Bat Species Richness and Community Composition along a Mega-transect in the Okavango River Basin
Abstract
1. Introduction
2. Materials and Methods
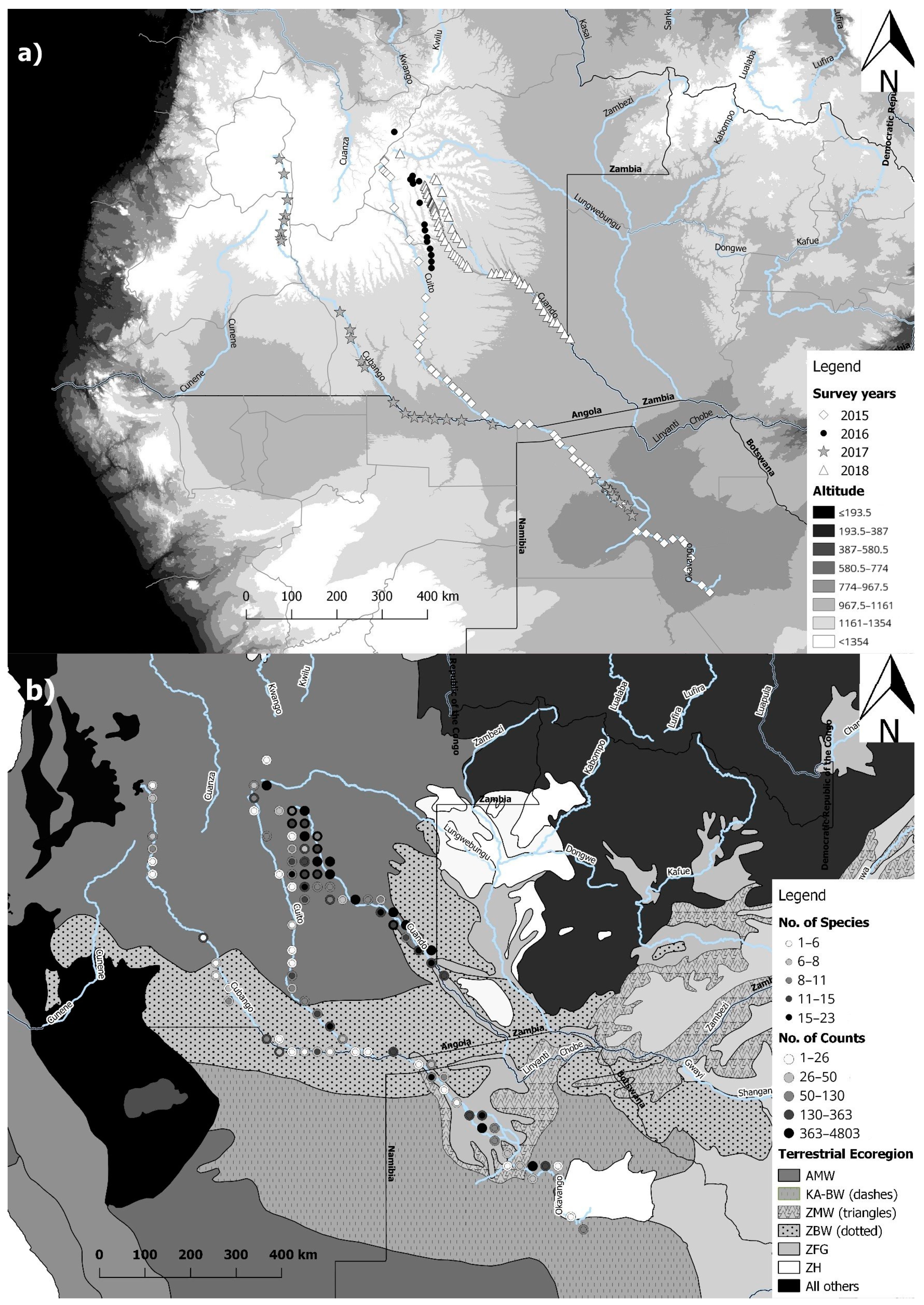
2.1. Acoustic Data Collection and Study Sites
2.2. Acoustic Call Analyses
2.3. GIS Analysis
2.4. Statistical Analysis
3. Results
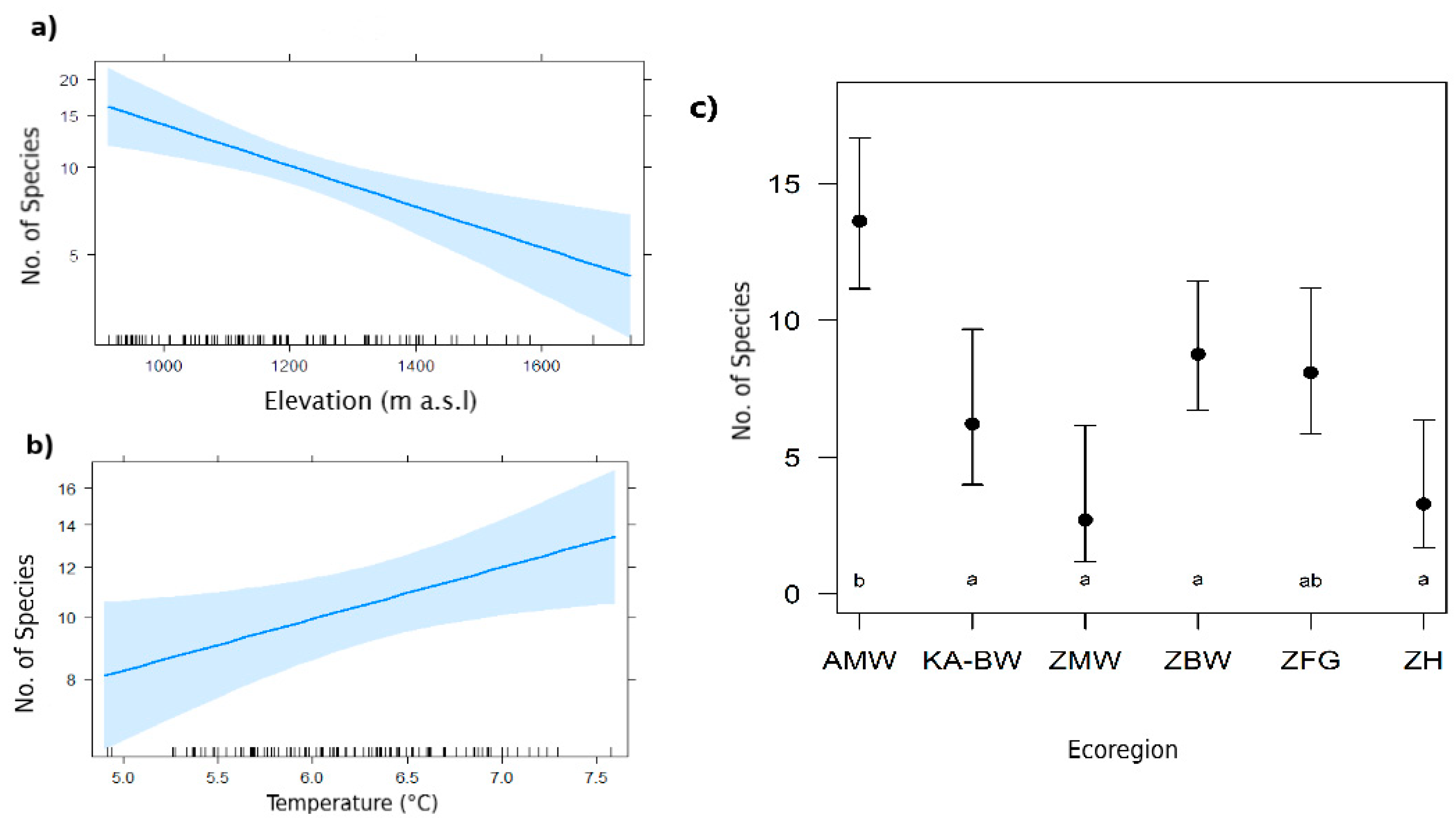
3.1. Predictors of Species Richness
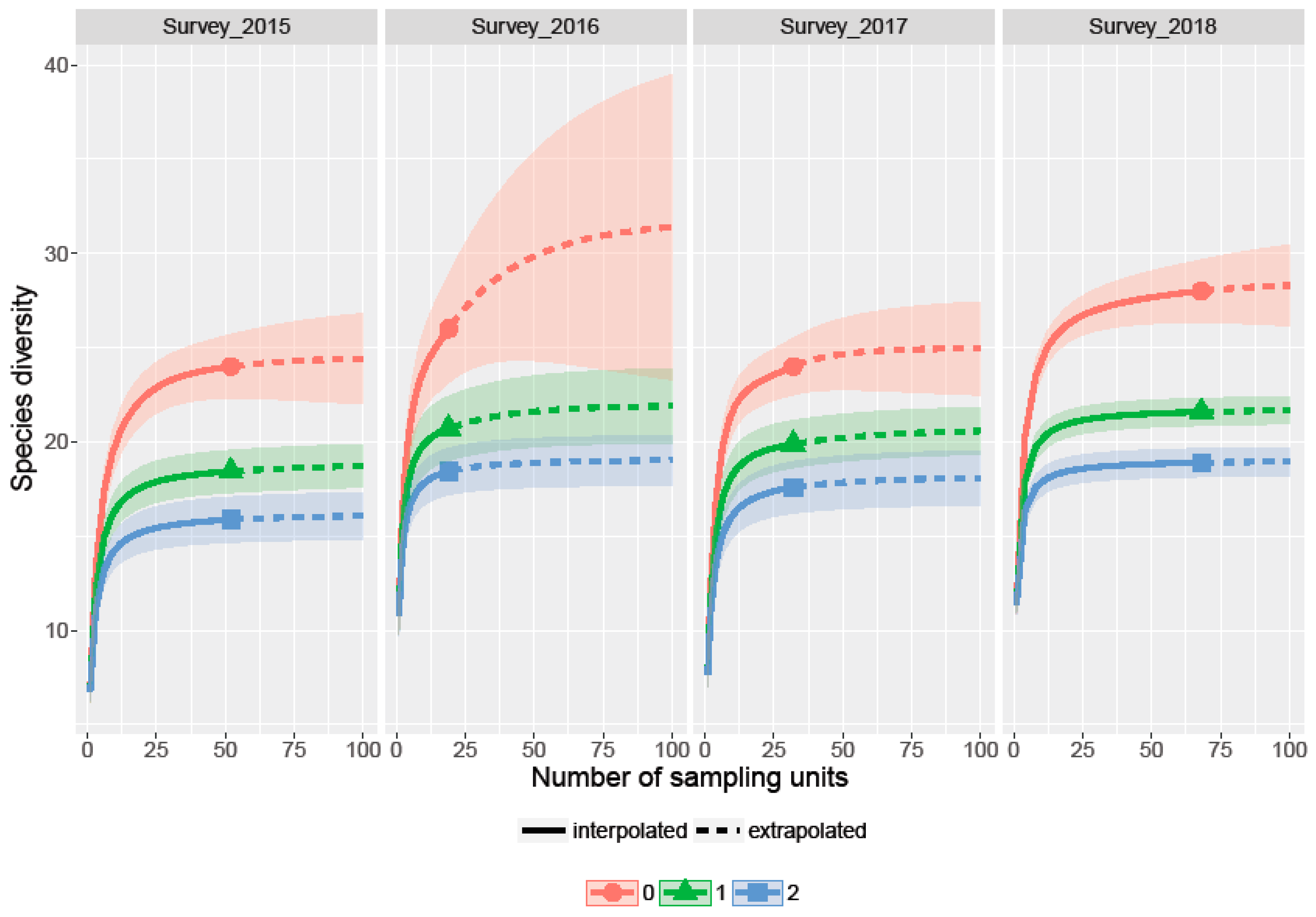
3.2. Richness, Diversity and Rarefaction
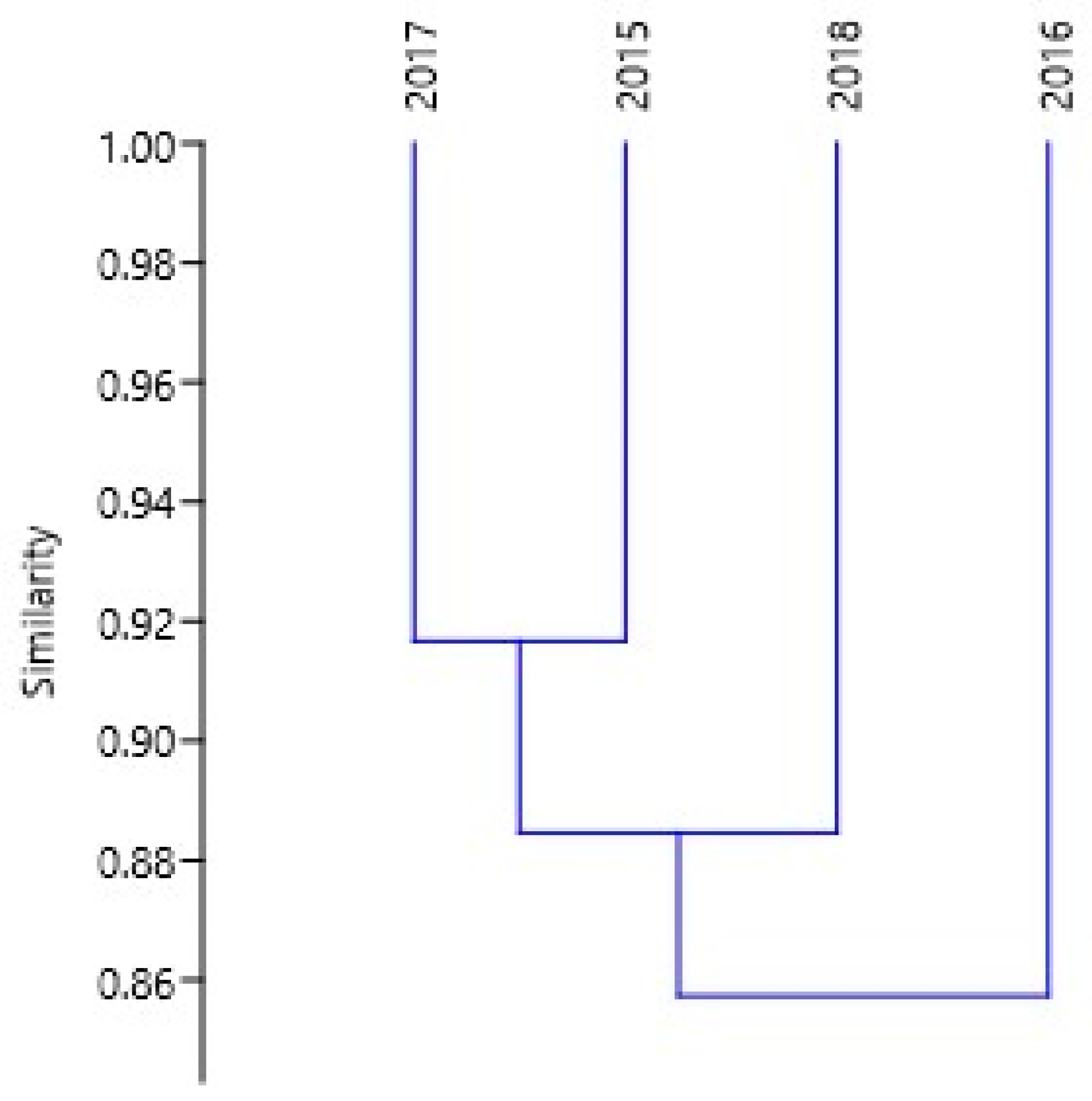
3.3. Community Structure
4. Discussion
4.1. Effect of Elevation, Climate and Terrestrial Ecoregion
4.2. Biogeographical Aspects
4.3. Bat Activity as a Control Variable
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Huntley, B.J.; Russo, V.; Lages, F.; Ferrand, N. Biodiversity of Angola: Science & Conservation: A Modern Synthesis; Springer: Cham, Switzerland, 2019; p. 552. ISBN 978-3-030-03083-4. [Google Scholar] [CrossRef]
