterranean ruderal plant: A review. Phyton 2014, 83, 251–262. [Google Scholar]
- Ounoughi, A.; Ramdani, M.; Lograda, T.; Chalard, P.; Figuérédo, G. Chemotypes and antibacterial activities of Inula viscosa essential oils from Algeria. Biodiversitas J. Biol. Divers. 2020, 21, 1504–1517. [Google Scholar] [CrossRef]
- Glanznig, A.; Kessal, O. Invasive Plants of National Importance and Their Legal Status by State and Territory; WWF-Australia: Sydney, Australia, 2004; pp. 19–21. [Google Scholar]
- Araniti, F.; Lupini, A.; Sunseri, F.; Abenavoli, M.R. Allelopatic Potential of Dittrichia viscosa (L.) W. Greuter Mediated by VOCs: A Physiological and Metabolomic Approach. PLoS ONE 2017, 12, e0170161. [Google Scholar] [CrossRef] [Green Version]
- Šugar, I. Vegetacijska Karta SR Hrvatske List 77; Botanički zavod PMF: Pula, Hrvatska, 1978. [Google Scholar]
- GBIF Secretariat. GBIF Backbone Taxonomy. 2021. Checklist Dataset. Available online: https://www.gbif.org/species/3101184 (accessed on 2 August 2021).
- Di Pietro, R.; Germani, D.; Fortini, P. A phytosociological investigation on the mixed hemycryptophitic and therophitic grasslands of the Cornicolani mountains (Lazio Region-central Italy). Plant Sociol. 2017, 54, 107–128. [Google Scholar] [CrossRef]
- Hulme, P. Weed risk assessment: A way forward or a waste of time? J. Appl. Ecol. 2011, 49, 10–19. [Google Scholar] [CrossRef]
- Grašič, M.; Anžlovar, S.; Strgulc Krajšek, S. Germination rate of stinkwort (Dittrichia graveolens (L.) Greuter) and false yellowhead (D. viscosa (L.) Greuter) in relation to salinity and the impact of their extracts on germination of selected plant species. In Proceedings of the 6th Slovenian Symposium on Plant Biology, Hoče, Maribor, Slovenia, 12 September 2014. [Google Scholar]
- Cramer, V.; Hobbs, J.R.; Standish, R. What’s new about old fields? Land abandonment and ecosystem assembly. Ecol. Evol. 2008, 23, 104–112. [Google Scholar] [CrossRef]
- Seabloom, E.; Williams, J.; Slayback, D.; Stoms, D.; Viers, H.J.; Dobson, A.P. Human impacts, plant invasion, and imperiled plant species in California. Ecol. Appl. 2006, 16, 1338–1350. [Google Scholar] [CrossRef] [Green Version]
- Brook, B.; Sodhi, S.N.; Bradshaw, C. Synergies among extinction drivers under global change. Ecol. Evol. 2008, 23, 453–460. [Google Scholar] [CrossRef]
- Vesperinas, E.S.; Moreno, A.G.; Elorza, M.S.; Sánchez, E.D.; Mata, D.S.; Gavilán, R. The Expansion of Thermophilic Plants in the Iberian Peninsula as a Sign of Climatic Change. In "Fingerprints" of Climate Change; Walther, G.R., Burga, C.A., Edwards, P.J., Eds.; Springer: Boston, MA, USA, 2001; pp. 163–184. [Google Scholar]
- Doroftei, M.; Anastasiu, P. Potential Impacts of Climate Change on Habitats and Their Effects on Invasive Plant Species in Danube Delta Biosphere Reserve, Romania. In Managing Protected Areas in Central and Eastern Europe Under Climate Change: Advances in Global Change Research; Rannow, S., Neubert, M., Eds.; Springer: Dordrecht, The Netherlands, 2014; pp. 267–278. [Google Scholar]
- Hoveka, L.N.; Bezeng, B.S.; Yessoufou, K.; Boatwrigh, J.S.; Van der Bank, M. Effects of climate change on the future distributions of the top five freshwater invasive plants in South Africa. S. Afr. J. 2016, 102, 33–38. [Google Scholar] [CrossRef]
- Hellmann, J.J.; Byers, J.E.; Bierwagen, B.G.; Dukes, J.S. Five potential consequences of climate change for invasive species. Biol. Conserv. 2008, 22, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Cronk, Q.C.B.; Fuller, J. Plant Invaders: The Threat to Natural Ecosystems. In Plant Invaders; Cronk, Q.C.B., Ed.; Earthscan Publications: London, UK, 1995. [Google Scholar]
