Snapshot of Macroalgae and Fish Assemblages in Temperate Reefs in the Southern European Atlantic Ecoregion
Abstract
1. Introduction
2. Materials and Methods
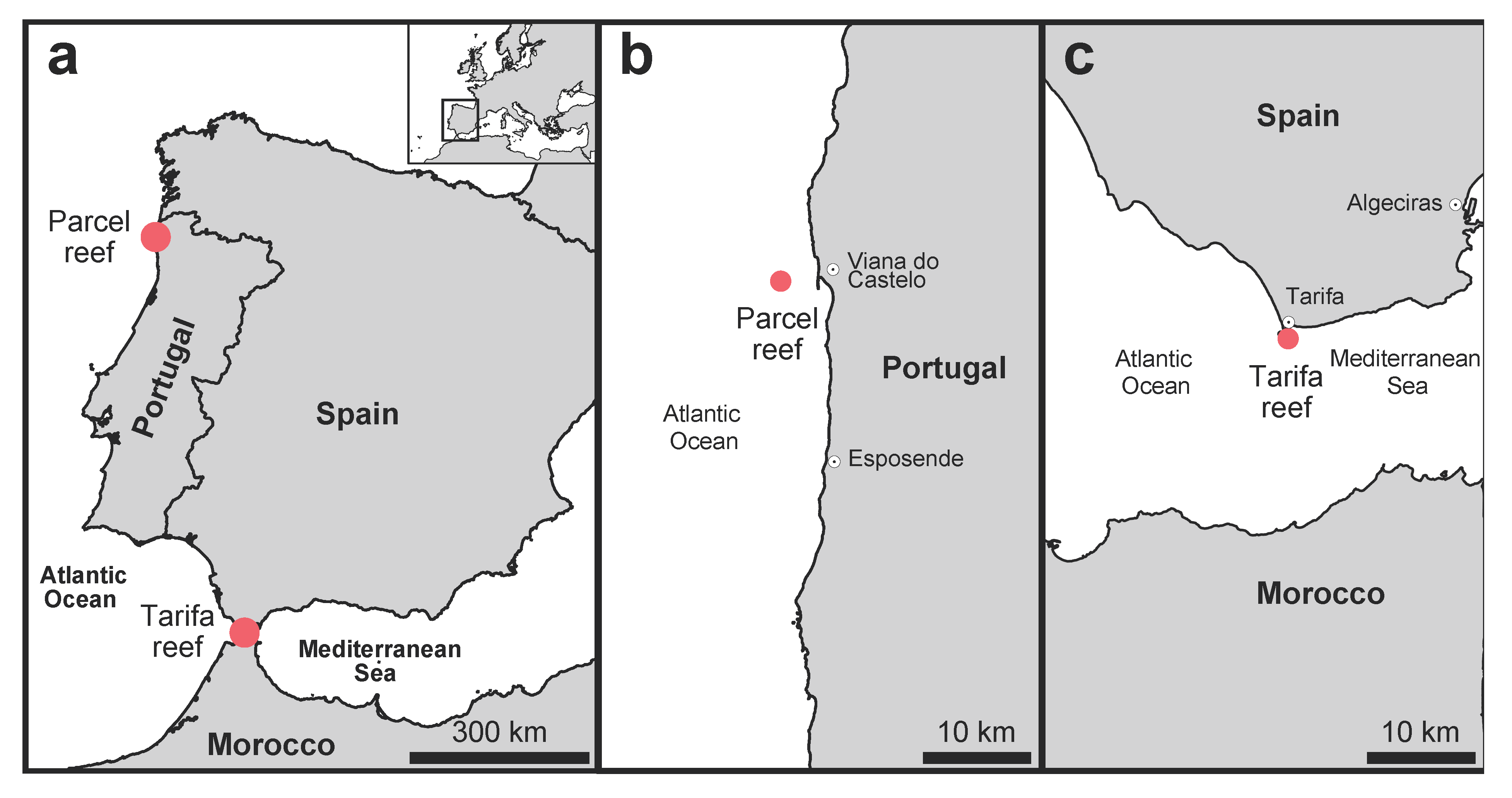
2.1. Study Locations and Abiotic Characterization
2.2. Community Data: Ichthyofauna and Algae Survey
3. Results
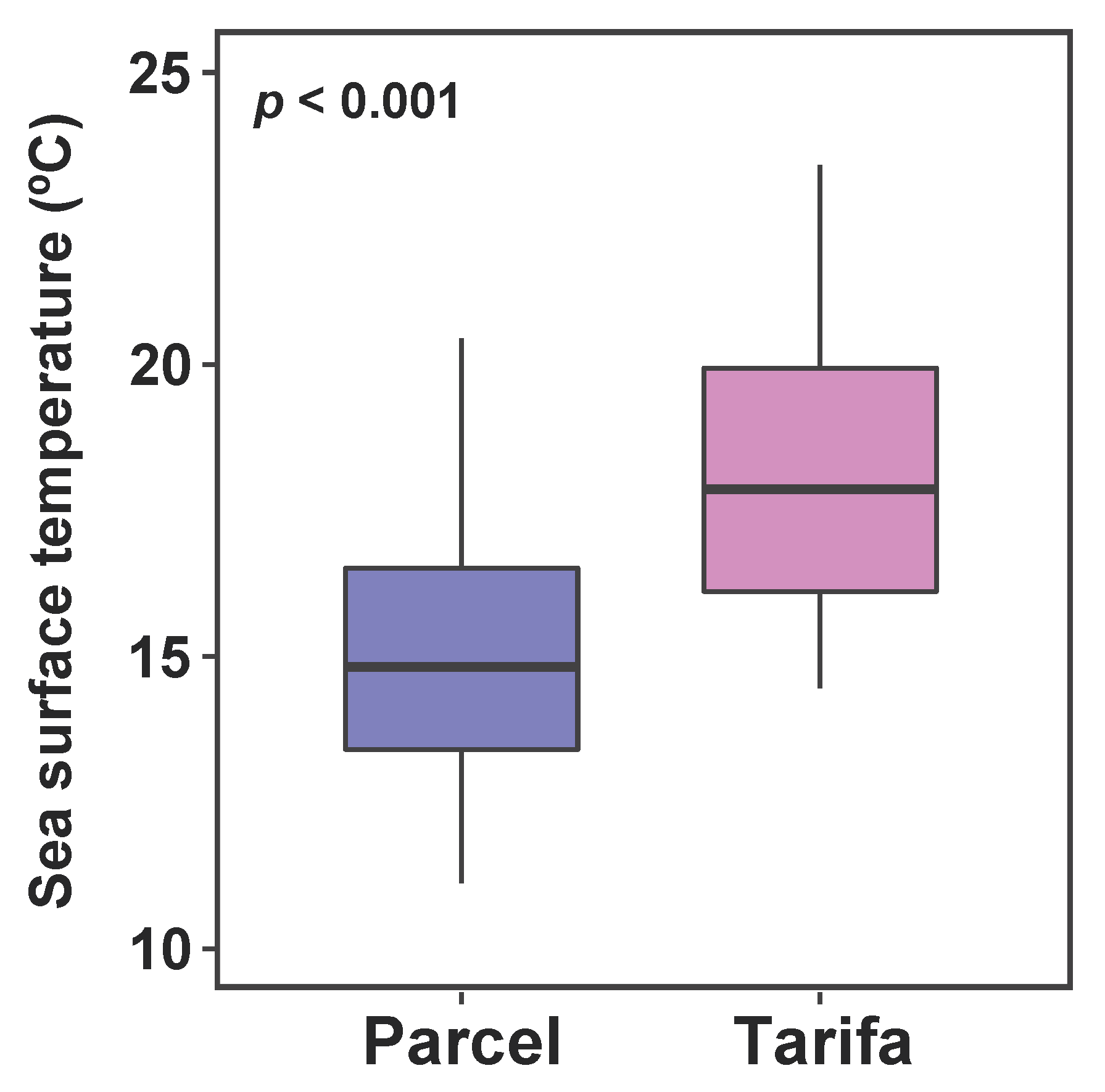
3.1. Sea Surface Temperature
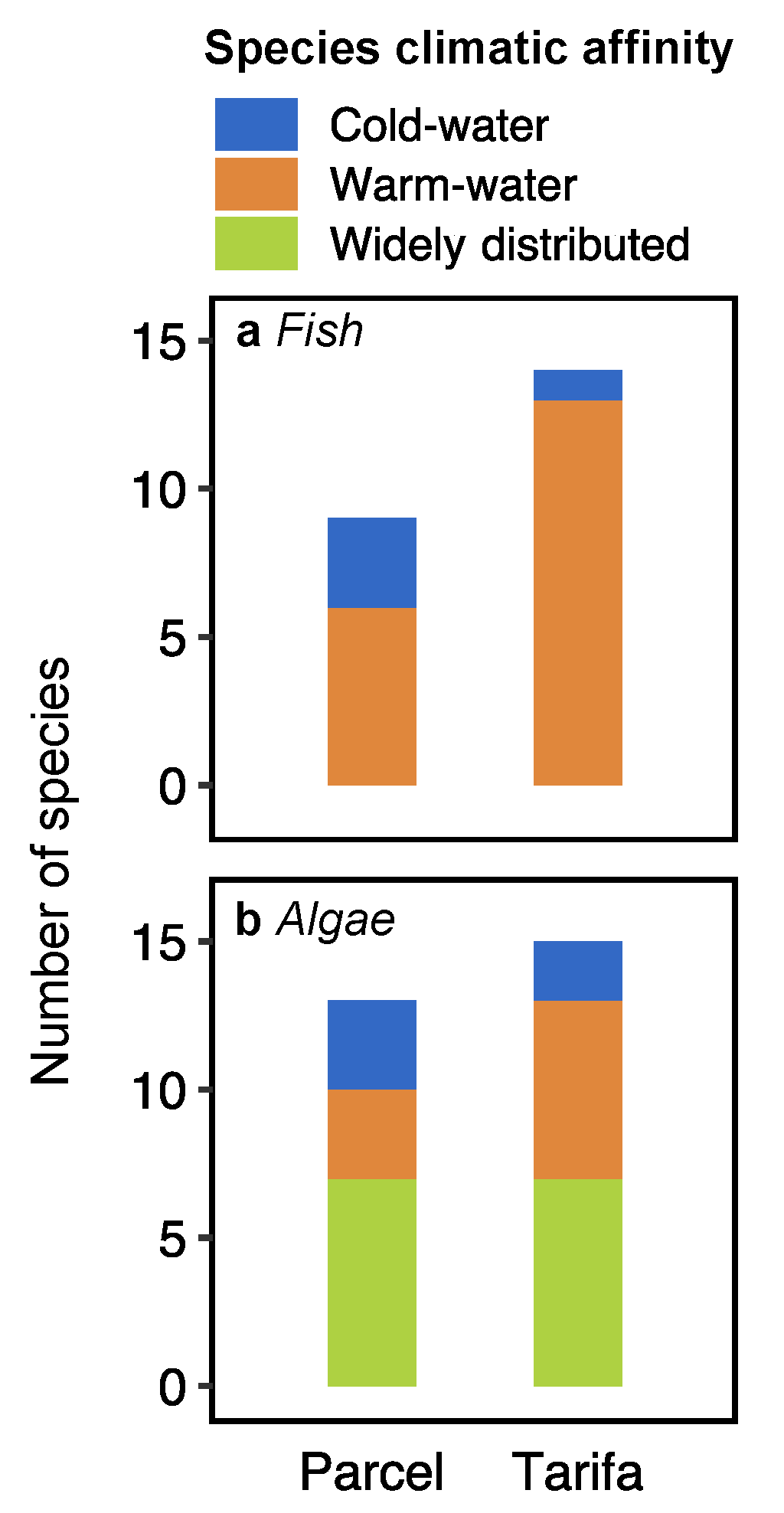
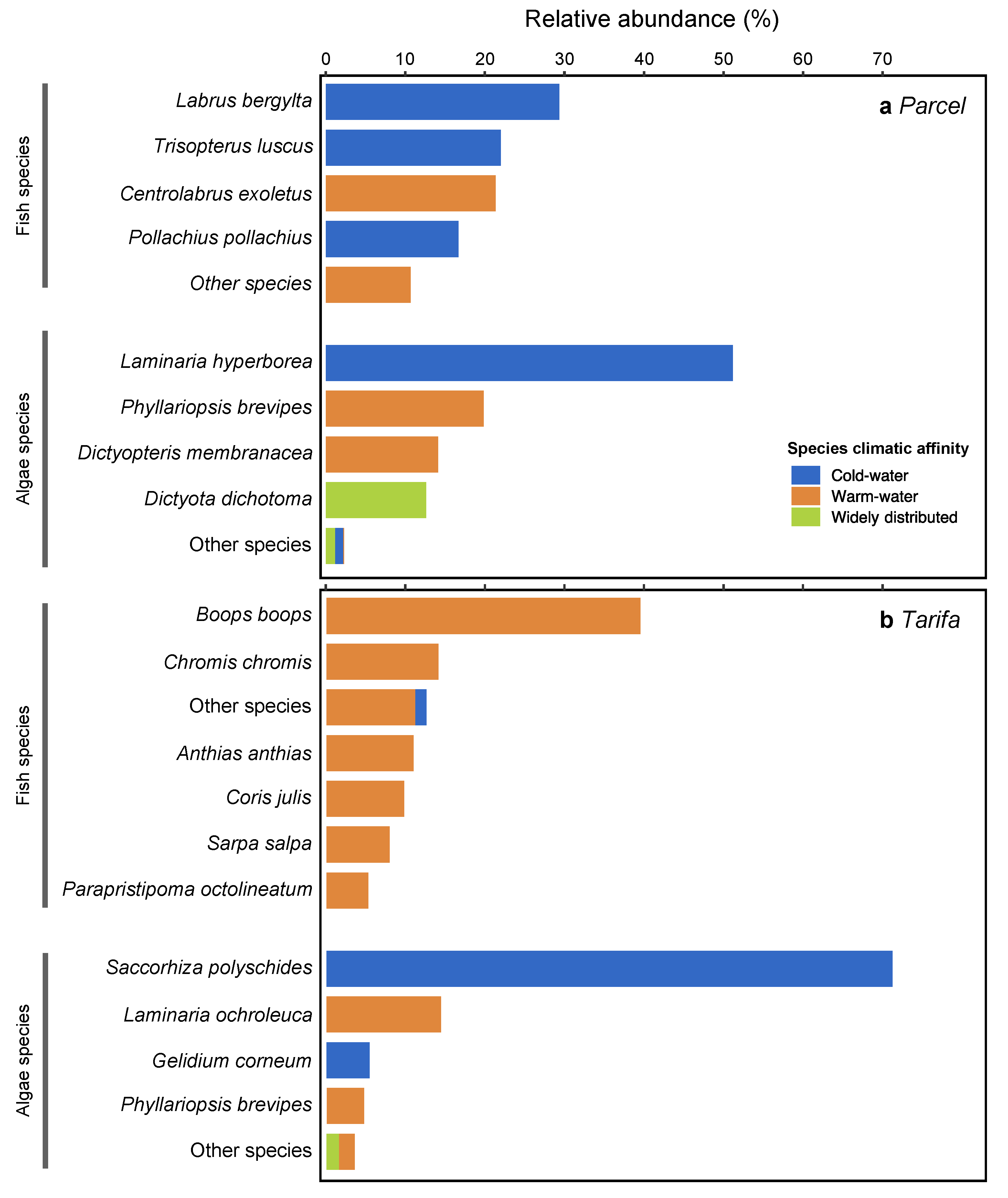
3.2. Fish and Algae Assemblages
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Southward, A.J.; Hawkins, S.J.; Burrows, M.T. Seventy years’ observations of changes in distribution and abundance of zooplankton and intertidal organisms in the western English Channel in relation to rising sea temperature. J. Therm. Biol. 1995, 20, 127–155. [Google Scholar] [CrossRef]
