Exploring Effective Conservation of Charismatic Flora: Orchids in Armenia as a Case Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Data Collection and Cleaning
2.2. Sampling Density and Species Richness Analysis
2.3. Species Distribution Models (SDMs) of Conservation Priority Species
3. Results
3.1. Sampling Density and Overall Orchid Diversity
3.2. Protected Area Network Comparisons
3.3. Potential Areas of Suitability and Protection
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WWF. Living Planet Report 2020—Bending the Curve of Biodiversity Loss; Almond, R.E.A., Grooten, M., Petersen, T., Eds.; WWF: Gland, Switzerland, 2020; ISBN 978-2-940529-99-5. [Google Scholar]
- IPBES. Global Assessment Report on Biodiversity and Ecosystem Services of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services; IPBES Secretariat: Bonn, Germany, 2019. [Google Scholar]
- Khapugin, A.A.; Kuzmin, I.V.; Silaeva, T.B. Anthropogenic drivers leading to regional extinction of threatened plants: Insights from regional Red Data Books of Russia. Biodivers. Conserv. 2020, 29, 2765–2777. [Google Scholar] [CrossRef]
- Le Roux, J.J.; Hui, C.; Castillo, M.L.; Iriondo, J.M.; Keet, J.H.; Khapugin, A.A.; Médail, F.; Rejmánek, M.; Theron, G.; Yannelli, F.A.; et al. Recent Anthropogenic Plant Extinctions Differ in Biodiversity Hotspots and Coldspots. Curr. Biol. 2019, 29, 2912–2918.e2. [Google Scholar] [CrossRef] [PubMed]
- Štípková, Z.; Kindlmann, P. Orchid Extinction over the Last 150 Years in the Czech Republic. Diversity 2021, 13, 78. [Google Scholar] [CrossRef]
- Kougioumoutzis, K.; Kokkoris, I.P.; Panitsa, M.; Strid, A.; Dimopoulos, P. Extinction Risk Assessment of the Greek Endemic Flora. Biology 2021, 10, 195. [Google Scholar] [CrossRef]
- Knapp, W.M.; Frances, A.; Noss, R.; Naczi, R.F.C.; Weakley, A.; Gann, G.D.; Baldwin, B.G.; Miller, J.; McIntyre, P.; Mishler, B.D.; et al. Vascular plant extinction in the continental United States and Canada. Conserv. Biol. 2021, 35, 360–368. [Google Scholar] [CrossRef]
- Nic Lughadha, E.; Bachman, S.P.; Leão, T.C.C.; Forest, F.; Halley, J.M.; Moat, J.; Acedo, C.; Bacon, K.L.; Brewer, R.F.A.; Gâteblé, G.; et al. Extinction risk and threats to plants and fungi. Plants People Planet 2020, 2, 389–408. [Google Scholar] [CrossRef]
- Gray, C.L.; Hill, S.L.; Newbold, T.; Hudson, L.N.; Börger, L.; Contu, S.; Hoskins, A.J.; Ferrier, S.; Purvis, A.; Scharlemann, J.P. Local biodiversity is higher inside than outside terrestrial protected areas worldwide. Nat. Commun. 2016, 7, 12306. [Google Scholar] [CrossRef] [Green Version]
- Aubele, D.T.; Wang, D.; Post, K.W.; Hahn, S.; Wincott, F. Effectiveness of Parks in Protecting Tropical Biodiversity. Nucleic Acids Res. 1997, 17, 5191. [Google Scholar]
- CBD. Zero Draft of Post—2020 Biodiversity Framework. 2020. Available online: https://www.cbd.int/conferences/post2020/wg2020-02/documents (accessed on 29 September 2021).
- Rodrigues, A.S.L.; Andelman, S.J.; Bakarr, M.I.; Boitani, L.; Brooks, T.M.; Cowling, R.M.; Fishpool, L.D.C.; da Fonseca, G.A.B.; Gaston, K.J.; Pilgrim, J.D.; et al. Effectiveness of the global protected area network in representing species diversity. Nature 2004, 428, 640–643. [Google Scholar] [CrossRef]
- Dobrowski, S.Z.; Littlefield, C.E.; Lyons, D.S.; Hollenberg, C.; Carroll, C.; Parks, S.A.; Abatzoglou, J.T.; Hegewisch, K.; Gage, J. Protected-area targets could be undermined by climate change-driven shifts in ecoregions and biomes. Commun. Earth Environ. 2021, 2, 198. [Google Scholar] [CrossRef]
- Beyer, H.L.; Venter, O.; Grantham, H.S.; Watson, J.E. Substantial losses in ecoregion intactness highlight urgency of globally coordinated action. Conserv. Lett. 2020, 13, e12692. [Google Scholar] [CrossRef]
- Mancheno, C.S.M.; Zazanashvili, N.; Beruchashvili, G. Effectiveness of the network of protected areas of the South Caucasus at representing terrestrial ecosystems after the dissolution of the Soviet Union. Environ. Conserv. 2017, 44, 158–165. [Google Scholar] [CrossRef]
- Delso, Á.; Fajardo, J.; Muñoz, J. Protected area networks do not represent unseen biodiversity. Sci. Rep. 2021, 11, 12275. [Google Scholar] [CrossRef]
