Abstract
In this work, the consequences of a local gorgonian coral mortality on the whole coralligenous assemblage were studied. A Before/After-Control/Impact sampling design was used: the structure of the coralligenous assemblage was compared before and after the gorgonian mortality event at the mortality site and two control sites. At the mortality site, a relevant decrease in alpha and beta diversity occurred, with a shift from a stratified assemblage characterized by gorgonians and other invertebrates to an assemblage dominated by algal turfs; conversely, neither significant variations of the structure nor decrease in biodiversity were observed at the control sites. The assemblage shift involved the main taxa in different times: in autumn 2018, a large proportion of the plexaurid coral Paramuricea clavata died, but no significant changes were observed in the structure of the remaining assemblage. Then, in autumn 2019, algal turfs increased significantly and, one year later, the abundance of the gorgonian Eunicella cavolini and bryozoans collapsed. Although the mechanisms of the assemblage shift following gorgonian loss will remain uncertain and a cause-effect relationship cannot be derived, results suggest the need for detecting signs of gorgonian forests stress in monitoring programs, which should be considered early indicators of their condition. in the coralligenous monitoring programs for detecting any sign of gorgonian forests stress which should be considered an early indicator of the assemblage condition.
1. Introduction
Mass mortality events (MMEs) in marine systems are of an increasing ecological concern, as their intensity and frequency are rising worldwide due to global changes mostly related to climate anomalies, such as sea surface temperature rise [1,2,3]. Consequences of mass mortality may be more serious than the mere loss of the concerned taxa, as some of them are habitat-forming species and their regression may lead to severe biodiversity erosion and changes in the whole system functioning [4,5]. The ability of marine systems to recover after MMEs is greatly variable due to differences in the taxa concerned, propagule availability, and the occurrence of further stressors [6,7,8]; thus, different systems may take different trajectories of community change following a MME [9].
In the Mediterranean Sea, MMEs have been first evidenced in the 1980s in the Western Basin and the Aegean Sea [10,11,12], but probably, the most recent impressive MME that has involved more than thirty species from different phyla has happened in 1999 along the French and Italian coasts [13,14]. Since then, other numbered MMEs have been reported of a range of scales, with respect to geographic extend and number of affected species [15,16,17]. Because MMEs have generally been associated with strong and recurrent marine heat waves (MHWs) [1,18,19,20], a collaborative data base has been created to examine the relationship between the thermal conditions and other environmental variables (such as the number of MHW days) possibly linked with thermal anomalies (e.g., occurrence of the proliferation of filamentous algal blooms and mucilage) and the MME [21].
The main Mediterranean MMEs, besides the mortality of the giant mussel Pinna nobilis (Linnaeus 1758) [22], concerned the coralligenous habitat [23], a typical Mediterranean biogenic subtidal seascape mainly produced by the accumulation of calcareous encrusting algae growing in dim light conditions and in relatively calm waters, mostly at depths of 30–130 m [24]. Calcareous encrusting algae constitute a secondary substrate where stratified assemblages including erect algae, sponges, bryozoans, gorgonian corals, and other alcyonaceans develop [24]. The maintenance of coralligenous organisms is likely related to relatively steady environmental conditions, and therefore they are particularly sensitive to local and global stressors [25,26,27,28,29]. MMEs have been described for many coralligenous species [30,31,32,33], but gorgonian corals and Paramuricea clavata (Risso, 1826) in particular appear to be mostly affected [34,35,36,37]. Indeed, Mediterranean gorgonians may be threatened by many local human pressures, such as pollution, sedimentation, introduced species, and mechanical destruction by fishing gears or scuba divers [38,39,40]. However, the most severe threats are often related to stressors acting at large scale, such as climatic anomalies [15,41,42], benthic mucilage [43,44,45], diseases [46,47], and synergisms among them [48]. Recovery of populations is slow and almost always partial [34,49,50,51,52]. Gorgonians are habitat-forming species whose presence is known to influence the structure of the associated assemblage and form an animal forest [53,54,55,56,57,58]. Thus, gorgonian loss is thought to induce changes in species composition of coralligenous assemblages and to reduce species diversity [42,59]. Although some common patterns have been described [42,59], the ecological consequences of gorgonian loss are variable [50,52,60,61] and far from being understood.
In this work, the diversity and structure of a coralligenous assemblage affected by a local gorgonian mortality (GM) was compared through time to nearby unaffected assemblages. Here, a Before/After-Control/Impact (BACI) sampling design [62,63] was used to compare the coralligenous assemblage before and after the GM event at the mortality site and two control sites. We hypothesized that the diversity of the coralligenous assemblages over time would change differently at the GM site rather than at the controls. Variations in taxa abundance following the mortality event was also evaluated to detect changes due to gorgonian loss, both identifying the taxa involved and measuring taxa vulnerability, by estimating the time needed to detect changes since GM.
2. Material and Methods
The study was done in the Marine Protected Area (MPA) of Capo Carbonara, located in South-Eastern Sardinia (Italy, Western Mediterranean). Three granite rocky bottom sites were considered: Cavoli Island (D, disturbed), where a widespread mortality of the gorgonian Paramuricea clavata was detected in late October 2018, and Serpentara and Santa Caterina (C1 and C2, controls Figure 1), where no mortality events have been recorded within the same period (LP, personal observation). The three sites are monitoring stations for the assessment of the MPA coralligenous reefs, and data before and after the GM event were therefore available. The D site was monitored annually between November 2016 and November 2020, while C1 and C2 were sampled in 2017 and 2020.
Figure 1.
Map of the study sites. D = disturbed site, C1 and C2 = control sites.
During each sampling period, three plots (4 m2 in size), tens of metres apart, were randomly selected on vertical substrates at about 33 m depth at each site. In each plot, 10 photographic samples of 0.2 m2 were collected [64]. Photographic samplings were analyzed to evaluate the percentage cover of the main taxa or morphological groups using ImageJ software [65]. Organisms easily detected by photographic samples were identified to the lowest possible taxonomic level, while those not easily recognizable were identified according to morphological groups [66].
The diversity of the assemblages was evaluated both as taxa/morphological group number per sample (alpha diversity) and as heterogeneity (beta diversity) calculated through the PERMDISP analysis (Primer 6 + PERMANOVA) [67], using the mean distance of photographic samples from centroids as a measure of the beta diversity of the system [67].
Temporal and spatial differences in the structure of assemblages were analyzed by a permutational analysis of variance (Primer6 + PERMANOVA) [68] based on a Bray–Curtis resemblance matrix of untransformed data. Data were not transformed in order to stress the importance of the abundance of taxa/groups in determining the differences among conditions. The alpha and beta diversity and the abundance of the main taxa were analysed by PERMANOVA based on Euclidean distance. The Pair-wise test was used to discriminate between levels of significant factors.
