Class Conflict: Diffuse Competition between Mammalian and Reptilian Predators
Abstract
1. Introduction
- Nocturnal lizards with generalist diets will experience the strongest competition with dasyurids;
- Diurnal lizards with generalist diets will experience weak competition with dasyurids;
- Ant- and termite-specialist lizards will experience weak or no competition with dasyurids; and
- Competition, if it occurs, will be via interference and be detectable as intraguild predation.
2. Materials and Methods
2.1. Study Site
2.2. Field Sampling
2.3. Diet Analysis
2.4. Statistical Analysis
3. Results
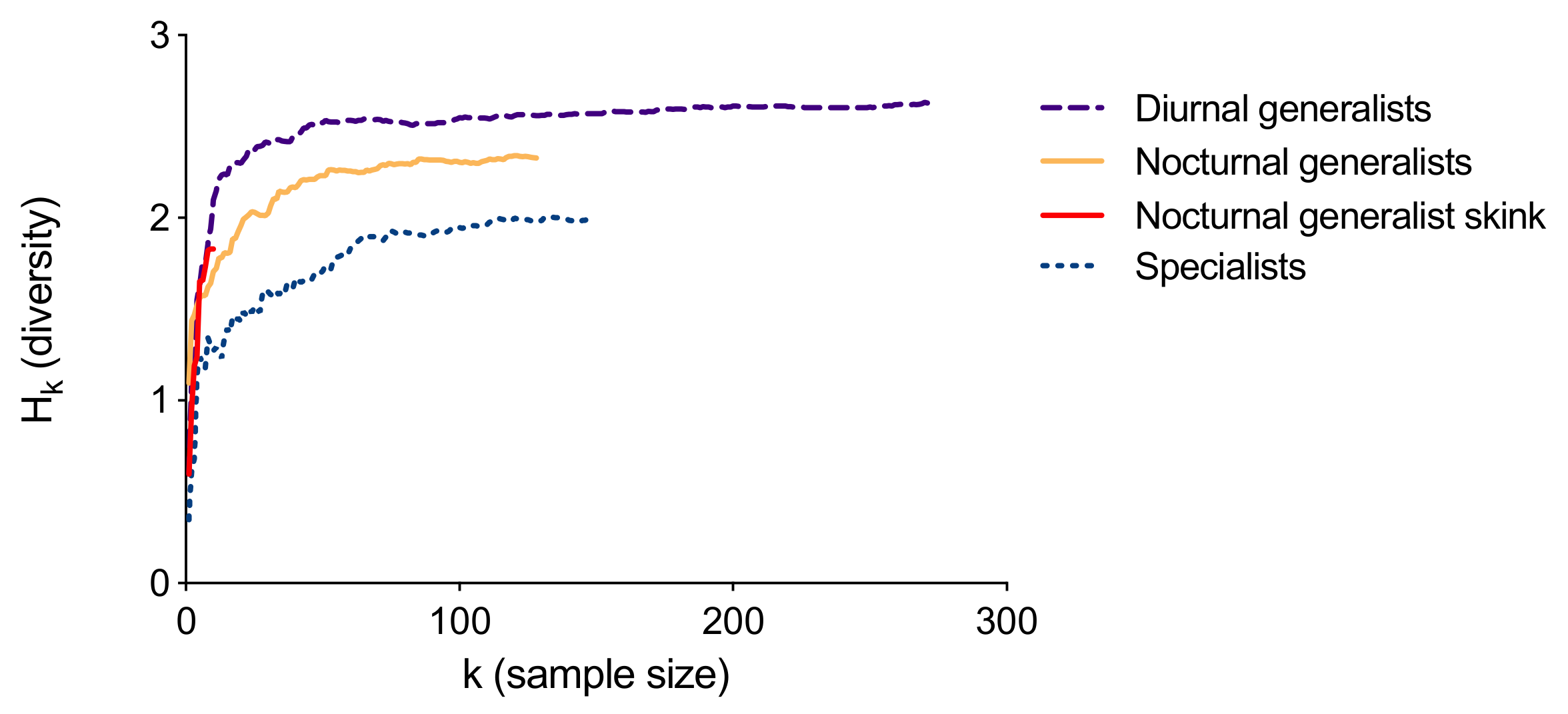
3.1. Diet Analyses
3.2. Predictions 1–3
3.3. Prediction 4
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sale, P.F. Overlap in resource use, and interspecific competition. Oecologia 1974, 17, 245–256. [Google Scholar] [CrossRef] [PubMed]
- Rosenzweig, M.L. Community structure in sympatric Carnivora. J. Mammal. 1966, 47, 602–612. [Google Scholar] [CrossRef]
- Dickman, C.R. Mechanisms of competition among insectivorous mammals. Oecologia 1991, 85, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Donadio, E.; Buskirk, S.W. Diet, morphology, and interspecific killing in Carnivora. Am. Nat. 2006, 167, 524–536. [Google Scholar] [CrossRef] [PubMed]
- Barabás, G.; Michalska-Smith, M.J.; Allesina, S. The effect of intra-and interspecific competition on coexistence in multispecies communities. Am. Nat. 2016, 188, E1–E12. [Google Scholar] [CrossRef]
- Stump, S.M. Multispecies coexistence without diffuse competition; or, why phylogenetic signal and trait clustering weaken coexistence. Am. Nat. 2017, 190, 213–228. [Google Scholar] [CrossRef]
- Morosinotto, C.; Villers, A.; Thomson, R.L.; Varjonen, R.; Korpimäki, E. Competitors and predators alter settlement patterns and reproductive success of an intraguild prey. Ecol. Monogr. 2017, 87, 4–20. [Google Scholar] [CrossRef]
- Zhang, J.; Qian, H.; Girardello, M.; Pellissier, V.; Nielsen, S.E.; Svenning, J.-C. Trophic interactions among vertebrate guilds and plants shape global patterns in species diversity. Proc. R. Soc. B Biol. Sci. 2018, 285, 20180949. [Google Scholar] [CrossRef]
- Moen, J. Diffuse competition: A diffuse concept. Oikos 1989, 54, 260–263. [Google Scholar] [CrossRef]
- Brown, J.S.; Kotler, B.P.; Mitchell, W.A. Competition between birds and mammals: A comparison of giving-up densities between crested larks and gerbils. Evolut. Ecol. 1997, 11, 757–771. [Google Scholar] [CrossRef]
- Kotler, B.P.; Brown, J.S. Mechanisms of coexistence of optimal foragers as determinants of local abundances and distributions of desert granivores. J. Mammal. 1999, 80, 361–374. [Google Scholar] [CrossRef]
- Rubbo, M.J.; Townsend, V.R., Jr.; Smyers, S.D.; Jaeger, R.G. The potential for invertebrate-vertebrate intraguild predation: The predatory relationship between wolf spiders (Gladicosa pulchra) and ground skinks (Scincella lateralis). Can. J. Zool. 2001, 79, 1465–1471. [Google Scholar] [CrossRef]