- Milchev, B.; Georgiev, V. The effect of N atura 2000 network on the Eurasian Eagle-owl (Bubo bubo) population in Southeast Bulgaria: Implications for conservation. Ornis Hung. 2021, 29, 170–178. [Google Scholar] [CrossRef]
- Koskela, J.; Lefèvre, F.; Schueler, S.; Kraigher, H.; Olrik, D.C.; Hubert, J.; Longauer, R.; Bozzano, M.; Yrjänä, L.; Alizoti, P.; et al. Translating conservation genetics into management: Pan-European minimum requirements for dynamic conservation units of forest tree genetic diversity. Biol. Conserv. 2013, 157, 39–49. [Google Scholar] [CrossRef] [Green Version]
- Oberosler, V.; Tenan, S.; Zipkin, E.F.; Rovero, F. Poor management in protected areas is associated with lowered tropical mammal diversity. Anim. Conserv. 2020, 23, 171–181. [Google Scholar] [CrossRef]
- Moreno-Saiz, J.C.; Albertos, B.; Ruiz-Molero, E.; Mateo, R.G. The European Union can afford greater ambition in the conservation of its threatened plants. Biol. Conserv. 2021, 261, 109231. [Google Scholar] [CrossRef]
- Guisan, A.; Tingley, R.; Baumgartner, J.B.; Naujokaitis-Lewis, I.; Sutcliffe, P.R.; Tulloch, A.I.T.; Regan, T.J.; Brotons, L.; Mcdonald-Madden, E.; Mantyka-Pringle, C.; et al. Predicting species distributions for conservation decisions. Ecol. Lett. 2013, 16, 1424–1435. [Google Scholar] [CrossRef] [PubMed]
- Balding, M.; Williams, K.J.H. Plant blindness and the implications for plant conservation. Conserv. Biol. 2016, 30, 1192–1199. [Google Scholar] [CrossRef] [PubMed]
- Govearts, R.; Bernet, P.; Kratochvil, K.; Gerlach, G.; Carr, G.; Alrich, P.; Pridgeon, A.M.; Pfahl, J.; Campacci, M.A.; Holland Baptista, D.; et al. World Checklist of Orchidaceae; The Board of Trustees of the Royal Botanic Gardens: Richmond, UK, 2008. [Google Scholar]
- Esposito, F.; Merckx, T.; Tyteca, D.; Esposito, F.; Merckx, T.; Tyteca, D. Noctuid moths as potential hybridization agents for Platanthera orchids. Lankesteriana 2017, 17, 383–393. [Google Scholar] [CrossRef] [Green Version]
- Ayasse, M.; Schiestl, F.P.; Paulus, H.F.; Ibarra, F.; Francke, W. Pollinator attraction in a sexually deceptive orchid by means of unconventional chemicals. Proc. R. Soc. Lond. B 2003, 270, 517–522. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paulus, H.F. Speciation, pattern recognition and the maximization of pollination: General questions and answers given by the reproductive biology of the orchid genus Ophrys. J. Comp. Physiol. A 2019, 205, 285–300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turco, A.; Medagli, P.; Wagensommer, R.P.; D’Emerico, S.; Gennaio, R.; Albano, A. A morphometric study on Ophrys sect. Pseudophrys in Apulia (Italy) and discovery of Ophrys japigiae sp. nov. (Orchidaceae). Plant Biosyst. Int. J. Deal. All Asp. Plant Biol. 2021. [Google Scholar] [CrossRef]
- Schiestl, F.P. On the success of a swindle: Pollination by deception in orchids. Naturwissenschaften 2005, 92, 255–264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calevo, J.; Voyron, S.; Adamo, M.; Alibrandi, P.; Perotto, S.; Girlanda, M. Can orchid mycorrhizal fungi be persistently harbored by the plant host? Fungal Ecol. 2021, 53, 101071. [Google Scholar] [CrossRef]
- Esposito, F.; Jacquemyn, H.; Waud, M.; Tyteca, D. Mycorrhizal fungal diversity and community composition in two closely related Platanthera (orchidaceae) species. PLoS ONE 2016, 11, e0164108. [Google Scholar] [CrossRef] [PubMed]
- Swarts, N.D.; Dixon, K.W. Terrestrial orchid conservation in the age of extinction. Ann. Bot. 2009, 104, 543–556. [Google Scholar] [CrossRef] [Green Version]
- Averyanov, L.V. Averyanov, L.V. A Review of the Genus Dactylorhiza. In Orchid Biology: Reviews and Perspectives; Arditti, J., Ed.; Timber Press, Inc.: Portland, OR, USA, 1990; Volume V, pp. 161–204. ISBN 0-88192-170-X. [Google Scholar]
- Khapugin, A.A. A global systematic review on orchid data in protected areas. Nat. Conserv. Res. 2020, 5, 19–33. [Google Scholar] [CrossRef]
- POWO Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew. Available online: https://powo.science.kew.org/ (accessed on 1 September 2021).