The structure of the D assemblage and the abundance of the main taxa were analyzed through a two-way model with the factors Time (2016, 2017, 2018, 2019, 2020, treated as fixed) and Plot (3 levels, treated as random and orthogonal). An nMDS was performed for a graphical representation of dissimilarities among assemblages through time.
To compare the D assemblage with the Cs between 2017 and 2020, a three-way PERMANOVA was used with Time (before vs. after, i.e., 2017 vs. 2020) and Site (D, C1 and C2) as fixed and orthogonal factors, while Plot (3 levels) as random and nested in Site. Gorgonians were not included in the analyses to test the hypothesis that their mortality affected the structure of assemblages. The same model of multivariate analysis was used for the alpha diversity, while beta diversity was analysed through a two-way model with Time and Site as fixed and orthogonal factors, while sampling plots were used as replicates.
3. Results
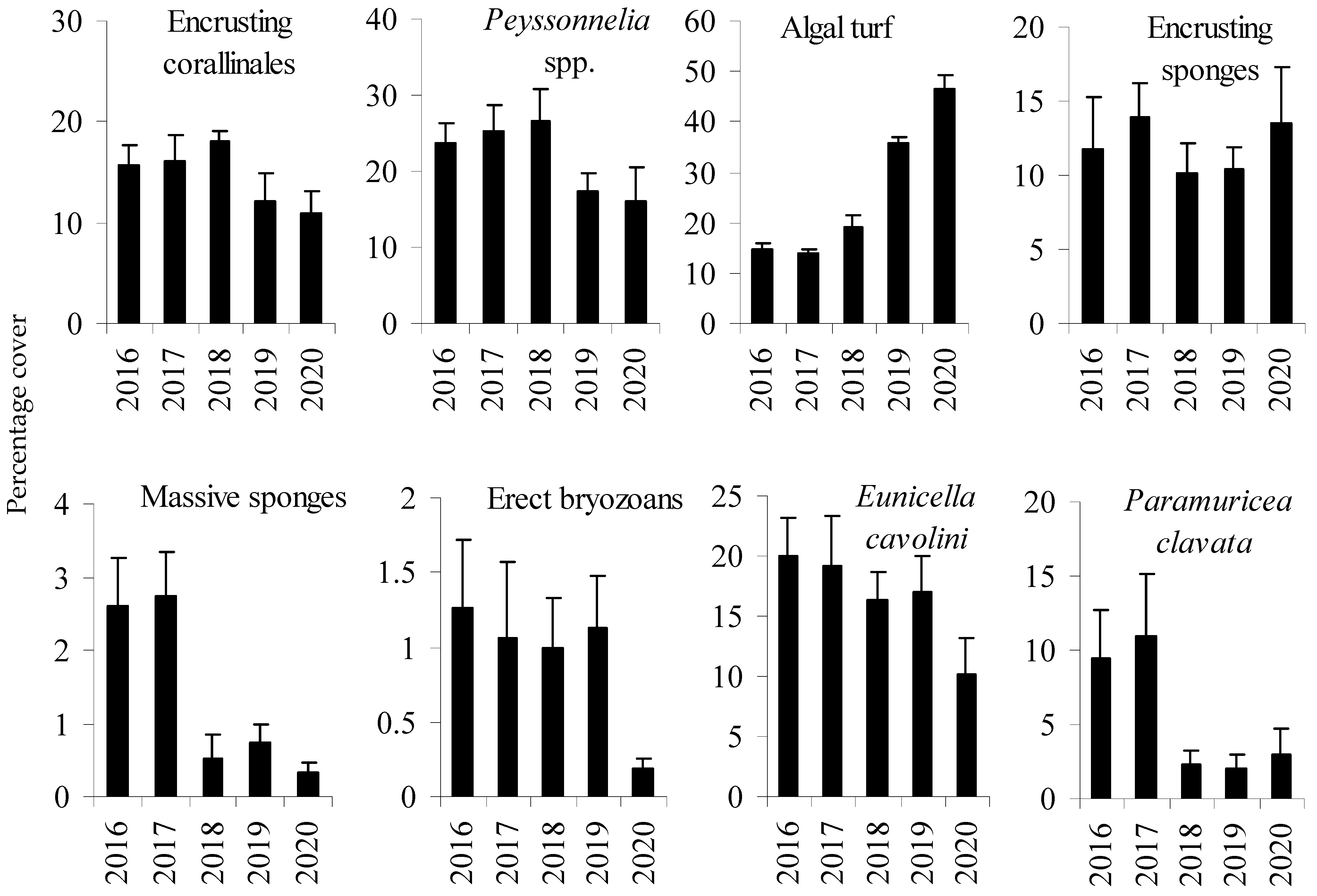
Before mortality (in 2016–2017) at the D site, the coralligenous assemblage was characterized by a stratified structure: the erect layer constituted by the gorgonians Paramuricea clavata and Eunicella cavolini (Koch, 1887), the intermediate layer by erect bryozoans (mostly Smittina cervicornis (Pallas, 1766)) and massive sponges, while the basal layer consisted of encrusting coralline algae, encrusting sponges, and prostrate red algae Peyssonnelia spp. (Figure 2). A small variability between 2016 and 2018 at D site in the structure of the coralligenous assemblage was identified (PERMANOVA, MDS and the Pair-wise test) compared to the variations found both in 2019 and 2020 (Table 1, Figure 3). Variations were mostly due to P. clavata that decreased between 2017 and 2018, the algal turf that increased between 2018 and 2019, and the erect bryozoans and E. cavolini that decreased between 2019 and 2020 (Table 1, Figure 2). The abundance of massive sponges, encrusting coralline algae and Peyssonnelia spp. also decreased during the study period, but variations were not significant (Table 1, Figure 2).
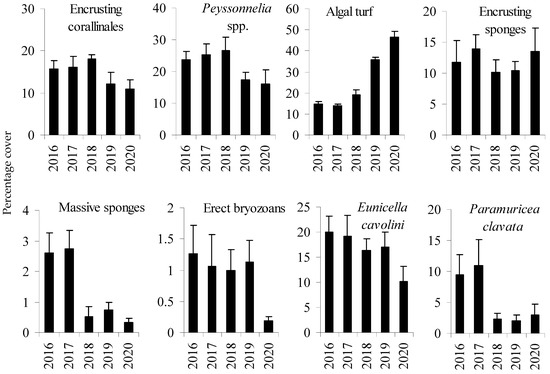
Figure 2.
Percentage cover (mean ± SE, n = 30) of the main taxa groups of the coralligenous assemblages at the D site throughout the whole the study period.

Table 1.
