Local-Scale Bat Guild Activity Differs with Rice Growth Stage at Ground Level in the Philippines
Abstract
:1. Introduction
2. Materials and Methods
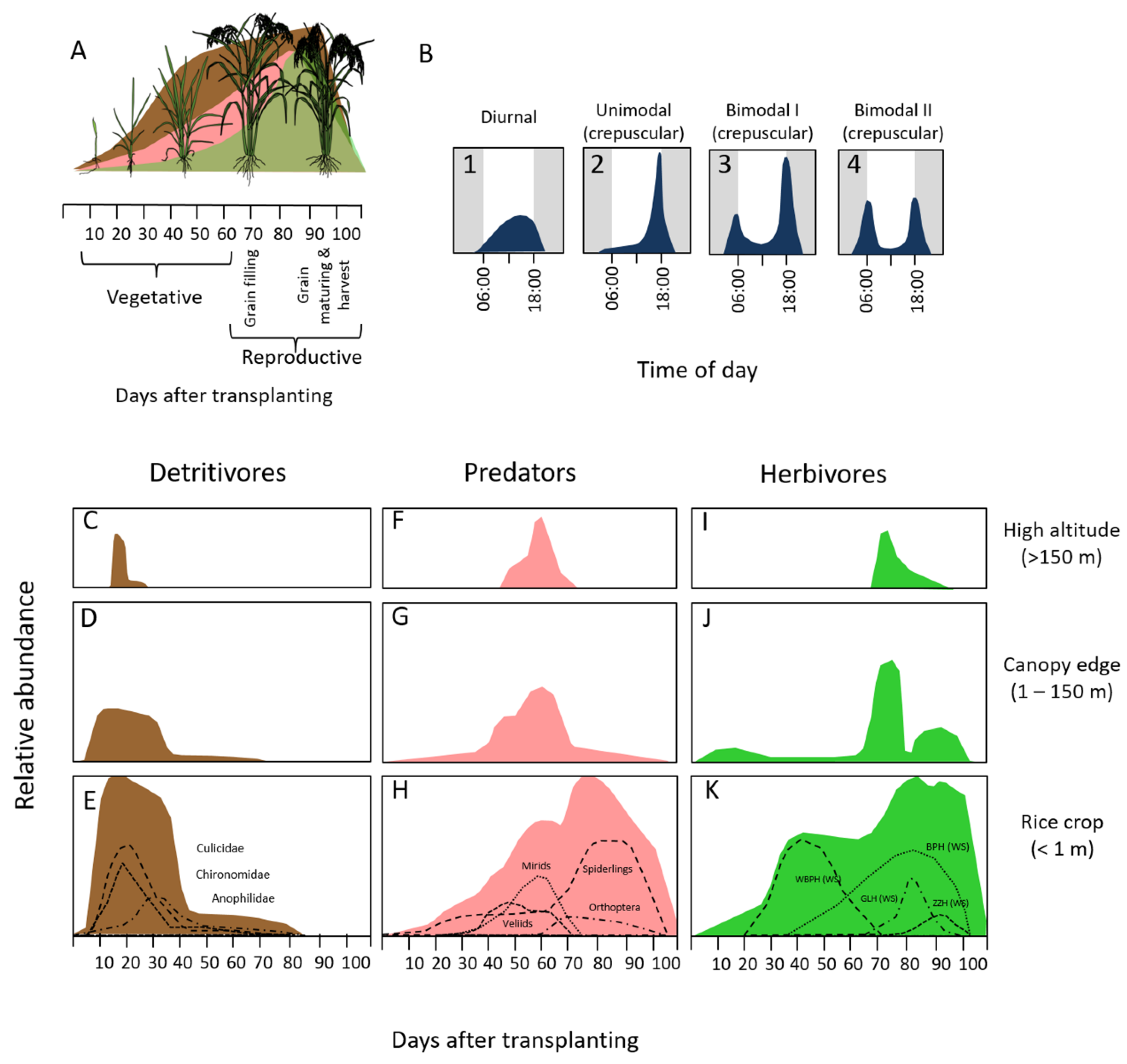
2.1. Field Site and Rice Growth Stages
2.2. Acoustic Sampling
2.3. Assignment of Bats to Functional Guilds
2.4. Analysis of Echolocation Calls
2.5. Bat Capture and Recording Reference Calls
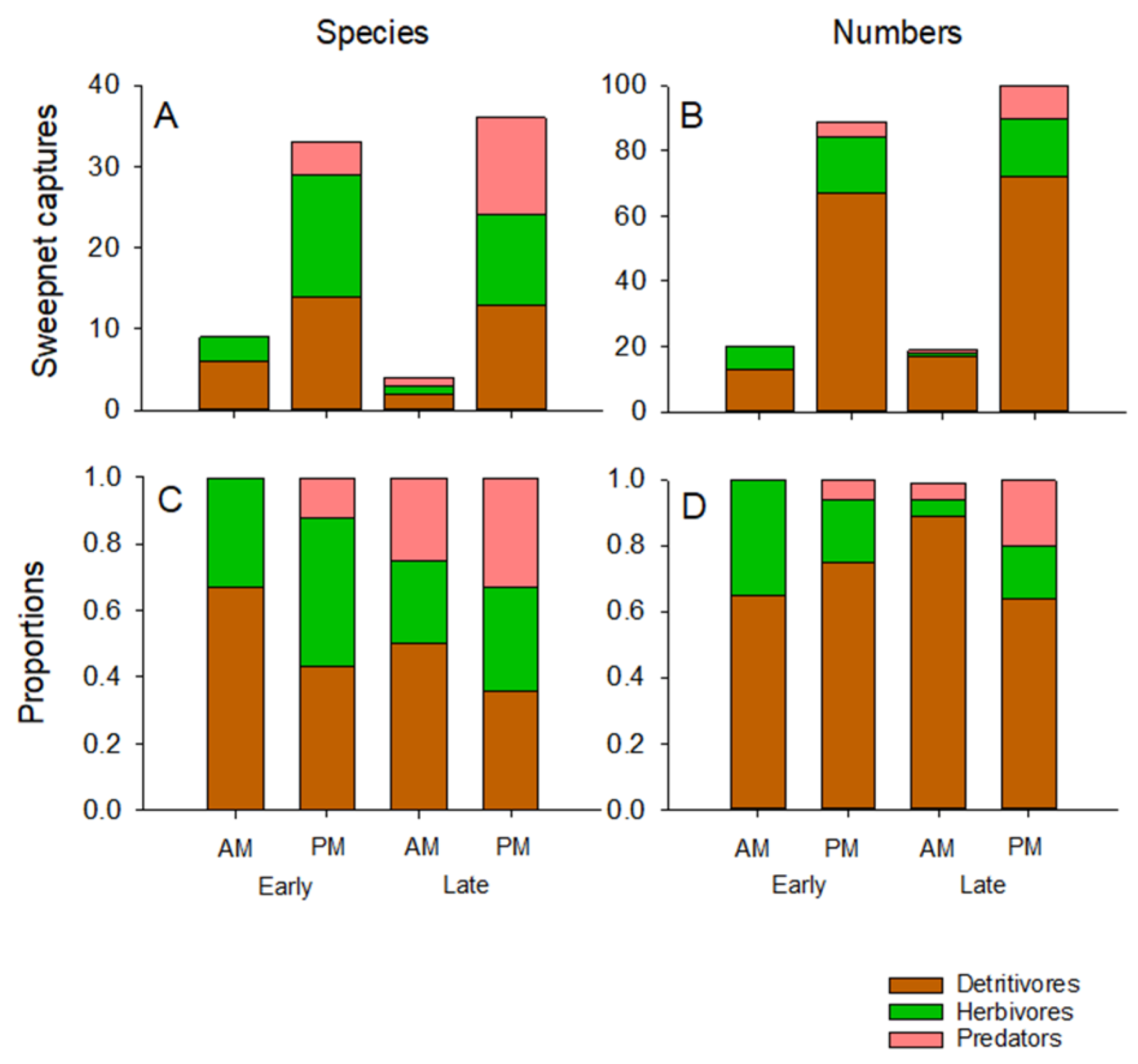
2.6. Arthropod Sampling
2.7. Statistical Analysis
3. Results
3.1. Bat Assemblage
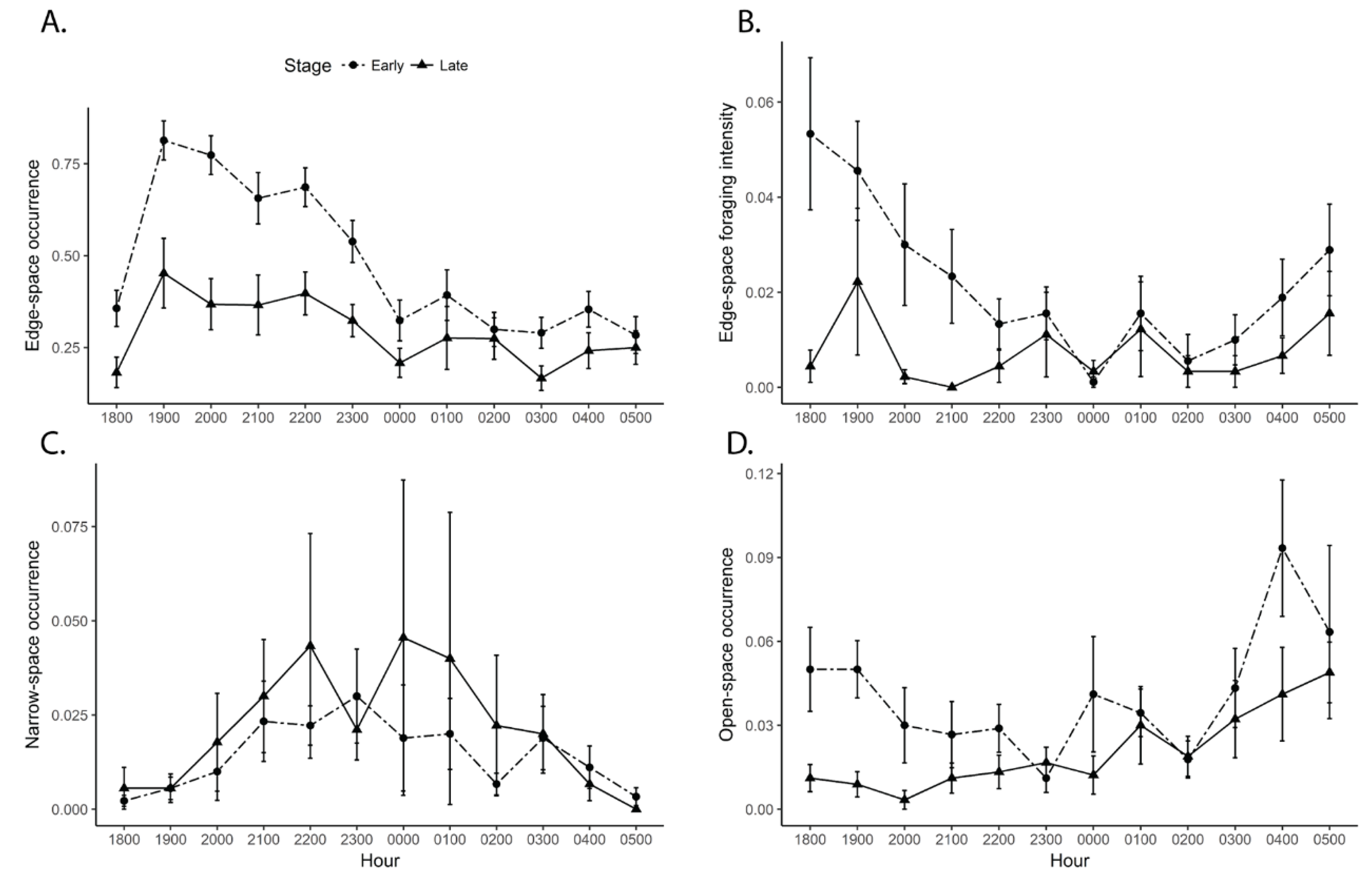
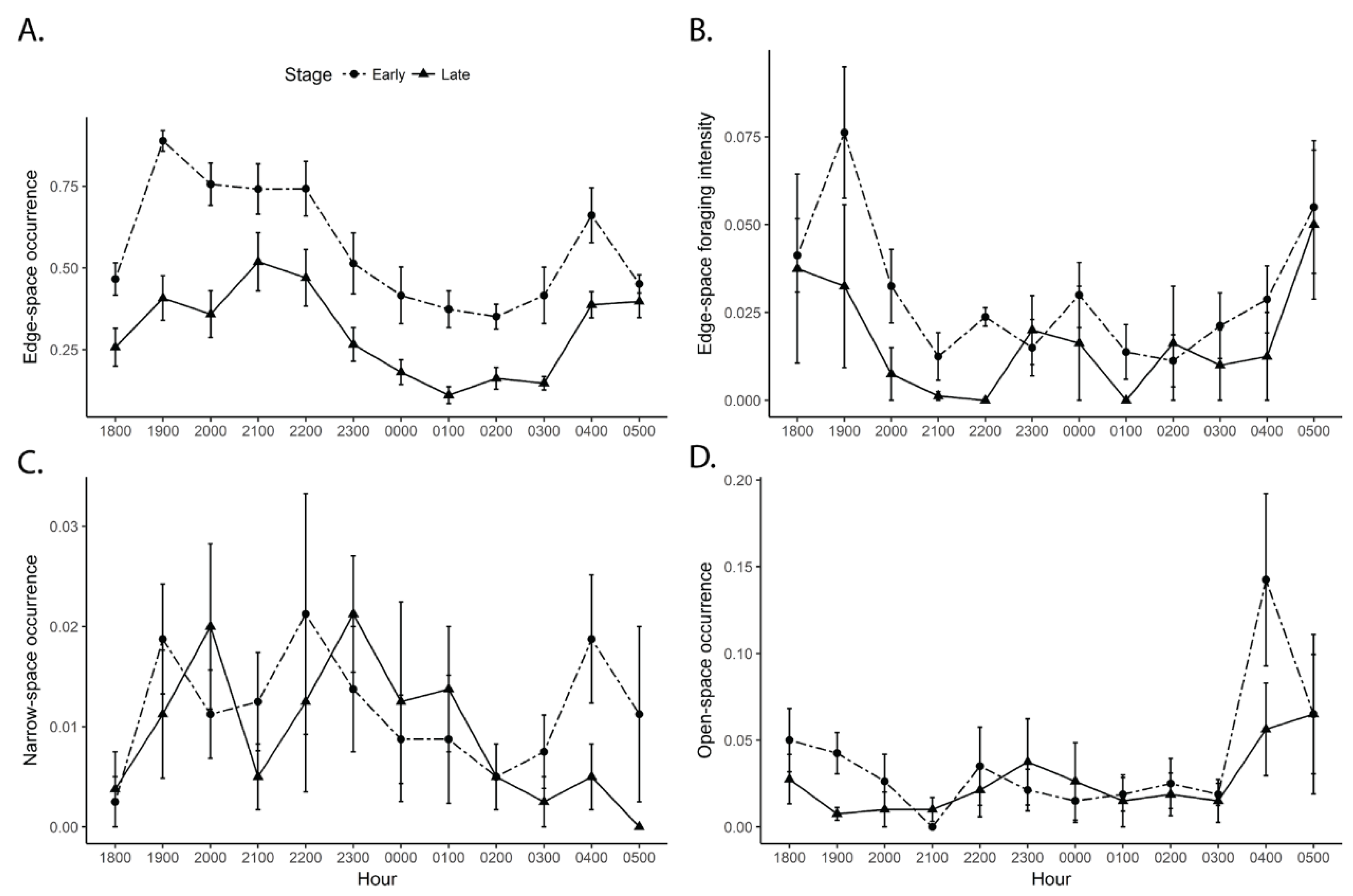
3.2. Bat Activity by Rice Stage and Time
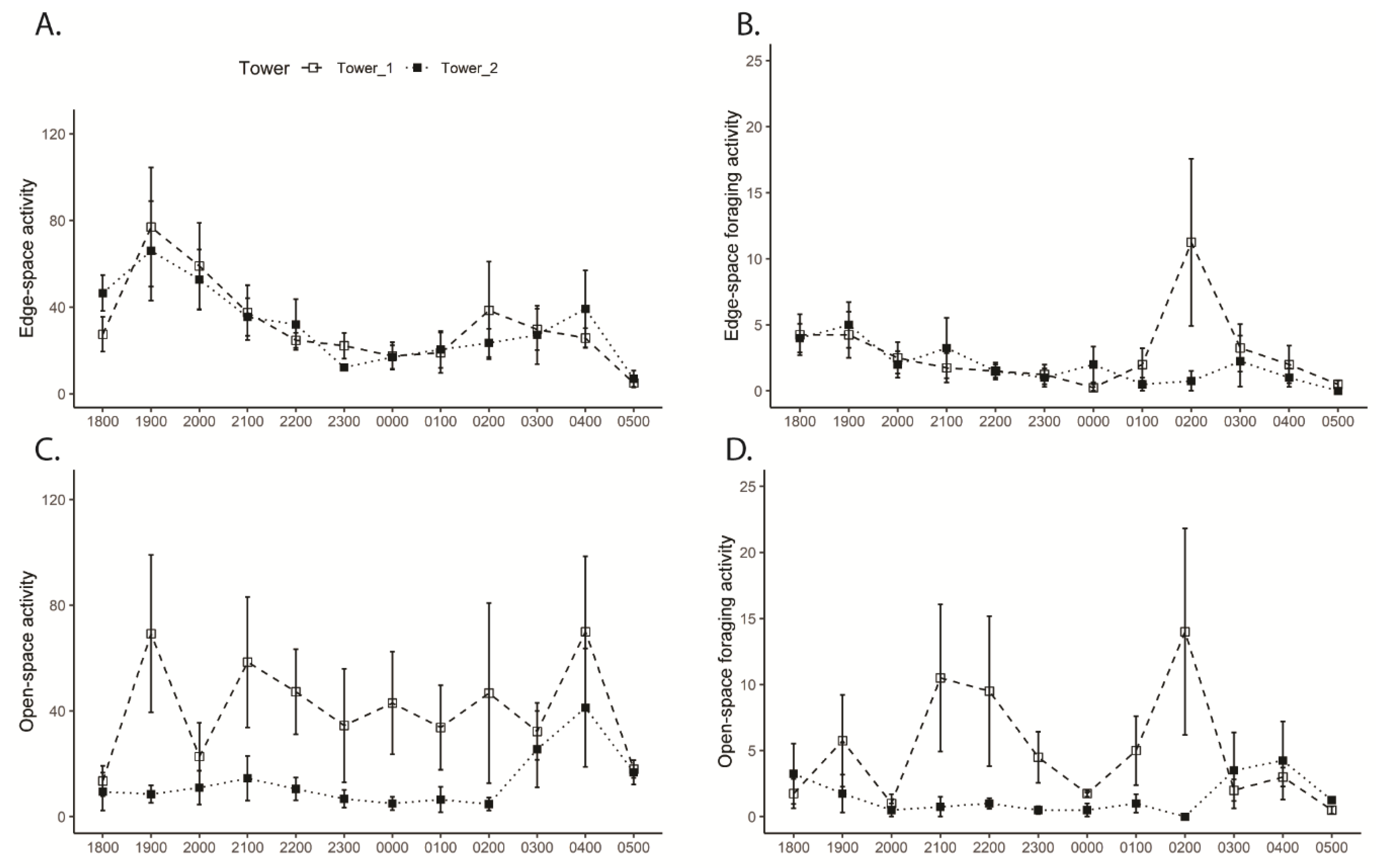
3.3. Tower Bat Activity
3.4. Aerial Arthropod Abundance and Composition by Stage
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Year | Night | Date | Site | Stage | No. of Detectors | Detector Type |
|---|---|---|---|---|---|---|
| 2015 | 1 | 27-July | 1 | Early | 1 | Anabat SD2 |
| 2015 | 1 | 27-July | 1 | Late | 1 | Anabat SD2 |
| 2015 | 1 | 27-July | 1 | Water | 1 | Anabat SD2 |
| 2015 | 2 | 28-July | 2 | Water | 1 | Anabat SD2 |
| 2015 | 2 | 28-July | 2 | Early | 1 | Anabat SD2 |
| 2015 | 2 | 28-July | 2 | Late | 1 | Anabat SD2 |
| 2015 | 3 | 29-July | 2 | Water | 1 | Anabat SD2 |
| 2015 | 3 | 29-July | 2 | Early | 1 | Anabat SD2 |
| 2015 | 3 | 29-July | 2 | Late | 1 | Anabat SD2 |
| 2015 | 4 | 30-July | 3 | Early | 2 | Anabat SD2 |
| 2015 | 4 | 30-July | 3 | Late | 2 | Anabat SD2 |
| 2015 | 5 | 31-July | 4 | Early | 1 | Anabat SD2 |
| 2015 | 5 | 31-July | 4 | Late | 2 | Anabat SD2 |
| 2015 | 6 | 3-August | 1 | Early | 2 | Anabat SD2 |
| 2015 | 6 | 3-August | 1 | Late | 2 | Anabat SD2 |
| 2015 | 7 | 4-August | 2 | Early | 2 | Anabat SD2 |
| 2015 | 7 | 4-August | 2 | Late | 2 | Anabat SD2 |
| 2015 | 8 | 5-August | 3 | Early | 2 | Anabat SD2 |
| 2015 | 8 | 5-August | 3 | Late | 2 | Anabat SD2 |
| 2015 | 9 | 7-August | 4 | Early | 2 | Anabat SD2 |
| 2015 | 9 | 7-August | 4 | Late | 2 | Anabat SD2 |
| 2016 | 1 | 21-July | 1 | Early | 1 | SM4Bat_U1 mic |
| 2016 | 1 | 21-July | 1 | Late | 1 | SM4Bat_U1 mic |
| 2016 | 2 | 22-July | 1 | Early | 1 | SM4Bat_U1 mic |
| 2016 | 2 | 22-July | 1 | Late | 1 | SM4Bat_U1 mic |
| 2016 | 3 | 26-July | 4 | Early | 1 | SM4Bat_U1 mic |
| 2016 | 3 | 26-July | 4 | Late | 1 | SM4Bat_U1 mic |
| 2016 | 4 | 27-July | 3 | Early | 1 | SM4Bat_U1 mic |
| 2016 | 4 | 27-July | 3 | Late | 1 | SM4Bat_U1 mic |
| 2016 | 5 | 28-July | 2 | Early | 1 | SM4Bat_U1 mic |
| 2016 | 5 | 28-July | 2 | Late | 1 | SM4Bat_U1 mic |
| 2016 | 6 | 29-July | 1 | Early | 1 | SM4Bat_U1 mic |
| 2016 | 6 | 29-July | 1 | Late | 1 | SM4Bat_U1 mic |
| 2016 | 7 | 31-July | 4 | Early | 1 | SM4Bat_U1 mic |
| 2016 | 7 | 31-July | 4 | Late | 1 | SM4Bat_U1 mic |
| 2016 | 8 | 1-August | 3 | Early | 1 | SM4Bat_U1 mic |
| 2016 | 8 | 1-August | 3 | Late | 1 | SM4Bat_U1 mic |
| 2016 | 9 | 3-August | 2 | Early | 1 | SM4Bat_U1 mic |
| 2016 | 9 | 3-August | 2 | Grass | 1 | SM4Bat_U1 mic |
| 2016 | 10 | 2-August | 3 | Water | 1 | SM4Bat_U1 mic |
| 2016 | 10 | 2-August | 3 | Grass | 1 | SM4Bat_U1 mic |
| 2016 | 11 | 4-August | Tower | 2 | SM4Bat_U1 mic | |
| 2016 | 12 | 5-August | Tower | 2 | SM4Bat_U1 mic | |
| 2016 | 13 | 6-August | Tower | 2 | SM4Bat_U1 mic | |