- Lima, F.P.; Ribeiro, P.A.; Queiroz, N.; Hawkins, S.J.; Santos, A.M. Do distributional shifts of northern and southern species of algae match the warming pattern? Glob. Chang. Biol. 2007, 13, 2592–2604. [Google Scholar] [CrossRef]
- Spalding, M.D.; Fox, H.E.; Allen, G.R.; Davidson, N.; Ferdaña, Z.A.; FINLAYSON, M.; Halpern, B.S.; Jorge, M.A.; Lombana, A.; Lourie, S.A.; et al. Marine Ecoregions of the World: A Bioregionalization of Coastal and Shelf Areas. Bioscience 2007, 57, 573. [Google Scholar] [CrossRef]
- Lüning, K. Seaweeds: Their Environment, Biogeography and Ecophysiology, by Klaus Lünin; John Wiley: New York, NY, USA, 1990; ISBN 978-0-471-62434-9. [Google Scholar]
- Wiencke, C.; Bischof, K. (Eds.) Seaweed Biology; Ecological Studies; Springer: Berlin/Heidelberg, Germany, 2012; Volume 219, ISBN 978-3-642-28450-2. [Google Scholar]
- Zarnetske, P.L.; Skelly, D.K.; Urban, M.C. Biotic multipliers of climate change. Science 2012, 336, 1516–1518. [Google Scholar] [CrossRef]
- Vergés, A.; McCosker, E.; Mayer-Pinto, M.; Coleman, M.A.; Wernberg, T.; Ainsworth, T.; Steinberg, P.D. Tropicalisation of temperate reefs: Implications for ecosystem functions and management actions. Funct. Ecol. 2019, 33, 1000–1013. [Google Scholar] [CrossRef]
- Poloczanska, E.S.; Brown, C.J.; Sydeman, W.J.; Kiessling, W.; Schoeman, D.S.; Moore, P.J.; Brander, K.; Bruno, J.F.; Buckley, L.B.; Burrows, M.T.; et al. Global imprint of climate change on marine life. Nat. Clim. Chang. 2013, 3, 919–925. [Google Scholar] [CrossRef]