- Potter, T.I.; Greenville, A.C.; Dickman, C.R. Assessing the potential for intraguild predation among taxonomically disparate micro-carnivores: Marsupials and arthropods. R. Soc. Open Sci. 2018, 5, 171872. [Google Scholar] [CrossRef] [PubMed]
- Potter, T.I.; Stannard, H.J.; Greenville, A.C.; Dickman, C.R. Understanding selective predation: Are energy and nutrients important? PLoS ONE 2018, 13, e0201300. [Google Scholar] [CrossRef]
- Darwin, C. On the Origin of Species by Means of Natural Selection; John Murray: London, UK, 1859. [Google Scholar]
- Elton, C. Animal Ecology; Sidgwick & Jackson: London, UK, 1927. [Google Scholar]
- MacArthur, R.H. Geographical Ecology: Patterns in the Distribution of Species; Harper & Row, Publishers, Inc.: New York, NY, USA, 1972. [Google Scholar]
- Case, T.J. Invasion resistance arises in strongly interacting species-rich model competition communities. Proc. Natl. Acad. Sci. USA 1990, 87, 9610–9614. [Google Scholar] [CrossRef]
- Freed, L.A.; Cann, R.L. Diffuse competition can be reversed: A case history with birds in Hawaii. Ecosphere 2014, 5, 147. [Google Scholar] [CrossRef]
- Mittelbach, G.G. Community Ecology; Sinauer: Sunderland, MA, USA, 2012. [Google Scholar]
- Roughgarden, J. Resource partitioning among competing species—A coevolutionary approach. Theor. Popul. Biol. 1976, 9, 388–424. [Google Scholar] [CrossRef]
- Jensen, P.G.; Humphries, M.M. Abiotic conditions mediate intraguild interactions between mammalian carnivores. J. Anim. Ecol. 2019, 88, 1305–1318. [Google Scholar] [CrossRef]
- Pianka, E.R. Biodiversity of Australian desert lizards. In Biodiversity and Terrestrial Ecosystems, Monographs Series; Peng, C., Chou, C., Eds.; Institute of Botany, Academia Sinica Monograph Series: Taipei, Taiwan, 1994; Volume 14, pp. 259–281. [Google Scholar]
- Dickman, C.R. Body size, prey size, and community structure in insectivorous mammals. Ecology 1988, 69, 569–580. [Google Scholar] [CrossRef]
- Dickman, C.R. An experimental study of competition between two species of dasyurid marsupials. Ecol. Monogr. 1986, 56, 221–241. [Google Scholar] [CrossRef]
- Vanak, A.T.; Fortin, D.; Thaker, M.; Ogden, M.; Owen, C.; Greatwood, S.; Slotow, R. Moving to stay in place: Behavioral mechanisms for coexistence of African large carnivores. Ecology 2013, 94, 2619–2631. [Google Scholar] [CrossRef] [PubMed]
- Wilson, S.D.; Keddy, P.A. Measuring diffuse competition along an environmental gradient: Results from a shoreline plant community. Am. Nat. 1986, 127, 862–869. [Google Scholar] [CrossRef]
- Bennett, J.A.; Lamb, E.G.; Hall, J.C.; Cardinal-McTeague, W.M.; Cahill, J.F. Increased competition does not lead to increased phylogenetic overdispersion in a native grassland. Ecol. Lett. 2013, 16, 1168–1176. [Google Scholar] [CrossRef]