Effects of Time in PERMANOVA for the Cavoli (D site) coralligenous assemblage and its main taxa/groups. Significant p-values are in bold.
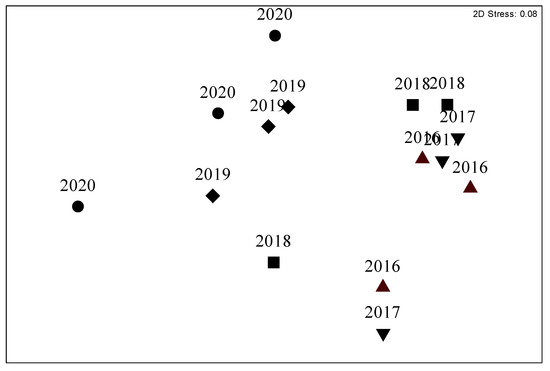
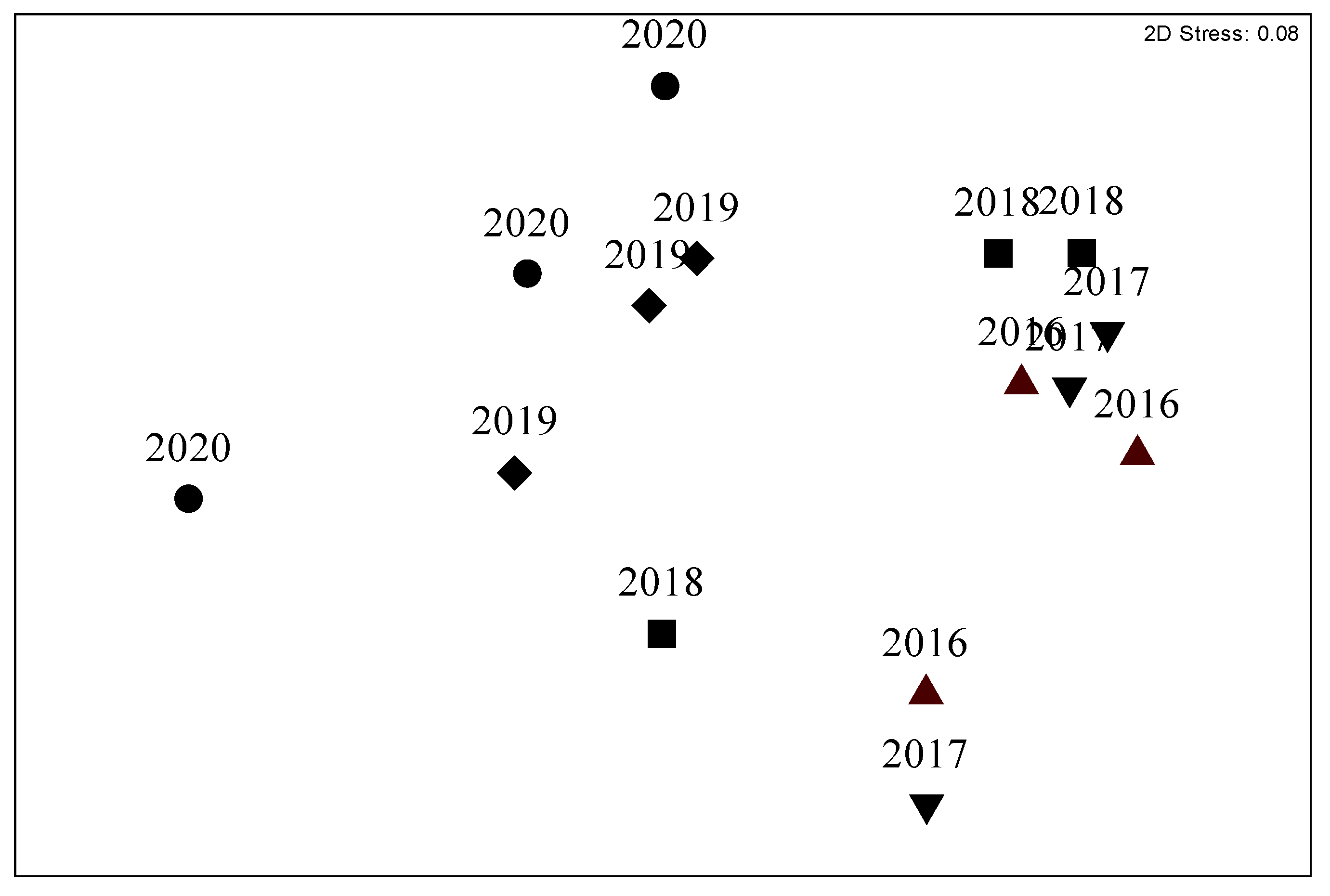
Figure 3.
MDS: temporal variation of similarities throughout the study period (from 2016 to 2020) of the Cavoli (D site) coralligenous assemblage.
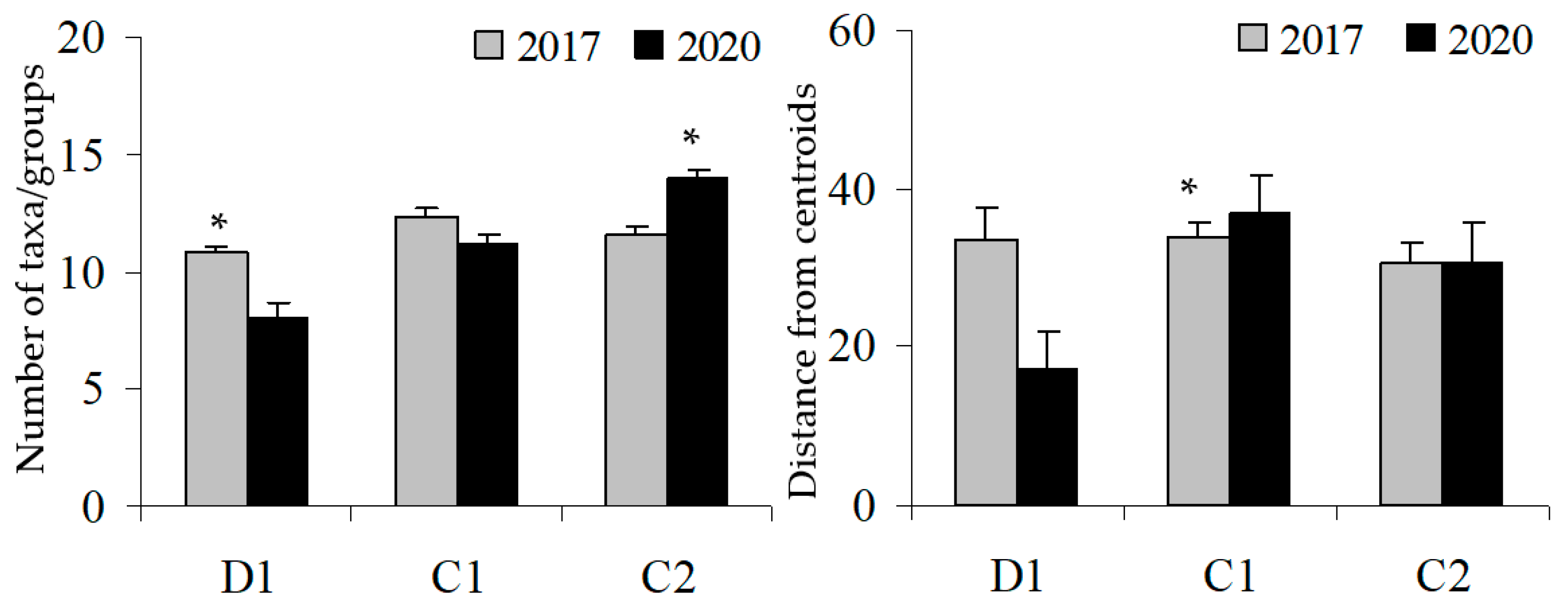
Based on the BACI design, on the before (2017) and after (2020) parameters, both the multivariate and univariate PERMANOVA analyses have found the interaction Time x Site to be significant (Table 2, Table 3 and Table 4). Particularly, the Pair-wise test showed that the structure of assemblages significantly varied at the D site from before to after GM, but not at the controls where assemblages did not change significantly through time (Table 2). The change in the structure of the coralligenous assemblage has led to a reduction of both alpha and beta diversity at the disturbed site; a different pattern was found at controls as alpha diversity increased in C2 and beta diversity did not change (Table 3 and Table 4; Figure 4).