- Rankou, H. Goodyera Macrophylla. Available online: https://www.iucnredlist.org/species/162070/5527443 (accessed on 29 September 2021). [CrossRef]
- Wagensommer, R.P.; Medagli, P.; Turco, A.; Perrino, E.V. IUCN red list evaluation of the orchidaceae endemic to apulia (Italy) and considerations on the application of the IUCN protocol to rare species. Nat. Conserv. Res. 2020, 5, 90–101. [Google Scholar] [CrossRef]
- Zazanashvili, N.; Sanadiradze, G.; Garforth, M.; Bitsadze, M.; Manvelyan, K.; Askerov, E.; Mousavi, M.; Krever, V.; Shmuk, V.; Kalem, S.; et al. Ecoregional Conservation Plan (ECP) for The Caucasus 2020 Edition; WWF, KfW: Tbilisi, Georgia, 2020. [Google Scholar]
- Schuerholz, G. Lake Arpi National Park and Sanctuaries: Akhuryan Gorge, Ardenis and Alvar; WWF, KfW: Gyumri, Armenia, 2009. [Google Scholar]
- Zazanashvili, N.; Mallon, D. Status and Protection of Globally Threatened Species in the Caucasus; Zazanashvili, N., Mallon, D., Eds.; Contour Ltd.: Tbilisi, Georgia, 2009. [Google Scholar]
- Mousavi, M.; Moqanaki, E.; Hamidi, A.K.; Breitenmoser, U.; Breitenmoser-Würsten, C.; Zazanashvili, N.; Heidelberg, A. Conservation of the Leopard in the Caucasus: Iran Chapter International Experts Workshop “Conservation of the Leopard in the Caucasus” Workshop Report; Tbilisi, 2014. Available online: https://www.researchgate.net/publication/274256377_Conservation_of_the_leopard_in_the_Caucasus_Iran_chapter (accessed on 29 September 2021).
- IUCN; UNEP-WCMC. The World Database on Protected Areas (WDPA). Available online: https://www.protectedplanet.net/en/thematic-areas/wdpa?tab=WDPA (accessed on 15 October 2021).
- Thiers, B. Index Herbariorium. Available online: http://sweetgum.nybg.org/science/ih/herbarium-list/?NamOrganisationAcronym=MW (accessed on 15 October 2021).
- Noé, N. GBIF Occurrences Plugin for QGIS 3. 2019. Available online: https://plugins.qgis.org/plugins/qgisgbifapi/ (accessed on 10 September 2021).
- GBIF.org Occurrence Download. Available online: https://www.gbif.org/occurrence/download/0014012-210819072339941 (accessed on 10 September 2021).
- Averianov, L.V.; Nersesian, A.A. Orchidaceae Juss. In Flora of Armenia V. 10; Takhtajan, A.L., Ed.; A. R. G. Gantner Verlag KG: Ruggell, Leichtenstein, 2001; pp. 165–225. [Google Scholar]
- Averianov, L.V. Orchidaceae Juss. In Conspectus Florae Caucasi; Takhtajan, A.L., Ed.; Saint-Petersburg University Press: Saint-Petersburg, Russia, 2006; pp. 84–101. [Google Scholar]
- WCVP World Checklist of Vascular Plants, Version 2.0. Facilitated by the Royal Botanic Gardens, Kew. Available online: http://wcvp.science.kew.org/ (accessed on 9 September 2021).
- Hijmans, R.J.; Guarino, L.; Jarvis, A.; O’Brian, R.; Mathur, P.; Busink, C.; Cruz, M.; Barrantes, I.; Rojas, E. DIVA-GIS: Freely Available Resource. Available online: www.diva-gis.org (accessed on 15 September 2021).
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Roeland, K.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, B.R.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package. R Package Version 2.5-7. 2020. Available online: https://CRAN.R-project.org/package=vegan (accessed on 20 September 2021).
- R Version 4.0.2 Copyright © The R Foundation for Statistical Computing Platform 2020. Available online: https://www.r-project.org/about.html (accessed on 10 October 2021).
- Phillips, S.J.; Dudik, M.; Schapire, R. Maxent Software for Modeling Species Niches and Distributions (Version 3.4.1). 2021. Available online: https://biodiversityinformatics.amnh.org/open_source/maxent/ (accessed on 15 September 2021).
- Deb, C.R.; Jamir, N.S.; Kikon, Z.P. Distribution Prediction Model of a Rare Orchid Species (Vanda bicolor Griff.) Using Small Sample Size. Am. J. Plant Sci. 2017, 8, 1388–1398. [Google Scholar] [CrossRef]
- Ma, X.; Chen, T.; Zhang, G.; Wang, R. Microbial community structure along an altitude gradient in three different localities. Folia Microbiol. 2004, 49, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Siles, J.A.; Margesin, R. Seasonal soil microbial responses are limited to changes in functionality at two Alpine forest sites differing in altitude and vegetation. Sci. Rep. 2017, 7, 2204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baumann, K.; Keune, J.; Wolters, V.; Jauker, F. Distribution and pollination services of wild bees and hoverflies along an altitudinal gradient in mountain hay meadows. Ecol. Evol. 2021, 11, 11345–11351. [Google Scholar] [CrossRef] [PubMed]
- Viana, T.A.; Martins, F.M.; Lourenço, A.P. The Orchid Bee Fauna (Hymenoptera: Apidae: Euglossini) of a Neotropical Savanna: An Efficient Protocol to Assess Bee Community and Diversity Along Elevational and Habitat Complexity Gradients. Neotrop. Entomol. 2021, 50, 748–758. [Google Scholar] [CrossRef]
- Hijmans, R.J.; Phillips, S.J.; Leathwick, J.R.; Elith, J. Dismo: Species Distribution Modeling, R Package Version 1.3-3; 2020. Available online: https://CRAN.R-project.org/package=dismo (accessed on 15 September 2021).