| 2016 | 14 | 7-August | Tower | 2 | SM4Bat_U1 mic |
Appendix B
| Guild/Taxon | Early | Late | ||
|---|---|---|---|---|
| PM | AM | PM | AM | |
| Detritivores | ||||
| Culicidae | 51 | 52 | 17 | |
| Chironomidae | 4 | 6 | 1 | |
| Formicidae | 1 | |||
| Hydrophilidae | 17 | 4 | ||
| Limnichidae | 1 | |||
| Sphaeroceridae | 1 | |||
| Tipulidae | 13 | |||
| Herbivores | ||||
| Chrysomelidae | 1 | |||
| Cicadellidae | 5 | 6 | 3 | |
| Recilia distincta | ||||
| Reilia dorsalis | ||||
| Crambidae | 1 | |||
| Cnaphalocrocis medinalis | ||||
| Delphacidae | 3 | 2 | ||
| Nilaparvata lugens | ||||
| Sogatella furafera | ||||
| Gryllidae | 1 | |||
| Nitidulidae | 2 | |||
| Noctuidae | 1 | 1 | ||
| Pentatonidae | 1 | |||
| Phalacridae | 3 | 8 | ||
| Pyralidae | 3 | 1 | 1 | |
| Predators | ||||
| Araneidae | 1 | |||
| Carabidae | 1 | |||
| Chlaenius | ||||
| Ceratopogonidae | 3 | 5 | ||
| Nilobezzia acanthipus | ||||
| Stilobezzia | ||||
| Phanobezzia | ||||
| Dytiscidae | 3 | |||
| Empididae | 2 | |||
| Lygaeidae | 2 | |||
| Miridae | 2 | |||
| Cyrtorhinus lividipennis | ||||
| Staphylinidae | 1 | |||
| Veliidae | 1 | |||
| Microvelia donglasi | ||||
| Total | 93 | 13 | 107 | 19 |
References
- Bambaradeniya, C.N.B.; Edirisinghe, J.P.; Silva, D.N.D.E.; Ranawana, K.B.; Wijekoon, S. Biodiversity associated with an irrigated rice agro-ecosystem in Sri Lanka. Biodivers. Conserv. 2004, 13, 1715–1753. [Google Scholar] [CrossRef]
- Shuman-Goodier, M.E.; Singleton, G.R.; Propper, C.R. Competition and pesticide exposure affect development of invasive (Rhinella marina) and native (Fejervarya vittigera) rice paddy amphibian larvae. Ecotoxicology 2017, 26, 1293–1304. [Google Scholar] [CrossRef] [PubMed]
- Holzer, K.A.; Bayers, R.P.; Nguyen, T.T.; Lawler, S.P. Habitat value of cities and rice paddies for amphibians in rapidly urbanizing Vietnam. J. Urban Ecol. 2017, 3, 1–12. [Google Scholar] [CrossRef]
- Bourdin, P.; Paris, T.; Serrano, F.; Smedley, R.; Hettel, G. Guide to the Birds of Philippine Rice Fields; International Rice Research Institute: Los Banos, Philippines, 2015; p. 116. [Google Scholar]
- Schoenly, K.G.; Justo, H.D.; Barrion, A.T.; Harris, M.K.; Bottrell, D.G. Analysis of invertebrate biodiversity in a Philippine farmer’s irrigated rice field. Environ. Entomol. 1998, 27, 1125–1136. [Google Scholar] [CrossRef]
- Way, M.J. The role of biodiversity in the dynamics and management of insect pests of tropical irrigated rice–A review The role of biodiversity in the dynamics and management of insect pests of tropical irrigated rice–A review. Bull. Entomol. Res. 2015, 567–587. [Google Scholar] [CrossRef]
- Horgan, F.G.; Ramal, A.F.; Bernal, C.C.; Villegas, J.M.; Stuart, A.M.; Almazan, M.L.P. Applying Ecological Engineering for Sustainable and Resilient Rice Production Systems. Procedia Food Sci. 2016, 6, 7–15. [Google Scholar] [CrossRef] [Green Version]
- Horgan, F.G.; Ramal, A.F.; Villegas, J.M.; Almazan, M.L.P.; Bernal, C.C.; Jamoralin, A.; Pasang, J.M.; Orboc, G.; Agreda, V.; Arroyo, C. Ecological engineering with high diversity vegetation patches enhances bird activity and ecosystem services in Philippine rice fields. Reg. Environ. Chang. 2017, 17, 1355–1367. [Google Scholar] [CrossRef]
- Pathak, M.D.; Khan, Z.R. Insect Pests of Rice; International Rice Research Institute: Manila, Philippines, 1994. [Google Scholar]
