Abstract
Amphibians are the most threatened vertebrate class based on the IUCN Red List. Their decline has been linked to anthropogenic activities, with wildfires being among the most conspicuous agents of habitat alterations affecting native amphibians. In 2011, the most destructive wildfire in Texas history occurred in the Lost Pines ecoregion of central Texas, USA, burning 39% of the 34,400 ha forest and drastically decreasing available habitats for many native wildlife species, including the green tree frog (Hyla cinerea). We investigated use of PVC pipes as artificial refuges for green tree frogs in different habitats within this post-fire pine forest. We monitored green tree frog use of small (diameter 38.1-mm, 1.5 inch) and large (diameter 50.8-mm, 2 inch) pipes located adjacent to, and 5 m from, ponds in burned and unburned areas over a 5-month period. We caught 227 frogs, 101 (24 adults and 77 juveniles) in burned and 126 (61 adults, 63 juveniles, and 2 unknown) in unburned areas. A relationship between pipe use by adults and/or juveniles and pipe location in burned versus unburned areas was found, but pipe use by adults and/or juveniles and pipe size were independent. Pipe use by adults and/or juveniles and pipe size were also independent. Juveniles were more frequently observed in pipes located adjacent to ponds. Our results confirmed that PVC pipes merit consideration as a simple, inexpensive, conservation tool to aid in restoration of green tree frog populations after high-severity wildfires. Such artificial refuges may be particularly important for survival of juveniles in severely altered post-fire habitats.
1. Introduction
Amphibians are important components of ecosystems throughout the temperate and tropical regions of the world [1]. They are considered as both predator and prey, and the cumulative biomass of amphibians shows they have key roles in community structure [2], energy flow [3], and nutrient cycling [4]. However, amphibian populations are declining rapidly, faster than those of birds and mammals, and the declines could lead to species’ extinction [5]. Although diseases such as chytridiomycosis [6,7], parasitism [8], toxic chemicals [9], increased levels of UVB light [9], and changing climatic conditions [10,11] impose negative impacts on amphibian populations, habitat loss due to urbanization or high-severity wildfires is the primary reason for local population extirpation [2,12]. This is because amphibian species have low migration rates, high mortality rates while moving across roadways, and narrow habitat tolerances [12]. With the number of threatened and endangered species of amphibians rising, researchers have been focusing not only on the issue of decline per se but also on conservation practices for sustaining amphibian species and their population sizes [13,14]. Providing artificial refuges—such as PVC pipes [15,16], bamboo [17], and coverboard [18]—is one of the conservation methods.
The Lost Pines ecoregion in central Texas is a unique 21 km belt of loblolly pines (Pinus taeda) which are closely related genetically to, but separated by more than 160 km from, the loblolly pines in the vast Piney Woods region of east Texas [19]. The ecoregion contains a variety of herpetofauna, including 12 species of amphibians [20]. The ecoregion was logged extensively during the 1800s and early 1900s, and fire suppression since the mid-1900s lead to the accumulation of heavy fuel loads [21]. In September 2011, a large wildfire burned 39% of the 34,400 ha of the Lost Pines and 96% of the area within the Bastrop State Park [21]. The fire started during a drought year and was fanned by strong winds from Tropical Storm Lee [22]. Recent studies [23] focused on the post-fire regrowth of vegetation and our photos taken in 2014 (Figure 1) show that more severely burned areas are still in the process of overstory vegetation recovery. Even though high-severity fires have been shown to increase nutrient levels and productivity in aquatic environments [24] and increase diversity of understory plants in terrestrial environments [25], recently-burned habitats lack the type of canopy cover needed by many amphibian species [21,26]. Hence, there is an urgent need to create artificial refuges until the habitat required by amphibian species has had time to restore itself.

Figure 1.
Slow vegetation response at post high-severity fire areas in Bastrop in 2014 (photos by Tomasz E. Koralewski).
The use of PVC pipes is a widely accepted technique for recreating refuges for hylid tree frogs [15,27]. For example, PVC pipes offer artificial refuge sites for many arboreal amphibian species that would otherwise seek shelter under bark or in natural cavities [28]. Campbell et al. found that observations of green tree frogs (Hyla cinerea), pinewoods tree frog (Hyla femoralis), and squirrel tree frog (Hyla squirella) in PVC pipes were 6- to 10-fold higher during dry seasons than during wet seasons, suggesting that pipe refugia provided shelter from the dry and cold conditions [29]. Although PVC pipes have been widely used as artificial refuges and as a sampling tool in the southeast USA [26,29,30,31,32], to our knowledge, studies have used PVC pipes to evaluate amphibian assemblages only under prescribed fire conditions [26,31].
In this study, we used the Bastrop County Complex fire, the most destructive wildfire in Texas history [33], as an unique opportunity, and focused on the green tree frog, which is an important component of the amphibian community in the Lost Pines ecoregion [20]. Although there are no indications of a range-wide decline, Corbett et al. (2003) have shown that green tree frog populations have declined where wildfires have occurred [34]. We investigated whether the use of PVC pipes by green tree frog adults and juveniles was related to: (1) pipe location in burned versus unburned areas, (2) pipe size (diameter), and/or (3) pipe location adjacent versus non-adjacent to ponds. Knowing these relationships will facilitate more cost-efficient conservation practices.
