Testing Bathymetric and Regional Patterns in the Southwest Atlantic Deep Sea Using Infaunal Diversity, Structure, and Function
Abstract
1. Introduction
- (1)
- Do deep-sea nematode structure and function at the SE Brazilian Margin follow the same bathymetric patterns as in other deep-sea areas?
- (2)
- Is there a marked difference between structural and functional nematode characteristics along bathymetric gradients, and what does that mean for our understanding of deep-sea infaunal communities?
- (3)
- What are the local or regional drivers of observed bathymetric patterns in deep-sea nematode structural biodiversity and function in this region of the SW Atlantic? Does the region stand out in the ways in which it maintains deep-sea biodiversity?
2. Materials and Methods
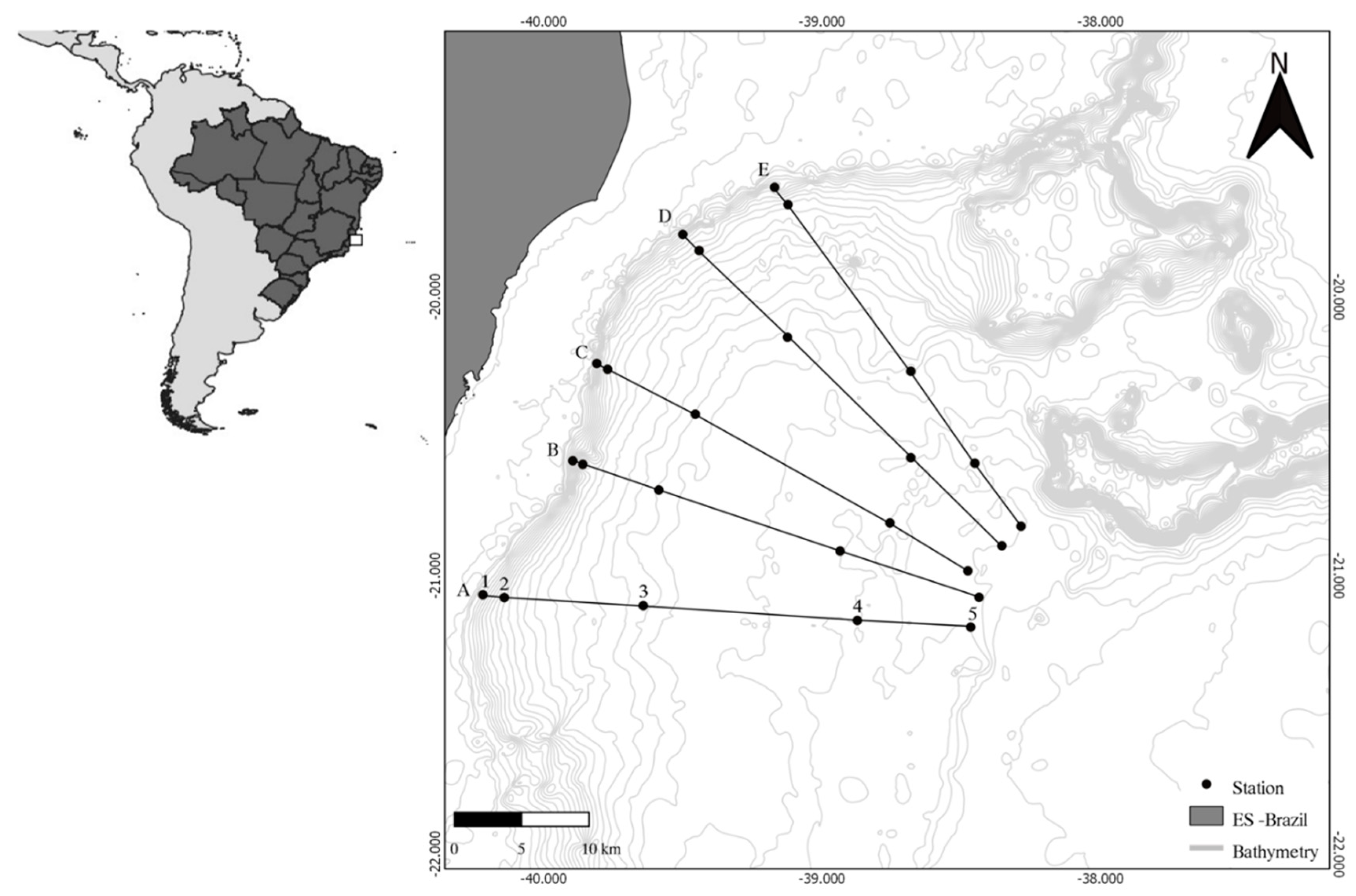
2.1. Study Area and Sampling
2.2. Nematode Sample Processing and Identification
2.3. Environmental Variables
2.4. Data Analysis
2.4.1. Structural and Functional Nematode Community Characteristics
2.4.2. Statistical Analyses
3. Results
3.1. Environmental Variables
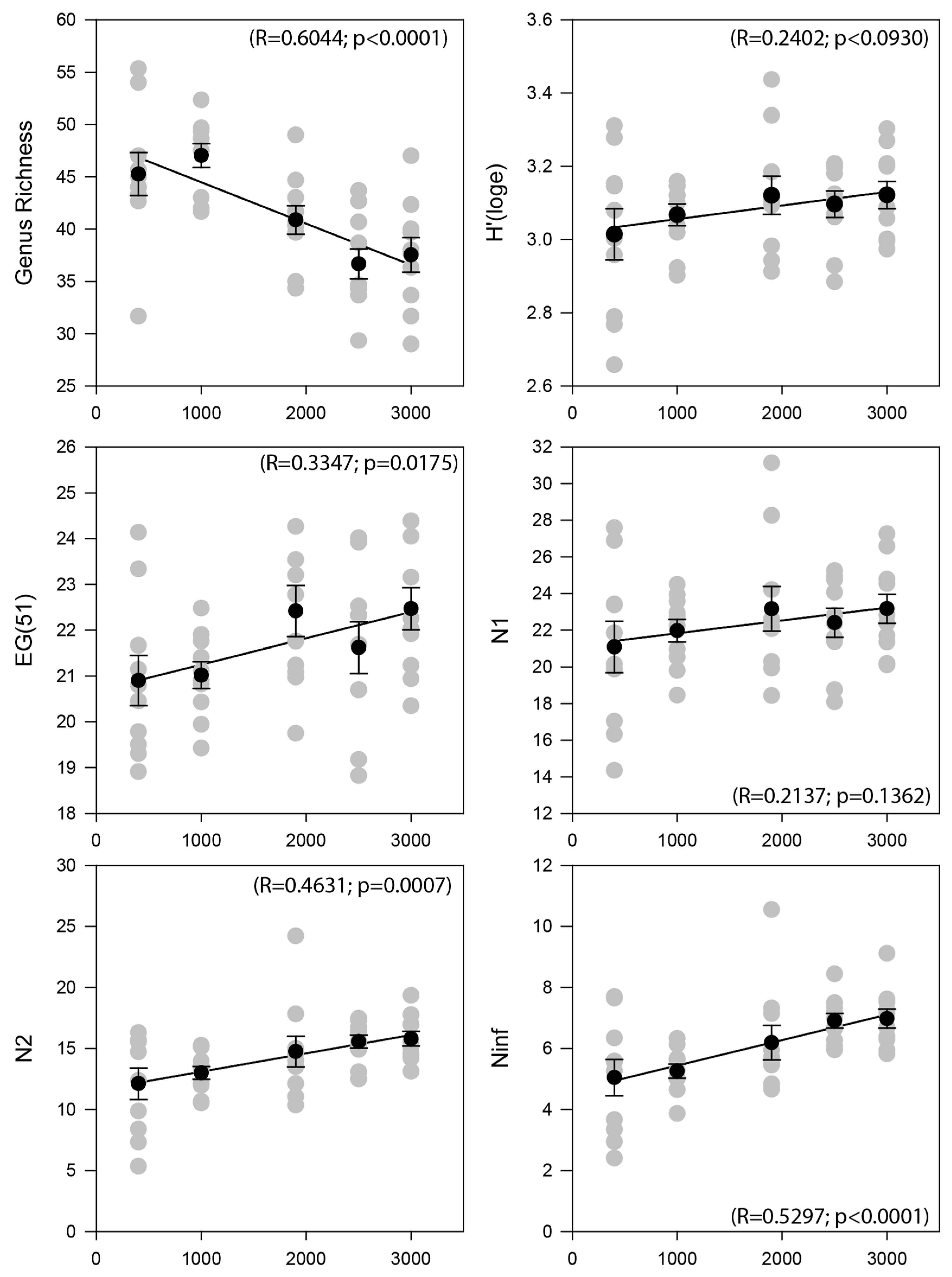
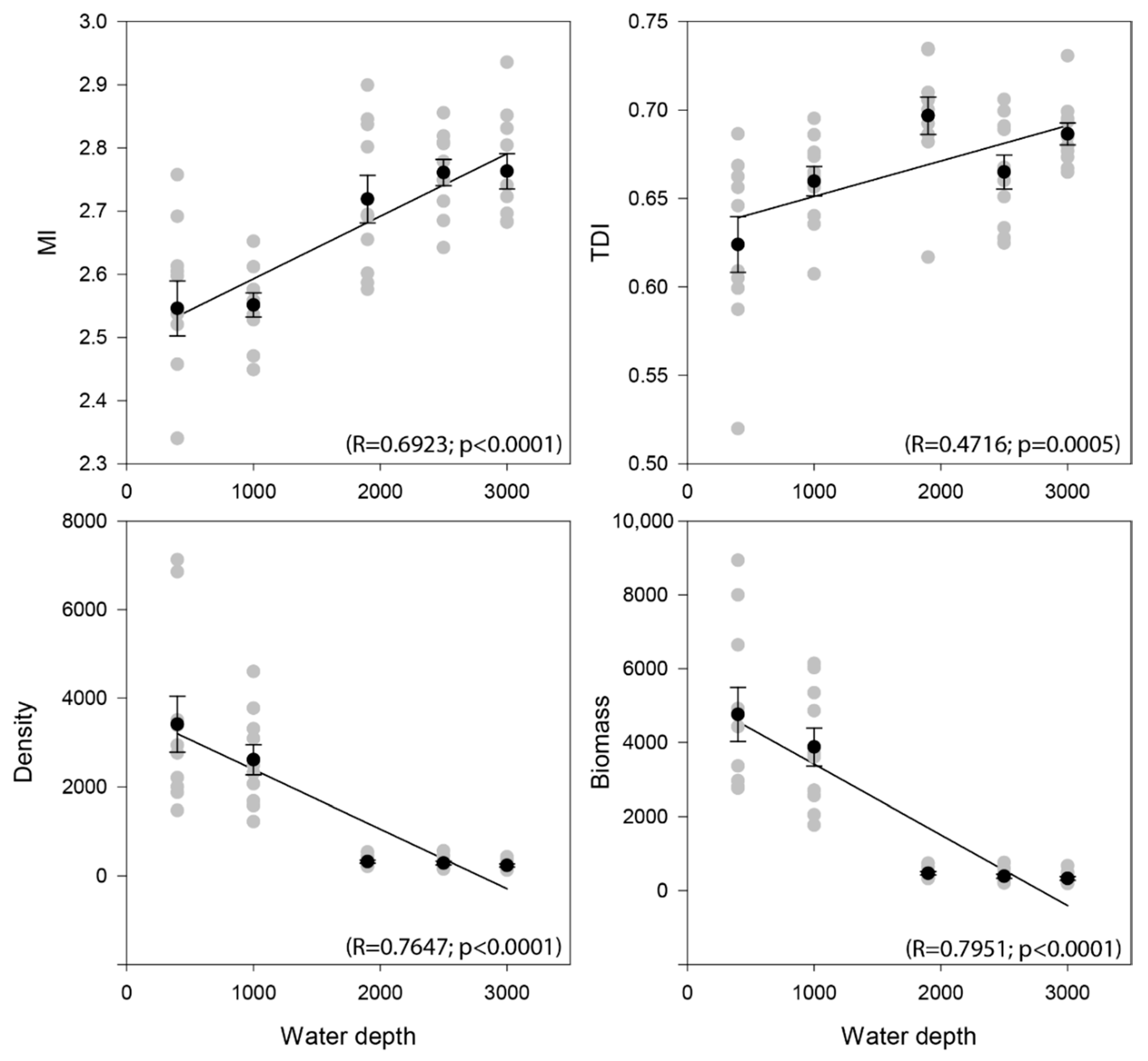
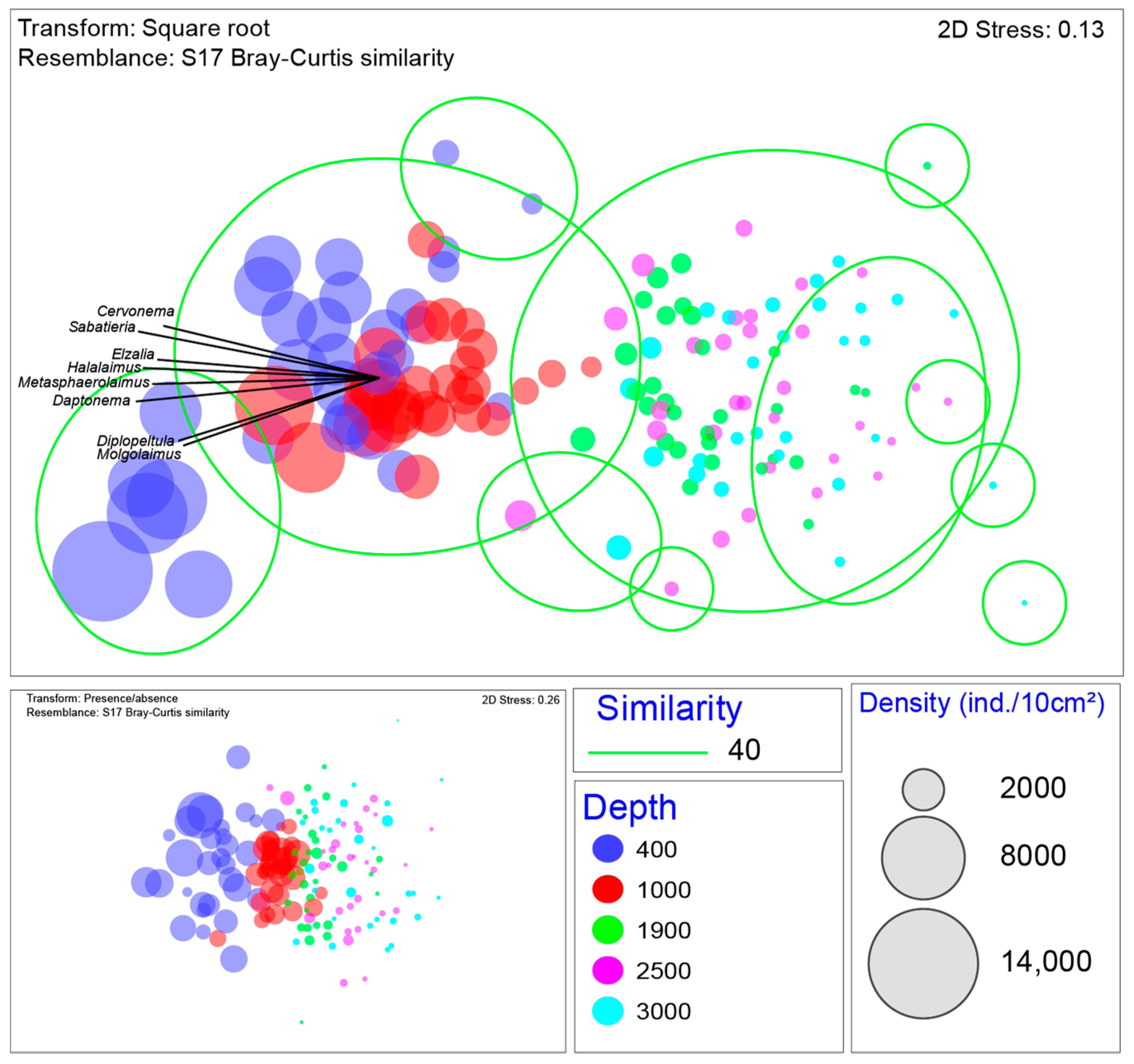
3.2. Nematode Community Characteristics and Bathymetrical Gradients
3.3. Relation of Environmental Variables with Nematode Community
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thurber, A.R.; Sweetman, A.K.; Narayanaswamy, B.E.; Jones, D.O.B.; Ingels, J.; Hansman, R.L. Ecosystem function and services provided by the deep sea. Biogeosciences 2014, 11, 3941–3963. [Google Scholar] [CrossRef]
- Danovaro, R.; Snelgrove, P.V.; Tyler, P. Challenging the paradigms of deep-sea ecology. Trends Ecol. Evol. 2014, 29, 465–475. [Google Scholar] [CrossRef]
- Snelgrove, P.V.; Thrush, S.F.; Wall, D.H.; Norkko, A. Real world biodiversity–ecosystem functioning: A seafloor perspective. Trends Ecol. Evol. 2014, 29, 398–405. [Google Scholar] [CrossRef]
- Rex, M.A.; Etter, R.J. Deep-Sea Biodiversity: Pattern and Scale; Harvard University Press: Cambridge, MA, USA, 2010; 354p. [Google Scholar]
- Rex, M.A.; Etter, R.J.; Morris, J.S.; Crouse, J.; McClain, C.R.; Johnson, N.A.; Stuart, C.T.; Deming, J.W.; Thies, R.; Avery, R. Global bathymetric patterns of standing stock and body size in the deep-sea benthos. Mar. Ecol. Prog. Ser. 2006, 317, 1–8. [Google Scholar] [CrossRef]
- Rosli, N.; Leduc, D.; Rowden, A.A.; Probert, P.K. Review of recent trends in ecological studies of deep-sea meiofauna, with focus on patterns and processes at small to regional spatial scales. Mar. Biodivers. 2018, 48, 13–34. [Google Scholar] [CrossRef]
- Snelgrove, P.V.R.; Smith, C.R. A riot of species in an environmental calm: The paradox of the species-rich deep-sea floor. In Oceanography and Marine Biology; Gibson, R.N., Barnes, M., Atkinson, R.J.A., Eds.; Taylor & Francis Ltd.: London, UK, 2002; Volume 40, pp. 311–342. [Google Scholar]
- Levin, L.A.; Dayton, P.K. Ecological theory and continental margins: Where shallow meets deep. Trends Ecol. Evol. 2009, 24, 606–617. [Google Scholar] [CrossRef]