- Settle, W.H.; Ariawan, H.; Astuti, E.T.; Cahyana, W.; Hakim, A.L.; Hindayana, D.; Lestari, A.S. Managing tropical rice pests through conservation of generalist natural enemies and alternative prey. Ecology 1996, 77, 1975–1988. [Google Scholar] [CrossRef]
- Savary, S.; Horgan, F.; Willocquet, L.; Heong, K.L. A review of principles for sustainable pest management in rice. Crop Prot. 2012, 32, 54–63. [Google Scholar] [CrossRef]
- Ohba, S.Y.; Huynh, T.T.T.; Le, L.L.; Ngoc, H.T.; Hoang, S.L.; Takagi, M. Mosquitoes and their potential predators in rice agroecosystems of the Mekong Delta, southern Vietnam. J. Am. Mosq. Control Assoc. 2011, 27, 384–392. [Google Scholar] [CrossRef]
- Roger, P.A.; Heong, K.L.; Teng, P.S. Biodiversity and sustainability of wetland rice production: Role and potential of microorganisms and invertebrates. In The Biodiversity of Microorganisms and Invertebrates: Its Role in Sustainable Agriculture; Hawksworth, D.L., Ed.; CAB International: Marseille, France, 1991; pp. 117–136. [Google Scholar]
- Normile, D. Vietnam turns back a “tsunami of pesticides. ” Science 2013, 341, 737–738. [Google Scholar] [CrossRef] [PubMed]
- Gurr, G.M.; Lu, Z.; Zheng, X.; Xu, H.; Zhu, P.; Chen, G.; Yao, X.; Cheng, J.; Zhu, Z.; Catindig, J.L.; et al. Multi-country evidence that crop diversification promotes ecological intensification of agriculture. Nat. Plants 2016, 2, 22–25. [Google Scholar] [CrossRef] [PubMed]
- McCracken, G.F.; Westbrook, J.K.; Brown, V.A.; Eldridge, M.; Federico, P.; Kunz, T.H. Bats Track and Exploit Changes in Insect Pest Populations. PLoS ONE 2012, 7, e43839. [Google Scholar] [CrossRef] [PubMed]
- Leelapaibul, W.; Bumrungrsi, S.; Pattanawiboon, A. Diet of wrinkle-lipped free-tailed bat (Tadarida plicata Buchannan, 1800) in central Thailand: Insectivorous bats potentially act as biological pest control agents. Acta Chiropterologica 2005, 7, 111–119. [Google Scholar] [CrossRef]
- Cleveland, C.J.; Betke, M.; Federico, P.; Frank, J.D.; Hallam, T.G.; Horn, J.; Lopez, J.D.; McCraken, G.F.; Medellin, R.A.; Moreno-Valdez, A.; et al. Economic value of the pest control service provided by Brazilian free-tailed bats in south-central Texas. Front. Ecol. Environ. 2006, 20, 207–219. [Google Scholar] [CrossRef]
- Federico, P.; Hallam, T.G.; McCracken, G.F.; Purucker, S.T.; Grant, W.E.; Correa-Sandoval, A.N.; Westbrook, J.K.; Medellín, R.A.; Cleveland, C.J.; Sansone, C.G.; et al. Brazilian free-tailed bats as insect pest regulaors in trangenic and conventional cotton crops. Ecol. Appl. 2008, 18, 826–837. [Google Scholar] [CrossRef] [PubMed]
- Kemp, J.; López-Baucells, A.; Rocha, R.; Wangensteen, O.S.; Andriatafika, Z.; Nair, A.; Cabeza, M. Bats as potential suppressors of multiple agricultural pests: A case study from Madagascar. Agric. Ecosyst. Environ. 2019, 269, 88–96. [Google Scholar] [CrossRef]
- Puig-Montserrat, X.; Torre, I.; López-Baucells, A.; Guerrieri, E.; Monti, M.M.; Ràfols-García, R.; Ferrer, X.; Gisbert, D.; Flaquer, C. Pest control service provided by bats in Mediterranean rice paddies: Linking agroecosystems structure to ecological functions. Mamm. Biol. 2015, 80, 237–245. [Google Scholar] [CrossRef]
