Abstract
Knowledge of subterranean fauna has mostly been derived from caves and streambeds, which are relatively easily accessed. In contrast, subterranean fauna inhabiting regional groundwater aquifers or the vadose zone (between surface soil layers and the watertable) is difficult to sample. Here we provide species lists for a globally significant subterranean fauna hotspot in the Robe Valley of the Pilbara region, Western Australia. This fauna was collected from up to 50 m below ground level using mining exploration drill holes and monitoring wells. Altogether, 123 subterranean species were collected over a distance of 17 km, comprising 65 troglofauna and 58 stygofauna species. Of these, 61 species were troglobionts and 48 stygobionts. The troglofauna occurs in small voids and fissures in mesas comprised mostly of an iron ore formation, while the stygofauna occurs in the alluvium of a river floodplain. The richness of the Robe Valley is not a localized aberration, but rather reflects the richness of the arid Pilbara region. While legislation in Western Australia has recognized the importance of subterranean fauna, mining is occurring in the Robe Valley hotspot with conditions of environmental approval that are designed to ensure species persistence.
1. Introduction
Subterranean habitats have little to no light, scarce organic matter, constantly high humidity, and a much more stable temperature than the surface [1]. Unsurprisingly, the vast majority of studies of troglofauna and stygofauna have been in caves that people can access or in streambeds (the hyporheic zone). Both types of habitats allow researchers relatively easy access to explore and collect animals [2,3]. It is often overlooked, however, that much more extensive habitats likely to support subterranean fauna occur worldwide. For stygofauna, these habitats occur in unconfined regional groundwater aquifers. For troglofauna, they occur in the vadose zone that extends from c. 2 m below the ground surface to the watertable. Abundant small spaces for subterranean animals are present in geologies ranging from detritals (e.g., colluvium, alluvium, scree, etc.) to sedimentary rocks, often at tens of meters depth [4,5,6]. Sampling troglofauna and stygofauna from these environments, which have no connection at the human scale to the surface, is obviously much more difficult than exploring large cave systems or digging in streambeds and the documentation of their biodiversity has lagged behind the general study of subterranean fauna.
Recognition that Western Australia supports rich stygofauna communities in arid zone aquifers began in the 1990s, with sampling at Cape Range on the mid-west coast and in the Pilbara and Yilgarn regions [7,8] (Figure 1). The richness of stygofauna across most Pilbara landscapes has been confirmed by a large regional survey [9,10] and many surveys associated with the environmental impact assessment of mining projects [11,12]. Recognition that equally rich troglofauna communities are present did not begin until stygofauna surveys in the Robe Valley during 2004 and 2005 also collected troglofauna, mostly schizomids [13,14]. Knowledge of both troglofauna and stygofauna in the Pilbara has continued to grow in the past 15 years, and it was recently estimated that 1511 troglofauna and 1329 stygofauna species occur in the region [5], with the caveat that the precision of these estimates is limited by a weak taxonomic framework for certain fauna groups.
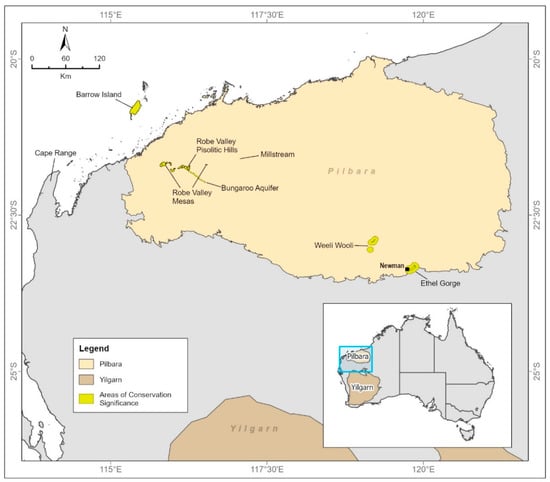
Figure 1.
The Pilbara region of Western Australia, showing subterranean fauna communities recognized as significant for conservation. The Robe Valley hotspot is at the western end of the Robe Valley Pisolitic Hills.
The richness of subterranean fauna in the Pilbara has been explained as partly a result of many aquatic and mesic-adapted terrestrial invertebrate species escaping the harshness of surface habitats during the Miocene aridification by moving underground, sometimes with dramatic subsequent radiation [15,16]. Schizomids are a well-documented case; these typically forest litter-dwelling animals moved into subterranean habitats as forest habitat was being lost, and then radiated in these habitats [17,18]. Equally impressive radiations have occurred, however, in stygofauna groups such as candonid ostracods [19], syncarids [20] and amphipods [21,22], which are essentially subterranean groups and would not have colonized from the surface. Information from a regional survey suggests that, as a result of these radiations, there are about 30% more stygofauna than surface aquatic invertebrate species in the Pilbara [23]. As far as we are aware, this greater richness of subterranean fauna is unique, but comparative data for other regions are scarce because of the rarity of published regional inventories.
While the Pilbara can be regarded as an area of globally significant radiation of subterranean fauna, here we focus on the richness of troglofauna and stygofauna in the western part of the Robe River catchment (Figure 2). We call this the Robe Valley, although it is a mostly flat landscape. The area has previously been identified as a hotspot for troglofauna [24] and stygofauna [25]. Cape Range, to the south-west of the Pilbara, the offshore Barrow Island, and the inland Ethel Gorge have also been identified as globally significant hotspots in, or near, the Pilbara ([24], Figure 1).
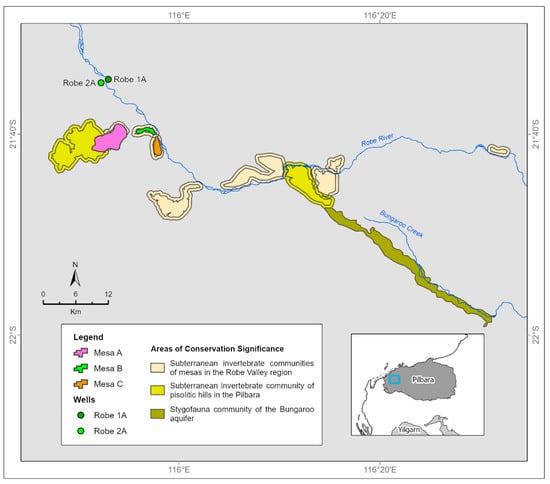
Figure 2.
Components of the Robe Valley subterranean fauna hotspot.