Table 2.
PERMANOVA on the coralligenous assemblage: Time (before and after GM, T1 and T2 GM), Site (D, C1 and C2) and Plot (3 levels). Significant p-values are in bold.

Table 3.
PERMANOVA on alpha diversity: Time (before and after GM, T1 and T2), Site (D, C1 and C2) and Plot (3 levels). Significant p-values are in bold.

Table 4.
PERMANOVA on beta diversity: Time (before and after GM, T1 and T2) and Site (D, C1 and C2). Significant p-values are in bold.
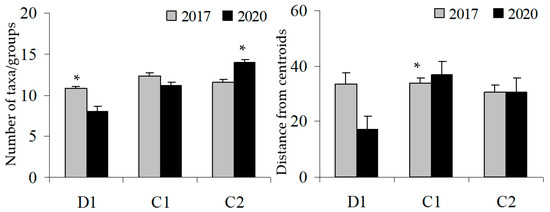
Figure 4.
Before (2017) and after (2020) GM: alpha (left) and beta diversity (right) at Cavoli (D) and at two control sites (C1 and C2). Asterisks indicate significant effects.
4. Discussion
The results of the study showed relevant differences in coralligenous assemblage structure and biodiversity patterns after the mortality event of the gorgonian Paramuricea clavata that in 2018 has happened in the Capo Carbonara MPA only at the D site. Important clues about the role of P. clavata on the whole coralligenous assemblage come from the evidence that the changes only concerned the site affected by the mortality event, while no significant variations of the structure and biodiversity decrease were observed at the control sites.
Indeed, the most relevant changes have occurred locally at the D site where a decrease in alpha and beta diversity was highlighted. Particularly, the modifications have involved the stratified structure as the assemblage characterized by large erect gorgonian corals and other invertebrates has shifted to an assemblage dominated by low-growth algal turfs. Overall, the assemblage shift has involved all the main taxa, although in different times and with different significance levels, and it has occurred across three years since the onset of the GM: in autumn 2018, a large proportion of P. clavata had died but no significant changes were observed in the structure of the remaining assemblage; in autumn 2019, algal turfs increased significantly and, one year later, the abundance of the gorgonian E. cavolini and bryozoans collapsed. This kind of successional pattern was previously described in a similar way [42,59]. Moreover, the temporal pattern of change depicted here is in agreement with previous observations [42] that highlighted delayed severe effects due to GM after 2.5 years (rather than the immediately), overall suggesting that the collapse of this habitat does not happen immediately.
The shift in a coralligenous assemblage following a gorgonian mortality resembles the collapse of a community following the loss of its foundation species [69,70,71]. The mechanisms (whether direct or indirect) involved in this shift are not easy to understand. A concurrent mortality of multiple taxa would be expected as consequence of a severe environmental stress as several macro-invertebrates and coralline algae, usually inhabiting gorgonian forests, are considered to be affected by the same factors as gorgonians [13,27,30,33]. However, different organisms may have different responses to stress, and sublethal effects or physiological stress may also occur, although the mortality happens in the subsequent years [50]. On the other hand, the lack of habitat-forming organisms may lead to further consequences: gorgonian forests can create complex three-dimension structures and offer protection to fragile calcareous colonies, supporting high species diversity [53,59]. Moreover, when the density of gorgonians is high, the altered environmental conditions (light, currents, sediment deposition rates, etc.) [53,72] offer several micro-habitats promoting high biodiversity [56]. Gorgonian forests may be considered structured habitat where smaller environmental modifications occur slower than in the surrounding ‘unforested’ environment, which provide positive effects on diversity, distribution, and abundance of a large proportion of organisms [53,56]. Finally, gorgonians are known to limit the settlement and spread of opportunistic species such as algal turfs and serpulid calcareous worms [56] and enhance the growth of calcareous algae and invertebrates [59]. Thus, the loss of gorgonians may explain itself the shift of assemblages dominated by erect invertebrates and encrusting algae to assemblages dominated by turf-forming species [42].
Nevertheless, a crucial role in this shift might be related to the spread of algal turfs which seems to be the first consequence of gorgonian loss [42]. In fact, turf algae have the capacity to affect the population dynamics of sessile invertebrates by reducing recruitment and juvenile survival [73]. The effects of turf algae on the invertebrate recruitment may be due either to a decrease of suitable substrate area for larval settlement and to algal overgrowth, as observed in coral reefs [74,75]. Thus, turfs, once established, can reduce the resilience of gorgonian forests preventing recruitment and stabilize an un-forested condition [73]. This effect may be more severe if invasive turf species are involved [40], such as the introduced Rhodophyta Womersleyella setacea (Hollenberg) R.E. Norris which has been described as a relevant component of coralligenous algal turfs in the Capo Carbonara MPA [76].