- Last, P.R.; White, W.T.; Gledhill, D.C.; Hobday, A.J.; Brown, R.; Edgar, G.J.; Pecl, G. Long-term shifts in abundance and distribution of a temperate fish fauna: A response to climate change and fishing practices. Glob. Ecol. Biogeogr. 2011, 20, 58–72. [Google Scholar] [CrossRef]
- Flores-Moya, A. Warm Temperate Seaweed Communities: A Case Study of Deep Water Kelp Forests from the Alboran Sea (SW Mediterranean Sea) and the Strait of Gibraltar. In Seaweed Biology; Wiencke, C., Bischof, K., Eds.; Springer: Berlin/Heidelberg, Germany, 2012; Volume 219, pp. 471–493. ISBN 978-3-642-28450-2. [Google Scholar]
- Acker, J.G.; Leptoukh, G. Online analysis enhances use of NASA Earth Science Data. Eos (Washington. DC.) 2007, 88. Available online: https://agupubs.onlinelibrary.wiley.com/doi/pdf/10.1029/2007EO020003 (accessed on 20 December 2019). [CrossRef]
- Tuya, F.; Cacabelos, E.; Duarte, P.; Jacinto, D.; Castro, J.; Silva, T.; Bertocci, I.; Franco, J.; Arenas, F.; Coca, J.; et al. Patterns of landscape and assemblage structure along a latitudinal gradient in ocean climate. Mar. Ecol. Prog. Ser. 2012, 466, 9–19. [Google Scholar] [CrossRef]
- Franco, J.N.; Wernberg, T.; Bertocci, I.; Duarte, P.; Jacinto, D.; Vasco-Rodrigues, N.; Tuya, F. Herbivory drives kelp recruits into “hiding” in a warm ocean climate. Mar. Ecol. Prog. Ser. 2015, 536, 1–9. [Google Scholar] [CrossRef]
- Tuya, F.; Wernberg, T.; Thomsen, M.S. The relative influence of local to regional drivers of variation in reef fishes. J. Fish Biol. 2011, 79, 217–234. [Google Scholar] [CrossRef] [PubMed]
- Froese, R.; Pauly, D. FishBase. Available online: www.fishbase.org (accessed on 20 December 2019).