2. Materials and Methods
2.1. Focal Species
Green tree frogs are an arboreal species commonly found in natural and suburban areas [35]. They are endemic to the southern region of the United States and can occur from Texas to Delaware [35]. Even though they are well adapted to urban environments, they prefer open canopy forests [36]. They are commonly found clinging to twigs, grasses, and low branches in cypress ponds, water lily prairies and marshes [37]. Green tree frogs are iteroparous. In the Lost Pines, their breeding season occurs from March to July and peaks in April or early May. They breed in permanent ponds and marshes and do not disperse far from water [35]. Most commonly, the maximum dispersal distance is <1.0 km [38]. Females produce clutch sizes of approximately 400 eggs, with pond vegetation providing a substrate for eggs and tadpoles [39]. Green tree frogs play a role in controlling populations of mosquitoes and other insects in the ecosystem [40]. With regard to diseases, Batrachochytrium dendrobatidis has not been detected in green tree frogs in the Lost Pines, however, B. dendrobatidis has been detected in gray tree frogs (Hyla versicolor) in the Lost Pines [41,42].
2.2. Study Area
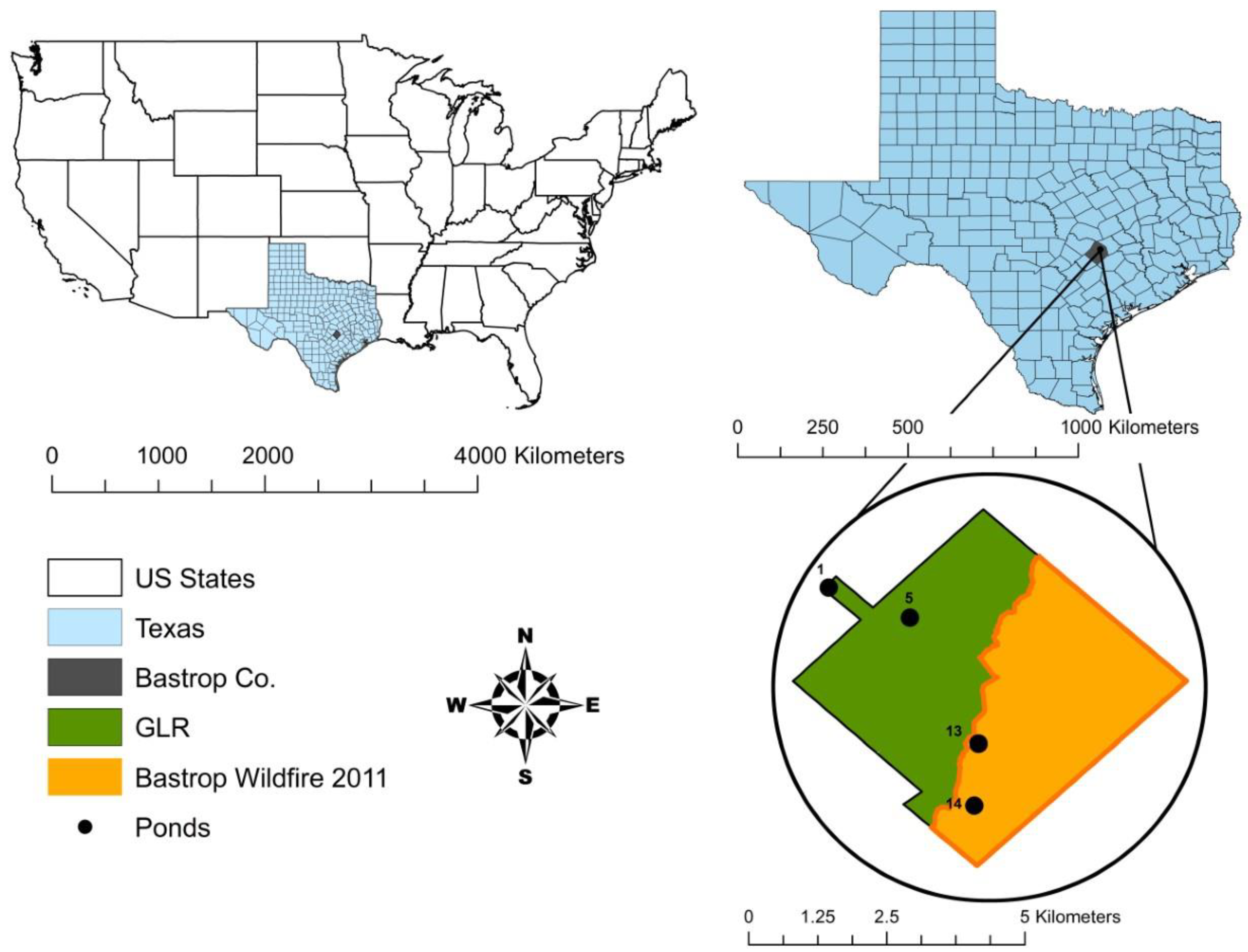
Our study area is located at the Griffith League Ranch, Bastrop, Texas, with centroid latitude and longitude of 30.196121 N, −97.229185 W (WGS84 coordinate system) and elevation of 184 m, within the Lost Pines ecoregion and the range of the 2011 Bastrop wildfire (Figure 2). The ranch is approximately 1948 ha. As a result of the 2011 wildfire, which burned for 55 days, 50.7% of the ranch was burned in September and another 4.1% was burned in October [21]. On the unburned areas, the overstory vegetation is dominated by loblolly pine, eastern red cedar (Juniperus virginiana), and post oak (Quercus stellata), and the understory is dominated by yaupon holly (Ilex vomitoria), American beautyberry (Callicarpa americana), and farkleberry (Vaccinium arboreum) [21]. On the burned areas, the wildfire resulted in a mean overstory tree mortality of 87% and a mean pole-sized tree mortality of 97%, which was categorized as a high-severity fire based on metrics designed by Ryan and Noste [43], Turner et al. [44], and Ryan [45]. Even though the overstory vegetation is recovering slowly on the burned areas, the understory is recovering more rapidly with poke weed (Phytolacca americana), panic grasses (Dicanthelium spp.), and sedges (Cyperus spp.) [46].