- Williams-Guillén, K.; Perfecto, I.; Vandermeer, J. Bats limit insects in a Neotropical agroforestry system. Science 2008, 320, 4–6. [Google Scholar] [CrossRef]
- Maine, J.J.; Boyles, J.G. Bats initiate vital agroecological interactions in corn. Proc. Natl. Acad. Sci. USA 2015, 112, 12438–12443. [Google Scholar] [CrossRef] [Green Version]
- Maas, B.; Clough, Y.; Tscharntke, T. Bats and birds increase crop yield in tropical agroforestry landscapes. Ecol. Lett. 2013, 16, 1480–1487. [Google Scholar] [CrossRef]
- Maslo, B.; Valentin, R.; Leu, K.; Kerwin, K.; Hamilton, G.C.; Bevan, A.; Fefferman, N.H.; Fonseca, D.M. Chirosurveillance: The use of native bats to detect invasive agricultural pests. PLoS ONE 2017, 12, e0173321. [Google Scholar] [CrossRef]
- Kingston, T. Research priorities for bat conservation in Southeast Asia: A consensus approach. Biodivers. Conserv. 2010, 19, 471–484. [Google Scholar] [CrossRef]
- Heaney, L.R.; Balete, D.S.; Rickart, E.A. The Mammals of Luzon Island: Biogeography and Natural History of a Philippine Fauna; Johns Hopkins University Press: Baltimore, MD, USA, 2016; ISBN 978-1-4214-1837-7. [Google Scholar]
- Schnitzler, H.-U.; Kalko, E.K.V. Echolocation by insect-eating bats. Bioscience 2001, 51, 557–569. [Google Scholar] [CrossRef]
- Denzinger, A.; Schnitzler, H. Bat guilds, a concept to classify the highly diverse foraging and echolocation behaviors of microchiropteran bats. Front. Physiol. 2013, 4, 1–15. [Google Scholar] [CrossRef]
- Srilopan, S.; Bumrungsri, S.; Jantarit, S. The wrinkle-lipped free-tailed bat (Chaerephon plicatus Buchannan, 1800) feeds mainly on brown planthoppers in rice fields of central Thailand. Acta chiropt. 2018, 20, 207–219. [Google Scholar] [CrossRef]
- Wanger, T.C.; Darras, K.; Bumrungsri, S.; Tscharntke, T.; Klein, A.M. Bat pest control contributes to food security in Thailand. Biol. Conserv. 2014, 171, 220–223. [Google Scholar] [CrossRef]
- Ruadreo, N.; Voigt, C.C.; Bumrungsri, S. Large dietary niche overlap of sympatric open-space foraging bats revealed by carbon and nitrogen stable isotopes. Acta Chiropterologica 2019, 20, 329. [Google Scholar] [CrossRef]
- Nguyen, T.N.; Ruangwiset, A.; Bumrungsri, S. Vertical stratification in foraging activity of Chaerephon plicatus (Molossidae, Chiroptera) in Central Thailand. Mamm. Biol. 2019, 96, 1–6. [Google Scholar] [CrossRef]
- Chhay, S. Cambodian bats: A review of farming practices and economic value of lesser Asiatic yellow house bat Scotophilus kuhlii (Leach, 1821), in Kandal and Takeo provinces, Cambodia. Cambodian J. Nat. Hist. 2012, 2, 164. [Google Scholar]
- Biscardi, S.; Russo, D.; Casciani, V.; Cesarini, D.; Mei, M.; Boitani, L. Foraging requirements of the endangered long-fingered bat: The influence of micro-habitat structure, water quality and prey type. J. Zool. 2007, 273, 372–381. [Google Scholar] [CrossRef]
- Perfect, T.J.; Cook, A.G. Diurnal periodicity of flight in some Delphacidae and Cicadellidae associated with rice. Ecol. Entomol. 1982, 7, 317–326. [Google Scholar] [CrossRef]
- Riley, J.R.; Reynolds, D.R.; Smith, A.D.; Edwards, A.S.; Zhang, X.X.; Cheng, X.N.; Wang, H.K.; Cheng, J.Y.; Zhai, B.P. Observations of the autumn migration of the rice leaf roller Cnaphalocrocis medinalis (Lepidoptera: Pyralidae) and other moths in eastern China. Bull. Entomol. Res. 1995, 85, 397–414. [Google Scholar] [CrossRef]