- Wheeler, A.C.; Merrett, N.R.; Quigley, D.T.G. Additional records and notes for Wheeler’s (1992) List of the Common and Scientific Names of Fishes of the British Isles. J. Fish Biol. 2004, 65, 1–40. [Google Scholar]
- Wernberg, T.; Kendrick, G.A.; Phillips, J.C. Regional differences in kelp-associated algal assemblages on temperate limestone reefs in south-western Australia. Divers. Distrib. 2003, 9, 427–441. [Google Scholar] [CrossRef]
- Alvarez, M.; Gallardo, T.; Ribera, M.A.; Garreta, A.G. A reassessment of Northern Atlantic seaweed biogeography. Phycologia 1988, 27, 221–223. [Google Scholar] [CrossRef]
- Guiry, M.D.; Guiry, G.M. AlgaeBase. Available online: www.algaebase.org (accessed on 20 December 2019).
- García-Sánchez, M.J.; Delgado-Huertas, A.; Fernández, J.A.; Flores-Moya, A. Photosynthetic use of inorganic carbon in deep-water kelps from the Strait of Gibraltar. Photosynth. Res. 2016, 127, 295–305. [Google Scholar] [CrossRef]
- Lemos, R.T.; Pires, H.O. The upwelling regime off the west Portuguese coast, 1941–2000. Int. J. Climatol. 2004, 24, 511–524. [Google Scholar] [CrossRef]
- Franco, J.N.; Tuya, F.; Bertocci, I.; Rodríguez, L.; Martínez, B.; Sousa-Pinto, I.; Arenas, F. The ‘golden kelp’ Laminaria ochroleuca under global change: Integrating multiple eco-physiological responses with species distribution models. J. Ecol. 2018, 106, 47–58. [Google Scholar] [CrossRef]
- Franco, J.N.; Wernberg, T.; Bertocci, I.; Jacinto, D.; Maranhão, P.; Pereira, T.; Martinez, B.; Arenas, F.; Sousa-Pinto, I.; Tuya, F. Modulation of different kelp life stages by herbivory: Compensatory growth versus population decimation. Mar. Biol. 2017, 164, 164. [Google Scholar] [CrossRef]
- Vergés, A.; Steinberg, P.D.; Hay, M.E.; Poore, A.G.B.; Campbell, A.H.; Ballesteros, E.; Heck, K.L.; Booth, D.J.; Coleman, M.A.; Feary, D.A.; et al. The tropicalization of temperate marine ecosystems: Climate-mediated changes in herbivory and community phase shifts. Proc. R. Soc. B Biol. Sci. 2014, 281, 20140846. [Google Scholar] [CrossRef]
- Wernberg, T.; Bennett, S.; Babcock, R.C.; de Bettignies, T.; Cure, K.; Depczynski, M.; Dufois, F.; Fromont, J.; Fulton, C.J.; Hovey, R.K.; et al. Climate-driven regime shift of a temperate marine ecosystem. Science 2016, 353, 169–172. [Google Scholar] [CrossRef]
- Raitsos, D.E.; Beaugrand, G.; Georgopoulos, D.; Zenetos, A.; Pancucci-Papadopoulou, A.M.; Theocharis, A.; Papathanassiou, E. Global climate change amplifies the entry of tropical species into the eastern Mediterranean Sea. Limnol. Oceanogr. 2010, 55, 1478–1484. [Google Scholar] [CrossRef]
- Bañón, R.; Fernández, A.; Arronte, J.C.; García, D.; Villamarín, A. New records of two southern fishes found in northern waters of the Iberian Peninsula. Acta Ichthyol. Piscat. 2017, 47, 401–405. [Google Scholar] [CrossRef][Green Version]