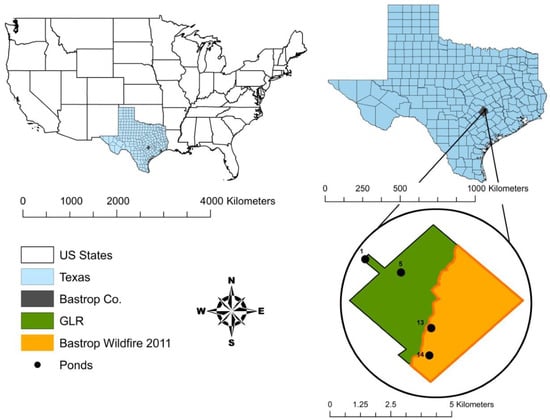
Figure 2.
Location of the study ponds in unburned (grey, ponds 1 and 5) and burned (checkered, ponds 13 and 14) areas of the Griffith League Ranch (GLR) in Bastrop County, Texas.
2.3. Methods
We conducted our study using ponds on the Griffith League ranch property. We first used preliminary data collected during call surveys conducted January through May 2015 to assess which ponds were likely to support breeding populations of green tree frogs. These ponds were selected based on accessibility (near roads), especially in the burned portion of the ranch where many ponds were not accessible or were in hazardous areas away from the roads. In May 2015, Pond 1, Pond 5, and Pond 13 each had approximately 20 adult males calling; therefore, we expected that there would be successful breeding in these ponds. The maximum number of frogs calling at Pond 14 was three. Hence, we selected these four ponds to conduct our study. Pond 1 and Pond 5 were located in the unburned areas and Pond 13 and Pond 14 were located in the burned areas.
Even though a total of 80 PVC pipes were established around the four selected ponds, only 60 pipes around three ponds were used because Pond 14 went dry in mid-July and stayed dry for the rest of the survey period. The 20 PVC pipes around Pond 14 provided no useful data with only one juvenile caught there. The snout to urostyle length and mass of the juvenile were 20.8 mm and 0.44 g. Five PVC pipes of 38.1-mm (1.5-inch) and 5 PVC pipes of 50.8-mm (2-inch) diameters were placed alternately around the perimeter of each pond, at the water edge, evenly distributed around the pond. The PVC pipes were staked into the ground. Another 5 PVC pipes of 38.1-mm diameter and 5 PVC pipes of 50.8-mm diameter were also placed alternately in the surrounding vegetation, 5 m away from pond, evenly distributed around as well. The PVC pipes were tied to a nearby tree with a zip tie. All pipes were five feet tall and vertically positioned at a 90-degree angle from the ground. Frogs can climb to the top of the pipe from the outside and then enter inside. Pipes that were placed in vegetation were tied to various types of trees present in the area, including 3 elms (Ulmus), 25 junipers (Juniperus), 3 loblolly pines, 4 oaks (Quercus), 5 willows (Salix), and 15 yaupons surrounded in a 5-m radius from pipes at Pond 1, 26 junipers, 15 loblolly pines, 1 willow, and 27 yaupons at Pond 5, and 1 elm, 12 dead oaks, 3 junipers, 10 loblolly pines, 7 oaks, 3 sumacs (Rhus), 1 willow, and 30 yaupons at Pond 13. We checked the pipes on 15 different occasions (days) from 21 June to 4 October 2015, with the frequency varying from once a week to once a month (i.e., September and October). Data collection included the locale of capture (specifying the pond and the pipe), snout to urostyle length (mm, using Tactix® stainless steel (150 mm) digital calipers with an accuracy of 0.0127 mm), and mass (g, using weigh gram scale (100 g) digital pocket scale with an accuracy of 0.01 g). Specimens exceeding 32 mm in snout to urostyle length were recorded as adults [47]. Upon arrival at each capture site, we recorded temperature and relative humidity (RH) using a Kestrel (Nielsen-Kellerman Company, Boothwyn, PA, USA).