- Kolar, C.S.; Lodge, D.M. Progress in invasion biology: Predicting invaders. Ecol. Evol. 2001, 16, 199–204. [Google Scholar] [CrossRef]
- Arim, M.; Abades, S.; Neill, P.; Lima, M.; Marquet, P. Spread dynamics of invasive species. Proc. Natl. Acad. Sci. USA 2006, 103, 374–378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sebert-Cuvillier, E.; Paccaut, F.; Chabrerie, O.; Endels, P.; Goubet, O.; Decocq, G. Local population dynamics of an invasive tree species with a complex life-history cycle: A stochastic matrix model. Ecol. Model. 2007, 201, 127–143. [Google Scholar] [CrossRef]
- Lind, E.M.; Parker, J.D. Novel Weapons Testing: Are Invasive Plants More Chemically Defended than Native Plants? PLoS ONE 2010, 5. [Google Scholar] [CrossRef]
- Bennett, A.E.; Thomsen, M.; Strauss, S.Y. Multiple mechanisms enable invasive species to suppress native species. Am. J. Bot. 2011, 98, 1086–1094. [Google Scholar] [CrossRef] [PubMed]
- Dormontt, E.E.; Lowe, A.J.; Prentis, P.J. Is rapid adaptive evolution important in successful invasions? In Fifty Years of Invasion Ecology The Legacy of Charles Elton; Richardson, D.M., Ed.; Wiley-Blackwell: Shichester, UK, 2011; pp. 175–193. [Google Scholar]
- Sladonja, B.; Sušek, M.; Guillermic, J. Review on invasive tree of heaven (Ailanthus altissima (Mill.) Swingle) conflicting values: Assessment of its ecosystem services and potential biological threat. J. Environ. Manag. 2015, 56, 1009–1034. [Google Scholar] [CrossRef]
- Millennium Ecosystem Assessment, Ecosystems and Human Well-being: Synthesis. Available online: https://www.millenniumassessment.org/documents/document.356.aspx.pdf (accessed on 13 October 2020).
- Kavallieratos, N.G.; Stathas, G.J.; Athanassiou, C.G.; Tomanović, Ž. Aphid parasitoids (Hymenoptera: Braconidae: Aphidiinae) on citrus: Seasonal abundance, association with the species of host plant, and sampling indices. Phytoparasitica 2002, 30, 231. [Google Scholar] [CrossRef]
- Cohen, Y.; Wang, W.; Ben-Daniel, B.H.; Ben-Daniel, Y. Extracts of Inula viscosa control downy mildew of grapes caused by Plasmopara viticola. Phytopathology 2006, 96, 417–424. [Google Scholar] [CrossRef] [Green Version]
- Jaber, L.R.; Araj, S.E.; Qasem, J.R. Compatibility of endophytic fungal entomopathogens with plant extracts for the management of sweetpotato whitefly Bemesia tabaci Gennadius (Homoptera: Aleyrodidae). Biol. Control. 2018, 117, 164–171. [Google Scholar] [CrossRef]
- Cobo, A.; González-Núñez, M.; Sánchez-Ramos, I.; Pascual, S. Selection of non-target tephritids for risk evaluation in classical biocontrol programmes against the olive fruit fly. J. Appl. Entomol. 2015, 139, 179–191. [Google Scholar] [CrossRef]
- Kožuharova, E.; Lebanova, H.; Getov, I.; Benbassat, N.; Kochmarov, V. Ailanthus altissima (Mill.) Swingle-a terrible invasive pest in Bulgaria or potential useful medicinal plant? Bothalia J. 2014, 44, 213–229. [Google Scholar]
- Macel, M.; de Vos, R.C.H.; Jansen, J.J.; van der Putten, W.H.; van Dam, N.M. Novel chemistry of invasive plants: Exotic species have more unique metabolomic profiles than native congeners. Ecol. Evol. 2014, 4, 2777–2786. [Google Scholar] [CrossRef] [PubMed]
- Maoz, M.; Neeman, I. Effect of Inula viscosa extract on chitin synthesis in dermatophytes and Candida albicans. J. Ethnopharmacol. 2000, 71, 479–482. [Google Scholar] [CrossRef]