- Wilby, A.; Lan, L.P.; Heong, K.L.; Huyen, N.P.D.; Quang, N.H.; Minh, N.V.; Thomas, M.B. Arthropod diversity and community structure in relation to land use in the Mekong Delta, Vietnam. Ecosystems 2006, 9, 538–549. [Google Scholar] [CrossRef]
- Schoenly, K.; Cohen, J.E.; Heong, K.L.; Litsinger, J.A.; Aquino, G.B.; Barrion, A.T.; Arida, G. Food web dynamics of irrigated rice fields at five elevations in Luzon, Philippines. Bull. Entomol. Res. 1996, 86, 451–466. [Google Scholar] [CrossRef]
- Aizpurua, O.; Budinski, I.; Georgiakakis, P.; Gopalakrishnan, S.; Ibañez, C.; Mata, V.; Rebelo, H.; Russo, D.; Szodoray-Parádi, F.; Zhelyazkova, V.; et al. Agriculture shapes the trophic niche of a bat preying on multiple pest arthropods across Europe: Evidence from DNA metabarcoding. Mol. Ecol. 2018, 27, 815–825. [Google Scholar] [CrossRef]
- Gonthier, D.J.; Ennis, K.K.; Farinas, S.; Hsieh, H.-Y.; Iverson, A.L.; Batary, P.; Rudolphi, J.; Tscharntke, T.; Cardinale, B.J.; Perfecto, I. Biodiversity conservation in agriculture requires a multi-scale approach. Proc. R. Soc. B Biol. Sci. 2014, 281, 20141358. [Google Scholar] [CrossRef] [Green Version]
- Toffoli, R.; Rughetti, M. Bat activity in rice paddies: Organic and conventional farms compared to unmanaged habitat. Agric. Ecosyst. Environ. 2017, 249, 123–129. [Google Scholar] [CrossRef]
- Fuentes-Montemayor, E.; Goulson, D.; Cavin, L.; Wallace, J.M.; Park, K.J. Fragmented woodlands in agricultural landscapes: The influence of woodland character and landscape context on bats and their insect prey. Agric. Ecosyst. Environ. 2013, 172, 6–15. [Google Scholar] [CrossRef]
- Hu, G.; Lu, M.H.; Tuan, H.A.; Liu, W.C.; Xie, M.C.; McInerney, C.E.; Zhai, B.P. Population dynamics of rice planthoppers, Nilaparvata lugens and Sogatella furcifera (Hemiptera, Delphacidae) in Central Vietnam and its effects on their spring migration to China. Bull. Entomol. Res. 2017, 107, 369–381. [Google Scholar] [CrossRef]
- Vu, Q.; Ramal, A.F.; Villegas, J.M.; Jamoralin, A.; Bernal, C.C.; Pasang, J.M.; Almazan, M.L.P.; Ramp, D.; Settele, J.; Horgan, F.G. Enhancing the parasitism of insect herbivores through diversification of habitat in Philippine rice fields. Paddy Water Environ. 2018, 16, 379–390. [Google Scholar] [CrossRef]
- Lu, Z.; Zhu, P.; Gurr, G.M.; Zheng, X.; Chen, G.; Heong, K.L. Rice Pest Management by Ecological Engineering : A Pioneering Attempt in China. In Rice Planthoppers: Ecology, Management, Socio Economics and Policy; Heong, K., Cheng, J., Escalada, M., Eds.; Springer Science+Business Media Dordrecht: Berlin, Germany, 2015; pp. 163–180. ISBN 9789401795357. [Google Scholar]
- Mogi, M.; Miyagi, I. Colonization of rice fields by mosquitoes (Diptera: Culicidae) and larvivorous predators in asynchronous rice cultivation areas in the Philippines. J. Med. Entomol. 1990, 27, 530–536. [Google Scholar] [CrossRef]
- Kenmore, P.E.; Carino, F.O.; Perez, C.A.; Dyck, V.A.; Gutierrez, A.P. Population regulation of the rice brown planthopper (Nilaparvata lugens Stål) within rice fields in the Philippines. J. Plant Prot. Trop. 1984, 1, 19–37. [Google Scholar]