- Fryar, S.C.; Booth, W.; Davies, J.; Hodgkiss, I.J.; Hyde, K. Evidence of in situ competition between fungi in freshwater. Fungal Diver. 2005, 18, 59–71. [Google Scholar]
- Holmer, L.; Stenlid, J. Diffuse competition for heterogeneous substrate in soil among six species of wood-decomposing basidiomycetes. Oecologia 1996, 106, 531–538. [Google Scholar] [CrossRef]
- Davidson, D.W. Some consequences of diffuse competition in a desert ant community. Am. Nat. 1980, 116, 92–105. [Google Scholar] [CrossRef]
- Davidson, D.W. An experimental study of diffuse competition in harvester ants. Am. Nat. 1985, 125, 500–506. [Google Scholar] [CrossRef]
- Pianka, E.R. Niche overlap and diffuse competition. Proc. Natl. Acad. Sci. USA 1974, 71, 2141–2145. [Google Scholar] [CrossRef]
- Pianka, E.R. Rarity in Australian desert lizards. Austral Ecol. 2014, 39, 214–224. [Google Scholar] [CrossRef]
- Bock, C.E.; Cruz, A., Jr.; Grant, M.C.; Aid, C.S.; Strong, T.R. Field experimental evidence for diffuse competition among southwestern riparian birds. Am. Nat. 1992, 104, 815–828. [Google Scholar] [CrossRef]
- Périquet, S.; Valeix, M.; Claypole, J.; Drouet-Hoguet, N.; Salnicki, J.; Mudimba, S.; Revilla, E.; Fritz, H. Spotted hyaenas switch their foraging strategy as a response to changes in intraguild interactions with lions. J. Zool. 2015, 297, 245–254. [Google Scholar] [CrossRef]
- Wikenros, C.; Stahlberg, S.; Sand, H. Feeding under high risk of intraguild predation: Vigilance patterns of two medium-sized generalist predators. J. Mammal. 2014, 95, 862–870. [Google Scholar] [CrossRef]
- Yarnell, R.W.; Phipps, W.L.; Burgess, L.P.; Ellis, J.A.; Harrison, S.W.; Dell, S.; MacTavish, D.; MacTavish, L.M.; Scott, D.M. The influence of large predators on the feeding ecology of two African mesocarnivores: The black-backed jackal and the brown hyaena. Afr. J. Wildl. Res. 2013, 43, 155–166. [Google Scholar] [CrossRef]
- Morin, P.J. Community Ecology, 2nd ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2011. [Google Scholar]
- Ayal, Y. Trophic structure and the role of predation in shaping hot desert communities. J. Arid Environ. 2007, 68, 171–187. [Google Scholar] [CrossRef]
- Polis, G.A. Complex trophic interactions in deserts: An empirical critique of food-web theory. Am. Nat. 1991, 138, 123–155. [Google Scholar] [CrossRef]
- Morton, S.R.; Stafford-Smith, D.M.; Dickman, C.R.; Dunkerley, D.L.; Friedel, M.H.; McAllister, R.R.J.; Reid, J.R.W.; Roshier, D.A.; Smith, M.A.; Walsh, F.J.; et al. A fresh framework for the ecology of arid Australia. J. Arid Environ. 2011, 75, 313–329. [Google Scholar] [CrossRef]
- Morton, S.R. Diversity of desert-dwelling mammals: A comparison of Australia and North America. J. Mammal. 1979, 60, 253–264. [Google Scholar] [CrossRef]