- Bresch, C.; Mailleret, L.; Muller, M.M.; Poncet, C.; Parolin, P. Invasive plants in the Mediterranean basin: Which traits do they share? J. Mediterr. Ecol. 2013, 12, 13–19. [Google Scholar]
- Al Hassan, M.; Chaura, J.; López-Gresa Pilar, M.; Borsai, O.; Daniso, E.; Donat, P.; Mayoral, O.; Vicente, O.; Boscaiu, M. Native-Invasive Plants vs. Halophytes in Mediterranean Salt Marshes: Stress Tolerance Mechanisms in Two Related Species. Front. Plant Sci. 2016, 7, 1438. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wacquant, P.J. Biogeographical and physiological aspects of the invasion by Dittrichia (ex-Inula) viscosa. In A Ruderal Species in the Mediterranean Basin; Greuter, W., Ed.; Kluwer: Dordrecht, The Netherlands, 1990; pp. 353–364. [Google Scholar]
- Wacquant, J.P.; Bouab, N. Nutritional differentiation within the species Dittrichia viscosa W. Greuter, between a population from a calcareous habitat and another from an acidic habitat. In Genetic Aspects of Plant Nutrition, Developments in Plant and Soil Sciences; Sarić, M.R., Loughman, B.C., Eds.; Springer: Dordrecht, The Netherlands, 1983; pp. 285–291. [Google Scholar]
- Parolin, P.; Scotta, I.M.; Bresch, C. Notes on the phenology of Dittrichia viscosa. J. Mediterr. Ecol. 2013, 12, 27–35. [Google Scholar]
- Aronson, J.A. HALOPH: A Data Base of Salt Tolerant Plants of the World; Office of Arid Land Studies, University of Arizona: Tucson, AZ, USA, 1989. [Google Scholar]
- Grašič, M.; Anžlovar, S.; Strgulc Krajšek, S. Germination rate of stinkwort (Dittrichia graveolens) and false yel-lowhead (D. viscosa) in relation to salinity. Acta. Biol. Slov. 2016, 59, 5–11. [Google Scholar]
- De Laurentis, N.; Losacco, V.; Milillo, M.A.; Lai, O. Chemical investigations of volatile constituents of Inula viscosa (L.) Aiton (Asteraceae) from different areas of Apulia, Southern Italy. Delpinoa 2002, 44, 115–119. [Google Scholar]
- Dor, E.; Hershenhorn, J. Allelopathic effects of Inula viscosa leaf extracts on weeds. Allelopathy. J. 2012, 30, 281–290. [Google Scholar]
- Levizou, E.; Karageorgou, P.G.K.; Manetas, Y. Inhibitory effects of water soluble leaf leachates from Dittrichia viscosa on lettuce root growth, statocyte development and graviperception. Flora-Morphol. Distrib. Funct. Ecol. Plants 2002, 197, 152–157. [Google Scholar] [CrossRef] [Green Version]
- Curadi, M.; Graifenberg, A.; Magnani, G.; Giustiniani, L. Growth and element allocation in tissues of Inula viscosa in sodic-saline conditions: A candidate for programs of desertification control. Arid. Land Res. Manag. 2005, 19, 257–265. [Google Scholar] [CrossRef]
- Murciego, A.M.; Sanchez, A.G.; Gonzalez, M.R.; Gil, E.P.; Gordillo, C.T.; Fernandez, J.C.; Triguero, T.B. Antimony distribution and mobility in topsoils and plants (Cytisus striatus, Cistus ladanifer and Dittrichia viscosa) from polluted Sb-mining areas in Extremadura (Spain). Environ. Pollut. 2007, 145, 15–21. [Google Scholar] [CrossRef]
- Perdikis, D.; Favas, C.; Lykouressis, D.; Fantinou, A. Ecological relationships between non-cultivated plants and insect predators in agroecosystems: The case of Dittrichia viscosa (Asteraceae) and Macrolophus melanotoma (Hemiptera: Miridae). Acta Oecol. 2007, 31, 299–306. [Google Scholar] [CrossRef]
- Pest Rating Proposals and Final Rating for Dittrichia viscosa. Available online: https://blogs.cdfa.ca.gov/Section3162/?tag=dittrichia-viscosa (accessed on 28 May 2020).