Although the mechanisms of the assemblage shift following gorgonian loss will remain uncertain and a cause-effect relationship cannot be derived, the present effort through a BACI approach has linked the loss of Paramuricea clavata with the decrease of biodiversity and structural complexity of coralligenous assemblages. Marine animal forests are declining worldwide with deep ecological consequences [77] mostly related to a shift towards less structurally complex systems [78]; the present study, in accordance with previous investigations [42,59], suggests the crucial role of animal forests in the maintenance of biodiversity of the Mediterranean coralligenous system. This study has documented the shift of assemblages two years after the GM, although long-term monitoring would be needed to detect the future development of the system, as different scenarios may be hypothesized: (i) the turf dominated assemblages can stabilize, (ii) gorgonians may recover their population or (iii) a new type of assemblage can develop, as high quality coralligenous reefs dominated by invertebrates or macroalgae may also occur in absence of an erect layer [79]. The trajectory of system evolution is expected to be strictly related to environmental conditions, as pressing stressors, such as climatic warming, thermal anomalies, and benthic mucilage, may preclude the spread of sensitive organisms, eroding the resilience of the system. Within the actual global change scenario, where environmental stress may be independent of local protection measures, raising the frequency of surveys is increasingly needed. Although the presence, size and status of gorgonians have already been proposed as descriptors of the ecological quality of coralligenous reefs [80,81,82]; the surveillance of gorgonian forest stress should be implemented, as they should be considered an important early indicator of the whole assemblage condition.
Author Contributions
Conceptualization: F.A., L.P., and G.C.; Formal Analysis: L.P.; Investigation: L.P., N.C., M.F.C., F.F., A.P., F.P., and P.S.; Funding Acquisition: F.A. and G.C.; Writing—Original Draft Preparation: L.P. and G.C. All authors have read and agreed to the published version of the manuscript.
Funding
The study has been funded by Italian Ministry of Education and Research: PRIN 2017 (MHHWBN) “Marine Habitats restoration in a climate change-impaired Mediterranean Sea (MAHRES)”.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data is available in the supplementary materials.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Crisci, C.; Bensoussan, N.; Romano, J.C.; Garrabou, J. Temperature anomalies and mortality events in marine communities: Insights on factors behind differential mortality impacts in the NW Mediterranean. PLoS ONE 2011, 6, e23814. [Google Scholar] [CrossRef]
- Eakin, M.; Liu, G.; Gomez, A.; De la Cour, J.; Heron, S.; Skirving, W. Global coral bleaching 2014–2017: Status and an appeal for observations. Reef Encount. 2016, 31, 20–26. [Google Scholar]
- Ereskovsky, A.; Ozerov, D.A.; Pantyulin, A.N.; Tzetlin, A.B. Mass mortality event of White Sea sponges as the result of high temperature in summer 2018. Polar Biol. 2019, 42, 2313–2318. [Google Scholar] [CrossRef]
- Hughes, T.P.; Kerry, J.T.; Baird, A.H.; Sean, R.; Connolly, S.R.; Dietzel, A.; Eakin, C.M.; Heron, S.F.; Hoey, A.S.; Hoogenboom, M.O.; et al. Global warming transforms coral reef assemblages. Nature 2018, 556, 492–506. [Google Scholar] [CrossRef] [PubMed]
- Jones, N.P.; Figueiredo, J.; Gilliam, D.S. Thermal stress-related spatiotemporal variations in high-latitude coral reef benthic communities. Coral Reefs 2020, 39, 1661–1673. [Google Scholar] [CrossRef]
- Morri, C.; Montefalcone, M.; Lasagna, R.; Gatti, G.; Rovere, A.; Parravicini, V.; Baldelli, G.; Colantoni, P.; Bianchi, C.N. Through bleaching and tsunami: Coral reef recovery in the Maldives. Mar. Poll. Bull. 2015, 98, 188–200. [Google Scholar] [CrossRef]
- Montefalcone, M.; Morri, C.; Bianchi, C.N. Influence of local pressures on Maldivian coral reef resilience following repeated bleaching events, and recovery perspectives. Front. Mar. Sci. 2020, 7, 587. [Google Scholar] [CrossRef]
- Robinson, J.P.W.; Wilson, S.K.; Graham, N.A.J. Abiotic and biotic controls on coral recovery 16 years after mass bleaching. Coral Reefs 2019, 38, 1255–1265. [Google Scholar] [CrossRef]
- Vercelloni, J.; Mengersen, K.; Ruggeri, F.; Caley, M.J. Improved coral population estimation reveals trends at multiple scales on Australia’s Great Barrier Reef. Ecosystems 2017, 20, 1337–1350. [Google Scholar] [CrossRef]
- Bavestrello, G.; Boero, F. Necrosi e rigenerazione in Eunicella cavolinii (Anthozoa, Cnidaria) in Mar Ligure. Bollet. Mus. Ist. Biol. Univ. Genova 1986, 52, 295–300. [Google Scholar]
- Gaino, E.; Pronzato, R. Ultrastructural evidence of bacterial damage to Spongia officinalis fibres (Porifera, Demospongiae). Diseases Aquat. Organ. 1989, 6, 67–74. [Google Scholar] [CrossRef]
- Voultsiadou, E.; Dailianis, T.; Antoniadou, C.; Vafidis, D.; Dounas, C.; Chintiroglou, C.C. Aegean bath sponges: Historical data and current status. Rev. Fish. Sci. 2011, 19, 34–51. [Google Scholar] [CrossRef]
- Cerrano, C.; Bavestrello, G.; Bianchi, C.N.; Cattaneo-Vietti, R.; Bava, S.; Morganti, C.; Morri, C.; Pico, P.; Sarà, G.; Schiaparelli, S.; et al. A catastrophic mass-mortality episode of gorgonians and other organisms in the Ligurian Sea (Northwestern Mediterranean), summer 1999. Ecol. Lett. 2000, 3, 284–293. [Google Scholar] [CrossRef]