- Sarmiento, J.L.; Gruber, N. Sinks for anthropogenic carbon. Phys. Today 2002, 55, 30–36. [Google Scholar] [CrossRef]
- Harris, P.T.; Whiteway, T. Global distribution of large submarine canyons: Geomorphic differences between active and passive continental margins. Mar. Geol. 2011, 285, 69–86. [Google Scholar] [CrossRef]
- McClain, C.R.; Barry, J.P. Habitat heterogeneity, disturbance, and productivity work in concert to regulate biodiversity in deep submarine canyons. Ecology 2010, 91, 964–976. [Google Scholar] [CrossRef]
- Shank, T.M. Seamounts Deep-Ocean Laboratories of Faunal Connectivity, Evolution, and Endemism. Oceanography 2010, 23, 108–122. [Google Scholar] [CrossRef]
- Giere, O. Meiobenthology: The Microscopic Motile Fauna of Aquatic Sediments, 2nd ed.; Springer: Berlin, Germany, 2009. [Google Scholar]
- Ingels, J.; Tchesunov, A.V.; Vanreusel, A. Meiofauna in the Gollum Channels and the Whittard Canyon, Celtic Margin--how local environmental conditions shape nematode structure and function. PLoS ONE 2011, 6, e20094. [Google Scholar] [CrossRef]
- Ingels, J.; Vanreusel, A. The importance of different spatial scales in determining structural and functional characteristics of deep-sea infauna communities. Biogeosciences 2013, 10, 4547–4563. [Google Scholar] [CrossRef]
- Soltwedel, T. Metazoan meiobenthos along continental margins: A review. Prog. Oceanogr. 2000, 46, 59–84. [Google Scholar] [CrossRef]
- Soltwedel, T.; Grzelak, K.; Hasemann, C. Spatial and Temporal Variation in Deep-Sea Meiofauna at the LTER Observatory HAUSGARTEN in the Fram Strait (Arctic Ocean). Diversity 2020, 12, 279. [Google Scholar] [CrossRef]
- Ingels, J.; Vanreusel, A.; Brandt, A.; Catarino, A.I.; David, B.; De Ridder, C.; Dubois, P.; Gooday, A.J.; Martin, P.; Pasotti, F.; et al. Possible effects of global environmental changes on Antarctic benthos: A synthesis across five major taxa. Ecol. Evol. 2012, 2, 453–485. [Google Scholar] [CrossRef]
- Zeppilli, D.; Sarrazin, J.; Leduc, D.; Arbizu, P.M.; Fontaneto, D.; Fontanier, C.; Gooday, A.J.; Kristensen, R.M.; Ivanenko, V.N.; Sørensen, M.V.; et al. Is the meiofauna a good indicator for climate change and anthropogenic impacts? Mar. Biodivers. 2015, 45, 505–535. [Google Scholar] [CrossRef]
- Zeppilli, D.; Leduc, D.; Fontanier, C.; Fontaneto, D.; Fuchs, S.; Gooday, A.J.; Goineau, A.; Ingels, J.; Ivanenko, V.N.; Kristensen, R.M.; et al. Characteristics of meiofauna in extreme marine ecosystems: A review. Mar. Biodivers. 2018, 48, 35–71. [Google Scholar] [CrossRef]
- Danovaro, R.; Gambi, C.; Dell’Anno, A.; Corinaidesi, C.; Fraschetti, S.; Vanreusel, A.; Vincx, M.; Gooday, A.J. Exponential decline of deep-sea ecosystem functioning linked to benthic biodiversity loss. Curr. Biol. 2008, 18, 1–8. [Google Scholar] [CrossRef]
- Moens, T.; Braeckman, U.; Derycke, S.; Fonseca, G.; Gallucci, F.; Gingold, R.; Guilini, K.; Ingels, J.; Leduc, D.; Vanaverbeke, J.; et al. Ecology of free-living nematodes. In Handbook of Zoology; Schmidt-Rhaesa, A., Ed.; De Gruyter: Berlin, Germany, 2013; Volume 2, pp. 109–152. [Google Scholar]