- Cooper-Bohannon, R.; Rebelo, H.; Jones, G.; Cotterill, F.P.D.; Monadjem, A.; Schoeman, M.C.; Taylor, P.J.; Park, K.J. Predicting bat distributions and diversity hotspots in Southern Africa. Hystrix 2016, 27, 1. [Google Scholar] [CrossRef]
- Schoeman, M.C.; Cotterill, F.; Taylor, P.J.; Monadjem, A. Using potential distributions to explore environmental correlates of bat species richness in southern Africa: Effects of model selection and taxonomy. Curr. Zool. 2013, 59, 279–293. [Google Scholar] [CrossRef]
- Herkt, K.M.B.; Barnikel, G.; Skidmore, A.K.; Fahr, J. A high-resolution model of bat diversity and endemism for continental Africa. Ecol. Modell. 2016, 320, 9–28. [Google Scholar] [CrossRef]
- Pio, D.V.; Engler, R.; Linder, H.P.; Monadjem, A.; Cotterill, F.P.; Taylor, P.J.; Schoeman, M.C.; Price, B.W.; Villet, M.H.; Eick, G. Climate change effects on animal and plant phylogenetic diversity in southern Africa. Glob. Chang. Biol. 2014, 20, 1538–1549. [Google Scholar] [CrossRef]
- Clausnitzer, V.; Kityo, R. Altitudinal distribution of rodents (Muridae and Gliridae) on Mt Elgon, Uganda. Trop. Zool. 2001, 14, 95–118. [Google Scholar] [CrossRef]
- Curran, M.; Kopp, M.; Beck, J.; Fahr, J. Species diversity of bats along an altitudinal gradient on Mount Mulanje, southern Malawi. J. Trop. Ecol. 2012, 28, 243–253. [Google Scholar] [CrossRef]
- Linden, V.M.; Weier, S.M.; Gaigher, I.; Kuipers, H.J.; Weterings, M.J.; Taylor, P.J. Changes of bat activity, species richness, diversity and community composition over an altitudinal gradient in the Soutpansberg range, South Africa. Acta Chiropt. 2014, 16, 27–40. [Google Scholar] [CrossRef]
- Taylor, P.; Munyai, A.; Gaigher, I.; Baxter, R. Afromontane small mammals do not follow the hump-shaped rule: Elevational variation in a tropical biodiversity hotspot (Soutpansberg Mountains, South Africa). J. Trop. Ecol. 2015, 31, 37–48. [Google Scholar] [CrossRef]
- Weier, S.M.; Linden, V.M.; Gaigher, I.; White, P.J.; Taylor, P.J. Changes of bat species composition over altitudinal gradients on northern and southern aspects of the Soutpansberg mountain range, South Africa. Mammalia 2017, 81, 49–60. [Google Scholar] [CrossRef]
- McCain, C.M. Elevational gradients in diversity of small mammals. Ecology 2005, 86, 366–372. [Google Scholar] [CrossRef]
- McCain, C.M. Could temperature and water availability drive elevational species richness patterns? A global case study for bats. Glob. Ecol. Biogeogr. 2007, 16, 1–13. [Google Scholar] [CrossRef]
- Okavango Wilderness Project. Available online: https://www.nationalgeographic.org/projects/okavango/ (accessed on 20 March 2020).