- Cook, A.G.; Perfect, T.J. The influence of immigration on population development of Nilaparvata lugens and Sogatella furcifera and its interaction with immigration by predators. Crop Prot. 1985, 4, 423–433. [Google Scholar] [CrossRef]
- Heong, K.L.; Aquino, G.B.; Barrion, A.T. Population dynamics of plant-and leafhoppers and their natural enemies in rice ecosystems in the Philippines. Crop Prot. 1992, 11, 371–379. [Google Scholar] [CrossRef]
- Rubia, E.G.; Ferrer, E.R.; Shepard, B.M. Biology and predatory behavior of Conocephalus longipennis (de Haan) (Orthoptera: Tettigoniidae). J. Plant Prot. Trop. 1990, 7, 47–54. [Google Scholar]
- Simpson, I.C.; Roger, P.A.; Oficial, R.; Grant, I.F. Effects of nitrogen fertilizer and pesticide management on floodwater ecology in a wetland ricefield I. Experimental design and dynamics of the photosynthetic aquatic biomass. Biol. Fertil. Soils 1994, 17, 129–137. [Google Scholar] [CrossRef]
- Victor, T.J.; Reuben, R. Effects of organic and inorganic fertilisers on mosquito populations in rice fields of southern India. Med. Vet. Entomol. 2000, 14, 361–368. [Google Scholar] [CrossRef]
- Mogi, M. Water management in rice cultivation and its relation to mosquito production in Japan. In Vector-Borne Disease Control in Humans through Rice Agroecosystem Management; WHO/FAO/UNEP, Ed.; International Rice Research Institute: Los Banos, Philippines, 1988; pp. 101–110. [Google Scholar]
- Mogi, M.; Miyagi, I.; Cabrera, B.D. Development and survival of immature mosquitoes (Dipera: Culicidae) in Philippine rice fields. J. Med. Entomol. 1984, 21, 283–291. [Google Scholar] [CrossRef]
- Riley, J.R.; Xia-Nian, C.; Xiao-Xi, Z.; Reynolds, D.R.; Guo-Min, X.; Smith, A.D.; Ji-Yi, C.; Ai-Dong, B.; Bao-Ping, Z. The long-distance migration of Nilaparvata lugens (Stål) (Delphacidae) in China: Radar observations of mass return flight in the autumn. Ecol. Entomol. 1991, 16, 471–489. [Google Scholar] [CrossRef]
- Combalicer, E.A.; Im, S. Change anomalies of hydrologic responses to climate variability and land-use changes in the Mt. Makiling forest reserve. J. Environ. Sci. Manag. 2012, 15, 1–13. [Google Scholar]
- Surlykke, A.; Kalko, E.K. V Echolocating bats cry out loud to detect their prey. PLoS ONE 2008, 3, e2036. [Google Scholar] [CrossRef]
- Goerlitz, H.R. Weather conditions determine attenuation and speed of sound: Environmental limitations for monitoring and analyzing bat echolocation. Ecol. Evol. 2018, 8, 5090–5100. [Google Scholar] [CrossRef] [Green Version]
- Agranat, I. Detecting Bats with Ultrasonic Microphones: Understanding the Effects of Microphone Variance and Placement on Detection Rates. 2014. Available online: https://www.wildlifeacoustics.com/images/pdfs/UltrasonicMicrophones.pdf (accessed on 30 May 2019).
- Norberg, U.M.; Rayner, J.M.V. Ecological morphology and flight in bats (Mammalia; Chiroptera): Wing adaptations, flight performance, foraging strategy and echolocation. Philos. Trans. R. Soc. London Ser. B Biol. Sci. 1987, 316, 335–427. [Google Scholar] [CrossRef]
- Sedlock, J.L. Inventory of insectivorous bats on Mount Makiling, Philippines using echolocation call signatures and a new tunnel trap. Acta Chiropterologica 2001, 3, 163–178. [Google Scholar]
- Sedlock, J.L.; Jose, R.P.; Vogt, J.M.; Paguntalan, L.M.J.; Cariño, A.B. A survey of bats in a karst landscape in the central Philippines. Acta Chiropterologica 2014, 16, 197–211. [Google Scholar] [CrossRef]