In addition to providing species lists for sites in the Robe Valley, we have three goals in this manuscript. One is to highlight the importance of vadose zone and groundwater aquifers for troglofauna and stygofauna. The second is to illustrate that, in many parts of the world, huge numbers of subterranean species remain to be discovered and described. The third is to point out that water extraction and mining, particularly for iron ore but also gold, base metals, lithium, potash and rare earth metals, may sometimes threaten the conservation of subterranean communities. At the same time, we recognize that it is the wells and exploration drill holes of these developments that provide the portal that has enabled the discovery of the otherwise inaccessible and hidden subterranean fauna of the Pilbara region.
2. Materials and Methods
We confirm previous identification of the Robe Valley as a subterranean fauna hotspot by compiling lists of species for discrete areas or sites within this hotspot. For troglofauna, we provide lists for three small mesas (Mesas A, B and C) on the west flank of the Robe River, approximately 47 km from the river mouth (Figure 2). The mesas represent topographically distinct features in the landscape (Figure 3), and their outlines as defined here can be seen in Figure 2. For stygofauna, we provide lists of species collected from two wells (Robe 2A and 1A) near the Robe River as it crosses the coastal plain, approximately 8 km north of the mesas. The wells are portals to a stygofauna community for which the geographical boundary is not obvious at the surface.

Figure 3.
Upper panel, Mesa A, showing hardcap on top of the mesa; inset A, a scrape net; inset B, a troglofauna trap. Lower panel, Robe 2A well showing steel collar set in concrete; inset A, a stygofauna net. Photos by Claire Stephenson, Mike Scanlon and Melanie Fulcher.
Troglofauna were sampled at the mesas for the purposes of environmental impact assessments and monitoring compliance with the conditions for mining. Most of the sampling was undertaken by Biota Environmental Sciences, although in some years other consultants (including Bennelongia) or mining company staff in conjunction with consultants, undertook the work. The predominant sampling method was trapping, whereby between one and four cylinders of PCV containing moist leaf material were suspended at varying depths in a series of vertical exploration drill holes. The drill holes (diameter 130 mm) had 2–3 m of casing at the surface (called a collar) to prevent the collapse of sand and loose rock into the hole, but the remainder of the hole was in direct contact with the surrounding substrate. Traps were left in place for 6–8 weeks. A smaller number of samples were collected by a technique called scraping, whereby a net was lowered to the bottom of the exploration hole and scraped along the substrate surface while being retrieved [26]. Approximately 1100 trap or scrape samples were collected from Mesa A (21.66072°S, 115.89547°E; hole depth range: 8–88 m) in 11 years of sampling between 2004 and 2017. This equates to about 450 sampling events (i.e., site/date combinations) because multiple traps were set in most holes, and from 2014 onwards holes were also scraped. Sampling effort was lower at the other two mesas, with approximately 400 samples and 180 sampling events during seven years between 2005 and 2016 at Mesa B (21.65894°S, 115.94547°E; depth range: 15–70 m) and 300 samples and 180 sampling events in three years of sampling between 2005 and 2017 at Mesa C (21.68928°S, 115.96206°E; depth range: 15–125 m). Troglofauna traps were set in an iron ore formation called channel iron deposit (CID) at depths between 5 m and 70 m below ground level, with the median depth being 20 m. Animals were recovered from depths up to 50 m.
Stygofauna were collected mainly by net hauling at Robe 2A (well G70730102, 21.58123S°S, 115.87047°E) and Robe 1A (well G70730101, 21.57542°S, 115.88266°E). These wells are 14 m and 23 m deep, with standing water level fluctuating between 4–9 m and 6–10 m below ground level, respectively. This level of groundwater fluctuation is characteristic of Pilbara floodplains. The wells had a steel collar extending approximately 1 m above and below the ground surface and a 100 mm diameter PVC casing inside the collar that extended to the end of the hole. The casing was slotted continuously below the watertable. A small, weighted plankton net, with a diameter only slightly smaller than the cased hole, was lowered to the base of the hole, agitated vigorously to stir ‘benthic’ fauna into the water column, and then retrieved. Two sizes of mesh (50 µm and 150 µm) were used in different hauls. Pump samples were also collected on two dates, whereby three times the bore volume was pumped through a 50 µm-mesh net [9]. Twelve sampling events occurred at Robe 2A in five years of sampling between 2002 and 2007, while eight sampling events occurred in the three years between 2002 and 2004 at Robe 1A.
Determining Species Numbers and Subterranean Affinity
The taxonomic framework for the identification of most groups of subterranean fauna in the Pilbara is poor. As a result, even when there was competent species-level discrimination by different survey teams, the compilation of species lists for the hotspots was difficult. Species-level discrimination was mostly achieved using morphology, often coupled with molecular sequencing, but also by sequencing alone. Very few species have been described to date, so most species were identified by codes linked to voucher specimens or molecular sequences. In a few cases, when Western Australian Museum staff work on a group and verified identifications, a museum voucher name was used. More often codes were generated by individual consultancies. Representative specimens of most putative species are lodged in the Western Australian Museum.
Another part of the process of compiling the species lists was determining the subterranean affinity of specimens. This is more important for troglofauna, because aquatic species collected from groundwater beneath a completely dry terrestrial surface rely entirely on subterranean habitat for persistence in that setting. On the other hand, scrapes and traps usually collect an order of magnitude more surface animals than troglofauna. While most surface drop-ins can be easily identified as such, some surface soil species lack pigmentation and eyes as a result of their specialization to life in topsoil and leaf litter, and it can difficult to distinguish between them and troglofauna. We considered species to be troglofauna if they are known only from scrapes or traps and possess some troglomorphic traits in addition to lack of pigmentation and eyes. Pauropods and symphylans are the groups with greatest potential for surface species to be treated as troglofauna but soil and litter sampling in the Pilbara has collected very few species of either group [26; Bennelongia, unpublished data], suggesting few species in these groups are drop-ins. Mites and collembolans were omitted from the lists, reflecting advice from the Western Australian Museum to the environmental impact assessment process many years ago that few species in these groups are subterranean. We recognise that this is incorrect for some species.
3. Environment and Geology
The Pilbara Craton is one of the oldest and most stable geological regions in the world, with basement rocks dating as far back as 3.5 billion years [27]. Uniquely, this basement rock in the form of granite and greenstone terrains remains largely exposed [28], particularly in the north of the craton [5]. Throughout the south, and to a lesser extent in the north, the basement rock is overlain by depositional sedimentary rocks from the Archaean to Proterozoic time periods [29]. The major ranges of hills in the Pilbara (i.e., the Hamersley and Chichester Ranges) consist largely of iron-rich sedimentary rocks, mostly banded iron formations. Less resistant rock formations (such as the dolomitic Wittenoom formation) have been highly weathered, with geologies such as CID being deposited in the resultant drainage lines [29].