- Phillips, S.J.; Anderson, R.P.; Dudík, M.; Schapire, R.E.; Blair, M.E. Opening the black box: An open-source release of Maxent. Ecography 2017, 40, 887–893. [Google Scholar] [CrossRef]
- Akaike, H. A New Look at the Statistical Model Identification. IEEE Trans. Automat. Contr. 1974, 19, 716–723. [Google Scholar] [CrossRef]
- Warren, D.L.; Seifert, S.N. Ecological niche modeling in Maxent: The importance of model complexity and the performance of model selection criteria. Ecol. Appl. 2011, 21, 335–342. [Google Scholar] [CrossRef] [Green Version]
- Tamanyan, K.; Fayvuh, G.; Nanagulyan, S. Higher Plants and Fungi. In The Red Data Book of Plants of the Republic of Armenia; Danielyan, T., Ed.; Printing-House of “Zangak-97”, LLC: Yerevan, Armenia, 2010; p. 592. [Google Scholar]
- Shahsavari, A. Flora of Iran. Part 57: Orchidaceae; Research Institute of Forests and Rangelands: Tehran, Iran, 2008. [Google Scholar]
- Akhalkatsi, M.; Arabuli, G.; Lorenz, R. Orchids as indicator species of forest disturbances on limestone quarry in Georgia (South Caucasus). J. Eur. Orchid. 2014, 46, 123–160. [Google Scholar]
- IUCN/SSC Orchid Specialist Group. Status Survey and Conservation Action Plan Orchids; IUCN: Gland, Switzerland; Cambridge, UK, 1996. [Google Scholar]
- Feeley, K.J.; Silman, M.R. Keep collecting: Accurate species distribution modelling requires more collections than previously thought. Divers. Distrib. 2011, 17, 1132–1140. [Google Scholar] [CrossRef]
- Meyer, C.; Weigelt, P.; Kreft, H. Multidimensional biases, gaps and uncertainties in global plant occurrence information. Ecol. Lett. 2016, 19, 992–1006. [Google Scholar] [CrossRef]
- Muñoz-Rodríguez, P.; Carruthers, T.; Wood, J.R.I.; Williams, B.R.M.; Weitemier, K.; Kronmiller, B.; Goodwin, Z.; Sumadijaya, A.; Anglin, N.L.; Filer, D.; et al. A taxonomic monograph of Ipomoea integrated across phylogenetic scales. Nat. Plants 2019, 5, 1136–1144. [Google Scholar] [CrossRef] [PubMed]