- Wardle, G.M.; Pavey, C.R.; Dickman, C.R. Greening of arid Australia: New insights from extreme years. Austral Ecol. 2013, 38, 731–740. [Google Scholar] [CrossRef]
- Fisher, D.O.; Dickman, C.R. Diets of insectivorous marsupials in arid Australia: Selection for prey type, size or hardness? J. Arid Environ. 1993, 25, 397–410. [Google Scholar] [CrossRef]
- Woolnough, A.P.; Carthew, S.M. Selection of prey by size in Ningaui yvonneae. Aust. J. Zool. 1996, 44, 319–326. [Google Scholar] [CrossRef]
- Chen, X.; Dickman, C.R.; Thompson, M.B. Diet of the mulgara, Dasycercus cristicauda (Marsupialia: Dasyuridae), in the Simpson Desert, central Australia. Wildl. Res. 1998, 25, 233–242. [Google Scholar] [CrossRef]
- Warnecke, L.; Körtner, G.; Burwell, C.J.; Turner, J.M.; Geiser, F. Short-term movement patterns and diet of small dasyurid marsupials in semiarid Australia. Aust. Mammal. 2012, 34, 49–54. [Google Scholar] [CrossRef]
- Baker, A.; Dickman, C.R. Secret Lives of Carnivorous Marsupials; CSIRO Publishing: Melbourne, Australia, 2018. [Google Scholar]
- Moss, G.L.; Croft, D.B. Behavioural mechanisms of microhabitat selection and competition among three species of arid zone dasyurid marsupial. Aust. J. Ecol. 1988, 13, 485–493. [Google Scholar] [CrossRef]
- Greenville, A.C.; Wardle, G.M.; Dickman, C.R. Desert mammal populations are limited by introduced predators rather than future climate change. R. Soc. Open Sci. 2017, 4, 170384. [Google Scholar] [CrossRef]
- Pianka, E.R.; Pianka, H.D. Comparative ecology of twelve species of nocturnal lizards (Gekkonidae) in the Western Australian desert. Copeia 1976, 88, 125–142. [Google Scholar] [CrossRef]
- James, C.D. Temporal variation in diets and trophic partitioning by coexisting lizards (Ctenotus: Scincidae) in central Australia. Oecologia 1991, 85, 553–561. [Google Scholar] [CrossRef]
- Gordon, C.E.; Dickman, C.R.; Thompson, M.B. Partitioning of temporal activity among desert lizards in relation to prey availability and temperature. Austral Ecol. 2010, 35, 41–52. [Google Scholar] [CrossRef]
- Dickman, C.R.; Haythornthwaite, A.S.; McNaught, G.H.; Mahon, P.S.; Tamayo, B.; Letnic, M. Population dynamics of three species of dasyurid marsupials in arid central Australia: A 10-year study. Wildl. Res. 2001, 28, 493–506. [Google Scholar] [CrossRef]
- Cogger, H.G. Reptiles and Amphibians of Australia, 7th ed.; CSIRO Publishing: Clayton, Australia, 2014. [Google Scholar]
- Bennett, A.F.; Ruben, J.A. Endothermy and activity in vertebrates. Science 1979, 206, 649–654. [Google Scholar] [CrossRef]
- Nagy, K.A. Field metabolic rate and food requirement scaling in mammals and birds. Ecol. Monogr. 1987, 57, 112–128. [Google Scholar] [CrossRef]
- Dickman, C.R. An experimental manipulation of the intensity of interspecific competition: Effects on a small marsupial. Oecologia 1986, 70, 536–543. [Google Scholar] [CrossRef]
- Pianka, E.R. The structure of lizard communities. Ann. Rev. Ecol. Syst. 1973, 4, 53–74. [Google Scholar] [CrossRef]