Despite geological stability, the Pilbara climate has changed substantially over the past 20 million years from cool and wet to hot and arid as Australia separated from Antarctica and began moving northwards [16]. The Pilbara now has very hot summers, moderate winters, and low but highly variable annual rainfall. Mean daily maximum temperatures exceed 35 °C from October to March, and the hottest recorded Pilbara day is 50.5 °C (at Mardie near the mouth of the Robe River in February 1998). Mean annual rainfall at Mardie is 275 mm, with 54% of this rain falling between January and March, mostly from cyclones or monsoonal rain. A further 33% falls between April and June, mostly from late cyclones or large cold fronts that extend to the Pilbara from the south at the start of winter. The main source of groundwater recharge is cyclones [30].
The prospective habitats for troglofauna in the Robe Valley, and the Pilbara generally, mostly comprise centimeter to millimeter-scale fissures and vugs. In some ways, these Pilbara habitats are similar to the small microcaverns radiating out from limestone caves, although limestone microcaverns often become blocked with detritus and are unsuitable for troglofauna [31]. The Pilbara habitats are perhaps more directly comparable with canga and other ferruginous formations in South America, although caves are more common in South American formations and are the focus of most studies [32].
The major troglofauna habitat in the Robe Valley lies within the flat-topped mesas that typically stand 30–50 m above the surrounding landscape. The flat top consists of laterised hardcap, with a thickness of up to 10 m (Figure 3). This cap has a near vertical face at the edges of the mesas, below which is a sloping face of detritals. The core geology of the mesas is CID. Details of the genesis of this CID in the palaeochannel of the Robe River are contentious, but it is agreed CID began to be laid down after sediment in-filled the Robe River palaeovalley during the Palaeogene, then larger areas of CID formed in the Miocene [33,34]. Subsequent surface flow events eroded the in-fill and left the mesas observed today [33]. The typical stratigraphy of the mesas consists of a weathered hardcap (loosely analogous to Brazilian canga) above layers of CID. The CID is predominately made up of iron-rich pisoids, which are 2–10 mm rounded bodies that are bonded together by a geothitised secondary matrix. Over time, this sedimentary rock has become weathered, principally through groundwater leaching, leaving vugs and other voids in the matrix. Some of the voids have gradually combined to form larger spaces, resulting in limited connectivity of spaces throughout the matrix. Most of these subterranean spaces now sit above the watertable and have been colonised by troglofauna. Mesa A has an approximate area of 5.7 km2, Mesa B 1.7 km2 and Mesa C 2.3 km2. Relative humidity in the subterranean habitats of Robe River mesas is 100% [5].
Near the surface, the troglofauna habitat in sections of the Robe River mesas, and in the Pilbara more generally, may be considered to have similarities with Milieu Souterrain Superficial (MSS) habitat, especially on valley slopes [35]. However, this is not the case at depth, where the troglofauna sampling was undertaken. The mesas, and Pilbara as a whole, mostly consist of consolidated rock habitat, including bedrock, rather than scree and loose rock.
The habitat for stygofauna in the Robe Valley is provided mainly by aquifers in CID or alluvium. Robe 2A and 1A lie in an alluvial aquifer associated with the active channel of the Robe River as it crosses the Pilbara coastal plain; CID occurs in the vicinity if not at the wells themselves. The surface provides little indication of the rich stygofauna community below (Figure 3). Depth to the watertable varies from 4–10 m, depending on season and year, with salinity in the top metre of groundwater being 2400–5500 µS/cm and temperature ranging from 30.5–32.4 °C during two years. The Robe River flows, on average, less than once a year and most of the riverbed is dry the remainder of the time. Flood volumes can be large (mean annual flow is 108 GL [36]).
4. Results
Altogether, 65 species of troglofauna have been collected from Mesas A, B and C, with 35 species in Mesa A, 30 species in Mesa B and 17 species in Mesa C (Table 1, Figure 4). Two species, the millipede Lophoturus madecassus and the pseudoscorpion Tyrannochthonius aridus, are also known from surface habitats and are troglophiles [37,38]. Two other species, the hemipteran Cixiidae sp. B02 and the dipteran Allopnyxia sp. B01, are treated as troglophiles. It has been shown genetically that they are widespread across the Pilbara. Adult Allopnyxia sp. B01 has a vestigial eye, consisting of a dark crescent with a few variable-sized ommatidia and, therefore, may have some surface dispersal. Cixiidae sp. B02 has obligate subterranean nymphs with eyes that become larger as they approach the adult stage. Adults are capable of surface dispersal (Bennelongia, unpublished data). All other species are troglobionts known only from Mesas A, B and C, and usually from only one of these mesas. They are extreme short-range endemics.

Table 1.
Troglofauna species collected from Mesas A, B and C. Records with an asterisk represent identifications above species level, but they are usually aligned with a related species to reduce table size. Species in bold have special conservation protection. Comments show species delimited using CO1 results, with divergence values from other species indicated where available. # indicates species are troglophiles, whereas all other species are likely troglobionts.

Figure 4.
Troglofauna at the mesas. (A) Trinemura ‘MesaA1’; (B) Ptinella sp. B01; (C) Japygidae sp.; (D) Troglarmadillo ‘Helix-ISA005’; (E) Allopnyxia sp. B01; (F) Tyrannochthonius aridus; (G) Draculoides sp.; (H) Prethopalpus scanloni. Photos by Jane McRae.
Only 12 of the 65 troglofauna species at the mesas are described, but assignments to at least 34 other species have been supported by sequencing, and another three have been through the process of receiving museum codes. Many additional species, such as the cockroach Nocticola ‘OES11’, the spider Trichocyclus sp. ‘MesaA’, various beetles, the diplurans Japygidae sp. and Projapygidae sp., and the centipedes Cormocephalus sp. and Geophilidae sp., are the single occurrence of their families in a mesa (Table 1), which ensures it is valid to recognize each of these taxa as representing at least one unit in the species list.