- Russo, D.; Voigt, C.C. The use of automated identification of bat echolocation calls in acoustic monitoring: A cautionary note for a sound analysis. Ecol. Indic. 2016, 66, 598–602. [Google Scholar] [CrossRef]
- Siemers, B.M.; Stilz, P.; Scchnitzler, H.-U. The acoustic advantage of hunting at low heights above water: Behavioural experiments on the European ‘trawling’ bats Myotis capaccinii, M. dasycneme and M. daubentonii. J. Exp. Biol. 2001, 204, 3843–3854. [Google Scholar] [CrossRef]
- Krauel, J.J.; Ratcliffe, J.M.; Westbrook, J.K.; McCracken, G.F. Brazilian free-tailed bats (Tadarida brasiliensis) adjust foraging behaviour in response to migratory moths. Can. J. Zool. 2018, 96, 513–520. [Google Scholar] [CrossRef]
- Miller, B.W. A method for determining relative activity of free flying bats using a new activity index for acoustic monitoring. Acta Chiropterologica 2001, 3, 93–105. [Google Scholar] [CrossRef]
- Ingle, N.R.; Heaney, L.R. A key to the bats of the Philippine Islands. Fieldiana Zool. New Ser. 1992, 69, 1–44. [Google Scholar]
- R Developer Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Sedlock, J.L.; Ingle, N.R.; Balete, D.S. Enhanced sampling of bat assemblages: A field test on Mount Banahaw, Luzon. Fieldiana Life Earth Sci. 2011, 2, 96–102. [Google Scholar] [CrossRef]
- Malmqvist, E.; Jansson, S.; Zhu, S.; Li, W.; Svanberg, K.; Svanberg, S.; Rydell, J.; Song, Z.; Bood, J.; Brydegaard, M.; et al. The bat-bird-bug battle: Daily flight activity of insects and their predators over a rice field revealed by high-resolution Scheimpflug Lidar. R. Soc. Open Sci. 2018, 5, 172303:1–172303:12. [Google Scholar] [CrossRef]
- Ancillotto, L.; Ariano, A.; Nardone, V.; Budinski, I.; Rydell, J.; Russo, D. Effects of free-ranging cattle and landscape complexity on bat foraging: Implications for bat conservation and livestock management. Agric. Ecosyst. Environ. 2017, 241, 54–61. [Google Scholar] [CrossRef]
- Fukui, D.; Murakami, M.; Nakano, S.; Aoi, T. Effect of emergent aquatic insects on bat foraging in a riparian forest. J. Anim. Ecol. 2006, 75, 1252–1258. [Google Scholar] [CrossRef]
- Adams, R.A.; Hayes, M.A. Water availability and successful lactation by bats as related to climate change in arid regions of western North America. J. Anim. Ecol. 2008, 77, 1115–1121. [Google Scholar] [CrossRef]
- Russo, D.; Ancillotto, L.; Cistrone, L.; Korine, C. The Buzz of Drinking on the Wing in Echolocating Bats. Ethology 2016, 122, 226–235. [Google Scholar] [CrossRef]
- Greif, S.; Siemers, B.M. Innate recognition of water bodies in echolocating bats. Nat. Commun. 2010, 1, 1–5. [Google Scholar] [CrossRef]
- Raju, K.H.K.; Sabesan, S.; Rajavel, A.R.; Subramanian, S.; Natarajan, R.; Thenmozhi, V.; Tyagi, B.K.; Jambulingam, P. A preliminary study to forecast Japanese encephalitis vector abundance in paddy growing area, with the aid of radar satellite images. Vector-Borne Zoonotic. Dis. 2016, 16, 117–123. [Google Scholar] [CrossRef]
- Russo, D.; Bosso, L.; Ancillotto, L. Novel perspectives on bat insectivory highlight the value of this ecosystem service in farmland: Research frontiers and management implications. Agric. Ecosyst. Environ. 2018, 266, 31–38. [Google Scholar] [CrossRef]