- Weeds Australia. Centre for Invasive Species Solutions, Canberra. Available online: https://profiles.ala.org.au/opus/weeds-australia/profile/Dittrichia%20viscosa (accessed on 18 June 2021).
- Zurayk, R.; Khoury, N.; Talhouk, S.; Baalbaki, R. Salinity-Heavy Metal Interactions in Four Salt-tolerant Plant Species. J. Plant Nutr. 2001, 24, 1773–1786. [Google Scholar] [CrossRef]
- Cafarchia, C.; De Laurentis, N.; Milillo, M.A.; Losacco, V.; Puccini, V. Fungistatic activity of a sesquiterpene lactone (tomentosin) isolated from fresh Inula viscosa (Asteraceae) flowers from the Puglia region. Parasitologia 2001, 43, 117–121. [Google Scholar]
- Passalacqua, N.G.; Guarrera, P.M.; De Fine, G. Contribution to the knowledge of the folk plant medicine in Calabria region (Southern Italy). Fitoterapia 2007, 78, 52–68. [Google Scholar] [CrossRef]
- Musthaba, S.M.; Athar, M.T.; Kamal, Y.T.; Baboota, S.; Javed, A.; Sayeed, A. Fast analysis and validation of rutin in anti-psoriatic ayurvedic formulation by HPLC. J. Liq. Chromatogr. Relat. Technol. 2011, 34, 446–455. [Google Scholar] [CrossRef]
- Seca, A.; Grigore, A.; Pinto, D.; Silva, A. The genus Inula and their metabolites: From ethnopharmacological to medicinal uses. J. Ethnopharmacol. 2014, 154, 286–310. [Google Scholar] [CrossRef] [Green Version]
- Merghoub, N.; El Btaouri, H.; Benbacer, L.; Gmouh, S.; Trentesaux, C.; Brassart, B.; Terryn, C.; Attaleb, M.; Madoulet, C.; Benjouad, A.; et al. Inula Viscosa Extracts Induces Telomere Shortening and Apoptosis in Cancer Cells and Overcome Drug Resistance. Nutr. Cancer 2016, 68, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Lounis, H.; Bergheim, I.; Bouhaimi, A.; Guigonis, J.M.; Belhamel, K. Anti-inflammatory and antioxidant activities of Inula viscosa and Senecio anteuphorbium. Orient. Pharm. Exp. Med. 2018, 18, 225–236. [Google Scholar] [CrossRef]
- Askin, C.T.; Aslantürk, T.S. Evaluation of Cytotoxicity and Genotoxicity of Inula viscosa Leaf Extracts with Allium Test. J. Biomed. Biotechnol. 2010, 8. [Google Scholar] [CrossRef] [Green Version]
- Omezzine, F.; Rinez, A.; Ladhari, A.; Farooq, M.; Haouala, R. Allelopathic potential of Inula viscosa against crops and weeds. Int. J. Agric. Biol. 2011, 13, 841–849. [Google Scholar]
- Oka, Y.; Ben-Daniel, B.H.; Cohen, Y. Nematicidal activity of powder and extracts of Inula viscosa. J. Nematol. 2001, 3, 735–742. [Google Scholar]
- Mansour, F.; Azaizeh, H.; Saad, B.; Tadmor, Y.; Abo-Moch, F.; Said, O. The potential of middle eastern flora as a source of new safe bio-acaricides to control Tetranychus cinnabarinus, the carmine spider mite. Phytoparasitica 2004, 32, 66–72. [Google Scholar] [CrossRef]
- Oka, Y.; Ben-Daniel, B.H.; Cohen, Y. Control of Meloidogyne javanica by formulations of Inula viscosa leaf extracts. J. Nematol. 2006, 38, 46. [Google Scholar] [CrossRef]
- Alexenizer, M.; Dorn, A. Screening of medicinal and ornamental plants for insecticidal and growth regulating activity. J. Pest Sci. 2007, 80, 205–215. [Google Scholar] [CrossRef]