- Korotkova, N.; Parolly, G.; Khachatryan, A.; Ghulikyan, L.; Sargsyan, H.; Akopian, J.; Borsch, T.; Gruenstaeudl, M. Towards resolving the evolutionary history of Caucasian pears (Pyrus, Rosaceae)—Phylogenetic relationships, divergence times and leaf trait evolution. J. Syst. Evol. 2018, 56, 35–47. [Google Scholar] [CrossRef]
- Cristescu, M.E. From barcoding single individuals to metabarcoding biological communities: Towards an integrative approach to the study of global biodiversity. Trends Ecol. Evol. 2014, 29, 566–571. [Google Scholar] [CrossRef] [PubMed]
- Vogel Ely, C.; Bordignon, S.A.D.L.; Trevisan, R.; Boldrini, I.I. Implications of poor taxonomy in conservation. J. Nat. Conserv. 2017, 36, 10–13. [Google Scholar] [CrossRef]
- Sheth, B.P.; Thaker, V.S. DNA barcoding and traditional taxonomy: An integrated approach for biodiversity conservation. Genome 2017, 60, 618–628. [Google Scholar] [CrossRef] [Green Version]
- Haque, M.D.M.; Nipperess, D.A.; Baumgartner, J.B.; Beaumont, L.J. A journey through time: Exploring temporal patterns amongst digitized plant specimens from Australia. Syst. Biodivers. 2018, 16, 604–613. [Google Scholar] [CrossRef]
- Hughes, A.C.; Orr, M.C.; Ma, K.; Costello, M.J.; Waller, J.; Provoost, P.; Yang, Q.; Zhu, C.; Qiao, H. Sampling biases shape our view of the natural world. Ecography 2021, 44, 1259–1269. [Google Scholar] [CrossRef]
- Fisher-Phelps, M.; Cao, G.; Wilson, R.M.; Kingston, T. Protecting bias: Across time and ecology, open-source bat locality data are heavily biased by distance to protected area. Ecol. Inform. 2017, 40, 22–34. [Google Scholar] [CrossRef]
- Asatryan, A. New data on distribution of some rare plant species (Pyrus gergerana Gladkova, P. daralagezi Mulk., P. voronovii Rubtzov, Orchis punctulata Steven ex Lindl.) in Armenia. Takhtajania 2018, 4, 51–56. [Google Scholar]
- Nersesyan, A. New data on families Apiaceae, Caryophyllaceae, Orchidaceae, Poaceae of the Armenian flora. Takhtajania 2018, 4, 53–56. [Google Scholar]
- Fayvush, G.; Tamanyan, K.; Kalahsyan, M.; Vitek, E. “Biodiversity Hotspots” in Armenia. Ann. Naturhist. Mus. Wien B 2013, 115, 11–20. [Google Scholar]