- Horta E Costa, B.; Assis, J.; Franco, G.; Erzini, K.; Henriques, M.; Gonçalves, E.J.; Caselle, J.E. Tropicalization of fish assemblages in temperate biogeographic transition zones. Mar. Ecol. Prog. Ser. 2014, 504, 241–252. [Google Scholar] [CrossRef]
- Encarnação, J.; Morais, P.; Baptista, V.; Cruz, J.; Teodósio, M.A. New evidence of marine fauna tropicalization offthe Southwestern Iberian Peninsula (Southwest Europe). Diversity 2019, 11, 1–11. [Google Scholar] [CrossRef]
- Araújo, R.; Sousa-Pinto, I.; Bárbara, I.; Quintino, V. Macroalgal communities of intertidal rock pools in the northwest coast of Portugal. Acta Oecologica 2006, 30, 192–202. [Google Scholar] [CrossRef]
- Hernández, I.; Bermejo, R.; Vergara, J.J.; Olivé, I.; Fernando, G.; Mourente, G.; de los Santos, C.B.; Pérez-lloréns, J.L. Estudio ficológico del intermareal y fondos submareales del sector oriental del Parque Natural del Estrecho, entre el puerto de Tarifa y Piedra Alta. MIGRES Rev. Ecol. 2011, 2, 1–10. [Google Scholar]
- Assis, J.; Coelho, N.C.; Lamy, T.; Valero, M.; Alberto, F.; Serrão, E.Á. Deep reefs are climatic refugia for genetic diversity of marine forests. J. Biogeogr. 2016, 43, 833–844. [Google Scholar] [CrossRef]
| Family | Species | Climatic Affinity | Parcel | Tarifa |
|---|---|---|---|---|
| Serranidae | Anthias anthias Linnaeus, 1758 | Wr | 15.7 ± 6.0 | |
| Sparidae | Boops boops Linnaeus, 1758 | Wr | 56.7 ± 22.5 | |
| Labridae | Coris julis Linnaeus, 1758 | Wr | 0.5 ± 1.2 | 14.0 ± 8.9 |
| Pomacentridae | Chromis chromis Linnaeus, 1758 | Wr | 20.2 ± 8.5 | |
| Labridae | Centrolabrus exoletus Linnaeus, 1758 | Wr | 5.3 ± 1.8 | |
| Labridae | Ctenolabrus rupestris Linnaeus, 1758 | Wr | 0.2 ± 0.4 | 2.2 ± 1.2 |
| Moronidae | Dicentrarchus labrax Linnaeus, 1758 | Wr | 1.0 ± 1.3 | |
| Sparidae | Diplodus cervinus Lowe, 1838 | Wr | 2.5 ± 0.8 | |
| Sparidae | Diplodus sargus Linnaeus, 1758 | Wr | 5.7 ± 1.6 | |
| Sparidae | Diplodus vulgaris Geoffroy Saint-Hilaire, 1817 | Wr | 0.3 ± 0.8 | 2.7 ± 2.1 |
| Labridae | Labrus bergylta Ascanius, 1767 | Cd | 7.3 ± 8.1 | |
| Mullidae | Mullus surmuletus Linnaeus, 1758 | Wr | 1.0 ± 1.3 | |
| Haemulidae | Parapristipoma octolineatum Valenciennes, 1833 | Wr | 7.5 ± 4.8 | |
| Gadidae | Pollachius pollachius Linnaeus, 1758 | Cd | 4.2 ± 8.0 | |
| Sparidae | Sarpa salpa Linnaeus, 1758 | Wr | 11.3 ± 6.9 | |
| Serranidae | Serranus cabrilla Linnaeus, 1758 | Wr | 1.0 ± 0.9 | |
| Labridae | Symphodus melops Linnaeus, 1758 | Wr | 0.7 ± 1.2 | 1.0 ± 1.1 |
| Gadidae | Trisopterus luscus Linnaeus, 1758 | Cd | 5.5 ± 7.9 | 2.0 + 4.9 |
| Order | Species | Climatic Affinity | Parcel | Tarifa |
|---|---|---|---|---|
| Class Phaeophyceae | ||||
| Desmaerstiales | Desmarestia ligulata (Stackhouse) J.V. Lamouroux, 1813 | Cd | 2.78 ± 6.80 | |
| Dictyotales | Dictyopteris polypodioides (A.P. De Candolle) J.V. Lamouroux, 1809 | Wr | 37.30 ± 28.28 | 1.29 ± 3.17 |