- Taylor, P.J.; Neef, G.; Keith, M.; Weier, S.; Monadjem, A.; Parker, D.M. Tapping into technology and the biodiversity informatics revolution: updated terrestrial mammal list of Angola, with new records from the Okavango Basin. Zookeys 2018, 10, 51–88. [Google Scholar] [CrossRef]
- Monadjem, A.; Taylor, P.J.; Cotterill, F.D.P.; Schoeman, M.C. Bats of Southern and Central Africa: A Biographic and Taxonomic Synthesis; Wits University Press: Johannesburg, South Africa, 2010; p. 596. ISBN 978-1-86814-508-9. [Google Scholar]
- Beja, P.; Vaz Pinto, P.; Verissimo, L.; Bersacola, E.; Fabiano, E.; Palmeirim, J.M.; Monadjem, A.; Monterroso, P.; Svensson, M.; Taylor, P.J. The Mammals of Angola. In Biodiversity of Angola; Huntley, B.J., Russo, V., Lages, F., Ferrand, N., Eds.; Springer: Cham, Switzerland, 2019; pp. 357–444. ISBN 978-3-030-03083-4. [Google Scholar] [CrossRef]
- Smith, A.; Schoeman, M.C.; Keith, M.; Erasmus, B.F.; Monadjem, A.; Moilanen, A.; Di Minin, E. Synergistic effects of climate and land-use change on representation of African bats in priority conservation areas. Ecol. Indic. 2016, 69, 276–283. [Google Scholar] [CrossRef]
- Goyder, D.J.; Barker, N.; Bester, S.P.; Frisby, A.; Janks, M.; Gonçalves, F.M. The Cuito catchment of the Okavango system: A vascular plant checklist for the Angolan headwaters. PhytoKeys 2018, 113, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Jacobs, D.S.; Kunz, T.H.; Willig, M.R.; Racey, P.A. Carpe noctem: the importance of bats as bioindicators. Endanger. Species Res. 2009, 8, 93–115. [Google Scholar] [CrossRef]
- Russo, D.; Jones, G. Bats as bioindicators: an introduction. Mamm. Biol. 2015, 80, 157–158. [Google Scholar] [CrossRef]
- Russo, D.; Voigt, C.C. The use of automated identification of bat echolocation calls in acoustic monitoring: A cautionary note for a sound analysis. Ecol. Indic. 2016, 66, 598–602. [Google Scholar] [CrossRef]
- Mickleburgh, S.P.; Hutson, A.M.; Racey, P.A. A review of the global conservation status of bats. Oryx 2002, 36, 18–34. [Google Scholar] [CrossRef]
- Jones, K.; Russ, J.; Bashta, A.; Bilhari, Z.; Catto, C.; Csosz, I.; Gorbachev, A.; Gyorfi, P.; Hughes, A.; Ivashkiv, I.; et al. Indicator bats program: A system for the global acoustic monitoring of bats. In Biodiversity Monitoring and Conservation: Bridging the Gap between Global Commitment and Local Action; Collen, B., Durant, S.M., Baillie, J.E.M., Pettorelli, N., Eds.; John Wiley and Sons, Ltd: Chichester West Sussex, UK, 2013; pp. 211–247. ISBN 978-1-4443-3291-9. [Google Scholar]
- Russo, D.; Ancillotto, L. Sensitivity of bats to urbanization: a review. Mamm. Biol. 2015, 80, 205–212. [Google Scholar] [CrossRef]
- Luck, G.W.; Smallbone, L.; Threlfall, C.; Law, B. Patterns in bat functional guilds across multiple urban centres in south-eastern Australia. Landsc. Ecol. 2013, 28, 455–469. [Google Scholar] [CrossRef]
- Linden, V.M.; Grass, I.; Tscharntke, T.; Weier, S.M.; Taylor, P.J. Bat guilds and their pest control in macadamia landscapes exhibit contrasting responses to natural habitat. Biol. Conserv. under review.
- Shapiro, J.T.; Monadjem, A.; Röder, T.; McCleery, R.A. Response of bat activity to land cover and land use in savannas is scale-, season-, and guild-specific. Biol. Conserv. 2020, 241, 108245. [Google Scholar] [CrossRef]
- Jung, K.; Threlfall, C.G. Urbanisation and Its Effects on Bats—A Global Meta-Analysis. In Bats in the Anthropocene: Conservation of Bats in a Changing World; Voigt, C.C., Kingston, T., Eds.; Springer: Cham, Switzerland, 2016; pp. 13–33. [Google Scholar] [CrossRef]
- ANABAT. Available online: http://users.lmi.net/corben/ (accessed on 19 December 2018).