- Sofou, K.; Isaakidis, D.; Spyros, A.; Büttner, A.; Giannis, A.; Katerinopoulos, H. Use of costic acid, a natural extract from Dittrichia viscosa, for the control of Varroa destructor, a parasite of the European honey bee. Beilstein J. Org. Chem. 2017, 13, 952–959. [Google Scholar] [CrossRef] [Green Version]
- Cohen, Y.; Baider, A.; Ben-Daniel, B.; Ben-Daniel, Y. Fungicidal preparations from Inula viscosa. Plant Prot. Sci. 2002, 38, 629–630. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Ben-Daniel, B.; Cohen, Y. Control of Plant Diseases by Extracts of Inula viscosa. Phytopathology 2004, 94, 1042–1047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Talib, W.; Abu Zarga, M.; Mahasneh, A. Antiproliferative, Antimicrobial and Apoptosis Inducing Effects of Compounds Isolated from Inula viscosa. Molecules 2012, 17, 3291–3303. [Google Scholar] [CrossRef]
- Al-Masri, M.I.; Sharawi, S.M.; Barakat, R.M. Effect of Clammy Inula (Inula viscose) Plant Extract in Combination with a Low Dose of the Fungicide Iprodione on Botrytis cinerea In Vitro and In Vivo. Am. J. Plant Sci. 2015, 6, 1519–1526. [Google Scholar] [CrossRef] [Green Version]
- Dominguez, M.; Madejón, P.; Madejón, E.; Díaz Blanco, M. Novel energy crops for Mediterranean contaminated lands: Valorization of Dittrichia viscosa and Silybum marianum biomass by pyrolysis. Chemosphere 2017, 186, 976–986. [Google Scholar] [CrossRef] [PubMed]
- Ater, M.; Lefèbvre, C.; Gruber, W.; Meerts, P. A phytogeochemical survey of the flora of ultramafic and adjacent normal soils in North Morocco. Plant Soil 2000, 218, 127–135. [Google Scholar] [CrossRef]
- Swaileh, K.; Hussein, R.; Abu-Elhaj, S. Assessment of Heavy Metal Contamination in Roadside Surface Soil and Vegetation from the West Bank. Arch. Environ. Contam. Toxicol. 2004, 23, 22–30. [Google Scholar] [CrossRef]
- Gisbert, C.; Almela, C.; Vélez, D.; López-Moya, J.R.; Serrano, R.; Montoro, R.; Navarro-Aviñó, J. Identification of As accumulation plant species growing on highly contaminated soils. Int. J. Phytoremediation 2008, 10, 183–184. [Google Scholar] [CrossRef]
- Conesa, H.; María-Cervantes, A.; Alvarez-Rogel, J.; González, M. Influence of soil properties on trace element availability and plant accumulation in a Mediterranean salt marsh polluted by mining wastes: Implications for phytomanagement. Sci. Total Environ. 2011, 409, 4470–4479. [Google Scholar] [CrossRef]
- Perez, C.; Martínez-Sánchez, M.; Martínez-López, S.; Bech, J.; Bolan, N. Distribution and bioaccumulation of arsenic and antimony in Dittrichia viscosa growing in mining-affected semiarid soils in southeast Spain. J. Geochem. Explor. 2012, 123, 128–135. [Google Scholar] [CrossRef]
- Pistelli, L.; D’Angiolillo, F.; Morelli, E.; Basso, B.; Rosellini, I.; Posarelli, M.; Barbafieri, M. Response of spontaneous plants from an ex-mining site of Elba island (Tuscany, Italy) to metal (loid) contamination. Environ. Sci. Pollut. Res. Int. 2017, 24, 7809–7820. [Google Scholar] [CrossRef] [PubMed]