Although the 60 PVC pipes may be considered as pseudo-replicates, given that green tree frogs could move into and out of the PVC pipes freely between sampling periods, we assumed the captures on any given day were not related to previous captures [48]. To further minimize pseudo-replication issues, we treated each survey day as a sampling unit and used data only for each individual’s first capture. We analyzed data on adults and juveniles separately. Since part of the data (from Pond 14) was excluded, three 2 × 2 contingency tables and chi-squared tests were used to test if there was a relationship between pipe use by adults and/or juveniles and (1) pipe location in burned versus unburned areas, (2) pipe size (diameter), and/or (3) pipe location adjacent versus non-adjacent to ponds. Specifically, the null hypotheses were that there were no relationships between adult or juvenile pipe use and (1), (2), or (3) above. We set the level of significance of all statistical tests at α = 0.05. All statistical analyses were conducted using R version 3.2.4 [49].
3. Results
The highest and lowest temperatures recorded during the study were 36.1 °C in August and 16.8 °C in October, with mean temperature declines from September (24.9 °C) to October (20.4 °C). The highest and lowest RH recorded were 88.65% in June and 46.33% in September, with mean RH decreasing from June (80.5%) to October (53.5%). The total number of green tree frogs recorded throughout the experiment was 227. There were 140 juveniles, 85 adults, and 2 with unknown life stage (which escaped during measuring) using PVC pipes. The mean (± standard error) snout to urostyle length and weight were 32.39 (±0.81) mm and 2.95 (±0.20) g, respectively, and the more specific measurement information from burned and unburned areas is in Table 1. The values of mean snout to urostyle length and mean weight of green tree frogs were greater in unburned areas than in the burned areas (Table 1).

Table 1.
Mean snout to urostyle length and mean weight of green tree frogs captured in burned versus unburned areas.
Adults were found more frequently in unburned areas while juveniles were observed with equal frequency in both areas (Table 2). Based on X2 test, there was a relationship between pipe use by adults and/or juveniles and pipe location in burned versus unburned areas (X2 = 22.1, df = 1, p-value = 0.000002621). However, pipe use by adults and/or juveniles and pipe size were independent (X2 = 0.1, df = 1, p-value = 0.71432; Table 3). Juveniles were caught more frequently than adults regardless of the pipe diameter. Similarly, pipe use by adults and/or juveniles and pipe location adjacent to, versus 5 m from, ponds were independent (X2 = 3.6, df = 1, p-value = 0.0563; Table 4). Juveniles were more frequently observed in pipes located adjacent to ponds.

Table 2.
Number of adult and juvenile green tree frogs captured in burned versus unburned areas.

Table 3.
Number of adult and juvenile green tree frogs captured in small versus large (diameter) PVC pipes.

Table 4.
Number of adult and juvenile green tree frogs captured in PVC pipes adjacent to, versus 5 m from, ponds.
4. Discussion
We used the Bastrop County Complex fire, the most destructive wildfire in Texas history, as a unique opportunity to investigate how green tree frogs utilized PVC pipes under ‘unprescribed’ fire conditions. Prescribed fire and wildfire have been defined differently by different countries and/or land management agencies [50,51]. However, in general, prescribed fire is the intentional use of wildland fire to meet resource management objectives [52], while wildfire is an unplanned, unwanted, uncontrolled fire in an area of combustible vegetation starting in rural areas and urban areas [53]. Amphibians as a group are taxonomically and ecologically diverse; thus, responses to fire are expected to vary widely among species and among geographic regions [54]. Studies have shown that prescribed fires have no or little effect on overall amphibian communities in a southern Appalachian upland hardwood forest [55] and in an oak/hickory forest in western Kentucky [56], whereas severe wildfires decrease amphibian geographic ranges and species richness in Glacier National Park, Montana [57] and in San Diego County, California, USA [58], respectively.
In this study, adult and juvenile green tree frogs used intentionally placed vertical PVC pipes in central Texas. Interestingly, juveniles were particularly abundant in the pipes and were often found in groups within the same pipe. A plausible inference that might be drawn from these observations is that juveniles used the PVC pipes as a part of a post-emergence survival strategy. However, little is known about the behavior of juvenile green tree frogs and post-emergence community structures.
Green tree frog juveniles in burned areas extensively used PVC pipes as artificial refuges in the lack of live woody vegetation and other natural cover structures. Unburned areas were surrounded by very thick understory shrub (e.g., yaupon) in combination with canopy pine trees providing abundant cover that tree frogs preferred over artificial cover structures (Figure 3). The abundant cover could explain the lower capture rates at unburned Pond 1. Moreover, prior to the 2011 fire, unburned Ponds 1 and burned 13 were very similar in their upland vegetation structures based on the survey conducted by Brown et al. [59], further supporting our hypothesis.

Figure 3.
Ponds sampled on the Griffith League Ranch in Bastrop County, Texas. Ponds 1 (top left) and 5 (top right) are located in the unburned areas (photos by Andrea Villamizar-Gomez and Ivana Mali). Ponds 13 (bottom left) and 14 (bottom right) are located in the burned areas (photos by Donald J. Brown and Ivana Mali).