- Rydell, J.; Nyman, S.; Eklöf, J.; Jones, G.; Russo, D. Testing the performances of automated identification of bat echolocation calls: A request for prudence. Ecol. Indic. 2017, 78, 416–420. [Google Scholar] [CrossRef]
- Taylor, P.J.; Sowler, S.; Schoeman, M.C.; Monadjem, A. Diversity of bats in the Soutpansberg and Blouberg Mountains of northern South Africa: complementarity of acoustic and non-acoustic survey methods. Afr. J. Wildl. Res. 2013, 43, 12–26. [Google Scholar] [CrossRef][Green Version]
- Happold, M.; Happold, D.C. Hedgehogs, Shrews and Bats. In Mammals of Africa Volume IV; Bloomsbury Publishing: London, UK, 2013; p. 800. ISBN 978-1-4081-2254-9. [Google Scholar]
- QGIS Geographic Information System. Available online: http://qgis.osgeo.org (accessed on 17 January 2018).
- Fleming, G. SACRS.md. Available online: https://gist.github.com/gubuntu/6403425 (accessed on 3 January 2018).
- WorldClim. Available online: https://www.worldclim.org/ (accessed on 17 December 2019).
- Olson, D.M.; Dinerstein, E.; Wikramanayake, E.D.; Burgess, N.D.; Powell, G.V.; Underwood, E.C.; D’amico, J.A.; Itoua, I.; Strand, H.E.; Morrison, J.C. Terrestrial Ecoregions of the World: A New Map of Life on EarthA new global map of terrestrial ecoregions provides an innovative tool for conserving biodiversity. BioScience 2001, 51, 933–938. [Google Scholar] [CrossRef]
- The R Project for Statistical Computing. Available online: https://www.R-project.org/ (accessed on 3 July 2017).
- Fitting linear mixed-effects models using lme4. Available online: https://arxiv.org/pdf/1406.5823.pdf (accessed on 13 January 2020).
- Performance: Assessment of Regression Models Performance. Available online: https://rdrr.io/cran/performance/ (accessed on 2 January 2019).
- Multi-Model Inference. Available online: https://CRAN.Rproject.org/package=MuMIn (accessed on 13 October 2018).
- Fox, J.; Hong, J. Effect displays in R for multinomial and proportional-odds logit models: Extensions to the effects package. J. Stat. Softw. 2009, 32, 1–24. [Google Scholar] [CrossRef]
- Hothorn, T.; Bretz, F.; Westfall, P. Simultaneous inference in general parametric models. J. Math Meth. Biosci. 2008, 50, 346–363. [Google Scholar] [CrossRef]
- Chao, A.; Gotelli, N.J.; Hsieh, T.C.; Sander, E.L.; Ma, K.H.; Colwell, R.K.; Ellison, A.M. Rarefaction and extrapolation with Hill numbers: a framework for sampling and estimation in species diversity studies. Ecol. Monogr. 2014, 84, 45–67. [Google Scholar] [CrossRef]
- Hsieh, T.; Ma, K.; Chao, A. iNEXT: an R package for rarefaction and extrapolation of species diversity (H ill numbers). Meth. Ecol. Evol. 2016, 7, 1451–1456. [Google Scholar] [CrossRef]
- Colwell, R.K.; Chao, A.; Gotelli, N.J.; Lin, S.-Y.; Mao, C.X.; Chazdon, R.L.; Longino, J.T. Models and estimators linking individual-based and sample-based rarefaction, extrapolation and comparison of assemblages. Plant Ecol. 2012, 5, 3–21. [Google Scholar] [CrossRef]
- Hsieh, T.; Ma, K.; Chao, A. iNEXT: iNterpolation and EXTrapolation for species diversity. R package version 2016, 2, 1–18. [Google Scholar]
- López-González, C.; Presley, S.J.; Lozano, A.; Stevens, R.D.; Higgins, C.L. Metacommunity analysis of Mexican bats: environmentally mediated structure in an area of high geographic and environmental complexity. J. Biogeogr. 2012, 39, 177–192. [Google Scholar] [CrossRef]
- Jaberg, C.; Guisan, A. Modelling the distribution of bats in relation to landscape structure in a temperate mountain environment. J. Appl. Ecol. 2001, 38, 1169–1181. [Google Scholar] [CrossRef]
- Monadjem, A.; Shapiro, J.T.; Mtsetfwa, F.; Reside, A.E.; McCleery, R.A. Acoustic Call Library and Detection Distances for Bats of Swaziland. Acta Chiropt. 2017, 19, 175–187. [Google Scholar] [CrossRef]