- Barbafieri, M.; Dadea, C.; Tassi, E.; Bretzel, F.; Fanfani, L. Uptake of heavy metals by native species growing in a mining area in Sardenia, Italy: Discovering native flora for phytoremediation. Int. J. Phytoremediation 2011, 13, 985–997. [Google Scholar] [CrossRef]
- Jimenez, M.N.; Bacchetta, G.; Casti, M.; Navarro, F.B.; Lallena, A.M.; Fernández-Ondoñoa, E. Potential use in phytoremediation of three plant species growing on contaminated mine-tailing soils in Sardinia. Ecol. Eng. 2011, 37, 392–398. [Google Scholar] [CrossRef]
- Perez-Fernandez, M.A.; Calvo-Magro, E.; Ferrrer-Castan, D. Simulation of germination of pioneer species along an experimental drought gradient. J. Environ. Biol. 2006, 27, 679–685. [Google Scholar] [PubMed]
- Gonzalez-Tejero, M.R.; Casares-Porcel, M.; Sánchez-Rojas, C.P.; Ramiro-Gutiérrez, J.M.; Mesa, J.; Pieroni, A.; Giusti, M.E.; Censorii, E.; de Pasquale, C.; Della, A.; et al. Medicinal plants in the Mediterranean area: Synthesis of the results of the project Rubia. J. Ethnopharmacol. 2008, 116, 341–357. [Google Scholar] [CrossRef] [PubMed]
- Akkawi, M.; Abbasi, I.; Jaber, S.; Abouremeleh, Q.; Nasereddin, A.; Lutgen, P. Investigation of Traditional Palestinian Medicinal Plant Inula viscosa as Potential Anti-malarial Agent. Br. J. Pharmacol. 2014, 5, 156–162. [Google Scholar] [CrossRef]
- Kawada, K.; Vovk, A.G.; Filatova, O.V.; Araki, M.; Nakamura, T.; Hayashi, I. Floristic composition and plant biomass production of steppe communities in the vicinity of Kharkiv, Ukraine. Grassl. Sci. 2005, 51, 205–213. [Google Scholar] [CrossRef]
- Zhang, T.; Guo, R.; Gao, S.; Guo, J.; Sun, W. Responses of Plant Community Composition and Biomass Production to Warming and Nitrogen Deposition in a Temperate Meadow Ecosystem. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Karageorgou, P.; Levizou, E.F.I.; Manetas, Y. The influence of drought, shade and availability of mineral nutrients on exudate phenolics of Dittrichia viscosa. Flora 2002, 197, 285–289. [Google Scholar] [CrossRef]
- Terradas, J. Mediterranean woody plant growth-forms, biomass and production in the eastern part of the Iberian Peninsula. Oecologia 1990, 10, 337–349. [Google Scholar]
- Brahmi-Chendouh, N.; Piccolella, S.; Crescente, G.; Pacifico, F.; Boulekbache-Makhlouf, L.; Hamri-Zeghichi, S.; Akkal, S.; Madani, K.; Pacifico, S. A nutraceutical extract from Inula viscosa leaves: UHPLC-HR-MS/MS based polyphenol profile, and antioxidant and cytotoxic activities. J. Food Drug Anal. 2019, 27, 692–702. [Google Scholar] [CrossRef] [PubMed]
- Trimech, I.; Weiss, E.K.; Chedea, V.; Marin, D.; Detsi, A.; Ioannou, E.; Roussis, V.; Kefalas, P. Evaluation of Anti-oxidant and Acetylcholinesterase Activity and Identification of Polyphenolics of the Invasive Weed Dittrichia viscosa. Phytochem. Anal. 2014, 25, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Lattanzio, V.; Lattanzino, V.M.T.; Cardinali, A. Role of phenolics in the resistance mechanisms of plants against fungal pathogens and insects. In Phytochemistry. Advances in Research; Imperato, F., Ed.; Research Signpost: Trivandrum, India, 2006; pp. 23–67. [Google Scholar]