The most difficult issue when compiling species lists was reconciling the multiple species names used in different surveys. Some problems were caused by different levels of identification (earlier surveys tended to have less taxonomic information available and hence less rigorous identifications). Others resulted from lack of clear diagnoses associated with each voucher name, leading to conflicting views about which species animals belonged to. For example, delimitation of the five species of Ideoblothrus at Mesa A in an area <6 km2 has been confused, particularly in relation to Ideoblothrus ‘MesaA’, which was described but not formally named [39]. The ‘type’ animal could not be sequenced, but some other animals identified by the Western Australian Museum as Ideoblothrus ‘MesaA’ appear genetically to be Ideoblothrus linnaei, which was described in the same paper. Whether some museum identifications were wrong, or whether Ideoblothrus ‘MesaA’ should be treated as Ideoblothrus linnaei, remains unclear. Delimitation is further complicated by co-occurrence with Ideoblothrus ‘MesaA2’, ‘MesaA3’ and ‘MesaA4’. Ideoblothrus ‘MesaA2’ and ‘MesaA3’ are separated from other species by a borderline 9% sequence divergence in CO1 (Table 1).
Five troglofauna species at Mesa A and one at Mesa B are listed in Western Australian species conservation legislation or associated mechanisms. These are two schizomids (Draculoides anachoretus and D. bythius) that are listed as Vulnerable under the Western Australian Biodiversity Conservation Act 2016 and three pseudoscorpions (Lagynochthonius asema, Tyrannochthonius ‘MesaA’ and Ideoblothrus ‘MesaA’) that are informally listed by the government conservation agency as Priority 1 species (defined as poorly known from few locations and potentially under threat). In addition, the troglofauna communities and their habitats at Mesas A, B and C, as well as nearby mesas in the Robe Valley, are listed informally by the conservation agency as a priority ecological community, namely the Priority 1 Subterranean invertebrate communities of mesas in the Robe valley region. Another Priority 1 troglofauna community, the Subterranean invertebrate community of pisolitic hills in the Pilbara, occurs immediately west of the mesa community (Figure 2). Priority 1 communities are poorly known but considered to have restricted occurrence and to be potentially threatened.
At least 58 species of stygofauna have been collected from the Robe 2A and 1A wells, with 46 species at Robe 2A and 31 species at Robe 1A (Table 2, Figure 5). All species collected are reliant on groundwater for their persistence at the site, although the cyclopoid copepod Apocyclops dengizicus is cosmopolitan and would usually be classed as a stygophile [40]. Twenty-nine of the species are described, largely through taxonomy funded in association with a large biodiversity survey of the Pilbara [41]. An additional species, the thermosbaenid Halosbaena sp. PL, is undescribed, but has been well studied genetically [42]. A lack of diagnostic information for the multiple names used (in different surveys) for syncarid taxa resulted in them being reduced to two species, Bathynellidae sp. and Atopobathynella ‘A’. This probably underestimates the number of syncarid species present. There is also taxonomic and ecological uncertainty associated with oligochaete records. The Enchytraeus, Phreodrilidae and Tubificidae species names in Table 2 represent morphological clades to which immature animals are assigned, and each clade potentially contains multiple species. The groundwater dependence of enchytraeids is uncertain, and they perhaps should be regarded as amphibious species [43], although collected commonly in stygofauna sampling in Western Australia and apparently subterranean. Similarly, some tubificids are stygophiles, and the groundwater dependence of rotifers, nematodes and the leech is unknown. In total, 48 species are recognised as stygobionts.

Table 2.
Stygofauna species collected from wells Robe 2A and Robe 1A. Records with an asterisk represent identifications above species level. # indicates species are stygophiles, ‡ indicates uncertain status; all other species are likely stygobionts.

Figure 5.
Stygofauna at Robe 2A and 1A. (A) Humphreyscandona fovea; (B) Wesniphargus ‘Helix-AMN004’; (C) Namanereis pilbarensis; (D) Kagalana tonde; (E) Microcerberidae sp.; (F) Eriopisidae gen. nov. ‘Helix AMM006’; (G) Nedsia ‘mcraei’ King & Cooper in press; (H) Halosbaena sp. PL.; (I) Peza sp. Photos by Jane McRae.
Unlike the mesas, which have unique assemblages of species associated with easily delineated topographic features, the spatial extent of the stygofauna hotspot is unclear. Of the four wells within 2 km of Robe 2A, only Robe 1A was sampled by the team that sampled Robe 2A. The other wells yielded few species, but Robe 2A and 1A appear to have the same rich stygofauna community, with 61% of the species collected at Robe 1A also found at the better sampled Robe 2A. In addition, five described ostracod and copepod species, three undescribed ostracods, two undescribed amphipod species, a rotifer and a nematode not found at Robe 2A were collected from Robe 1A. The amphipods include a new and distinctive lineage of Eriopisidae that is widespread in coastal parts of the Pilbara and the low-lying Fortescue Valley, as well as a species of Wesniphargus, representing a family (Neoniphargidae) that is relatively uncommon in the Pilbara (Table 2, Figure 5).
Some described stygobiont species have relatively large linear ranges, e.g., the polychaete Namanereis pilbarensis (c. 500 km), the calanoid copepod Stygoridgewayia trispinosa (>300 km), the cyclopid copepods Halicyclops rochai (>300 km), Diacyclops species (400–600 km) and Orbuscyclops westaustraliensis (>800 km), as well as the harpacticoid copepods Pseudectinosoma galassiae (>300 km), Schizopera roberiverensis (c. 300 km), Megastygonitocrella species (600–700 km) and Parastenocaris jane (600 km, Bennelongia unpublished data). Ostracods tend to have smaller ranges, e.g., Deminutiocandona aenigma (c. 200 km), Humphreyscandona species (80–190 km), Pierrecandona posteriorrecta (100 km), Pilbaracandona rosa (120 km) and Areacandona species (50–60 km). Most other species can be considered short-range endemics [9].
The relatively small amount of comprehensive genetic work done on stygofauna (rather than comparisons of individual specimens a few kilometres apart to determine whether they are conspecific) suggests it is likely that some, if not all, of the stygofauna species currently regarded as widespread are, in fact, either cryptic species complexes or have very high intraspecific variability (or both). For example, what was previously regarded as Halosbaena tulki is now regarded as comprising up to five different species, including Halosbaena sp. PL [42].
No stygofauna species from Robe 2A and 1A are listed for protection under conservation legislation, and neither is the community. However, the community of an area to the southeast of the mesas, namely the Priority 1 Stygofauna community of the Bungaroo aquifer, has been listed (Figure 2). It contains at least 17 (30%) of the species present in the Robe 2A and 1A wells.