Green tree frogs are relatively large hylids that range in snout-vent length from 32–64 mm [37]. Initially, we hypothesized that there would be a relationship between PVC pipe use by adults and/or juveniles and pipe size (diameter), however, we found no such relationship. A factor that may have influenced this is the small difference in pipe sizes, and preferences may have been detected if the difference in pipe diameters had been greater. Zacharow et al. (2003) have shown that occupancy can vary among pipes of three different sizes (19, 51, and 77 mm), which indicates that tree frogs are selective in their use of PVC pipes [15]. It is possible that microclimate within the pipe plays an important role in their use. Zacharow et al. (2003) indicated that occupancy could vary among pipes of different shapes and also could depend on whether a cap is present on the end to retain moisture [15]. Moreover, a biomechanical perspective might provide some physics-based arguments for pipe size preferences. Hill et al. (2018) have shown that tree frog climbing performance scales inversely with pipe diameter [60]. Hill et al. (2018) and Langowski et al. (2019) have shown that an increased surface roughness can challenge the attachment abilities of tree frogs [60,61]. Endlein et al. (2013) have shown that excessive substrate wetting can prevent functioning of tree frog adhesion mechanisms. Perhaps pipe sizes, their roughness, and their substrate wetting conditions in our study were such that they did not affect the ability of green tree frogs to grip the pipes [62]. In the future, it may be useful to assess the microclimates within pipes of different sizes in comparison to the outside environment and to observe the bio-adhesion ability of tree frogs to gain a deeper understanding of the biomechanics involved and thus improve future designs of synthetic tree frog refuges.
Even though adults showed no preference for pipes located adjacent to—versus 5 m from—ponds, juveniles showed a clear preference for pipes located adjacent to ponds. This perhaps resulted from the relatively short dispersal distances of emerging juvenile hylid frogs [35]. However, the potential caveat could be that the pipes farther away from ponds covered larger areas, hence the chance of frogs encountering them was lower. If we would have measured the pond size, we could have corrected the number of captures in view of the increased area sampled by the pipes located 5 m from the ponds.
Some limitations to the present study that are important to note include: (1) the lack of sufficient pond site replicates, particularly relatively inaccessible burned areas; (2) the lack of long-term data collection; (3) the lack of pre- and post-burn tree frog population size estimates at each pond site; (4) the lack of vegetation plot sampling data from the different habitat types; and (5) the exclusion of other amphibian species from the survey. The lack of sufficient pond site replicates limited our ability to interpret the statistically significant differences that we reported in terms of practical significance within a tree frog conservation context. The lack of long-term data collection limited our ability to calculate the probabilities of detection and occupancy across seasons. This information would help us know more about green tree frog population recovery after the high-severity wildfire. The lack of pre- and post-burn tree frog population size estimates and the lack of vegetation plot sampling data from the different habitat types limited our ability to tell if differences in frog use of PVC pipes among pond sites were confounded with vegetation differences among pond sites. It is also important to note that this study was conducted following a high-severity wildfire, and that the severely burned areas were still in the process of restoration [23]. Thus, our results may not be generally applicable to all post-burn management scenarios. Indeed, low-severity fires can help maintain suitable habitat conditions for many amphibian species by reducing canopy cover and thus creating favorable light regimes, temperatures, and hydro periods [31,32,63]. Given their preference for open canopy forests, low-severity fires also might benefit green tree frogs. Even though our focus was on how green tree frogs utilize PVC pipes, and the number of other amphibian species utilizing the PVC pipes was low (count of gray tree frog, Hyla versicolor, was 39), it is important to consider surveying other amphibian species to learn their spatial segregation, coexistence, and competition [29,64].
A further consideration within a management context is the potential impact of non-target captures [28,65]. We did not observe non-target captures in the present study, but significant southern flying squirrel (Glaucomys volans) mortality in PVC pipes placed in trees has been observed in Georgia [28], and dead birds have been found in open PVC mine markers in New Mexico [65]. Nonetheless, we would suggest that PVC pipes merit consideration as a simple, inexpensive, conservation tool to aid in the restoration of green tree frogs during restoration periods after high-severity wildfires.
Author Contributions
This study was conceived and designed by I.M. and M.R.J.F.; T.S., K.F., A.V.-G. and I.M. conducted the experiments; T.S., K.F. and A.V.-G. organized the data; H.-H.W., I.M. and T.S. conducted statistical analyses; and H.-H.W., I.M. and W.E.G. led the writing. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