- Schratzberger, M.; Ingels, J. Meiofauna matters: The roles of meiofauna in benthic ecosystems. J. Exp. Mar. Biol. Ecol. 2018, 502, 12–25. [Google Scholar] [CrossRef]
- Mokievsky, V.O.; Udalov, A.A.; Azovskii, A.I. Quantitative distribution of meiobenthos in deep-water zones of the World Ocean. Oceanology 2007, 47, 797–813. [Google Scholar] [CrossRef]
- Netto, S.A.; Gallucci, F.; Fonseca, G.F.C. Meiofauna communities of continental slope and deep-sea sites off SE Brazil. Deep Sea Res. Part I Oceanogr. Res. Pap. 2005, 52, 845–859. [Google Scholar] [CrossRef]
- Bernardino, A.F.; Berenguer, V.; Ribeiro-Ferreira, V.P. Bathymetric and regional changes in benthic macrofaunal assemblages on the deep Eastern Brazilian margin, SW Atlantic. Deep Sea Res. Part I Oceanogr. Res. Pap. 2016, 111, 110–120. [Google Scholar] [CrossRef]
- Almada, G.V.D.B.; Bernardino, A.F. Conservation of deep-sea ecosystems within offshore oil fields on the Brazilian margin, SW Atlantic. Biol. Conserv. 2017, 206, 92–101. [Google Scholar] [CrossRef]
- Mokievsky, V.; Azovsky, A. Re-evaluation of species diversity patterns of free-living marine nematodes. Mar. Ecol. Prog. Ser. 2002, 238, 101–108. [Google Scholar] [CrossRef]
- Vanreusel, A.; Fonseca, G.; Danovaro, R.; da Silva, M.C.; Esteves, A.M.; Ferrero, T.; Gad, G.; Galtsova, V.; Gambi, C.; Genevois, V.D.; et al. The contribution of deep-sea macrohabitat heterogeneity to global nematode diversity. Mar. Ecol. Evol. Perspect. 2010, 31, 6–20. [Google Scholar] [CrossRef]
- Ingels, J.; Billett, D.S.M.; Kiriakoulakis, K.; Wolff, G.A.; Vanreusel, A. Structural and functional diversity of Nematoda in relation with environmental variables in the Setúbal and Cascais canyons, Western Iberian Margin. Deep Sea Res. Part II Top. Stud. Oceanogr. 2011, 58, 2354–2368. [Google Scholar] [CrossRef]
- Vanaverbeke, J.; Steyaert, M.; Vanreusel, A.; Vincx, M. Nematode biomass spectra as descriptors of functional changes due to human and natural impact. Mar. Ecol. Prog. Ser. 2003, 249, 157–170. [Google Scholar] [CrossRef]
- Levin, L.A. Oxygen minimum zone benthos: Adaptation and community response to hypoxia. Oceanogr. Mar. Biol. 2003, 41, 1–45. [Google Scholar]
- Danovaro, R.; Carugati, L.; Corinaldesi, C.; Gambi, C.; Guilini, K.; Pusceddu, A.; Vanreusel, A. Multiple spatial scale analyses provide new clues on patterns and drivers of deep-sea nematode diversity. Deep Sea Res. Part II Top. Stud. Oceanogr. 2013, 92, 97–106. [Google Scholar] [CrossRef]
- Gambi, C.; Vanreusel, A.; Danovaro, R. Biodiversity of nematode assemblages from deep-sea sediments of the Atacama Slope and Trench (South Pacific Ocean). Deep Sea Res. Part I Oceanogr. Res. Pap. 2003, 50, 103–117. [Google Scholar] [CrossRef]
- Román, S.; Vanreusel, A.; Ingels, J.; Martin, D. Nematode community zonation in response to environmental drivers in Blanes Canyon (NW Mediterranean). J. Exp. Mar. Biol. Ecol. 2018, 502, 111–128. [Google Scholar] [CrossRef]