5. Discussion
The voids in groundwater aquifers and the vadose zone are amongst the least accessible continental habitats for fauna. Hence, it is unsurprising they harbour a subterranean fauna that is not as well characterized as that of streambeds and caves. Sampling yields are low, especially for troglofauna, where the combination of scraping and trapping often yields less than one animal per site and one species for every four sites sampled [26]. Net hauling for stygofauna usually yields about 50 animals and three species per site in areas where stygofauna is abundant [10]. A major contributing factor to these low yields is the disconnection between the sampler on the surface and the animals many metres below in a hidden matrix. In this situation, acquiring fine scale habitat and ecological information to improve sampling yields is difficult. Use of packers and optical viewers [44] can provide information on habitat preferences in small areas but is too expensive for large-scale deployment. Despite these difficulties, sampling of wells and drill holes has enabled documentation of a globally significant number of subterranean fauna species in the Robe Valley. Altogether, 123 species have been recorded over a distance of 17 km. Troglofauna sampling was concentrated in an area of approximately 16 km2. The spatial extent of the stygofauna community documented at Robe 2A and 1A is unknown but probably <4 km2. Of the 123 subterranean species collected, 61 species are considered troglobionts and at least 48 species are stygobionts.
When comparing species richness of the Robe Valley with other subterranean fauna hotspots, some of our assignments of species as troglobionts may seem controversial to readers who think of subterranean animals outside caves as deep soil fauna. However, all troglofauna sampling occurred in CID, which is consolidated rock. Unlike caves, where animals can position themselves along a subterranean gradient according to their ecological requirements, nearly all species collected in the Robe Valley mesas spend their full life cycles in environmental conditions typical of the deep sections of caves [5]. This is also seen in stygofauna, for which there is relatively little overlap in the assemblages of the hyporheic zone and regional aquifers [45]. We treat the stygofauna species that occur in both habitats as stygophiles.
Recognition of the Robe Valley as a hotspot for subterranean fauna is not new. The troglofauna community at Mesa A has previously been identified as a hotspot with 24 troglofauna species [24]. Additional data for Mesa A and the inclusion of Mesas B and C species explains the much larger number of species recognised in Table 1. The stygofauna community at Robe 2A has also previously been recognized as a hotspot with 32 stygobiont [25] or 54 stygofauna species [24], depending on how survey data were interpreted [10]. The stygobiont list [25] excluded rotifers, worms and mites, and the stygofauna list [24] included some taxa that are treated here as duplicate names. Table 2 includes data from well Robe 1A.
The landscape of the Pilbara region as a whole (approximately 250,000 km2) is estimated to support 1511 troglofauna and 1329 stygofauna species [5], nearly all of which we classify as troglobionts or stygobionts. The subterranean fauna richness of the Robe Valley does not stand out in a major way from many other parts of the Pilbara, with a compilation of impact assessment surveys for troglofauna between 2007 and 2016 showing that the two major ranges of hills in the Pilbara to the east of the Robe Valley (Hamersley and Chichester) are also important for troglofauna [12]. In fact, survey data suggest the richest parts of the Hamersley Range contain a greater density of troglofauna species than the Robe Valley (Bennelongia, unpublished data). Broadscale stygofauna survey of the Pilbara between 2001 and 2005 identified nine areas, including the Robe Valley, as important for stygofauna [10]. Subsequent compilation of impact assessment surveys for stygofauna between 2007 and 2016 identified additional areas in the eastern Pilbara as important [12]. Overall, the Pilbara region appears to have a significance for subterranean fauna similar to that of the Dinaric karst in Europe, which has historically been regarded as the most speciose region globally [46,47]. The Pilbara has an estimated 1511 mostly troglobiont species [5] compared with 995 troglobionts in the Dinaric karst [46], and 1329 mostly stygobiont species [10] compared with 680 stygobionts in the Dinaric karst [46]. The Postojna-Planina cave system, the richest hotspot in the Dinaric karst, supports 71 stygobiont and 45 troglobiont species [48], which is similar to the numbers for the Robe Valley hotspot.
As is the case in most areas outside Western Europe and North America [49], especially when considering fauna outside caves, the majority of troglofauna and stygofauna species in the Pilbara are undescribed [10,26]. This is less pronounced for stygofauna because of extensive taxonomic work on ostracods [19] and copepods [50]. With the current state of taxonomy and the complexities of species delimitation for subterranean species, some described Pilbara species are known, or likely, to be species complexes in which several cryptically different species hide under a single name [42,51,52]. On the other hand, the disjunct occurrence of the well-studied cave eel Ophisternon candidum in the Pilbara and surrounds [53] highlights that care should be taken when assuming large-range taxa are species complexes.
The lack of formal species description holds back conservation in two ways. Firstly, accurately determining the number of species in an area is difficult without sound taxonomy. A confusion of names arises when relatively little time can be spent during inventory surveys delimiting near-cryptic species, and when various surveys independently apply their own codes for the same undescribed species. This confusion can create doubts about the validity of species counts, which in turn reduces the credibility of calls to conserve areas or communities. Molecular sequencing is very useful for assigning individual animals, especially if juvenile or physically damaged, to described or vouchered species for which sequence information is available. It can also assist with species delimitation, but it does not replace formal taxonomy or the use of morphology when recognising new species, especially among subterranean animals where intraspecific genetic distances are highly variable and often large [18,52,54]. In situations where a species is encountered only once in an area and is subsequently being compared to a single animal from another area, it can be difficult to determine genetically whether the animals represent different species, different evolutionarily significant units, or just a genetically variable species [55]. Only additional sampling between the two areas or across the possible range of the species can clarify the issue. Furthermore, different genes seem to work better for different taxa and over-splitting into different genetic species, rather than lumping of morphologically cryptic species, seems to be a risk if the ‘wrong’ gene is selected for sequencing [56]. In this context, we point out that COI sequencing without morphological examination of animals has produced more taxa than expected for pseudoscorpions at Mesa A (Ideoblothrus, 5 species) and Mesa B (Chthoniidae, 3 species). While this may have led to a slight overestimate of troglofauna species richness in the Robe Valley, other factors such as sampling adequacy are likely to have caused underestimation.
The second way in which lack of species description obstructs conservation is by hampering the accumulation of ecological knowledge. Species descriptions provide a framework for the collation of information about the ecology and occurrence of different subterranean groups. Synthesis of this knowledge enables more informed decision-making in the environmental impact assessment process and supports future management and conservation of subterranean fauna [57].