- Perez, T.; Garrabou, J.; Sartoretto, S.; Harmelin, J.G.; Francour, P.; Vacelet, J. Mass mortality of marine invertebrates: An unprecedent event in the North Occidental Mediterranean. CR Acad. Sci. Paris 2000, 323, 853–865. [Google Scholar] [CrossRef]
- Garrabou, J.; Coma, R.; Bensoussan, N.; Bally, M.; Chevaldonné, P.; Cigliano, M.; Diaz, D.; Harmelin, J.G.; Gambi, M.C.; Kersting, D.K.; et al. Mass mortality in Northwestern Mediterranean rocky benthic communities: Effects of the 2003 heat wave. Glob. Chang. Biol. 2009, 15, 1090–1103. [Google Scholar] [CrossRef]
- Rivetti, I.; Fraschetti, S.; Lionello, P.; Zambianchi, E.; Boero, F. Global warming and mass mortalities of benthic invertebrates in the Mediterranean Sea. PLoS ONE 2014, 9, e115655. [Google Scholar] [CrossRef]
- Marbà, N.; Gabriel, J.; Agusti, S.; Girard, C.; Duarte, C.M. Footprints of climate change on Mediterranean Sea biota. Front. Mar. Sci. 2015, 2, 56. [Google Scholar] [CrossRef]
- Kersting, D.K.; Bensoussan, N.; Linares, C. Long-term responses of the endemic reef-builder Cladocora caespitosa to Mediterranean warming. PLoS ONE 2013, 8, e70820. [Google Scholar] [CrossRef] [PubMed]
- Turicchia, E.; Abbiati, M.; Sweet, M.; Ponti, M. Mass mortality hits gorgonian forests at Montecristo Island. Dis. Aquat. Org. 2018, 131, 79–85. [Google Scholar] [CrossRef]
- Bensoussan, N.; Chiggiato, J.; Buongiorno Nardelli, B.; Pisano, A.; Garrabou, J. Insights on 2017 Marine Heat Waves in the Mediterranean Sea. In Copernicus Marine Service Ocean State, Report 3. J. Operat. Oceanogr. 2019. [Google Scholar] [CrossRef]
- Garrabou, J.; Gómez-Gras, D.; Ledoux, J.-B.; Linares, C.; Bensoussan, N.; López-Sendino, P.; Bazairi, H.; Espinosa, F.; Ramdani, M.; Grimes, S.; et al. Collaborative database to track mass mortality events in the Mediterranean Sea. Front. Mar. Sci. 2019, 6, 707. [Google Scholar] [CrossRef]
- Özalp, H.B.; Kersting, D.K. A pan-Mediterranean extinction? Pinna nobilis mass mortality has reached the Turkish straits system. Mar. Biodivers. 2020, 50, 81. [Google Scholar]
- Kruzic, P.; Rodic, P. Impact of climate changes on coralligenous community in the Adriatic Sea. In Proceedings of the Second Mediterranean Symposium of Coralligenous and Other Calcareous Bioconcretions, Portoroz, Slovenia, 29–30 October 2014; RACSPA: Tunis, Tunisia, 2014; pp. 100–105. [Google Scholar]
- Ballesteros, E. Mediterranean coralligenous assemblages: A synthesis of present knowledge. Oceanogr. Mar. Biol. 2006, 44, 123–195. [Google Scholar]
- Ezzat, L.; Merle, P.L.; Furla, P.; Buttler, A.; Ferrier-Pagès, C. The response of the Mediterranean gorgonian Eunicella singularis to thermal stress is independent of its nutritional regime. PLoS ONE 2013, 8, e64370. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.; Cohu, S.; Vignot, C.; Zimmerman, G.; Gattuso, J.P. One-year experiment on the physiological response of the Mediterranean crustose coralline alga, Lithophyllum cabiochae, to elevated pCO2 and temperature. Ecol. Evol. 2013, 3, 676–693. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Prieto, C. Light and temperature requirements for survival, growth and reproduction of the crustose coralline Lithophyllum stictaeforme from the Mediterranean Sea. Bot. Mar. 2016, 59, 95–104. [Google Scholar] [CrossRef]
- Pagès-Escolà, M.; Hereu, B.; Garrabou, J.; Montero-Serra, I.; Gori, A.; Gómez-Gras, D.; Figuerola, B.; Linares, C. Divergent responses to warming of two common co-occurring Mediterranean bryozoans. Sci. Rep. 2018, 8, 17455. [Google Scholar] [CrossRef]
- Gómez-Gras, D.; Linares, C.; de Caralt, S.; Cebrian, E.; Frleta-Valić, M.; Montero-Serra, I.; Pagès-Escolà, M.; López-Sendino, P.; Garrabou, J. Response diversity in Mediterranean coralligenous assemblages facing climate change: Insights from a multispecific thermotolerance experiment. Ecol. Evol. 2019, 9, 4168–4180. [Google Scholar] [CrossRef]
- Garrabou, J.; Perez, T.; Sartoretto, S.; Harmelin, J.G. Mass mortality event in red coral Corallium rubrum populations in the Provence Region (France, NW Mediterranean). Mar. Ecol. Prog. Ser. 2001, 217, 263–272. [Google Scholar] [CrossRef]
- Cebrian, E.; Uriz, M.J.; Garrabou, J.; Ballesteros, E. Sponge mass mortalities in a warming Mediterranean Sea: Are Cyanobacteria harboring species worse off? PLoS ONE 2011, 6, e20211. [Google Scholar]
- Cocito, S.; Sgorbini, S. Long-term trend in substratum occupation by a clonal, carbonate bryozoan in a temperate rocky reef in times of thermal anomalies. Mar. Biol. 2013, 161, 17–27. [Google Scholar] [CrossRef]
- Hereu, B.; Kersting, D.K. Diseases of coralline algae in the Mediterranean Sea. Coral Reefs 2016, 35, 713. [Google Scholar] [CrossRef]
- Cerrano, C.; Bavestrello, G. Medium-term effects of die-off of rocky benthos in the Ligurian Sea. What can we learn from gorgonians? Chem. Ecol. 2008, 24, 73–82. [Google Scholar] [CrossRef]
- Coma, R.; Linares, C.; Ribes, M.; Diaz, D.; Garrabou, J.; Ballesteros, E. Consequences of a mass mortality in populations of Eunicella singularis (Cnidaria:Octocorallia) in Menorca (NW Mediterranean). Mar. Ecol. Prog. Ser. 2006, 327, 51–60. [Google Scholar] [CrossRef]
- Huete-Stauffer, C.; Vielmini, I.; Palma, M.; Navone, A.; Panzalis, P.; Vezzulli, L.; Misic, C.; Cerrano, C. Paramuricea clavata (Anthozoa, Octocorallia) loss in the Marine Protected Area of Tavolara (Sardinia, Italy) due to a mass mortality event. Mar. Ecol. Evol. Persp. 2011, 32, 107–116. [Google Scholar] [CrossRef]