- Linden, V.M.G.; Grass, I.; Joubert, E.; Tscharntke, T.; Weier, S.M.; Taylor, P.J.; Struebig, M. Ecosystem services and disservices by birds, bats and monkeys change with macadamia landscape heterogeneity. J. Appl. Ecol. 2019, 56, 2069–2078. [Google Scholar] [CrossRef]
- Schoeman, M.C.; Monadjem, A. Community structure of bats in the savannas of southern Africa: Influence of scale and human land use. Hystrix 2018, 29, 3–10. [Google Scholar]
- Foord, S.H.; Swanepoel, L.H.; Evans, S.W.; Schoeman, C.S.; Erasmus, B.F.N.; Schoeman, M.C.; Keith, M.; Smith, A.; Mauda, E.V.; Maree, N.; et al. Animal taxa contrast in their scale-dependent responses to land use change in rural Africa. PLoS ONE 2018, 13, 5. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.; Threlfall, C.G. Trait-dependent tolerance of bats to urbanization: a global meta-analysis. Proc. R Soc. B 2018, 285, 20181222. [Google Scholar] [CrossRef]
- MacSwiney G, M.C.; Clarke, F.M.; Racey, P.A. What you see is not what you get: the role of ultrasonic detectors in increasing inventory completeness in Neotropical bat assemblages. J. Appl. Ecol. 2008, 45, 1364–1371. [Google Scholar] [CrossRef]
- Adams, R.; Kwiecinski, G. Sonar Surveys for Bat Species Richness and Activity in the Southern Kalahari Desert, Kgalagadi Transfrontier Park, South Africa. Diversity 2018, 10, 103. [Google Scholar] [CrossRef]
- Parker, D.M.; Bernard, R.T.F. The use of acoustic detectors for assessing bat species richness and functional activity in a South African National Park. Mammalia 2018, 83, 53–63. [Google Scholar] [CrossRef]
- Adams, A.M.; Jantzen, M.K.; Hamilton, R.M.; Fenton, M.B. Do you hear what I hear? Implications of detector selection for acoustic monitoring of bats. Meth. Ecol. Evol. 2012, 3, 992–998. [Google Scholar] [CrossRef]
- Wildlife Acoustics. Available online: https://www.wildlifeacoustics.com/images/documentation/SM4-BAT-ZC-USER-GUIDE.pdf (accessed on 24 February 2020).
- Harrison, X.A.; Donaldson, L.; Correa-Cano, M.E.; Evans, J.; Fisher, D.N.; Goodwin, C.E.; Robinson, B.S.; Hodgson, D.J.; Inger, R. A brief introduction to mixed effects modelling and multi-model inference in ecology. PeerJ 2018, 6, e4794. [Google Scholar] [CrossRef] [PubMed]
- Vellend, M.; Verheyen, K.; Flinn, K.M.; Jacquemyn, H.; Kolb, A.; Van Calster, H.; Peterken, G.; Graae, B.J.; Bellemare, J.; Honnay, O. Homogenization of forest plant communities and weakening of species–environment relationships via agricultural land use. J. Ecol. 2007, 95, 565–573. [Google Scholar] [CrossRef]
| Family | Species (Species Code) | Functional Group | Overlap Species | Occurrence in Angola | Caught during 2015–2018 | No. Recorded Calls/No. Recording Nights |
|---|---|---|---|---|---|---|
| Emballanuridae | Taphozous mauritianus (Tmaurit) | Open-air | C. pumilus, T. aegyptiaca | Specimen | No | 199/33 |
| Hipposideridae | Macronycteris vitattus (Hvit) | Clutter | None | Specimen | No | 5/4 |
| Rhinolophidae | Rhinolophus fumigatus (Rfum) | Clutter | None | Specimen | No | 1/1 |
| Miniopteridae | Miniopterus cf. fraterculus (Mfrat) | Clutter-edge | M. fraterculus, H. anchietae | No | No | 514/93 |
| Miniopterus natalensis (Mnatal) | Clutter-edge | P. rusticus, H. anchietae | Specimen | No | 56/24 | |
| Molossidae | Chaerephon ansorgei (Cansor) | Open-air | T. ventralis, C. nigeriae, T. fulminans | Specimen | No | 1265/75 |
| Chaerephon nigeriae (Cnig) | Open-air | T. ventralis, C. ansorgei, T. fulminans | Literature | Yes | 738/60 | |
| Chaerephon pumilus (Cpum) | Open-air | T. aegyptiaca, M. condylurus, T. mauritianus | Specimen | No | 991/79 | |
| Mops condylurus (Mcond) | Open-air | C. pumilus, T. aegyptiaca | Specimen | No | 293/61 | |