All amphibians were collected under the TPWD permit no. SPR-0102-191 and IACUC no. 0417-0513-08.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgments
The study was supported by the 2015–2016 Undergraduate Research Fund of the Department of Wildlife and Fisheries Sciences, Texas A&M University, the 2015–2016 LAUNCH: Undergraduate Research Scholars, Texas A&M University, and the 2015–2016 ABS Conservation Scholars Fund, Texas A&M University. We also thank three anonymous reviewers for their time and effort.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Halliday, T. Why amphibians are important. Int. Zoo Yearb. 2008, 42, 7–14. [Google Scholar] [CrossRef]
- Ficetola, G.F.; De Bernardi, F. Amphibians in a human-dominated landscape: The community structure is related to habitat features and isolation. Biol. Conserv. 2004, 119, 219–230. [Google Scholar] [CrossRef]
- Regester, K.J.; Lips, K.R.; Whiles, M.R. Energy flow and subsidies associated with the complex life cycle of ambystomatid salamanders in ponds and adjacent forest in southern Illinois. Oecologia 2006, 147, 303–314. [Google Scholar] [CrossRef]
- Beard, K.H.; Vogt, K.A.; Kulmatiski, A. Top-down effects of a terrestrial frog on forest nutrient dynamics. Oecologia 2002, 133, 583–593. [Google Scholar] [CrossRef] [PubMed]
- Monastersky, R. Biodiversity: Life—A status report. Nat. News 2014, 516, 158. [Google Scholar] [CrossRef] [PubMed]
- Longcore, J.E.; Pessier, A.P.; Nichols, D.K. Batrachochytrium dendrobatidis gen. et sp. nov., a chytrid pathogenic to amphibians. Mycologia 1999, 91, 219–227. [Google Scholar] [CrossRef]
- Berger, L.; Speare, R.; Daszak, P.; Green, D.E.; Cunningham, A.A.; Goggin, C.L.; Slocombe, R.; Ragan, M.A.; Hyatt, A.D.; McDonald, K.R.; et al. Chytridiomycosis causes amphibian mortality associated with population declines in the rain forests of Australia and Central America. Proc. Natl. Acad. Sci. USA 1998, 95, 9031–9036. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnson, P.T.; Chase, J.M. Parasites in the food web: Linking amphibian malformations and aquatic eutrophication. Ecol. Lett. 2004, 7, 521–526. [Google Scholar] [CrossRef]
- Blaustein, A.R.; Romansic, J.M.; Kiesecker, J.M.; Hatch, A.C. Ultraviolet radiation, toxic chemicals and amphibian population declines. Divers. Distrib. 2003, 9, 123–140. [Google Scholar] [CrossRef] [Green Version]
- Araújo, M.B.; Thuiller, W.; Pearson, R.G. Climate warming and the decline of amphibians and reptiles in Europe. J. Biogeogr. 2006, 33, 1712–1728. [Google Scholar] [CrossRef]
- McMenamin, S.K.; Hadly, E.A.; Wright, C.K. Climatic change and wetland desiccation cause amphibian decline in Yellowstone National Park. Proc. Natl. Acad. Sci. USA 2008, 105, 16988–16993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cushman, S.A. Effects of habitat loss and fragmentation on amphibians: A review and prospectus. Biol. Conserv. 2006, 128, 231–240. [Google Scholar] [CrossRef]
- Dodd, C.K. Amphibian Ecology and Conservation: A Handbook of Techniques; Oxford University Press: Oxford, UK, 2010. [Google Scholar]
- Park, I.-K.; Park, D.; Borzée, A. Defining conservation requirements for the Suweon treefrog (Dryophytes suweonensis) using species distribution models. Diversity 2021, 13, 69. [Google Scholar] [CrossRef]
- Zacharow, M.; Barichivich, W.J.; Dodd, C.K., Jr. Using ground-placed PVC pipes to monitor hylid treefrogs: Capture biases. Southeast. Nat. 2003, 2, 575–590. [Google Scholar] [CrossRef]
- Pereira-Ribeiro, J.; Ferreguetti, Á.C.; Bergallo, H.G.; Rocha, C.F.D. Use of polyvinyl chloride pipes (PVC) as potential artificial shelters for amphibians in a coastal plain forest of southeastern Brazil. J. Coast. Conserv. 2017, 21, 327–331. [Google Scholar] [CrossRef]
- Whittaker, L.; Whitworth, A.; Fowler, A.; Brent-Smith, M.; Beirne, C.; Villacampa, J. Bamboo traps as refugia for Pristimantis olivaceus (Anura: Craugastoridae) and as breeding site for Osteocephalus castaneicola (Anura: Hylidae). Phyllomedusa J. Herpetol. 2015, 14, 157–161. [Google Scholar] [CrossRef] [Green Version]
- Pittman, S.E.; Dorcas, M.E. Catawba River corridor coverboard program: A citizen science approach to amphibian and reptile inventory. J. N. C. Acad. Sci. 2006, 122, 142–151. [Google Scholar]
- Bilan, M.V.; Hogan, C.T.; Carter, H.B. Stomatal opening, transpiration, and needle moisture in loblolly pine seedlings from two Texas seed sources. For. Sci. 1977, 23, 457–462. [Google Scholar]
- Brown, D.J.; Swannack, T.M.; Dixon, J.R.; Forstner, M.R. Herpetofaunal survey of the Griffith League Ranch in the lost pines ecoregion of Texas. Tex. J. Sci. 2011, 63, 101–112. [Google Scholar]
- Brown, D.J.; Duarte, A.; Mali, I.; Jones, M.C.; Forstner, M.R. Potential impacts of a high severity wildfire on abundance, movement, and diversity of herpetofauna in the Lost Pines ecoregion of Texas. Herpetol. Conserv. Biol. 2014, 9, 192–205. [Google Scholar]
- Justice, C.J. The effect of prescribed burns and wildfire on vegetation in Bastrop State Park, TX. In Proceedings of the AGU Fall Meeting Abstracts, San Francisco, CA, USA, 15–19 December 2014; p. 0537. [Google Scholar]
- Lee, R.J.; Chow, T.E. Post-wildfire assessment of vegetation regeneration in Bastrop, Texas, using Landsat imagery. GIScience Remote Sens. 2015, 52, 609–626. [Google Scholar] [CrossRef]