- Danovaro, R.; Gambi, C.; Lampadariou, N.; Tselepides, A. Deep-sea nematode biodiversity in the Mediterranean basin: Testing for longitudinal, bathymetric and energetic gradients. Ecography 2008, 31, 231–244. [Google Scholar] [CrossRef]
- Román, S.; Lins, L.; Ingels, J.; Romano, C.; Martin, D.; Vanreusel, A. Role of spatial scales and environmental drivers in shaping nematode communities in the Blanes Canyon and its adjacent slope. Deep Sea Res. Part I Oceanogr. Res. Pap. 2019, 146, 62–78. [Google Scholar] [CrossRef]
- Leduc, D.; Rowden, A.A.; Pilditch, C.A.; Maas, E.W.; Probert, P.K. Is there a link between deep-sea biodiversity and ecosystem function? Mar. Ecol. 2013, 34, 334–344. [Google Scholar] [CrossRef]
- Bianchelli, S.; Gambi, C.; Pusceddu, A.; Danovaro, R. Trophic conditions and meiofaunal assemblages in the Bari Canyon and the adjacent open slope (Adriatic Sea). Chem. Ecol. 2008, 24, 101–109. [Google Scholar] [CrossRef]
- Gambi, C.; Danovaro, R. Biodiversity and life strategies of deep-sea meiofauna and nematode assemblages in the Whittard Canyon (Celtic margin, NE Atlantic Ocean). Deep Sea Res. Part I Oceanogr. Res. Pap. 2016, 108, 13–22. [Google Scholar] [CrossRef]
- França, R.L.; Del Rey, A.C.; Tagliari, C.V.; Brandão, J.R.; Fontanelli, P.R. Bacia do Espírito Santo. Bol. Geociências Petrobras 2007, 15, 501–509. [Google Scholar]
- Boland, G.S.; Rowe, G.T. Deep-sea benthic sampling with the GOMEX box corer. Limnol. Oceanogr. 1991, 36, 1015–1020. [Google Scholar] [CrossRef]
- Somerfield, P.J.; Warwick, R.M. Meiofauna Techniques, Methods for the Study of Marine Benthos; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2013; pp. 253–284. [Google Scholar]
- Somerfield, P.J.; Warwick, R.M.; Moens, T. Meiofauna techniques. In Methods for the Study of Marine Benthos; Eleftheriou, E., McIntyre, A., Eds.; Blackwell Science Ltd.: Oxford, UK, 2005; pp. 229–272. [Google Scholar]
- De Grisse, A.T. Redescription ou modification de quelques techniques utilisées dans l’étude des nématodes phytoparasitaires. Meded. Rijks Fak. Landbowwetenschappen Gent 1969, 34, 351–369. [Google Scholar]
- Cobb, N.A. Notes on Nemas. Contrib. A Sci. Nematol. 1917, 5, 117–128. [Google Scholar]
- Platt, H.M.; Warwick, R.M. Free-Living Marine Nematodes. Part I. British Enoplids: Pictorial Key to World Genera and Notes for the Identification of British Species; Cambridge University Press: Cambridge, UK, 1983. [Google Scholar]
- Platt, H.M.; Warwick, R.M. Free-Living Marine Nematodes. Part II. British Chromadorids: Pictorial Key to World Genera and Notes for the Identification of British Species; Brill, E.J., Ed.; W. Backhuys: Leiden, The Netherlands, 1988. [Google Scholar]
- Warwick, R.M.; Platt, H.M.; Somerfield, P.J. Free-Living Marine Nematodes Part III. Monohysterids. Synopses of the British Fauna (New Series); Field Studies Council: Shrewsbury, MA, USA, 1998; Volume 53, p. 296. [Google Scholar]
- Guilini, K.; Bezerra, T.; Eisendle-Flöckner, U.; Deprez, T.; Fonseca, G.; Holovachov, O.; Leduc, D.; Miljutin, D.; Moens, T.; Sharma, J. NeMys: World Database of Free-Living Marine Nematodes. 2016. Available online: http://nemys.ugent.be (accessed on 24 June 2016).