| Dictyota dichotoma (Hudson) J.V. Lamouroux, 1809 | Wd | 33.35 ± 17.23 | 1.34 ± 1.55 | |
| Fucales | Cystoseira tamariscifolia (Hudson) Papenfuss, 1950 | Wr | 21.58 ± 43.53 | |
| Laminariales | Laminaria hyperborea (Gunnerus) Foslie, 1884 | Cd | 135.34 ± 109.27 | |
| Lamiraria ochroleuca Bachelot de la Pylaie, 1824 | Wr | 191.67 ± 187.55 | ||
| Sphacelariales | Halopteris filicina (Grateloup) Kützing, 1843 | Wr | 1.18 ± 1.86 | |
| Sporochnales | Carpomitra costata (Stackhouse) Batters, 1902 | Wr | 1.58 ± 3.86 | |
| Tilopteridales | Phyllariopsis brevipes (C. Agardh) E.C. Henry and G.R. South, 1897 | Wr | 52.48 ± 55.47 | 62.50 ± 50.20 |
| Saccorhiza polyschides (Lightfoot) Batters, 1902 | Cd | 949.00 ± 1306.94 | ||
| Class Florideophyceae | ||||
| Rhodymeniales | Rhodymenia holmesii Ardissone, 1893 | Wr | 0.16 ± 0.28 | |
| Rhodymenia pseudopalmata (J.V. Lamouroux) P.C. Silva, 1952 | Wd | 0.19 ± 0.22 | ||
| Ceramiales | Acrosorium ciliolatum (Harvey) Kylin, 1924 | Wd | 1.59 ± 1.36 | |
| Chondria dasyphylla (Woodward) C. Agardh, 1817 | Wd | 0.34 ± 0.82 | ||
| Cryptopleura ramose (Hudson) L. Newton, 1931 | Wd | 0.30 ± 0.47 | ||
| Hypoglossum hypoglossoides (Stackhouse) Collins and Hervey, 1917 | Wd | 0.10 ± 0.00 | 0.70 ± 0.05 | |
| Pterothamnion plumula (J. Ellis) Nägeli, 1855 | Cd | 0.02 ± 0.05 | ||
| Gelidiales | Peyssonnelia coriacea Feldmann, 1941 | Wd | 3.01 ± 7.37 | |
| Gigartinales | Gelidium corneum (Hudson) J.V. Lamouroux, 1813 | Cd | 71.97 ± 97.31 | |
| Metacallophyllis laciniata (Hudson) A. Vergés and L. Le Gall, 2017 | Wd | 0.23 ± 0.57 | ||
| Sphaerococcus coronopifolius Stackhouse, 1797 | Wd | 14.17 ± 29.90 | ||
| Nemaliales | Scinaia furcellata (Turner) J. Agardh, 1851 | Wd | 1.59 ± 3.12 | |
| Plocamiales | Plocamium cartilagineum (Linnaeus) P.S. Dixon, 1967 | Wd | 0.56 ± 0.80 | 0.69 ± 1.07 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franco, J.N.; Arenas, F.; Sousa-Pinto, I.; de los Santos, C.B. Snapshot of Macroalgae and Fish Assemblages in Temperate Reefs in the Southern European Atlantic Ecoregion. Diversity 2020, 12, 26. https://doi.org/10.3390/d12010026
Franco JN, Arenas F, Sousa-Pinto I, de los Santos CB. Snapshot of Macroalgae and Fish Assemblages in Temperate Reefs in the Southern European Atlantic Ecoregion. Diversity. 2020; 12(1):26. https://doi.org/10.3390/d12010026
Chicago/Turabian StyleFranco, João N., Francisco Arenas, Isabel Sousa-Pinto, and Carmen B. de los Santos. 2020. "Snapshot of Macroalgae and Fish Assemblages in Temperate Reefs in the Southern European Atlantic Ecoregion" Diversity 12, no. 1: 26. https://doi.org/10.3390/d12010026
APA StyleFranco, J. N., Arenas, F., Sousa-Pinto, I., & de los Santos, C. B. (2020). Snapshot of Macroalgae and Fish Assemblages in Temperate Reefs in the Southern European Atlantic Ecoregion. Diversity, 12(1), 26. https://doi.org/10.3390/d12010026