- Mikulič-Petkovšek, M.; Štampar, F.; Veberić, R. Accumulation of phenolic compounds in apple in response to infection by the scab pathogen, Venturia inaequalis. Physiol. Mol. Plant Pathol. 2009, 74, 60–67. [Google Scholar] [CrossRef]
- Orcaray, L.; Igal, M.; Zabalza, A.; Royuela, M. Role of Exogenously Supplied Ferulic and p-Coumaric Acids in Mimicking the Mode of Action of Acetolactate Synthase Inhibiting Herbicides. J. Agric. Food Chem. 2011, 59, 10162–10168. [Google Scholar] [CrossRef]
- Dayan, F.E.; Duke, S.O. Natural compounds as next-generation herbicides. J. Plant Physiol. 2014, 166, 1090–1105. [Google Scholar] [CrossRef] [Green Version]
- Pyšek, P.; Richardson, D.M. Invasive plants. In Encyclopedia of Ecology; Jørgensen, S.E., Ed.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 2011–2020. [Google Scholar]
- Pyšek, P.; Richardson, M.D.; Rejmanek, M.; Webster, G.L.; Williamson, M.; Kirschner, J. Alien plants in checklists and floras: Towards better communication between taxonomists and ecologists. Taxon 2004, 53, 131–143. [Google Scholar] [CrossRef]
- James, L.; Evans, J.; Ralphs, M.; Child, R. Noxious Range Weeds; Westview Press: San Francisco, CA, USA, 1991; pp. 420–428. [Google Scholar]
- Pyšek, P.; Richardson, D.M. Invasive Species, Environmental Change and Management, and Health. Annu. Rev. Environ. Resour. 2010, 35, 25–55. [Google Scholar] [CrossRef] [Green Version]
- Niemelä, J. From systematics to conservation-carabidologists do it all. Ann. Zool. Fenn. 1996, 33, 1–4. [Google Scholar]
- Jefferies, R. Allochthonous inputs: Integrating population changes and food-web dynamics. Ecol. Evol. 2000, 15, 19–22. [Google Scholar] [CrossRef]
- Meiners, S.J. Native and exotic plant species exhibit similar population dynamics during succession. Ecology 2007, 88, 1098–1104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burgiel, S.W.; Muir, A.A. Invasive Species, Climate Change and Ecosystem-Based Adaptation: Addressing Multiple Drivers of Global Change; Global Invasive Species Programme (GISP): Washington, DC, USA; Nairobi, Kenya, 2010. [Google Scholar]
- Wijesundara, D. Can native plants become invasive? Ceylon J. Sci. 2017, 46, 1–2. [Google Scholar] [CrossRef] [Green Version]
- Plieninger, T.; Hui, C.; Gaertner, M.; Huntsinger, L.L. The Impact of Land Abandonment on Species Richness and Abundance in the Mediterranean Basin: A Meta-Analysis. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Mikulič, K.; Radović, A.; Kati, V.; Jelaska, S. Effects of land abandonment on bird communities of smallholder farming landscapes in post-war Croatia: Implications for conservation policies. Community Ecol. 2014, 15, 169–179. [Google Scholar] [CrossRef]
- Benayas, J.M.R.; Artins, A.; Nicolau, J.M.; Schulz, J.J. Abandonment of agricultural land: An overview of drivers and consequences. CAB. Rev. Perspect. Agric. Vet. Sci. Nutr. Nat. Resour. 2007, 2, 14. [Google Scholar] [CrossRef] [Green Version]