- Fois, M.; Cuena-Lombraña, A.; Fenu, G.; Cogoni, D.; Bacchetta, G. Does a correlation exist between environmental suitability models and plant population parameters? An experimental approach to measure the influence of disturbances and environmental changes. Ecol. Indic. 2018, 86, 1–8. [Google Scholar] [CrossRef]
- Hosseinzadeh, M.S.; Fois, M.; Zangi, B.; Kazemi, S.M. Predicting past, current and future habitat suitability and geographic distribution of the Iranian endemic species Microgecko latifi (Sauria: Gekkonidae). J. Arid Environ. 2020, 183. [Google Scholar] [CrossRef]
- Abdelaal, M.; Fois, M.; Fenu, G.; Bacchetta, G. Using MaxEnt modeling to predict the potential distribution of the endemic plant Rosa arabica Crép. in Egypt. Ecol. Inform. 2019, 50, 68–75. [Google Scholar] [CrossRef]
- Fourcade, Y.; Engler, J.O.; Rödder, D.; Secondi, J. Mapping species distributions with MAXENT using a geographically biased sample of presence data: A performance assessment of methods for correcting sampling bias. PLoS ONE 2014, 9, e97122. [Google Scholar] [CrossRef] [Green Version]
- Wan, J.; Wang, C.; Han, S.; Yu, J. Planning the priority protected areas of endangered orchid species in northeastern China. Biodivers. Conserv. 2014, 23, 1395–1409. [Google Scholar] [CrossRef]
- Štípková, Z.; Kindlmann, P. Factors determining the distribution of orchids—A review with examples from the Czech Republic. Eur. J. Environ. Sci. 2021, 11, 21–30. [Google Scholar] [CrossRef]
- Schwager, P.; Berg, C. Global warming threatens conservation status of alpine EU habitat types in the European Eastern Alps. Reg. Environ. Chang. 2019, 19, 2411–2421. [Google Scholar] [CrossRef] [Green Version]
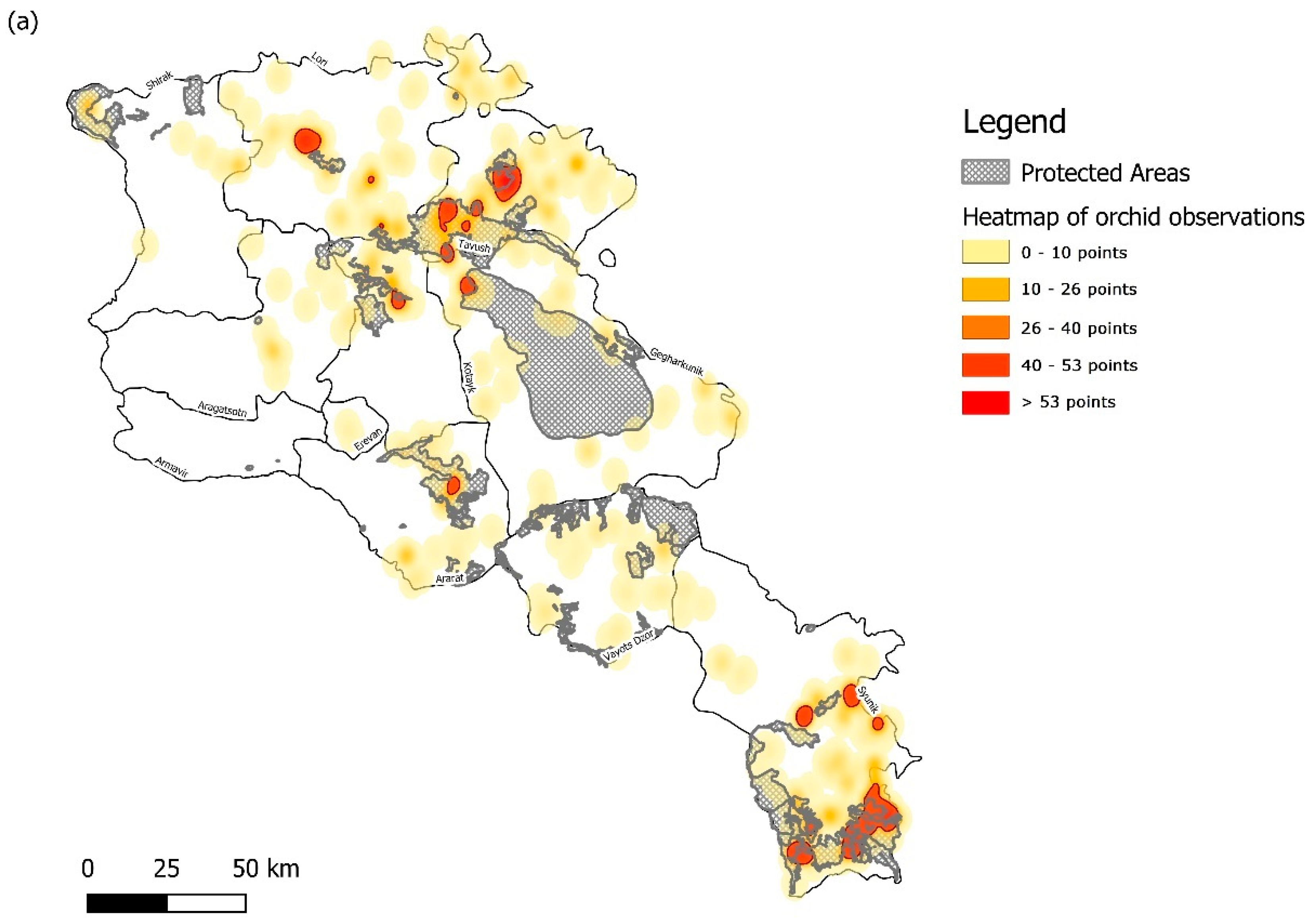
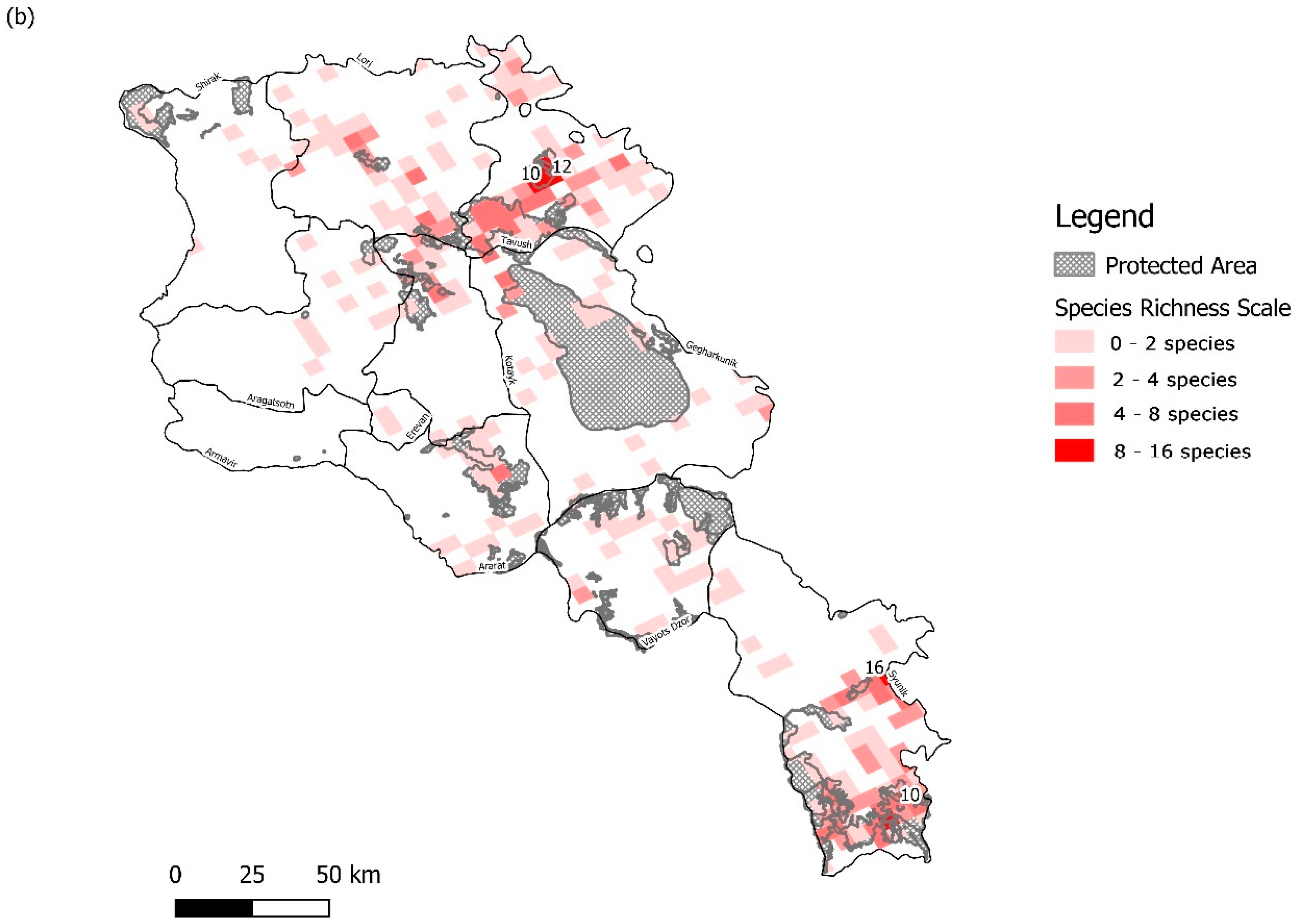
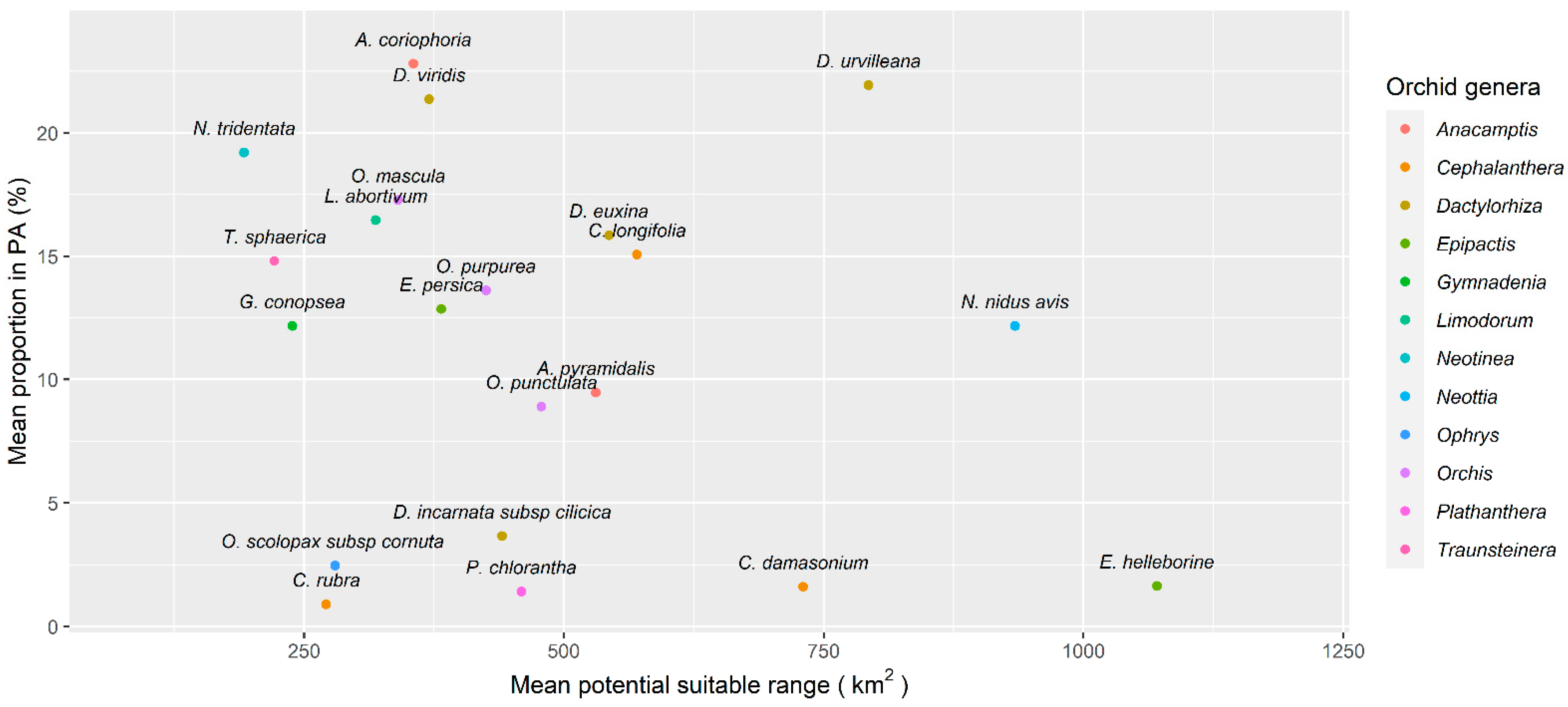
| Protected Area | Orchid Species Richness | Orchid Diversity Index | Reported Area (km2) | Designation | IUCN Category |
|---|---|---|---|---|---|
| Shirak | |||||
| Lake Arpi | 3 | 0.85 | 212 | National Park | II |
| Lori | |||||
| Gyulagarak | 4 | 2.41 | 25.8 | Sanctuary | IV |
| Zikatar | 1 | 0.00 | 1.50 | Sanctuary | IV |
| Margahovit | 4 | 1.36 | 33.7 | Sanctuary | IV |