- Wieser, W. Beziehungen zwischen Mundhöhlengestalt, Ernährungsweise und Vorkommen bei freilebenden marinen Nematoden. Ark. Zool. 1953, 2, 439–484. [Google Scholar]
- Bongers, T. The maturity index: An ecological measure of environmental disturbance based on nematode species composition. Oecologia 1990, 83, 14–19. [Google Scholar] [CrossRef]
- Bongers, T.; Alkemade, R.; Yeates, G.W. Interpretation of disturbance-induced maturity decrease in marine nematode assemblages by means of the maturity index. Mar. Ecol. Prog. Ser. 1991, 76, 135–142. [Google Scholar] [CrossRef]
- Bongers, T.; Ferris, H. Nematode community structure as a bioindicator in environmental monitoring. Trends Ecol. Evol. 1999, 14, 224–228. [Google Scholar] [CrossRef]
- Suguio, K. Introdução a Sedimentologia; Edgard Blucher/EDUSP: Sao Paulo, Brazil, 1973; p. 317. [Google Scholar]
- Hedges, J.L.; Stern, J.H. Carbon and nitrogen determination of carbonate-containing solids. Limnol. Oceanogr. 1984, 29, 657–663. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N. PRIMER v7: User Manual/Tutorial; PRIMER-E: Plymouth, UK, 2015. [Google Scholar]
- Heip, C.; Herman, P.; Soetaert, K. Indices of Diversity and Evenness. Oceanis 1998, 24, 61–87. [Google Scholar]
- Bongers, T. The Maturity Index, the evolution of nematode life history traits, adaptive radiation and cp-scaling. Plant Soil 1999, 212, 13–22. [Google Scholar] [CrossRef]
- Andrassy, I. The determination of volume and weight of nematodes. Acta Zool. Acad. Sci. Hung. 1956, 2, 1–15. [Google Scholar]
- Valentin, J. The Cabo Frio Upwelling System, Brazil, Coastal Marine Ecosystems of Latin America; Springer: Berlin, Germany, 2001; pp. 97–105. [Google Scholar]
- Schmid, C.; Schäfer, H.; Zenk, W.; Podestá, G. The Vitória eddy and its relation to the Brazil Current. J. Phys. Oceanogr. 1995, 25, 2532–2546. [Google Scholar] [CrossRef]
- De Léo, F.C.; Pires-Vanin, A.M.S. Benthic megafauna communities under the influence of the South Atlantic Central Water intrusion onto the Brazilian SE shelf: A comparison between an upwelling and a non-upwelling ecosystem. J. Mar. Syst. 2006, 60, 268–284. [Google Scholar] [CrossRef]
- Sumida, P.Y.G.; Yoshimaga, M.Y.; Ciotti, A.M.; Gaeta, S.A. Benthic response to upwelling events off the SE Brazilian coast. Mar. Ecol. Prog. Ser. 2005, 291, 35–42. [Google Scholar] [CrossRef]
- Quintana, C.O.; Bernardino, A.F.; de Moraes, P.C.; Valdemarsen, T.; Sumida, P.Y.G. Effects of coastal upwelling on the structure of macrofaunal communities in SE Brazil. J. Mar. Syst. 2015, 143, 120–129. [Google Scholar] [CrossRef]
- Carreira, R.S.; Araújo, M.P.; Costa, T.L.F.; Ansari, N.R.; Pires, L.C.M. Lipid biomarkers in deep sea sediments from the Campos Basin, SE Brazilian continental margin. Org. Geochem. 2010, 41, 879–884. [Google Scholar] [CrossRef]
- Yoshinaga, M.Y.; Sumida, P.Y.G.; Wakeham, S.G. Lipid biomarkers in surface sediments from an unusual coastal upwelling area from the SW Atlantic Ocean. Org. Geochem. 2008, 39, 1385–1399. [Google Scholar] [CrossRef]
- Mokievsky, V.O.; Udalov, A.A.; Azovsky, A.I. On the quantitative distribution of meiobenthos on the shelf of the world ocean. Oceanology 2004, 44, 99–109. [Google Scholar]
- Jensen, P. Measuring carbon content in nematodes. Helgol. Meeresunters. 1984, 38, 83–86. [Google Scholar]
- Bett, B.J.; Vanreusel, A.; Vincx, M.; Soltwedel, T.; Pfannkuche, O.; Lambshead, P.J.D.; Gooday, A.J.; Ferrero, T.; Dinet, A. Sampler Bias in the Quantitative Study of Deep-Sea Meiobenthos. Mar. Ecol. Prog. Ser. 1994, 104, 197–203. [Google Scholar] [CrossRef]
- Durrieu De Madron, X.; Weatherly, G. Circulation, transport and bottom boundary layers of the deep currents in the Brazil Basin. J. Mar. Res. 1994, 52, 583–638. [Google Scholar] [CrossRef]
- Stramma, L.; England, M. On the water masses and mean circulation of the South Atlantic Ocean. J. Geophys. Res. Oceans 1999, 104, 20863–20883. [Google Scholar] [CrossRef]
- Tietjen, J.H. Ecology and distribution of deep-sea meiobenthos off North-Carolina. Deep Sea Res. 1971, 18, 941–957. [Google Scholar] [CrossRef]
- Leduc, D.; Rowden, A.A.; Bowden, D.A.; Probert, P.K.; Pilditch, C.A.; Nodder, S.D. Unimodal relationship between biomass and species richness of deep-sea nematodes: Implications for the link between productivity and diversity. Mar. Ecol. Prog. Ser. 2012, 454, 53–64. [Google Scholar] [CrossRef]
- Jensen, P. Nematode Assemblages in the Deep-Sea Benthos of the Norwegian Sea. Deep Sea Res. 1988, 35, 1173–1184. [Google Scholar] [CrossRef]
- Dinet, A.; Vivier, M.H. Le meiobenthos abyssal du Golfe de Gascogne II. Les peuplements de nématodes et leur diversité spécifique. Cah. Biol. Mar. 1979, 18, 109–123. [Google Scholar]
- Lambshead, P.J.D.; Tietjen, J.; Ferrero, T.; Jensen, P. Latitudinal diversity gradients in the deep sea with special reference to North Atlantic nematodes. Mar. Ecol. Prog. Ser. 2000, 194, 159–167. [Google Scholar] [CrossRef]
- Grzelak, K.; Kotwicki, L.; Hasemann, C.; Soltwedel, T. Bathymetric patterns in standing stock and diversity of deep-sea nematodes at the long-term ecological research observatory HAUSGARTEN (Fram Strait). J. Mar. Syst. 2017, 172, 160–177. [Google Scholar] [CrossRef]