Recognition of conservation values of subterranean fauna has strongly influenced land management at Mesas A, B and C, where high conservation values coincide with high economic value of the subterranean habitat. A compromise between fauna conservation and economic development means that Mesa A is currently being mined for iron ore, while future mining of Mesas B and C has been approved by government [58]. However, at all three mesas an outer ‘shell’ approximately 80–200 m wide will remain un-mined the full way around the mesa in recognition of the importance of protecting troglofauna values. These shells should provide a sufficient volume of mesa to ensure persistence of the troglofauna species restricted to each mesa. There has been extensive monitoring at Mesa A since mining began (reported to the Environmental Protection Authority) to ensure that all troglofauna species known from that mesa are persisting in the un-mined shell. The documentation of stygofauna values has also been important, with early consideration of a borefield in the vicinity of Mesa 2a and 1A abandoned. These trade-offs are an example of a process that is likely to become increasingly important worldwide as awareness grows of the occurrence of subterranean fauna outside caves and, at the same time, there is increased demand for the products of mining.
Author Contributions
Conceptualization, H.L.C., S.A.H., B.A.B.; data curation, H.L.C.; writing—original draft preparation, S.A.H., B.A.B., H.L.C.; writing—review and editing, S.A.H., B.A.B.; visualization, H.L.C., S.A.H. All authors have read and agreed to the published version of the manuscript.
Funding
Funding for manuscript preparation was provided by Bennelongia Environmental Consultants.
Institutional Review Board Statement
Not applicable.
Acknowledgments
We thank the editors for inviting us to contribute to this special issue and accommodating our late contribution. We also thank the consultants who collected most of the data on which this paper is based. Vitor Marques assisted with mapping; Claire Stephenson, Mike Scanlon, Melanie Fulcher and Jane McRae provided photographs.
Conflicts of Interest
Bennelongia Environmental Consultants has sometimes undertaken monitoring of stygofauna and troglofauna in the Robe Valley for the Robe River Mining Company, including at Mesas A and B.
References
- Moldovan, O.T.; Kovác, L.; Halse, S. Cave Ecology; Springer Nature: Cham, Switzerland, 2018; p. 545. [Google Scholar]
- Culver, D.C.; Deharveng, L.; Bedos, A.; Lewis, J.J.; Madden, M.; Reddell, J.R.; Sket, B.; Trontelj, P.; White, D. The mid-latitude biodiversity ridge in terrestrial cave fauna. Ecography 2006, 29, 120–128. [Google Scholar] [CrossRef]
- Schneider, K.; Culver, D.C. Estimating subterranean species richness using intensive sampling and rarefaction curves in a high density cave region in West Virginia. J. Cave Karst Stud. 2004, 66, 39–45. [Google Scholar]
- Hahn, H.J.; Fuchs, A. Distribution patterns of groundwater communities across aquifer types in south-western Germany. Freshwater Biol. 2009, 54, 848–860. [Google Scholar] [CrossRef]
- Halse, S.A. Subterranean fauna of the arid zone. In On the Ecology of Australia’s Arid Zone; Lambers, H., Ed.; Springer Nature: Cham, Switzerland, 2018; p. 388. [Google Scholar]
- Pipan, T.; López, H.; Oromí, P.; Polak, S.; Culver, D.C. Temperature variation and the presence of troglobionts in terrestrial shallow subterranean habitats. J. Nat. Hist. 2010, 45, 253–273. [Google Scholar] [CrossRef]
- Pesce, G.L.; de Laurentiis, P.; Humphreys, W.F. Copepods from ground waters of Western Australia. II. The genus Halicyclops (Crustacea, Copepoda, Cyclopidae). Rec. West. Aust. Mus. 1996, 18, 77–85. [Google Scholar]
- Humphreys, W.F. Relict stygofauna’s living in sea salt, karst and calcrete habitats in arid northwestern Australia contain many ancient lineages. In The Other 99%: The Conservation and Biodiversity of Invertebrates; Ponder, W., Lunney, D., Eds.; Royal Zoological Society of New South Wales: Sydney, Australia, 1999; pp. 219–227. [Google Scholar]
- Eberhard, S.M.; Halse, S.A.; Williams, M.R.; Scanlon, M.D.; Cocking, J.S.; Barron, H.J. Exploring the relationship between sampling efficiency and short range endemism for groundwater fauna in the Pilbara region, Western Australia. Freshwater Biol. 2009, 54, 885–901. [Google Scholar] [CrossRef]
- Halse, S.A.; Scanlon, M.D.; Cocking, J.S.; Barron, H.J.; Richardson, J.B.; Eberhard, S.M. Pilbara stygofauna: Deep groundwater of an arid landscape contains globally significant radiation of biodiversity. Rec. West. Aust. Mus. Suppl. 2014, 78, 443–483. [Google Scholar] [CrossRef]
- Humphreys, G.; Alexander, J.; Harvey, M.S.; Humphreys, W.F. The subterranean fauna of Barrow Island, north-western Australia: 10 years on. Rec. West. Aust. Mus. Suppl. 2013, 83, 145–158. [Google Scholar] [CrossRef][Green Version]
- Mokany, K.; Harwood, T.D.; Halse, S.A.; Ferrier, S. Riddles in the dark: Assessing diversity patterns for cryptic subterranean fauna of the Pilbara. Divers. Distrib. 2019, 25, 240–254. [Google Scholar] [CrossRef]
- Biota. Mesa A and Robe Valley Mesas Troglobitic Fauna Survey; Biota Environmental Sciences: Leederville, Australia, 2006. Available online: https://www.epa.wa.gov.au/sites/default/files/PER_documentation/Vegetation%20and%20Flora%20and%20Fauna_Biota%202006f%20Subterranean%20Fauna%20Assessment.pdf (accessed on 9 September 2021).