- Keddy, P.A. Competition, 2nd ed.; Springer: Dordrecht, The Netherlands, 2001. [Google Scholar]
- Greenville, A.C.; Wardle, G.M.; Nguyen, V.; Dickman, C.R. Spatial and temporal synchrony in reptile population dynamics in variable environments. Oecologia 2016, 182, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Hallett, J.G.; Pimm, S.L. Direct estimation of competition. Am. Nat. 1979, 113, 593–600. [Google Scholar] [CrossRef]
- Rosenzweig, M.L.; Abramsky, Z.; Kotler, B. Can interaction coefficients be determined from census data? Oecologia 1985, 66, 194–198. [Google Scholar] [CrossRef]
- Purdie, R. Land Systems of the Simpson Desert Region; CSIRO Division of Water and Land Resources: Melbourne, Australia, 1984. [Google Scholar]
- Dickman, C.R.; Mahon, P.S.; Masters, P.; Gibson, D.F. Long-term dynamics of rodent populations in arid Australia: The influence of rainfall. Wildl. Res. 1999, 26, 389–403. [Google Scholar] [CrossRef]
- Dickman, C.R.; Greenville, A.C.; Beh, C.-L.; Tamayo, B.; Wardle, G.M. Social organization and movements of desert rodents during population “booms” and “busts” in central Australia. J. Mammal. 2010, 91, 798–810. [Google Scholar] [CrossRef]
- Letnic, M.; Dickman, C.R. Boom means bust: Interactions between the El Niño/Southern Oscillation (ENSO), rainfall and the processes threatening mammal species in arid Australia. Biodivers. Conserv. 2006, 15, 3847–3880. [Google Scholar] [CrossRef]
- Friend, G.R.; Smith, G.T.; Mitchell, D.S.; Dickman, C.R. Influence of pitfall and drift fence design on capture rates of small vertebrates in semi-arid habitats of Western-Australia. Aust. Wildl. Res. 1989, 16, 1–10. [Google Scholar] [CrossRef]
- Bytheway, J.P.; Dickman, C.R. Identifying the Diets of Insectivorous Vertebrates: A Photographic Reference Guide of Invertebrate Parts; Desert Ecology Research Group, School of Biological Sciences, University of Sydney: Sydney, Australia, 2014. [Google Scholar] [CrossRef]
- Dickman, C.R.; Huang, C. The reliability of fecal analysis as a method for determining the diet of insectivorous mammals. J. Mammal. 1988, 69, 108–113. [Google Scholar] [CrossRef]
- Angelici, F.M.; Luiselli, L.; Rugiero, L. Food habits of the green lizard, Lacerta bilineata, in central Italy and a reliability test of faecal pellet analysis. Ital. J. Zool. 1997, 64, 267–272. [Google Scholar] [CrossRef]
- Greenville, A.C.; Wardle, G.M.; Nguyen, V.; Dickman, C.R. Population dynamics of desert mammals: Similarities and contrasts within a multispecies assemblage. Ecosphere 2016, 7, e01343. [Google Scholar] [CrossRef]
- Haynes, R. Resource partitioning and demography of twelve sympatric skinks (Ctenotus) in the Simpson Desert. Honours Thesis, The University of Sydney, Sydney, Australia, 1996. [Google Scholar]