- Danovaro, R.; Fabiano, M.; Dellacroce, N. Labile organic matter and microbial biomass in deep-sea sediments (Eastern Mediterranean Sea). Deep Sea Res. Part I Oceanogr. Res. Pap. 1993, 40, 953–965. [Google Scholar] [CrossRef]
- Clark, M.R.; Bowden, D.A. Seamount biodiversity: High variability both within and between seamounts in the Ross Sea region of Antarctica. Hydrobiologia 2015, 761, 161–180. [Google Scholar] [CrossRef]
- Zeppilli, D.; Bongiorni, L.; Cattaneo, A.; Danovaro, R.; Santos, R.S. Meiofauna assemblages of the Condor Seamount (North-East Atlantic Ocean) and adjacent deep-sea sediments. Deep Sea Res. Part II Top. Stud. Oceanogr. 2013, 98, 87–100. [Google Scholar] [CrossRef]
- Zeppilli, D.; Bongiorni, L.; Serrão Santos, R.; Vanreusel, A. Changes in Nematode Communities in Different Physiographic Sites of the Condor Seamount (North-East Atlantic Ocean) and Adjacent Sediments. PLoS ONE 2014, 9, e115601. [Google Scholar] [CrossRef]
- Sanders, H.L. Benthic marine diversity and stability-time hypothesis. Brookhaven Symp. Biol. 1969, 22, 71–80. [Google Scholar]
- Grassle, J.F.; Sanders, H.L. Life histories and role of disturbance. Deep Sea Res. 1973, 20, 643–659. [Google Scholar]
- Soetaert, K.; Heip, C. Nematode assemblages of deep-sea and shelf break sites in the North-Atlantic and Mediterranean-Sea. Mar. Ecol. Prog. Ser. 1995, 125, 171–183. [Google Scholar] [CrossRef]
- Sanders, H.L. Marine benthic diversity: A comparative study. Am. Nat. 1968, 102, 243–282. [Google Scholar] [CrossRef]
- Fonseca, G.; Soltwedel, T. Deep-sea meiobenthic communities underneath the marginal ice zone off Eastern Greenland. Polar Biol. 2007, 30, 607–618. [Google Scholar] [CrossRef]
- Lambshead, P.J.D.; Brown, C.J.; Ferrero, T.J.; Mitchell, N.J.; Smith, C.R.; Hawkins, L.E.; Tietjen, J. Latitudinal diversity patterns of deep-sea marine nematodes and organic fluxes—A test from the central equatorial Pacific. Mar. Ecol. Prog. Ser. 2002, 236, 129–135. [Google Scholar] [CrossRef]
- Danovaro, R.; Croce, N.D.; Eleftheriou, A.; Fabiano, M.; Papadopoulou, N.; Smith, C.; Tselepides, A. Meiofauna of the deep Eastern Mediterranean Sea: Distribution and abundance in relation to bacterial biomass, organic matter composition and other environmental factors. Prog. Oceanogr. 1995, 36, 329–341. [Google Scholar] [CrossRef]
- Danovaro, R.; Dinet, A.; Duineveld, G.; Tselepides, A. Benthic response to particulate fluxes in different trophic environments: A comparison between the Gulf of Lions-Catalan Sea (western-Mediterranean) and the Cretan Sea (eastern-Mediterranean). Prog. Oceanogr. 1999, 44, 287–312. [Google Scholar] [CrossRef]

| Campaign | Water Depth | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 400 | 1000 | 1900 | 2500 | 3000 | |||||||
| SD | SD | SD | SD | SD | |||||||
| Average grain size | 1 | 5.64 | 0.82 | 6.05 | 0.38 | 5.78 | 0.74 | 4.66 | 0.71 | 4.59 | 0.46 |
| 2 | 6.02 | 0.73 | 6.62 | 0.84 | 5.74 | 0.59 | 4.96 | 0.69 | 5.07 | 0.50 | |
| Gravel | 1 | 0.000 | 0.001 | 0.001 | 0.001 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 | 0.000 |
| 2 | 0.001 | 0.001 | 0.000 | 0.000 | 0.002 | 0.001 | 0.002 | 0.001 | 0.000 | 0.000 | |
| Very coarse sand | 1 | 0.002 | 0.002 | 0.001 | 0.000 | 0.002 | 0.000 | 0.002 | 0.001 | 0.002 | 0.001 |
| 2 | 0.003 | 0.003 | 0.002 | 0.001 | 0.003 | 0.001 | 0.005 | 0.002 | 0.002 | 0.001 | |
| Coarse sand | 1 | 0.005 | 0.007 | 0.003 | 0.001 | 0.009 | 0.002 | 0.020 | 0.007 | 0.019 | 0.009 |
| 2 | 0.009 | 0.009 | 0.006 | 0.002 | 0.010 | 0.002 | 0.028 | 0.017 | 0.027 | 0.015 | |
| Medium sand | 1 | 0.022 | 0.034 | 0.011 | 0.004 | 0.030 | 0.003 | 0.102 | 0.045 | 0.086 | 0.042 |
| 2 | 0.030 | 0.036 | 0.022 | 0.007 | 0.033 | 0.009 | 0.126 | 0.094 | 0.130 | 0.080 | |
| Fine sand | 1 | 0.087 | 0.097 | 0.018 | 0.009 | 0.042 | 0.005 | 0.134 | 0.043 | 0.115 | 0.037 |
| 2 | 0.082 | 0.087 | 0.035 | 0.016 | 0.045 | 0.018 | 0.141 | 0.073 | 0.147 | 0.091 | |
| Very fine sand | 1 | 0.110 | 0.080 | 0.034 | 0.022 | 0.038 | 0.008 | 0.107 | 0.041 | 0.108 | 0.020 |
| 2 | 0.103 | 0.062 | 0.048 | 0.025 | 0.040 | 0.015 | 0.108 | 0.063 | 0.101 | 0.044 | |
| Total sand | 1 | 0.226 | 0.194 | 0.067 | 0.033 | 0.122 | 0.016 | 0.366 | 0.109 | 0.331 | 0.103 |
| 2 | 0.226 | 0.178 | 0.113 | 0.046 | 0.131 | 0.041 | 0.409 | 0.218 | 0.406 | 0.228 | |
| Silt | 1 | 0.503 | 0.244 | 0.623 | 0.104 | 0.692 | 0.052 | 0.422 | 0.175 | 0.487 | 0.110 |
| 2 | 0.520 | 0.180 | 0.624 | 0.047 | 0.628 | 0.137 | 0.417 | 0.203 | 0.443 | 0.287 | |
| Clay | 1 | 0.270 | 0.055 | 0.310 | 0.128 | 0.186 | 0.056 | 0.213 | 0.090 | 0.182 | 0.019 |
| 2 | 0.253 | 0.113 | 0.263 | 0.050 | 0.240 | 0.174 | 0.173 | 0.047 | 0.150 | 0.071 | |
| Total organic matter | 1 | 1.323 | 0.325 | 1.303 | 0.448 | 1.14 | 0.474 | 0.679 | 0.272 | 0.895 | 0.257 |
| 2 | 1.448 | 0.408 | 1.608 | 0.465 | 1.346 | 0.174 | 0.271 | 0.271 | 1.022 | 0.216 | |
| Carbonate | 1 | 35.105 | 22.599 | 19.098 | 10.739 | 52.818 | 3.011 | 53.410 | 16.030 | 47.975 | 41.548 |
| 2 | 37.711 | 10.944 | 31.807 | 8.154 | 66.417 | 19.607 | 56.499 | 5.590 | 73.227 | 19.217 | |
| Salinity | 1 | 34.978 | 0.064 | 34.384 | 0.022 | 34.939 | 0.005 | 34.792 | 0.237 | 34.881 | 0.0143 |
| 2 | 34.980 | 0.060 | 34.389 | 0.018 | 34.937 | 0.006 | 34.912 | 0.006 | 34.892 | 0.004 | |
| Temperature | 1 | 11.362 | 0.450 | 3.885 | 0.119 | 3.763 | 0.107 | 2.713 | 0.117 | 2.433 | 0.058 |