- Harvey, M.S.; Berry, O.; Edward, K.L.; Humphreys, G. Molecular and morphological systematics of hypogean schizomids (Schizomida:Hubbardiidae) in semiarid Australia. Invertebr. Syst. 2008, 22, 167–194. [Google Scholar] [CrossRef]
- Guzik, M.T.; Austin, A.D.; Cooper, S.J.B.; Harvey, M.S.; Humphreys, W.F.; Bradford, T.; Eberhard, S.M.; King, R.A.; Leys, R.; Muirhead, K.A.; et al. Is the Australian subterranean fauna uniquely diverse? Invertebr. Syst. 2010, 24, 407–418. [Google Scholar] [CrossRef]
- Byrne, M.; Yeates, D.K.; Joseph, L.; Kearney, M.; Bowler, J.; Williams, A.J.; Cooper, S.; Donnellan, S.C.; Keogh, S.; Leys, R.; et al. Birth of a biome: Insights into the assembly and maintenance of the Australian arid zone biota. Mol. Ecol. 2008, 17, 4398–4417. [Google Scholar] [CrossRef] [PubMed]
- Abrams, K.M.; Huey, J.A.; Hillyer, M.J.; Humphreys, W.F.; Didham, R.K.; Harvey, M.S. Too hot to handle: Cenozoic aridification drives multiple independent incursions of Schizomida (Hubbardiidae) into hypogean environments. Mol. Phylogenet. Evol. 2019, 139, 106532. [Google Scholar] [CrossRef]
- Harms, D.; Curran, M.K.; Klesser, R.; Finston, T.L.; Halse, S.A. Speciation patterns in complex subterranean environments: A case study using short-tailed whipscorpions (Schizomida: Hubbardiidae). Biol. J. Linn. Soc. 2018, 125, 355–367. [Google Scholar] [CrossRef]
- Karanovic, I. Candoninae (Ostracoda) from the Pilbara region in Western Australia. Crustaceana Monogr. 2007, 7, 1–432. [Google Scholar]
- Matthews, E.F.; Abrams, K.M.; Cooper, S.J.B.; Huey, J.A.; Hillyer, M.J.; Humphreys, W.F.; Austin, A.D.; Guzik, M.T. Scratching the surface of subterranean biodiversity: Molecular analysis reveals a diverse and previously unknown fauna of Parabathynellidae (Crustacea: Bathynellacea) from the Pilbara, Western Australia. Mol. Phylogenet. Evol. 2020, 142, 106643. [Google Scholar] [CrossRef]
- Finston, T.L.; Johnson, M.S.; Humphreys, W.F.; Eberhard, S.M.; Halse, S.A. Cryptic speciation in two widespread subterranean amphipod genera reflects historical drainage patterns in an ancient landscape. Mol. Ecol. 2007, 16, 355–365. [Google Scholar] [CrossRef]
- King, R.A.; Fagan-Jefferies, E.; Bradford, T.M.; Stringer, D.M.; Finston, T.L.; Halse, S.A.; Eberhard, S.M.; Humphreys, G.; Humphreys, W.F.; Austin, A.D.; et al. Cryptic diversity Down Under: Defining species in the subterranean amphipod genus Nedsia Barnard and Williams (Hadzioidea: Eriopisidae) from the Pilbara, Western Australia. Invertebr. Syst. 2021, in press. [Google Scholar]
- Pinder, A.M.; Halse, S.A.; Shiel, R.J.; McRae, J.M. An arid zone awash with diversity: Patterns in the distribution of aquatic invertebrates in the Pilbara region of Western Australia. Rec. West. Aust. Mus. Suppl. 2010, 78, 205–246. [Google Scholar] [CrossRef]
- Eberhard, S.M.; Howarth, F.G. Undara lava cave fauna in tropical Queensland with an annotated list of Australian subterranean biodiversity hotspots. Diversity 2021, 13, 326. [Google Scholar] [CrossRef]
- Deharveng, L.; Bedos, A. Biodiversity in the tropics. In Encyclopedia of Caves, 3rd ed.; White, W.B., Culver, D.C., Pipan, T., Eds.; Academic Press: New York, NY, USA, 2019; pp. 146–162. [Google Scholar]
- Halse, S.A.; Pearson, G.B. Troglofauna in the vadose zone: Comparison of scraping and trapping results and sampling adequacy. Subterr. Biol. 2014, 13, 17–34. [Google Scholar] [CrossRef]
- Buick, R.; Thornett, J.R.; McNaughton, N.J.; Smith, J.B.; Barley, M.E.; Savage, M. Record of emergent continental crust ~3.5 billion years ago in the Pilbara Craton of Australia. Nature 1995, 375, 574–577. [Google Scholar] [CrossRef]
- Johnson, D.P. The Geology of Australia; Cambridge University Press: London, UK, 2004. [Google Scholar]
- Johnson, S.L.; Wright, A.H. Central Pilbara Groundwater Study; Hydrogeological Record Series; Report HG 8; Water and Rivers Commission Resource Science Division: East Perth, WA, Australia, 2001; p. 124. [Google Scholar]
- Skrzypek, G.; Dogramaci, S.; Page, G.F.M.; Rouillard, A.; Grierson, P.F. Unique stable isotope signatures of large cyclonic events as a tracer of soil moisture dynamics in the semiarid subtropics. J. Hydrol. 2019, 578, 124124. [Google Scholar] [CrossRef]
- Howarth, F.G.; Moldovan, O.T. The ecological classifications of cave animals and their adaptations. In Cave Ecology; Moldovan, O.T., Kovac, L., Halse, S., Eds.; Springer Nature: Cham, Switzerland, 2018; pp. 41–67. [Google Scholar]
- Ferreira, R.L.; de Oliveira, M.P.A.; Silva, M.S. Subterranean biodiversity in ferruginous landscapes. In Cave Ecology; Moldovan, O.T., Kovac, L., Halse, S., Eds.; Springer Nature: Cham, Switzerland, 2018; pp. 435–447. [Google Scholar]
- Morris, R.C.; Ramanaidou, E.R. Genesis of the channel iron deposits (CID) of the Pilbara region, Western Australia. Australian Aust. J. Earth Sci. 2007, 54, 733–756. [Google Scholar] [CrossRef]
- Danišík, M.; Evans, N.J.; Ramanaidou, E.R.; McDonald, B.J.; Mayers, C.; McInnes, B.I.A. (U–Th)/He chronology of the Robe River channel iron deposits, Hamersley Province, Western Australia. Chem. Geol. 2013, 354, 150–162. [Google Scholar] [CrossRef]
- Mammola, S.; Giachino, P.M.; Piano, E.J.A.; Barberis, M.; Badino, G.; Isaia, M. Ecology and sampling techniques of an understudied subterranean habitat: The Milieu Souterrain Superficiel (MSS). Sci. Nat. 2016, 103, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Department of Water. Ecological Water Requirements of the Lower Robe River; Environmental Water Report Series 22; Department of Water: Perth, Australia, 2012; p. 38. [Google Scholar]