- Pianka, E.R. Ecology and Natural History of Desert Lizards; Princeton University Press: Princeton, NJ, USA, 1986. [Google Scholar]
- Brillouin, L. Science and Information Theory; Academic Press: New York, NY, USA, 1956. [Google Scholar]
- Pielou, E.R. Ecological Diversity; Wiley InterScience: New York, NY, USA, 1975. [Google Scholar]
- Koenker, R.; Bassett, G., Jr. Regression quantiles. Econom. J. Econom. Soc. 1978, 46, 33–50. [Google Scholar] [CrossRef]
- Cade, B.S.; Terrell, J.W.; Schroeder, R.L. Estimating effects of limiting factors with regression quantiles. Ecology 1999, 80, 311–323. [Google Scholar] [CrossRef]
- Cade, B.S.; Noon, B.R. A gentle introduction to quantile regression for ecologists. Front. Ecol. Environ. 2003, 1, 412–420. [Google Scholar] [CrossRef]
- Clarke, K.R.; Warwick, R.M. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation; Plymouth Marine Laboratory: Plymouth, UK, 1994. [Google Scholar]
- Anderson, M.J.; Underwood, A.J. Effects of substratum on the recruitment and development of an intertidal estuarine fouling assemblage. J. Exp. Mar. Biol. Ecol. 1994, 184, 217–236. [Google Scholar] [CrossRef]
- Quinn, G.P.; Keough, M.K. Experimental Design and Data Analysis for Biologists; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Drury, R. The physiology and behaviour of a nocturnal desert gecko, Nephrurus levis. Honours Thesis, The University of Sydney, Sydney, Australia, 1995. [Google Scholar]
- Charnov, E.L.; Orians, G.H.; Hyatt, K. Ecological implications of resource depression. Am. Nat. 1976, 110, 247–259. [Google Scholar] [CrossRef]
- Kwok, A.B.C.; Wardle, G.M.; Greenville, A.C.; Dickman, C.R. Long-term patterns of invertebrate abundance and relationships to environmental factors in arid Australia. Austral Ecol. 2016, 41, 480–491. [Google Scholar] [CrossRef]
- Gibb, H.; Grossman, B.F.; Dickman, C.R.; Decker, O.; Wardle, G.M. Long-term responses of desert ant assemblages to climate. J. Anim. Ecol. 2019, 88, 1549–1563. [Google Scholar] [CrossRef]
- Schoener, T.W. The compression hypothesis and temporal resource partitioning. Proc. Natl. Acad. Sci. USA 1974, 71, 4169–4172. [Google Scholar] [CrossRef]
- Masters, P. The mulgara Dasycercus cristicauda (Marsupialia: Dasyuridae) at Uluru National Park, Northern Territory. Aust. Mammal. 1998, 20, 403–407. [Google Scholar]
- Pavey, C.R.; Burwell, C.J.; Körtner, G.; Geiser, F. Trophic ecology of marsupial predators in arid Australia following reshaping of predator assemblages. J. Mammal. 2018, 99, 1128–1136. [Google Scholar] [CrossRef]
- Case, T.J.; Gilpin, M.E. Interference competition and niche theory. Proc. Natl. Acad. Sci. USA 1974, 71, 3073–3077. [Google Scholar] [CrossRef] [PubMed]
- Dickman, C.R. Vagrants in the desert. Nat. Aust. 1996, 25, 54–62. [Google Scholar]
- Ewer, R. Some observations on the killing and eating of prey by two dasyurid marsupials: The mulgara, Dasycercus cristicauda, and the Tasmanian devil, Sarcophilus harrisi. Z. Tierpsychol. 1969, 26, 23–38. [Google Scholar] [CrossRef]