| 2 | 11.292 | 0.533 | 3.810 | 0.171 | 3.788 | 0.031 | 2.957 | 0.288 | 2.300 | 0.122 | |
| Water Depth (m) | GR | EG(51) | H’ | N1 | N2 | Ninf | TDI | MI | Density | Biomass |
|---|---|---|---|---|---|---|---|---|---|---|
| 400 | 45.27 | 20.90 | 3.01 | 21.08 | 12.11 | 5.04 | 0.62 | 2.55 | 3406 | 4755.12 |
| 1000 | 47.00 | 21.00 | 3.06 | 21.87 | 12.84 | 5.19 | 0.66 | 2.55 | 2542 | 3801.94 |
| 1900 | 40.87 | 22.42 | 3.12 | 23.16 | 14.74 | 6.19 | 0.70 | 2.72 | 318 | 467.37 |
| 2500 | 36.67 | 21.62 | 3.10 | 22.40 | 15.56 | 6.90 | 0.66 | 2.76 | 286 | 385.42 |
| 3000 | 37.52 | 22.44 | 3.12 | 23.12 | 15.75 | 6.97 | 0.69 | 2.77 | 236 | 330.63 |
| Genus Richness | df | SS | MS | Pseudo-F | P(perm) | Perms | √ECV | ECV | ECV% | PERMDISP |
| WD | 4 | 2.42 × 103 | 6.05 × 102 | 15.555 | 0.0001 | 9953 | 4.4079 | 19.43 | 23.7 | 0.891 |
| TR | 4 | 1.30 × 102 | 3.25 × 101 | 0.83502 | 0.5041 | 9948 | - | 0.655 | ||
| SE | 1 | 3.98 × 102 | 3.98 × 102 | 10.227 | 0.0018 | 9841 | 2.2209 | 4.9325 | 6.0 | 0.759 |
| WD × TR | 16 | 1.22 × 103 | 7.63 × 101 | 1.9598 | 0.0230 | 9917 | 2.5279 | 6.3905 | 7.8 | |
| WD × SE | 4 | 1.04 × 102 | 2.59 × 101 | 0.66503 | 0.6184 | 9942 | - | |||
| TR × SE | 4 | 2.61 × 102 | 6.53 × 101 | 1.6768 | 0.1628 | 9947 | 1.3443 | 1.807 | 2.2 | |
| WD × TR × SE | 16 | 1.11 × 103 | 6.93 × 101 | 1.7814 | 0.0447 | 9918 | 3.2257 | 10.405 | 12.7 | |
| Residual | 97 | 3.78 × 103 | 3.89 × 101 | 6.2389 | 38.924 | 47.5 | ||||
| Total | 146 | 9.50 × 103 | ||||||||
| EG(51) | ||||||||||
| WD | 4 | 6.41 × 101 | 1.60 × 101 | 3.9089 | 0.0046 | 9951 | 0.64 | 0.41 | 6.4 | 0.083 |
| TR | 4 | 5.54 × 101 | 1.38 × 101 | 3.3761 | 0.0118 | 9945 | 0.58 | 0.33 | 5.2 | 0.456 |
| SE | 1 | 2.73 × 101 | 2.73 × 101 | 6.6496 | 0.0119 | 9838 | 0.56 | 0.32 | 5.0 | 0.458 |
| WD × TR | 16 | 1.09 × 102 | 6.84 | 1.6683 | 0.0639 | 9910 | 0.68 | 0.47 | 7.3 | |
| WD × SE | 4 | 2.23 × 101 | 5.57 | 1.3588 | 0.2527 | 9935 | 0.32 | 0.10 | 1.6 | |
| TR × SE | 4 | 1.26 × 101 | 3.16 | 0.77126 | 0.5433 | 9955 | ||||
| WD × TR × SE | 16 | 9.72 × 101 | 6.08 | 1.4824 | 0.1234 | 9926 | 0.82 | 0.68 | 10.6 | |
| Residual | 97 | 3.98 × 102 | 4.10 | 2.02 | 4.10 | 64.0 | ||||
| Total | 146 | 7.92 × 102 | ||||||||
| H’ | ||||||||||
| WD | 4 | 2.40 × 10−1 | 5.99 × 10−2 | 1.4822 | 0.2128 | 9944 | 0.03 | 0.00 | 1.1 | 0.239 |
| TR | 4 | 3.84 × 10−1 | 9.60 × 10−2 | 2.3763 | 0.0557 | 9953 | 0.04 | 0.00 | 3.3 | 0.751 |
| SE | 1 | 2.20 × 10−1 | 2.20 × 10−1 | 5.4377 | 0.0235 | 9829 | 0.05 | 0.00 | 4.2 | 0.879 |
| WD × TR | 16 | 1.07 | 6.71 × 10−2 | 1.66 | 0.0670 | 9913 | 0.07 | 0.00 | 7.8 | |
| WD × SE | 4 | 1.50 × 10−1 | 3.74 × 10−2 | 0.92655 | 0.4523 | 9954 | ||||
| TR × SE | 4 | 1.12 × 10−1 | 2.80 × 10−2 | 0.6927 | 0.5948 | 9938 | ||||
| WD × TR × SE | 16 | 1.03 | 6.41 × 10−2 | 1.587 | 0.0913 | 9924 | 0.09 | 0.01 | 14.0 | |
| Residual | 97 | 3.92 | 4.04 × 10−2 | 0.20 | 0.04 | 69.5 | ||||
| Total | 146 | 7.17 | ||||||||
| N1 | df | SS | MS | Pseudo-F | P(perm) | Perms | √ECV | ECV | ECV% | PERMDISP |
| WD | 4 | 9.14 × 101 | 2.28 × 101 | 1.348 | 0.2595 | 9953 | 0.44967 | 0.20 | 0.8 | 0.314 |
| TR | 4 | 1.84 × 102 | 4.59 × 101 | 2.7105 | 0.0355 | 9948 | 0.99693 | 0.99 | 3.9 | 0.727 |
| SE | 1 | 1.21 × 102 | 1.21 × 102 | 7.1471 | 0.0084 | 9848 | 1.196 | 1.43 | 5.6 | 0.495 |
| WD × TR | 16 | 4.33 × 102 | 2.70 × 101 | 1.5958 | 0.0875 | 9910 | 1.314 | 1.73 | 6.7 | |
| WD × SE | 4 | 8.36 × 101 | 2.09 × 101 | 1.234 | 0.3035 | 9947 | 0.52145 | 0.27 | 1.1 | |
| TR × SE | 4 | 5.95 × 101 | 1.49 × 101 | 0.87742 | 0.4845 | 9955 | ||||
| WD × TR × SE | 16 | 4.66 × 102 | 2.92 × 101 | 1.7208 | 0.0533 | 9908 | 2.0439 | 4.18 | 16.2 | |
| Residual | 97 | 1.64 × 103 | 1.69 × 101 | 4.1162 | 16.943 | 65.8 | ||||
| Total | 146 | 3.10 × 103 | ||||||||
| N2 | ||||||||||
| WD | 4 | 3.07 × 102 | 7.67 × 101 | 5.9462 | 0.0002 | 9952 | 1.4791 | 2.19 | 9.9 | 0.089 |
| TR | 4 | 1.76 × 102 | 4.39 × 101 | 3.4023 | 0.0117 | 9954 | 1.0308 | 1.06 | 4.8 | 0.518 |
| SE | 1 | 5.80 × 101 | 5.80 × 101 | 4.494 | 0.0389 | 9840 | 0.78669 | 0.62 | 2.8 | 0.367 |
| WD × TR | 16 | 4.41 × 102 | 2.76 × 101 | 2.1385 | 0.0114 | 9909 | 1.5848 | 2.51 | 11.4 | |
| WD × SE | 4 | 6.23 × 101 | 1.56 × 101 | 1.2084 | 0.3128 | 9953 | 0.42934 | 0.18 | 0.8 | |
| TR × SE | 4 | 3.07 × 101 | 7.68 | 0.59574 | 0.6695 | 9957 | ||||
| WD × TR × SE | 16 | 3.27 × 102 | 2.05 × 101 | 1.586 | 0.0868 | 9926 | 1.608 | 2.59 | 11.7 | |
| Residual | 97 | 1.25 × 103 | 1.29 × 101 | 3.5914 | 12.90 | 58.5 | ||||
| Total | 146 | 2.67 × 103 | ||||||||
| Ninf | ||||||||||
| WD | 4 | 9.45 × 101 | 2.36 × 101 | 8.0934 | 0.0001 | 9960 | 0.84253 | 0.71 | 13.8 | 0.103 |
| TR | 4 | 2.88 × 101 | 7.20 | 2.4667 | 0.0506 | 9931 | 0.38311 | 0.15 | 2.8 | 0.099 |
| SE | 1 | 3.08 | 3.08 | 1.0545 | 0.3045 | 9826 | 0.046741 | 0.0022 | 0.0 | 0.536 |
| WD × TR | 16 | 1.01 × 102 | 6.32 | 2.166 | 0.0112 | 9896 | 0.76285 | 0.58 | 11.3 | |
| WD × SE | 4 | 1.98 × 101 | 4.95 | 1.6952 | 0.1579 | 9948 | 0.37301 | 0.14 | 2.7 | |
| TR × SE | 4 | 5.02 | 1.26 | 0.43039 | 0.7853 | 9952 | ||||
| WD × TR × SE | 16 | 7.73 | 4.83 | 1.6552 | 0.0733 | 9925 | 0.8087 | 0.65 | 12.7 | |
| Residual | 97 | 2.83 × 102 | 2.92 | 1.7082 | 2.92 | 56.6 | ||||
| Total | 146 | 6.17 × 102 | ||||||||