- Spencer, C.N.; Gabel, K.O.; Hauer, F.R. Wildfire effects on stream food webs and nutrient dynamics in Glacier National Park, USA. For. Ecol. Manag. 2003, 178, 141–153. [Google Scholar] [CrossRef]
- Alfaro-Sánchez, R.; Camarero, J.J.; López-Serrano, F.R.; Sánchez-Salguero, R.; Moya, D.; De Las Heras, J. Positive coupling between growth and reproduction in young post-fire Aleppo pines depends on climate and site conditions. Int. J. Wildland Fire 2015, 24, 507–517. [Google Scholar] [CrossRef] [Green Version]
- Schurbon, J.M.; Fauth, J.E. Effects of prescribed burning on amphibian diversity in a southeastern US national forest. Conserv. Biol. 2003, 17, 1338–1349. [Google Scholar] [CrossRef]
- Moulton, C.; Fleming, W.; Nerney, B. The use of PVC pipes to capture hylid frogs. Herpetol. Rev. 1996, 27, 186–187. [Google Scholar]
- Borg, C.K.; Hoss, S.K.; Smith, L.L.; Conner, L.M. A method for preventing flying squirrel mortality in PVC pipe treefrog refugia. Wildl. Soc. Bull. 2004, 32, 1313–1315. [Google Scholar] [CrossRef]
- Campbell, K.R.; Campbell, T.S.; Johnson, S.A. The use of PVC pipe refugia to evaluate spatial and temporal distributions of native and introduced treefrogs. Fla. Sci. 2010, 73, 78–88. [Google Scholar]
- Elston, L.M.; Waddle, J.H.; Rice, K.G.; Percival, H.F. Co-occurrence of invasive Cuban treefrogs and native treefrogs in PVC pipe refugia. Herpetol. Rev. 2013, 44, 406–409. [Google Scholar]
- Moseley, K.R.; Castleberry, S.B.; Schweitzer, S.H. Effects of prescribed fire on herpetofauna in bottomland hardwood forests. Southeast. Nat. 2003, 2, 475–486. [Google Scholar] [CrossRef]
- Langford, G.J.; Borden, J.A.; Major, C.S.; Nelson, D.H. Southern Mississippi pine savanna. Herpetol. Conserv. Biol. 2007, 2, 135–143. [Google Scholar]
- Stambaugh, M.C.; Creacy, G.; Sparks, J.; Rooney, M. Three centuries of fire and forest vegetation transitions preceding Texas’ most destructive wildfire: Lost Pines or lost oaks? For. Ecol. Manag. 2017, 396, 91–101. [Google Scholar] [CrossRef]
- Corbett, L.; Andersen, A.; Müller, W. Terrestrial vertebrates. Fire Trop. Savannas 2003, 169, 126–152. [Google Scholar]
- Pham, L.; Boudreaux, S.; Karhbet, S.; Price, B.; Ackleh, A.S.; Carter, J.; Pal, N. Population estimates of Hyla cinerea (Schneider) (Green Tree Frog) in an urban environment. Southeast. Nat. 2007, 6, 203–216. [Google Scholar] [CrossRef]
- Horn, S.; Hanula, J.L.; Ulyshen, M.D.; Kilgo, J.C. Abundance of green tree frogs and insects in artificial canopy gaps in a bottomland hardwood forest. Am. Midl. Nat. 2005, 153, 321–326. [Google Scholar] [CrossRef]
- Conant, R.; Collins, J.T. A Field Guide to Reptiles & Amphibians: Eastern and Central North America; Houghton Mifflin Harcourt: Boston, MA, USA, 1998; Volume 12. [Google Scholar]
- Semlitsch, R.D. Differentiating migration and dispersal processes for pond-breeding amphibians. J. Wildl. Manag. 2008, 72, 260–267. [Google Scholar] [CrossRef]
- Garton, J.S.; Brandon, R.A. Reproductive ecology of the green treefrog, Hyla cinerea, in southern Illinois (Anura: Hylidae). Herpetologica 1975, 31, 150–161. [Google Scholar]
- Ritchie, S.A.; Montague, C.L. Simulated populations of the black salt march mosquito (Aedes taeniorhynchus) in a Florida mangrove forest. Ecol. Model. 1995, 77, 123–141. [Google Scholar] [CrossRef]
- Villamizar-Gomez, A.; Forstner, M.R.; Suriyamongkol, T.; Forks, K.N.; Grant, W.E.; Wang, H.-H.; Mali, I. Prevalence of Batrachochytrium dendrobatidis in two sympatric treefrog species, Hyla cinerea and Hyla versicolor. Herpetol. Rev. 2016, 47, 601–605. [Google Scholar]
- Villamizar-Gomez, A.; Wang, H.-H.; Peterson, M.R.; Grant, W.E.; Forstner, M.R. Environmental determinants of Batrachochytrium dendrobatidis and the likelihood of further dispersion in the face of climate change in Texas, USA. Dis. Aquat. Org. 2021, 146, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Ryan, K.; Noste, N. Evaluating prescribed fires. In Proceedings of the Symposium and Workshop on Wilderness Fire, Missoula, MT, USA, 15–18 November 1983; pp. 230–238. [Google Scholar]
- Turner, M.G.; Hargrove, W.W.; Gardner, R.H.; Romme, W.H. Effects of fire on landscape heterogeneity in Yellowstone National Park, Wyoming. J. Veg. Sci. 1994, 5, 731–742. [Google Scholar] [CrossRef] [Green Version]
- Ryan, K.C. Dynamic interactions between forest structure and fire behavior in boreal ecosystems. Silva. Fenn. 2002, 36, 13–39. [Google Scholar] [CrossRef] [Green Version]
- Brown, D.J.; Ferrato, J.R.; White, C.J.; Mali, I.; Forstner, M.R.J.; Simpson, T.R. Short-term changes in summer and winter resident bird communities following a high severity wildfire in a southern USA mixed pine/hardwood forest. For. Ecol. Manag. 2015, 350, 13–21. [Google Scholar] [CrossRef]
- Boughton, R.G.; Staiger, J.; Franz, R. Use of PVC pipe refugia as a sampling technique for hylid treefrogs. Am. Midl. Nat. 2000, 144, 168–177. [Google Scholar] [CrossRef]
- Morrison, D.A.; Morris, E.C. Pseudoreplication in experimental designs for the manipulation of seed germination treatments. Austral Ecol. 2000, 25, 292–296. [Google Scholar] [CrossRef]
- R-Core-Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Fire Executive Council. Guidance for Implementation of Federal Wildland Management Policy; US Department of Agriculture and US Department of Interior: Washington, DC, USA, 2009.