- Körtner, G.; Pavey, C.R.; Geiser, F. Thermal biology, torpor, and activity in free-living mulgaras in arid zone Australia during the winter reproductive season. Phys. Biochem. Zool. 2008, 81, 442–451. [Google Scholar] [CrossRef]
- Fisher, D.O.; Dickman, C.R. Body size-prey relationships in insectivorous marsupials: Tests of three hypotheses. Ecology 1993, 74, 1871–1883. [Google Scholar] [CrossRef]
- Greenville, A.C.; Dickman, C.R. The ecology of Lerista labialis (Scincidae) in the Simpson Desert: Reproduction and diet. J. Arid Environ. 2005, 60, 611–625. [Google Scholar] [CrossRef]
- Pianka, E.R. Desert lizard diversity: Additional comments and some data. Am. Nat. 1989, 134, 344–364. [Google Scholar] [CrossRef]
- Morton, S.R.; James, C.D. The diversity and abundance of lizards in arid Australia: A new hypothesis. Am. Nat. 1988, 132, 237–256. [Google Scholar] [CrossRef]
- Crowther, M.S.; Blacket, M.J. Biogeography and speciation in the Dasyuridae: Why are there so many kinds of dasyurids. In Predators with Pouches: The Biology of Carnivorous Marsupials; Jones, M.E., Dickman, C.R., Archer, M., Eds.; CSIRO Publishing: Melbourne, Australia, 2003; pp. 124–130. [Google Scholar]
- Byrne, M.; Yeates, D.K.; Joseph, L.; Kearney, M.; Bowler, J.; Williams, M.A.L.; Cooper, S.; Donnellan, S.C.; Keogh, J.S.; Leys, R.; et al. Birth of a biome: Insights into the assembly and maintenance of the Australian arid zone biota. Mol. Ecol. 2008, 17, 4398–4417. [Google Scholar] [CrossRef] [PubMed]
- Aplin, K.P. Ten million years of rodent evolution in Australasia: Phylogenetic evidence and a speculative historical biogeography. In Evolution and Biogeography of Australasian Vertebrates; Merrick, J.R., Archer, M., Hickey, G.M., Lee, M.S.Y., Eds.; Auscipub: Oatlands, Australia, 2006; pp. 707–744. [Google Scholar]
- Lee, A.K.; Baverstock, P.R.; Watts, C.H.S. Rodents—The late invaders. In Ecological Biogeography of Australia; Keast, A., Ed.; Junk: The Hague, The Netherlands, 1981; Volume 2, pp. 1521–1554. [Google Scholar]
- Dickman, C.; Wardle, G.; Foulkes, J.; de Preu, N. Desert complex environments. In Biodiversity and Environmental Change: Monitoring, Challenges and Direction; Lindenmayer, D., Burns, E., Thurgate, N., Lowe, A., Eds.; CSIRO Publishing: Melbourne, Australia, 2014; pp. 379–438. [Google Scholar]
- Dickman, C.R. Patterns in the structure and diversity of marsupial carnivore communities. In Patterns in the Structure of Mammalian Communities; Morris, D.W., Abramsky, Z., Fox, B.J., Willig, M.R., Eds.; Texas Tech University Press: Lubbock, TX, USA, 1989; pp. 241–251. [Google Scholar]
- Dickman, C.R. Distributional ecology of dasyurid marsupials. In Predators with Pouches: The Biology of Carnivorous Marsupials; Jones, M.E., Dickman, C.R., Archer, M., Eds.; CSIRO Publishing: Melbourne, Australia, 2003; pp. 318–331. [Google Scholar]