- Jackson, W.J.; Argent, R.M.; Bax, N.J.; Bui, E.; Clark, G.F.; Coleman, S.; Cresswell, I.D.; Emmerson, K.M.; Evans, K.; Hibberd, M.F.; et al. Overview: Invasive species are a potent, persistent and widespread threat to Australia’s environment. In Australia State of the Environment 2016; Australian Government Department of the Environment and Energy: Canberra, Australia, 2016. [Google Scholar]
- Yasumura, Y.; Crumpton-Taylor, M.; Fuentes, S.P.; Harberd, N. Step-by-Step Acquisition of the Gibberellin-DELLA Growth-Regulatory Mechanism during Land-Plant Evolution. Curr. Biol. 2007, 17, 1225–1230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ratnayake, R.M.C.S. Why plant species become invasive? Characters Related to Successful Biological Invasion. In Proceedings of the Conference National Symposium on Invasive alien Species, Colombo, Sri Lanka, 11 November 2014. [Google Scholar]
- Vilà, M.; Hulme, P. Impact of Biological Invasions on Ecosystem Services; Springer International Publishing: Cham, Switzerland, 2016. [Google Scholar]
- Grice, A.C.; Friedel, M.H.; Marshall, N.A.; Van Klinken, R.D. Tackling Contentious Invasive Plant Species: A Case Study of Buffel Grass in Australia. Environ. Manag. 2012, 49, 285–294. [Google Scholar] [CrossRef]
- Boari, A.; Vurro, M.; Calabrese, G.; Nesma, M.; Mahmoud, Z.; Cazzato, E.; Fracchiolla, M. Evaluation of Dittrichia viscosa (L.) Greuter Dried Biomass for Weed Management. Plants 2021, 10, 147. [Google Scholar] [CrossRef] [PubMed]

| Category | Example of Service Provided by D. viscosa | References |
|---|---|---|
| Provisioning | Medicinal | [31,48,49,50,51,52,53] |
| Weed management | [4,8,54,55] | |
| Pest control | [40,56,57,58,59,60] | |
| Antifungal control | [31,61,62,63,64] | |
| Biomass production | [65] | |
| Regulating | Bioindicator | [36,66,67] |
| Bioaccumulator | [36,68,69,70,71] | |
| Phytoremediation | [36,43,55,72,73] | |
| Desertification control | [33,42,74] | |
| Cultural | Traditional use | [34] |
| Biogeographical component | [51,75,76] | |
| Supporting | Primary production | [77,78] |
| Nutrient cycling | [41,79] | |
| Soil formation | [80] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sladonja, B.; Poljuha, D.; Krapac, M.; Uzelac, M.; Mikulic-Petkovsek, M. Dittrichia viscosa: Native-Non Native Invader. Diversity 2021, 13, 380. https://doi.org/10.3390/d13080380
Sladonja B, Poljuha D, Krapac M, Uzelac M, Mikulic-Petkovsek M. Dittrichia viscosa: Native-Non Native Invader. Diversity. 2021; 13(8):380. https://doi.org/10.3390/d13080380
Chicago/Turabian StyleSladonja, Barbara, Danijela Poljuha, Marin Krapac, Mirela Uzelac, and Maja Mikulic-Petkovsek. 2021. "Dittrichia viscosa: Native-Non Native Invader" Diversity 13, no. 8: 380. https://doi.org/10.3390/d13080380
APA StyleSladonja, B., Poljuha, D., Krapac, M., Uzelac, M., & Mikulic-Petkovsek, M. (2021). Dittrichia viscosa: Native-Non Native Invader. Diversity, 13(8), 380. https://doi.org/10.3390/d13080380