| Tavush | |||||
| Ijevan | 14 | 2.41 | 59.1 | Sanctuary | IV |
| Gandzakar | 1 | 0.00 | 68.1 | Sanctuary | IV |
| Dilijan | 17 | 2.50 | 377 | National Park | II |
| Akhnabat Yew Grove | 1 | 0.00 | 0.25 | Sanctuary | IV |
| Gegharkunik | |||||
| Sevan | 6 | 1.55 | 1474 | National Park | II |
| Juniper Open Woodland | 2 | 0.69 | 33.1 | Sanctuary | IV |
| Kotayk | |||||
| Arzakan-Meghradzor | 4 | 1.21 | 135 | Sanctuary | IV |
| Ararat | |||||
| Khosrov Forest | 6 | 1.50 | 232 | State Reserve | Ia |
| Vayots Dzor | |||||
| Jermuk Forest | 1 | 0.00 | 38.7 | Sanctuary | IV |
| Herher Open Woodland | 1 | 0.00 | 61.4 | Sanctuary | IV |
| Syunik | |||||
| Arevik | 17 | 2.30 | 344 | National Park | II |
| Boghaqar | 1 | 0.00 | 27.3 | Sanctuary | IV |
| Shikahogh | 13 | 2.26 | 121 | State Reserve | Ia |
| Zangezur | 2 | 0.69 | 259 | Sanctuary | IV |
| Goris | 1 | 0.00 | 18.5 | Sanctuary | IV |
| Plane Grove | 1 | 0.00 | 0.64 | Sanctuary | IV |
| Species | Presence Records | Test Records | Features | Regularization Multiplier | AICc Value | Training AUC | Test AUC | SD | Variable Contribution (%) | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bio5 | Bio18 | Bio17 | Alt | |||||||||
| Anacamptis coriophora | 24 | 6 | hlq | 1 | 477 | 0.720 | 0.612 | 0.084 | 98.5 | 0.1 | 1.3 | 0.1 |
| Anacamptis pyramidalis | 28 | 7 | hlq | 5 | 482 | 0.716 | 0.844 | 0.054 | 0.0 | 0.0 | 0.0 | 100 |
| Cephalanthera damasonium | 16 | 4 | hlq | 2 | 285 | 0.860 | 0.866 | 0.050 | 0.0 | 35.3 | 0.0 | 64.6 |
| Cephalanthera longifolia | 11 | 3 | lq | 1 | 197 | 0.819 | 0.783 | 0.024 | 0.0 | 18.5 | 0.0 | 81.5 |
| Cephalanthera rubra | 33 | 8 | hlq | 3 | 601 | 0.834 | 0.779 | 0.060 | 3.5 | 13.1 | 0.0 | 83.5 |
| Dactylorhiza euxina | 15 | 4 | hlq | 2 | 299 | 0.686 | 0.698 | 0.102 | 0.0 | 0.4 | 0.0 | 99.6 |
| Dactylorhiza incarnata subsp. cilicica | 30 | 8 | hlq | 3 | 574 | 0.795 | 0.710 | 0.084 | 20.5 | 50.2 | 0.0 | 29.4 |
| Dactylorhiza urvilleana | 45 | 11 | hlq | 5 | 859 | 0.738 | 0.646 | 0.064 | 56.2 | 13.6 | 30.1 | 0.0 |