- Teixido, N.; Casas, E.; Cebrian, E.; Linares, C.; Garrabou, J. Impacts on coralligenous outcrop biodiversity of a dramatic coastal storm. PLoS ONE 2013, 8, e53742. [Google Scholar] [CrossRef]
- Bavestrello, G.; Cerrano, C.; Zanzi, D.; Cattaneo-Vietti, R. Damage by fishing activities in the gorgonian coral Paramuricea clavata in the Ligurian Sea. Aquat. Conserv. 1997, 7, 253–262. [Google Scholar] [CrossRef]
- Coma, R.; Pola, E.; Ribes, M.; Zabala, M. Long-term assessment of temperate octocoral mortality patterns, protected vs. unprotected areas. Ecol. Appl. 2004, 14, 1466–1478. [Google Scholar] [CrossRef]
- Cebrian, E.; Linares, C.; Marschal, C.; Garrabou, J. Exploring the effects of invasive algae on the persistence of gorgonian populations. Biol. Invasions 2012, 14, 2647–2656. [Google Scholar] [CrossRef]
- Bianchi, C.N.; Azzola, A.; Bertolino, M.; Betti, F.; Bo, M.; CattaneoVietti, R.; Cocito, S.; Montefalcone, M.; Morri, C.; Oprandi, A.; et al. Consequences of the marine climate and ecosystem shift of the 1980–90s on the Ligurian Sea biodiversity (NW Mediterranean). Eur. Zool. J. 2019, 86, 458–487. [Google Scholar] [CrossRef]
- Verdura, J.; Linares, C.; Ballesteros, E.; Coma, R.; Uriz, M.J.; Bensoussan, N.; Cebrian, E. Biodiversity loss in a Mediterranean ecosystem due to an extreme warming event unveils the role of an engineering gorgonian species. Sci. Rep. 2019, 9, 5911. [Google Scholar] [CrossRef] [PubMed]
- Mistri, M.; Ceccherelli, V.U. Effects of a mucilage event on the Mediterranean gorgonian Paramuricea clavata. 1. Short term impacts at the population and colony levels. It. J. Zool. 1996, 63, 221–230. [Google Scholar] [CrossRef]
- Giuliani, S.; Virno Lamberti, C.; Sonni, C.; Pellegrini, D. Mucilage impact on gorgonians in the Tyrrhenian Sea. Sci. Total Environ. 2005, 353, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Piazzi, L.; Atzori, F.; Cadoni, N.; Cinti, M.F.; Frau, F.; Ceccherelli, G. Benthic mucilage blooms threaten coralligenous reefs. Mar. Environ. Res. 2018, 140, 145–151. [Google Scholar] [CrossRef]
- Martin, Y.; Bonnefort, J.L.; Chancerelle, L. Gorgonians mass mortality during the 1999 late summer in french Mediterranean coastal waters: The bacterial hypothesis. Water Res. 2002, 36, 779–782. [Google Scholar] [CrossRef]
- Vezzulli, L.; Previati, M.; Pruzzo, C.; Marchese, A.; Bourne, D.G.; Cerrano, C. Vibrio infections triggering mass mortality events in a warming Mediterranean Sea. Environ. Microbiol. 2010, 12, 2007–2019. [Google Scholar] [CrossRef]
- Vezzulli, L.; Pezzati, E.; Huete-Stauffer, C.; Pruzzo, C.; Cerrano, C. 16SrDNA pyrosequencing of the Mediterranean gorgonian Paramuricea clavata reveals a link among alterations in bacterial holobiont members, anthropogenic influence and disease outbreaks. PLoS ONE 2013, 8, e67745. [Google Scholar] [CrossRef]
- Mistri, M.; Ceccherelli, V.U. Effects of a mucilage event on the Mediterranean gorgonian Paramuricea clavata. 2. Population recovery after two years. It. J. Zool. 1996, 63, 231–236. [Google Scholar] [CrossRef]
- Linares, C.; Coma, R.; Diaz, D.; Zabala, M.; Hereu, B.; Dantart, L. Immediate and delayed effects of a mass mortality event on gorgonian population dynamics and benthic community structure in the NW Mediterranean Sea. Mar. Ecol. Prog. Ser. 2005, 305, 127–137. [Google Scholar] [CrossRef]
- Cupido, R.; Cocito, S.; Sgorbini, S.; Bordone, A.; Santangelo, G. Response of a gorgonian (Paramuricea clavata) population to mortality events: Recovery or loss? Aquat. Conserv. Mar. Freshw. Ecosyst. 2008, 18, 984–992. [Google Scholar] [CrossRef]
- Bramanti, L.; Bendetti, M.C.; Cupido, S.; Priori, C.; Erra, F.; Iannelli, M.; Santangelo, G. Demography of animal forests: The example of Mediterranean gorgonians. In Marine Animal Forests; Rossi, S., Bramanti, L., Gori, A., Orejas Saco del Valle, C., Eds.; Springer: Cham, Switzerland, 2017. [Google Scholar] [CrossRef]
- Cerrano, C.; Danovaro, R.; Gambi, C.; Pusceddu, A.; Riva, A.; Schiaparelli, S. Gold coral (Savalia savaglia) and gorgonian forests enhance benthic biodiversity and ecosystem functioning in the mesophotic zone. Biodivers. Conserv. 2010, 19, 153–167. [Google Scholar] [CrossRef]
- Casas-Güell, E.; Teixidó, N.; Garrabou, J.; Cebrian, E. Structure and biodiversity of coralligenous assemblages over broad spatial and temporal scales. Mar. Biol. 2015, 162, 901–912. [Google Scholar] [CrossRef]
- Ponti, M.; Grech, D.; Mori, M.; Perlini, R.A.; Ventra, V.; Panzalis, P.A.; Cerrano, C. The role of gorgonians on the diversity of vagile benthic fauna in Mediterranean rocky habitats. Mar. Biol. 2016, 163, 1–14. [Google Scholar] [CrossRef]
- Ponti, M.; Turicchia, E.; Ferro, F.; Cerrano, C.; Abbiati, M. The understorey of gorgonian forests in mesophotic temperate reefs. Aquat. Conserv. Mar. Freshw. Ecosyst. 2018, 28, 1153–1166. [Google Scholar] [CrossRef]
- Gori, A.; Bavestrello, G.; Grinyó, J.; Dominguez-Carrió, C.; Ambroso, S.; Bo, M. Animal forests in deep coastal bottoms and continental shelf of the Mediterranean Sea. In Marine Animal Forests; Rossi, S., Bramanti, L., Gori, A., Orejas, C., Eds.; Springer: Cham, Switzerland, 2017. [Google Scholar] [CrossRef]
- Rossi, S.; Bramanti, L.; Gori, A.; Orejas, C. An overview of the animal forests of the world. In Marine Animal Forests; Rossi, S., Bramanti, L., Gori, A., Orejas, C., Eds.; Springer: Cham, Switzerland, 2017. [Google Scholar] [CrossRef]
- Ponti, M.; Perlini, R.A.; Ventra, V.; Grech, D.; Abbiati, M.; Cerrano, C. Ecological shifts in Mediterranean coralligenous assemblages related to gorgonian forest loss. PLoS ONE 2014, 9, e102782. [Google Scholar] [CrossRef] [PubMed]