- Dickman, C.R. Micro-carnivores: The ecological role of small dasyurid predators in Australia. In Carnivores of Australia: Past, Present and Future; Glen, A.S., Dickman, C.R., Eds.; CSIRO Publishing: Melbourne, Australia, 2014; pp. 241–262. [Google Scholar]
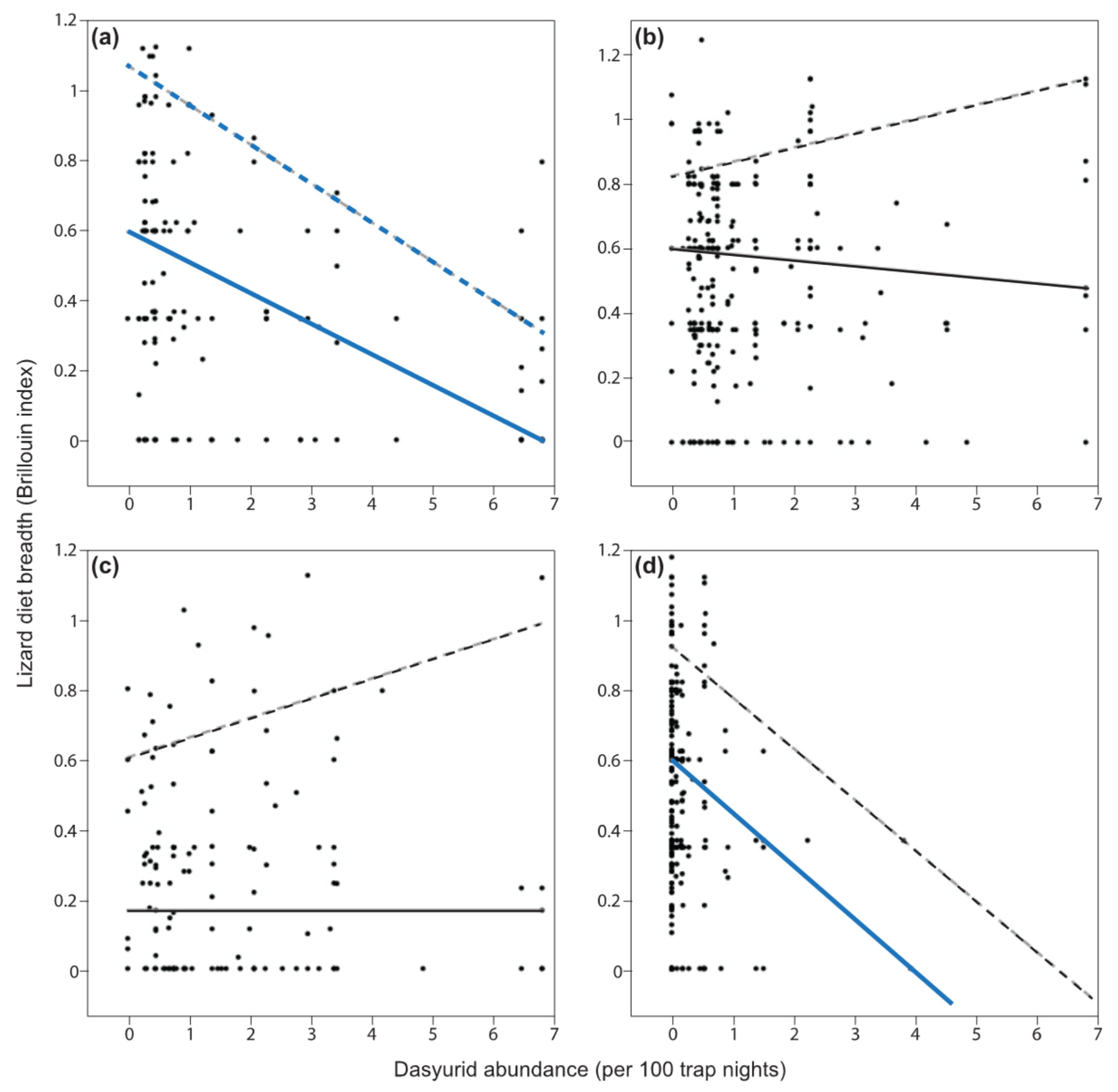
| Prediction Category—Lizard Group | 0.5 Quantile | 0.9 Quantile | ||||||
|---|---|---|---|---|---|---|---|---|
| Dasyurid Group | Estimate | CI | t-Value | p | Estimate | CI | t-Value | p |
| Nocturnal generalists | ||||||||
| All dasyurids | −0.087 | −0.102–0.072 | −11.478 | <0.0001 * | −0.112 | −0.150–0.073 | −5.739 | <0.0001 * |
| Sminthopsis youngsoni | −0.111 | −0.131–0.091 | −10.933 | <0.0001 * | −0.136 | −0.176–0.097 | −6.799 | <0.0001 * |
| Dasycercus blythi | −0.197 | −0.612–0.219 | −0.937 | 0.3506 | 0.157 | −0.232–0.545 | 0.799 | 0.4256 |
| Nocturnal generalist skink | ||||||||
| All dasyurids | 0.018 | −0.673–0.708 | 0.060 | 0.9539 | 0.171 | −0.978–1.320 | 0.343 | 0.7406 |
| Sminthopsis youngsoni | −0.959 | −4.413–2.496 | −0.640 | 0.5402 | −1.751 | −4.563–1.061 | −1.436 | 0.189 |
| Dasycercus blythi | 0.049 | −0.948–1.047 | 0.114 | 0.9121 | 0.235 | −2.530–3.000 | 0.196 | 0.8494 |
| Diurnal generalists | ||||||||
| All dasyurids | −0.018 | −0.076–0.040 | −0.607 | 0.5441 | 0.044 | −0.002–0.090 | 1.880 | 0.0612 |
| Sminthopsis youngsoni | 0.039 | −0.031–0.109 | 1.103 | 0.2712 | 0.057 | −0.001–0.116 | 1.932 | 0.0545 |
| Dasycercus blythi | −0.152 | −0.232–0.072 | −3.746 | 0.0002 * | −0.145 | −0.516–0.226 | −0.771 | 0.4416 |
| Specialists | ||||||||
| All dasyurids | <0.001 | −0.044–0.044 | <0.001 | 1.0000 | 0.056 | −0.028–0.140 | 1.321 | 0.1886 |
| Sminthopsis youngsoni | 0.000 | −0.058–0.058 | 0.000 | 1.0000 | 0.094 | −0.033–0.222 | 1.467 | 0.1445 |
| Dasycercus blythi | −0.042 | −0.236–0.151 | −0.433 | 0.6654 | 0.181 | −0.256–0.618 | 0.819 | 0.4143 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dickman, C.R.; Greenville, A.C.; Wardle, G.M.; Bytheway, J.P. Class Conflict: Diffuse Competition between Mammalian and Reptilian Predators. Diversity 2020, 12, 355. https://doi.org/10.3390/d12090355
Dickman CR, Greenville AC, Wardle GM, Bytheway JP. Class Conflict: Diffuse Competition between Mammalian and Reptilian Predators. Diversity. 2020; 12(9):355. https://doi.org/10.3390/d12090355
Chicago/Turabian StyleDickman, Christopher R., Aaron C. Greenville, Glenda M. Wardle, and Jenna P. Bytheway. 2020. "Class Conflict: Diffuse Competition between Mammalian and Reptilian Predators" Diversity 12, no. 9: 355. https://doi.org/10.3390/d12090355
APA StyleDickman, C. R., Greenville, A. C., Wardle, G. M., & Bytheway, J. P. (2020). Class Conflict: Diffuse Competition between Mammalian and Reptilian Predators. Diversity, 12(9), 355. https://doi.org/10.3390/d12090355