| Dactylorhiza viridis | 11 | 3 | lq | 1 | 207 | 0.835 | 0.740 | 0.173 | 85.3 | 8.8 | 5.9 | 0.0 |
| Epipactis helleborine | 14 | 4 | lq | 1 | 256 | 0.810 | 0.751 | 0.088 | 0.0 | 26.1 | 0.0 | 73.9 |
| Epipactis persica | 9 | 2 | l | 3 | 162 | 0.781 | 0.572 | 0.030 | 0.0 | 0.0 | 0.0 | 100 |
| Gymnadenia conopsea | 24 | 6 | hlq | 3 | 459 | 0.663 | 0.597 | 0.083 | 0.0 | 0.0 | 0.0 | 100 |
| Limodorum abortivum | 11 | 3 | lq | 1 | 192 | 0.857 | 0.908 | 0.017 | 0.0 | 0.0 | 12.2 | 87.8 |
| Neotinea tridentata | 15 | 4 | hlq | 2 | 280 | 0.894 | 0.804 | 0.059 | 79.0 | 2.5 | 18.4 | 0.0 |
| Neottia nidus-avis | 16 | 4 | hlq | 2 | 270 | 0.782 | 0.799 | 0.083 | 0.0 | 83.5 | 1.6 | 14.9 |
| Ophrys scolopax subsp. cornuta | 9 | 2 | l | 1 | 169 | 0.834 | 0.839 | 0.011 | 68.7 | 0.0 | 0.1 | 31.2 |
| Orchis mascula | 42 | 11 | hlq | 2 | 813 | 0.740 | 0.607 | 0.085 | 20.9 | 40.3 | 10.5 | 28.3 |
| Orchis punctulata | 9 | 2 | l | 1 | 170 | 0.853 | 0.660 | 0.087 | 90.8 | 0.0 | 9.2 | 0.0 |
| Orchis purpurea | 9 | 2 | l | 2 | 160 | 0.774 | 0.840 | 0.035 | 0.0 | 0.0 | 0.0 | 100 |
| Platanthera chlorantha | 23 | 6 | hlq | 3 | 447 | 0.702 | 0.590 | 0.122 | 0.0 | 83.8 | 0.0 | 16.2 |
| Traunsteinera sphaerica | 7 | 2 | l | 1 | 135 | 0.770 | 0.766 | 0.108 | 49.8 | 50.2 | 0.0 | 0.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faruk, A.; Papikyan, A.; Nersesyan, A. Exploring Effective Conservation of Charismatic Flora: Orchids in Armenia as a Case Study. Diversity 2021, 13, 624. https://doi.org/10.3390/d13120624
Faruk A, Papikyan A, Nersesyan A. Exploring Effective Conservation of Charismatic Flora: Orchids in Armenia as a Case Study. Diversity. 2021; 13(12):624. https://doi.org/10.3390/d13120624
Chicago/Turabian StyleFaruk, Aisyah, Astghik Papikyan, and Anush Nersesyan. 2021. "Exploring Effective Conservation of Charismatic Flora: Orchids in Armenia as a Case Study" Diversity 13, no. 12: 624. https://doi.org/10.3390/d13120624
APA StyleFaruk, A., Papikyan, A., & Nersesyan, A. (2021). Exploring Effective Conservation of Charismatic Flora: Orchids in Armenia as a Case Study. Diversity, 13(12), 624. https://doi.org/10.3390/d13120624