| Community | df | SS | MS | Pseudo-F | P(perm) | Perms | √ECV | ECV | ECV% | PERMDISP |
| WD | 4 | 1.10 × 105 | 2.75 × 104 | 23.008 | 0.0001 | 9890 | 30.06 | 903.58 | 35.8 | 0.001 |
| TR | 4 | 1.22 × 104 | 3.04 × 103 | 2.538 | 0.0001 | 9745 | 7.9461 | 63.14 | 2.5 | 0.001 |
| SE | 1 | 4.24 × 103 | 4.24 × 103 | 3.544 | 0.0001 | 9877 | 6.4675 | 41.829 | 1.7 | 0.758 |
| WD × TR | 16 | 3.79 × 104 | 2.37 × 103 | 1.978 | 0.0001 | 9607 | 14.151 | 200.25 | 7.9 | |
| WD × SE | 4 | 7.73 × 103 | 1.93 × 103 | 1.614 | 0.0001 | 9770 | 7.1013 | 50.429 | 2.0 | |
| TR × SE | 4 | 6.55 × 103 | 1.64 × 103 | 1.368 | 0.0044 | 9768 | 5.4943 | 30.187 | 1.2 | |
| WD × TR × SE | 16 | 2.09 × 104 | 1.31 × 103 | 1.091 | 0.0864 | 9576 | 6.1016 | 37.23 | 1.5 | |
| Residual | 97 | 1.16 × 105 | 1.20 × 103 | 34.6 | 1197.2 | 47.4 | ||||
| Total | 146 | 3.16 × 105 | ||||||||
| Density | ||||||||||
| WD | 4 | 2.72 × 108 | 6.79 × 107 | 74.399 | 0.0001 | 9952 | 1515.70 | 2297500.00 | 53.5 | 0.001 |
| TR | 4 | 2.14 × 107 | 5.36 × 106 | 5.8736 | 0.0002 | 9935 | 390.58 | 152550.00 | 3.6 | 0.003 |
| SE | 1 | 3.68 × 106 | 3.68 × 106 | 4.0337 | 0.0439 | 9849 | 195.00 | 38025.00 | 0.9 | 0.296 |
| WD × TR | 16 | 8.54 × 107 | 5.34 × 106 | 5.8485 | 0.0001 | 9928 | 870.01 | 756910.00 | 17.6 | |
| WD × SE | 4 | 8.76 × 106 | 2.19 × 106 | 2.3993 | 0.0462 | 9956 | 295.97 | 87598.00 | 2.0 | |
| TR × SE | 4 | 6.38 × 106 | 1.60 × 106 | 1.7485 | 0.1429 | 9953 | 216.46 | 46856.00 | 1.1 | |
| WD × TR × SE | 16 | 1.09 × 107 | 6.81 × 105 | 0.74603 | 0.7592 | 9921 | ||||
| Residual | 97 | 8.85 × 107 | 9.13 × 105 | 955.35 | 912690.00 | 21.3 | ||||
| Total | 146 | 4.93 × 108 | ||||||||
| Biomass | ||||||||||
| WD | 4 | 5.52 × 108 | 1.38 × 108 | 69.193 | 0.0001 | 9957 | 2158.7 | 4.66 × 106 | 56.7 | 0.001 |
| TR | 4 | 3.82 × 107 | 9.55 × 106 | 4.7935 | 0.0012 | 9956 | 509.15 | 2.59 × 105 | 3.2 | 0.0055 |
| SE | 1 | 1.62 × 107 | 1.62 × 107 | 8.112 | 0.0046 | 9835 | 441.16 | 1.95 × 105 | 2.4 | 0.049 |
| WD × TR | 16 | 1.07 × 108 | 6.70 × 106 | 3.3614 | 0.0003 | 9893 | 897.12 | 8.05 × 105 | 9.8 | |
| WD × SE | 4 | 2.55 × 107 | 6.38 × 106 | 3.2014 | 0.0155 | 9953 | 548.52 | 3.01 × 105 | 3.7 | |
| TR × SE | 4 | 5.37 × 106 | 1.34 × 106 | 0.67401 | 0.6193 | 9936 | ||||
| WD × TR × SE | 16 | 2.18 × 107 | 1.36 × 106 | 0.68245 | 0.8083 | 9918 | ||||
| Residual | 97 | 1.93 × 108 | 1.99 × 106 | 1411.6 | 1.99 × 106 | 24.3 | ||||
| Total | 146 | 9.55 × 108 | ||||||||
| TDI | df | SS | MS | Pseudo-F | P(perm) | Perms | √ECV | ECV | ECV% | PERMDISP |
| WD | 4 | 0.094438 | 0.023609 | 11.582 | 0.0001 | 9950 | 0.027199 | 7.40 × 10−4 | 20.0 | 0.0082 |
| TR | 4 | 0.0081714 | 0.0020429 | 1.0021 | 0.4104 | 9949 | 0.00038559 | 1.49 × 10−7 | 0.0 | 0.245 |
| SE | 1 | 0.012813 | 0.012813 | 6.2853 | 0.0132 | 9851 | 0.012164 | 1.48 × 10−4 | 4.0 | 0.3828 |
| WD × TR | 16 | 0.068936 | 0.0043085 | 2.1135 | 0.0132 | 9916 | 0.019704 | 3.88 × 10−4 | 10.5 | |
| WD × SE | 4 | 0.0013595 | 0.00033987 | 0.16672 | 0.9557 | 9952 | ||||
| TR × SE | 4 | 0.0055643 | 0.0013911 | 0.6824 | 0.6011 | 9953 | ||||
| WD × TR × SE | 16 | 0.050779 | 0.0031737 | 1.5569 | 0.1002 | 9926 | 0.019706 | 3.88 × 10−4 | 10.5 | |
| Residual | 97 | 0.19774 | 0.0020385 | 0.04515 | 2.04 × 10−3 | 55.0 | ||||
| Total | 146 | 0.44222 | ||||||||
| MI | ||||||||||
| WD | 4 | 1.4123 | 0.35308 | 24.576 | 0.0001 | 9963 | 0.10778 | 1.16 × 10−2 | 34.4 | 0.0026 |
| TR | 4 | 0.18301 | 0.045752 | 3.1846 | 0.0166 | 9960 | 0.032808 | 1.08 × 10−3 | 3.2 | 0.0501 |
| SE | 1 | 0.20478 | 0.20478 | 14.254 | 0.0007 | 9839 | 0.051137 | 2.62 × 10−3 | 7.8 | 0.0163 |
| WD × TR | 16 | 0.60817 | 0.03801 | 2.6457 | 0.0025 | 9921 | 0.063594 | 4.04 × 10−3 | 12.0 | |
| WD × SE | 4 | 0.038332 | 0.0095831 | 0.66704 | 0.6119 | 9949 | ||||
| TR × SE | 4 | 0.050984 | 0.012746 | 0.88719 | 0.4733 | 9948 | ||||
| WD × TR × SE | 16 | 0.19922 | 0.012451 | 0.86668 | 0.6099 | 9938 | ||||
| Residual | 97 | 1.3936 | 0.014367 | 0.11986 | 1.44 × 10−2 | 42.6 | ||||
| Total | 146 | 4.1575 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
dos Santos, G.A.P.; Silva, A.C.; Esteves, A.M.; Ribeiro-Ferreira, V.P.; Neres, P.F.; Valdes, Y.; Ingels, J. Testing Bathymetric and Regional Patterns in the Southwest Atlantic Deep Sea Using Infaunal Diversity, Structure, and Function. Diversity 2020, 12, 485. https://doi.org/10.3390/d12120485
dos Santos GAP, Silva AC, Esteves AM, Ribeiro-Ferreira VP, Neres PF, Valdes Y, Ingels J. Testing Bathymetric and Regional Patterns in the Southwest Atlantic Deep Sea Using Infaunal Diversity, Structure, and Function. Diversity. 2020; 12(12):485. https://doi.org/10.3390/d12120485
Chicago/Turabian Styledos Santos, Giovanni A. P., Alexsandra C. Silva, André M. Esteves, Venina P. Ribeiro-Ferreira, Patricia. F. Neres, Yirina Valdes, and Jeroen Ingels. 2020. "Testing Bathymetric and Regional Patterns in the Southwest Atlantic Deep Sea Using Infaunal Diversity, Structure, and Function" Diversity 12, no. 12: 485. https://doi.org/10.3390/d12120485
APA Styledos Santos, G. A. P., Silva, A. C., Esteves, A. M., Ribeiro-Ferreira, V. P., Neres, P. F., Valdes, Y., & Ingels, J. (2020). Testing Bathymetric and Regional Patterns in the Southwest Atlantic Deep Sea Using Infaunal Diversity, Structure, and Function. Diversity, 12(12), 485. https://doi.org/10.3390/d12120485