- Edward, K.L.; Harvey, M.S. Short-range endemism in hypogean environments: The pseudoscorpion genera Tyrannochthonius and Lagynochthonius (Pseudoscorpiones: Chthoniidae) in the semiarid zone of Western Australia. Invertebr. Syst. 2008, 22, 259–293. [Google Scholar] [CrossRef]
- Car, C.A.; Megan, S.; Huynh, C.; Harvey, M.S. The millipedes of Barrow Island, Western Australia (Diplopoda). Rec. West. Aust. Mus. Suppl. 2013, 83, 209–219. [Google Scholar] [CrossRef]
- Harvey, M.S.; Leng, M.C. Further observations on Ideoblothrus (Pseudoscorpiones: Syarinidae) from subterranean environments in Australia. Rec. West. Aust. Mus. 2008, 24, 381–386. [Google Scholar] [CrossRef][Green Version]
- Sket, B. Can we agree on an ecological classification of subterranean animals? J. Nat. Hist. 2008, 42, 1549–1563. [Google Scholar] [CrossRef]
- McKenzie, N.L.; van Leeuwen, S.; Pinder, A.M. Introduction to the Pilbara biodiversity survey, 2002–2007. Rec. West. Aust. Mus. Supppl. 2009, 78, 3–89. [Google Scholar] [CrossRef]
- Page, T.J.; Hughes, J.M.; Real, K.M.; Stevens, M.I.; King, R.A.; Humphreys, W.F. Allegory of a cave crustacean: Systematic and biogeographic reality of Halosbaena (Peracarida: Thermosbaenacea) sought with molecular data at multiple scales. Mar. Biodivers. 2016, 48, 1185–1202. [Google Scholar] [CrossRef]
- Des Châtelliers, M.C.; Juget, J.; Lafont, M.; Martin, P. Subterranean aquatic Oligochaeta. Freshwater Biol. 2009, 54, 678–690. [Google Scholar] [CrossRef]
- Sorensen, J.P.R.; Maurice, L.; Edwards, F.K.; Lapworth, D.J.; Read, D.S.; Allen, D.; Butcher, A.S.; Newbold, L.K.; Townsend, B.R.; Williams, P.J. Using boreholes as windows into groundwater ecosystems. PLoS ONE 2013, 8, e70264. [Google Scholar] [CrossRef] [PubMed]
- Halse, S.A.; Scanlon, M.D.; Cocking, J.S. Do Springs Provide a Window to the Groundwater Fauna of the Australian Arid Zone? In Balancing the Groundwater Budget, Proceedings of an International Groundwater Conference, Darwin, Australia, 12–17 May 2002; Yinfoo, D., Ed.; International Association of Hydrologists: Wallingford, UK, 2002; p. 12. [Google Scholar]
- Sket, B.; Paragamian, K.; Trontelj, P. A census of the obligate subterranean fauna of the Balkan Peninsula. In Balkan Biodiversity; Pattern and Process in the European Hotspot; Griffiths, H.I., Kryštufek, B., Reed, J.M., Eds.; Springer: Dordrecht, The Netherlands, 2004; pp. 309–332. [Google Scholar]
- Deharveng, L.; Gibert, J.; Culver, D.C. Biodiversity in Europe. In Encyclopedia of Caves, 3rd ed.; White, W.B., Culver, D.C., Pipan, T., Eds.; Academic Press: New York, NY, USA, 2019; pp. 136–145. [Google Scholar]
- Zagmajster, M.; Polak, S.; Fišer, C. Postojna-Planina cave system in Slovenia, a hotspot of subterranean biodiversity and a cradle of speleobiology. Diversity 2021, 13, 271. [Google Scholar] [CrossRef]
- Pipan, T.; Deharveng, L.; Culver, D.C. Hotspots of subterranean biodiversity. Diversity 2020, 12, 209. [Google Scholar] [CrossRef]
- Karanovic, T. Subterranean copepods (Crustacea, Copepoda) from the Pilbara region in Western Australia. Rec. West. Aust. Mus. Suppl. 2006, 70, 1–239. [Google Scholar] [CrossRef]
- Karanovic, T.; Djurakic, M.; Eberhard, S.M. Cryptic species or inadequate taxonomy? Implementation of 2D geometric morphometrics based on integumental organs as landmarks for delimitation and description of copepod taxa. Syst. Biol. 2016, 65, 304–327. [Google Scholar] [CrossRef]
- Trotter, A.J.; McRae, J.M.; Main, D.C.; Finston, T.L. Speciation in fractured rock landforms: Towards understanding the diversity of subterranean cockroaches (Dictyoptera: Nocticolidae: Nocticola) in Western Australia. Zootaxa 2017, 4232, 361–375. [Google Scholar] [CrossRef]
- Moore, G.I.; Humphreys, W.F.; Foster, R. New populations of the rare subterranean blind cave eel Ophisternon candidum (Synbranchidae) reveal recent historical connections throughout north-western Australia. Mar. Freshwater Res. 2018, 69, 1517–1524. [Google Scholar] [CrossRef]
- Perina, G.; Camacho, A.I.; Huey, J.; Horwitz, P.; Koenders, A. Understanding subterranean variability: The first genus of Bathynellidae (Bathynellacea, Crustacea) from Western Australia described through a morphological and multigene approach. Invertebr. Syst. 2018, 32, 423–447. [Google Scholar] [CrossRef]
- Coates, D.J.; Byrne, M.; Moritz, C. Genetic diversity and conservation units: Dealing with the species-population continuum in the age of genomics. Front. Ecol. Evol. 2018, 6. [Google Scholar] [CrossRef]
- Wesener, T.; Voigtländer, K.; Decker, P.; Oeyen, J.P.; Spelda, J. Barcoding of Central European Cryptops centipedes reveals large interspecific distances with ghost lineages and new species records from Germany and Austria (Chilopoda, Scolopendromorpha). ZooKeys 2016, 564, 21–46. [Google Scholar] [CrossRef] [PubMed]
- Halse, S. Conservation and impact assessment of subterranean fauna in Australia. In Cave Ecology; Moldovan, O.T., Kovacz, L., Halse, S., Eds.; Springer Nature: Gland, Switzerland, 2018; pp. 479–493. [Google Scholar]
- Environmental Protection Authority. Report and Recommendations of the Environmental Protection Authority: Mesa A Hub Revised Proposal. Report 1640. Environmental Protection Authority, Perth, Australia. 2019. Available online: https://www.epa.wa.gov.au/sites/default/files/EPA_Report/Mesa%20A%20Hub%20Revised%20Proposal%20-%20EPA%20Report.pdf (accessed on 9 September 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).