- Cerrano, C.; Arillo, A.; Azzini, F.; Calcinai, B.; Castellano, L.; Muti, C.; Valisano, L.; Zega, G.; Bavestrello, G. Gorgonian population recovery after a mass mortality event. Aquat. Conserv. 2005, 15, 147–157. [Google Scholar] [CrossRef]
- Santangelo, G.; Cupido, R.; Cocito, S.; Bramanti, L.; Priori, C.; Erra, F.; Iannelli, M. Effects of increased mortality on gorgonian corals (Cnidaria, Octocorallia): Different demographic features may lead affected populations to unexpected recovery and new equilibrium points. Hydrobiologia 2015, 759, 171–187. [Google Scholar] [CrossRef]
- Underwood, A.J. On beyond BACI: Sampling designs that might reliably detect environmental disturbances. Ecol. Appl. 1994, 4, 3–15. [Google Scholar] [CrossRef]
- Benedetti-Cecchi, L. Beyond BACI: Optimization of environmental sampling designs through monitoring and simulation. Ecol. Appl. 2001, 11, 783–799. [Google Scholar] [CrossRef]
- Piazzi, L.; Gennaro, P.; Montefalcone, M.; Bianchi, C.N.; Cecchi, E.; Morri, C.; Serena, F. STAR: An integrated and standardized procedure to evaluate the ecological status of coralligenous reefs. Aquat. Conserv. Mar. Freshw. Ecosys. 2019, 29, 189–201. [Google Scholar] [CrossRef]
- Cecchi, E.; Gennaro, P.; Piazzi, L.; Ricevuto, E.; Serena, F. Development of a new biotic index for ecological status assessment of Italian coastal waters based on coralligenous macroalgal assemblages. Eur. J. Phycol. 2014, 49, 298–312. [Google Scholar] [CrossRef]
- Piazzi, L.; Gennaro, P.; Cecchi, E.; Serena, F.; Bianchi, C.N.; Morri, C.; Montefalcone, M. Integration of ESCA index through the use of sessile invertebrates. Sci. Mar. 2017, 81, 1–8. [Google Scholar]
- Anderson, M.J.; Ellingsen, K.E.; McArdle, B.H. Multivariate dispersion as a measure of beta diversity. Ecol. Lett. 2006, 9, 683–693. [Google Scholar] [CrossRef] [PubMed]
- Anderson, M.J. A new method for non-parametric multivariate analysis of variance. Austral Ecol. 2001, 26, 32–46. [Google Scholar]
- Benedetti-Cecchi, L.; Pannacciulli, F.; Bulleri, F.; Moschella, P.; Airoldi, L.; Relini, G.; Cinelli, F. Predicting the consequences of anthropogenic disturbance: Large-scale effects of loss of canopy algae on rocky shores. Mar. Ecol. Progr. Ser. 2001, 214, 137–150. [Google Scholar] [CrossRef]
- Montefalcone, M.; Vassallo, P.; Gatti, G.; Parravicini, V.; Paoli, C.; Morri, C.; Bianchi, C.N. The exergy of a phase shift: Ecosystem functioning loss in seagrass meadows of the Mediterranean Sea. Estuar. Coast. Shelf Sci. 2015, 156, 186–194. [Google Scholar] [CrossRef]
- Carugati, L.; Gatto, B.; Rastelli, E.; Lo Martire, M.; Coral, C.; Greco, S.; Danovaro, R. Impact of mangrove forests degradation on biodiversity and ecosystem functioning. Sci. Rep. 2018, 8, 13298. [Google Scholar] [CrossRef]
- Valisano, L.; Notari, F.; Mori, M.; Cerrano, C. Temporal variability of sedimentation rates and mobile fauna inside and outside a gorgonian garden. Mar. Ecol. 2016, 37, 1303–1314. [Google Scholar] [CrossRef]
- Linares, C.; Cebrian, E.; Coma, R. Effects of turf algae on recruitment and juvenile survival of gorgonian corals. Mar. Ecol. Prog. Ser. 2012, 452, 81–88. [Google Scholar] [CrossRef]
- Birrell, C.L.; McCook, L.J.; Willis, B.L. Effects of algal turfs and sediment on coral settlement. Mar. Pollut. Bull. 2005, 51, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Arnold, S.N.; Steneck, R.S.; Mumby, P.J. Running the gauntlet: Inhibitory effects of algal turfs on the processes of coral recruitment. Mar. Ecol. Prog. Ser. 2010, 414, 91–105. [Google Scholar] [CrossRef]
- Piazzi, L.; Atzori, F.; Cadoni, N.; Cinti, M.F.; Frau, F.; Ceccherelli, G. Monitoring non-indigenous macroalgae in a Mediterranean MPA: Lessons from a short-temporal variability of pristine habitats invasion. Ocean. Coast. Manag. 2021, in press. [Google Scholar] [CrossRef]
- Piazzi, L.; Gennaro, P.; Cecchi, E.; Bianchi, C.N.; Cinti, F.; Gatti, G.; Guala, I.; Morri, C.; Sartoretto, F.; Serena, F.; et al. Ecological Status of Coralligenous Assemblages: Ten years of application of the ESCA index from local to wide scale validation. Ecol. Ind. 2021, 121, 107077. [Google Scholar] [CrossRef]
- Rossi, S. The destruction of the ‘animal forests’ in the oceans: Towards an over-simplification of the benthic ecosystems. Ocean Coast Manag. 2013, 84, 77–85. [Google Scholar] [CrossRef]
- Boudouresque, C.F.; Blanfuné, A.; Personnic, S.; Ruitton, S.; Thibaut, T.; Verlaque, M. Where seaweed forests meet animal forests: The examples of macroalgae in coral reefs and the Mediterranean coralligenous ecosystem. In Marine Animal Forests; Rossi, S., Bramanti, L., Gori, A., Orejas, C., Eds.; Springer: Cham, Switzerland, 2016. [Google Scholar] [CrossRef]
- Gatti, G.; Bianchi, C.N.; Morri, C.; Montefalcone, M.; Sartoretto, S. Coralligenous reefs state along anthropized coasts: Application and validation of the COARSE index, based on a rapid visual assessment (RVA) approach. Ecol. Ind. 2015, 52, 567–576. [Google Scholar] [CrossRef]
- Ferrigno, F.; Russo, G.F.; Sandulli, R. Coralligenous Bioconstructions Quality Index (CBQI): A synthetic indicator to assess the status of different types of coralligenous habitats. Ecol. Ind. 2017, 82, 271–279. [Google Scholar] [CrossRef]
- Enrichetti, F.; Bo, M.; Morri, C.; Montefalcone, M.; Toma, M.; Bavestrello, G.; Tunesi, L.; Canese, S.; Giusti, M.; Salvati, E.; et al. Assessing the environmental status of temperate mesophotic reefs: A new, integrated methodological approach. Ecol. Ind. 2019, 102, 218–229. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).