- Tedim, F.; Leone, V.; Amraoui, M.; Bouillon, C.; Coughlan, M.R.; Delogu, G.M.; Fernandes, P.M.; Ferreira, C.; McCaffrey, S.; McGee, T.K.; et al. Defining Extreme Wildfire Events: Difficulties, Challenges, and Impacts. Fire 2018, 1, 9. [Google Scholar] [CrossRef] [Green Version]
- Hunter, M.E.; Robles, M.D. Tamm review: The effects of prescribed fire on wildfire regimes and impacts: A framework for comparison. For. Ecol. Manag. 2020, 475, 118435. [Google Scholar] [CrossRef]
- CIFFC Glossary Task Team and Training Working Group. Canadian Wildland Fire Management Glossary; Canadian Interagency Forest Fire Centre: Winnipeg, MB, Canada, 2017. [Google Scholar]
- Pilliod, D.S.; Bury, R.B.; Hyde, E.J.; Pearl, C.A.; Corn, P.S. Fire and amphibians in North America. For. Ecol. Manag. 2003, 178, 163–181. [Google Scholar] [CrossRef]
- Greenberg, C.H.; Waldrop, T.A. Short-term response of reptiles and amphibians to prescribed fire and mechanical fuel reduction in a southern Appalachian upland hardwood forest. For. Ecol. Manag. 2008, 255, 2883–2893. [Google Scholar] [CrossRef]
- Hromada, S.J.; Howey, C.A.F.; Dickinson, M.B.; Perry, R.W.; Roosenburg, W.M.; Gienger, C.M. Response of reptile and amphibian communities to the reintroduction of fire in an oak/hickory forest. For. Ecol. Manag. 2018, 428, 1–13. [Google Scholar] [CrossRef]
- Hossack, B.R.; Lowe, W.H.; Corn, P.S. Rapid increases and time-lagged declines in amphibian occupancy after wildfire. Conserv. Biol. 2013, 27, 219–228. [Google Scholar] [CrossRef]
- Rochester, C.J.; Brehme, C.S.; Clark, D.R.; Stokes, D.C.; Hathaway, S.A.; Fisher, R.N. Reptile and amphibian responses to large-scale wildfires in southern California. J. Herpetol. 2010, 44, 333–351. [Google Scholar] [CrossRef]
- Brown, D.J.; Nowlin, W.H.; Ozel, E.; Mali, I.; Episcopo, D.; Jones, M.C.; Forstner, M.R. Comparison of short term low, moderate, and high severity fire impacts to aquatic and terrestrial ecosystem components of a southern USA mixed pine/hardwood forest. For. Ecol. Manag. 2014, 312, 179–192. [Google Scholar] [CrossRef]
- Hill, I.D.C.; Dong, B.; Barnes, W.J.P.; Ji, A.; Endlein, T. The biomechanics of tree frogs climbing curved surfaces: A gripping problem. J. Exp. Biol. 2018, 221, jeb168179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langowski, J.K.A.; Rummenie, A.; Pieters, R.P.M.; Kovalev, A.; Gorb, S.N.; van Leeuwen, J.L. Estimating the maximum attachment performance of tree frogs on rough substrates. Bioinspir. Biomim. 2019, 14, 025001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Endlein, T.; Barnes, W.J.P.; Samuel, D.S.; Crawford, N.A.; Biaw, A.B.; Grafe, U. Sticking under wet conditions: The remarkable attachment abilities of the Torrent frog, Staurois guttatus. PLoS ONE 2013, 8, e73810. [Google Scholar] [CrossRef] [Green Version]
- Hossack, B.R.; Pilliod, D.S. Amphibian responses to wildfire in the western United States: Emerging patterns from short-term studies. Fire Ecol. 2011, 7, 129–144. [Google Scholar] [CrossRef]
- Borzée, A.; Choi, Y.; Kim, Y.E.; Jablonski, P.G.; Jang, Y. Interspecific variation in seasonal migration and brumation behavior in two closely related species of treefrogs. Front. Ecol. Evol. 2019, 7, 55. [Google Scholar] [CrossRef] [Green Version]
- Brattstrom, B.H. Wildlife mortalities in PVC claim posts. Wildl. Soc. Bull. 1995, 23, 765–766. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).