| Mops midas (Mmidas) | Open-air | C. nigeriae, O. martiensseni | Predicted | No | 49/15 | |
| Otomops martiensseni (Omops) | Open-air | Mop. midas | Specimen | No | 105/14 | |
| Tadarida aegyptiaca (Taegypt) | Open-air | T. mauritianus, C. pumilus | Specimen | No | 2367/94 | |
| Vespertilionidae | Eptesicus hottentotus (Ehott) | Clutter-edge | S. dingani, M. welwitschii | Specimen | No | 83/28 |
| Neoromicia anchietae (Hanch) | Clutter-edge | M. natalensis | Specimen | Yes | 273/58 | |
| Kerivoula argentata (Karg) | Clutter | Unknown Vesper75 | Predicted | No | 1069/72 | |
| Laephotis angolensis (Lang) | Clutter-edge | P. hesperidus | Specimen | Yes | 155/33 | |
| Myotis welwitschii (Mwel) | Clutter-edge | M. bocagii | Specimen | Yes | 12/7 | |
| Mimetillus thomasi | Clutter-edge | Not applicable | Specimen | Yes | No reference calls available | |
| Neoromica zuluensis (Nzulu) | Clutter-edge | P. hesperidus | Specimen | Yes | 2186/106 | |
| Neoromicia capensis (Ncap) | Clutter-edge | S. viridis, S. hindei/albigula, N. schlieffeni, Vansonia rueppellii | Specimen | Yes | 656/114 | |
| Neoromicia nana (Nnana) | Clutter-edge | Not applicable | Specimen | No | 224/46 | |
| Nycticeinops schlieffeni (Nschlief) | Clutter-edge | S. viridis, S. hindei/albigula, N. capensis | Specimen | No | 3909/157 | |
| Pipistrellus cf hesperidus (Phesp) | Clutter-edge | N. zuluensis, L. angolensis | Predicted | Yes | 9418/144 | |
| Pipistrellus rusticus (Prust) | Clutter-edge | M. natalensis | Predicted | Yes | 826/64 | |
| Scotophilus dinganii (Sding) | Clutter-edge | E. hottentotus, L. botswanae | Specimen | No | 146/70 | |
| Scotophilus leucogaster | Clutter-edge | Possibly N. schlieffeni, S. viridis, N. capensis | Specimen | Yes | No reference calls available |
| Title | Estimate | SE | Z | p-Value |
|---|---|---|---|---|
| Intercept (Angolan Miombo Woodlands) | 2.61 | 0.10 | 25.54 | 0.000 |
| Kalahari Acacia-Baikiaea woodlands | −0.78 | 0.27 | −2.82 | 0.004 |
| Zambezian and Mopane woodlands | −1.61 | 0.44 | −3.60 | 0.000 |
| Zambezian Baikiaea woodlands | −0.44 | 0.13 | −3.21 | 0.001 |
| Zambezian flooded grasslands | −0.52 | 0.21 | −2.43 | 0.014 |
| Zambezian halophytics | −1.42 | 0.37 | −3.79 | 0.000 |
| Elevation | −0.30 | 0.08 | −3.54 | 0.000 |
| Minimum Temperature | 0.10 | 0.04 | 2.30 | 0.021 |
| Terrestrial Ecoregion | Status | Precipitation |
|---|---|---|
| Angolan Miombo Woodlands | Vulnerable | 800 to 1400 mm |
| Kalahari Acacia-Baikiaea woodlands | Vulnerable | Up to 800 |
| Zambezian and Mopane woodlands | Vulnerable | Up to 800 |
| Zambezian Baikiaea woodlands | Vulnerable | Up to 800 |
| Zambezian flooded grasslands | Relatively Stable/Intact | 800 to 1000 mm |
| Zambezian halophytics | Vulnerable | 450 and 500 mm |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weier, S.M.; Keith, M.; Neef, G.G.; Parker, D.M.; Taylor, P.J. Bat Species Richness and Community Composition along a Mega-transect in the Okavango River Basin. Diversity 2020, 12, 188. https://doi.org/10.3390/d12050188
Weier SM, Keith M, Neef GG, Parker DM, Taylor PJ. Bat Species Richness and Community Composition along a Mega-transect in the Okavango River Basin. Diversity. 2020; 12(5):188. https://doi.org/10.3390/d12050188
Chicago/Turabian StyleWeier, Sina M., Mark Keith, Götz G. Neef, Daniel M. Parker, and Peter J. Taylor. 2020. "Bat Species Richness and Community Composition along a Mega-transect in the Okavango River Basin" Diversity 12, no. 5: 188. https://doi.org/10.3390/d12050188
APA StyleWeier, S. M., Keith, M., Neef, G. G., Parker, D. M., & Taylor, P. J. (2020). Bat Species Richness and Community Composition along a Mega-transect in the Okavango River Basin. Diversity, 12(5), 188. https://doi.org/10.3390/d12050188