- Huang, F.; Subramanyam, B. Effects of delayed mating on reproductive performance of Plodia interpunctella (Hübner) (Lepidoptera: Pyralidae). J. Stored Prod. Res. 2003, 39, 53–63. [Google Scholar] [CrossRef]
- Dominik, C.; Seppelt, R.; Horgan, F.G.; Marquez, L.; Settele, J.; Václavík, T. Regional-scale effects override the influence of fine-scale landscape heterogeneity on rice arthropod communities. Agric. Ecosyst. Environ. 2017, 246, 269–278. [Google Scholar] [CrossRef]
- Settele, J.; Settle, W.H. Conservation biological control: Improving the science base. Proc. Natl. Acad. Sci. 2018, 115, 8241–8243. [Google Scholar] [CrossRef] [Green Version]
- Rothschild, G.H.L. The biology and ecology of rice stem borers in Sarawak (Malaysian Borneo). J. Appl. Ecol. 1971, 8, 287–322. [Google Scholar] [CrossRef]
- Shepard, B.M.; Barrion, A.T.; Litsinger, J.A. Rice-Feeding Insects of Tropical Asia; International Rice Research Institute: Manila, Los Baños, Philippines, 1995. [Google Scholar]
- Fullard, J.H.; Napoleone, N. Diel flight periodicity and the evolution of auditory defences in the macrolepidoptera. Anim. Behav. 2001, 62, 349–368. [Google Scholar] [CrossRef]
- Sedlock, J.L. Autecology and the conservation of insectivorous bats on Mt. Makiling. Silliman J. 2002, 42, 163–201. [Google Scholar]
- Shi, L.; Feng, J.; Liu, Y.; Ye, G.; Zhu, X. Is food resource partitioning responsible for deviation of echolocation call frequencies from allometry in Rhinolophus macrotis? Acta Theriol. Warsz. 2009, 54, 371–382. [Google Scholar] [CrossRef]
- ter Hofstede, H.M.; Ratcliffe, J.M. Evolutionary escalation: The bat-moth arms race. J. Exp. Biol. 2016, 219, 1589–1602. [Google Scholar] [CrossRef]
- Barber, J.R.; Conner, W.E. Acoustic mimicry in a predator-prey interaction. Proc. Natl. Acad. Sci. USA 2007, 104, 9331–9334. [Google Scholar] [CrossRef]
- Corcoran, A.J.; Conner, W.E. Predator counteradaptations: Stealth echolocation overcomes insect sonar-jamming and evasive-manoeuvring defences. Anim. Behav. 2017, 132, 291–301. [Google Scholar] [CrossRef]
- Wei, L.; Han, N.; Zhang, L.; Helgen, K.M.; Parsons, S.; Zhou, S.; Zhang, S. Wing morphology, echolocation calls, diet and emergence time of black-bearded tomb bats (Taphozous melanopogon, Emballonuridae) from southwest China. Acta Chiropterologica 2008, 10, 51–59. [Google Scholar] [CrossRef]

| Guild/Species | Species Code | Forearm Length (mm) | Mass (g) | Peak Frequency (kHz) | Duration (ms) | Bandwidth (kHz) | |
|---|---|---|---|---|---|---|---|
| Open space | |||||||
| Taphozous melanopogan | Tame | 61.0–65.0 1 | 23.0–28.0 1 | 26.0–30.0 2 | 3.7–20.2 2 | 2.0–6.0 2 | |
| Edge space | |||||||
| Miniopterus australis | Miau | 34.5–39.6 3 | 4.8–6.3 3 | 62.0–73.0 2 | 2.5–13.0 2 | 7.0–29.0 2 | |
| Miniopterus schreibersi | Misc | 40.1–45.7 3 | 4.7–12.6 3 | 45.6 ± 0.7 4 | |||
| Miniopterus tristis | Mitr | 51.0–54.0 1 | 16.0–21.0 1 | 34.6 ± 0.8 4 | |||
| Pipistrellus javanicus | Pija | 31.7–35.0 5 | 5.3–8.0 5 | 42.9–50.7 6 | 1.4–6.4 6 | 9.8–23.4 6 | |
| Scotophilus kuhlii | Scku | 47.1–51.5 5 | 17.5–26.0 5 | 38.0–46.0 7 | 3.5–12.0 7 | 7.0–34.0 7 | |
| Narrow space | |||||||
| Hipposideros diadema | Hidi | 77.0–88.0 1 | 33.0–54.0 1 | 68.0–70.0 8 | 8.0–14.3 8 | 0.0–10.7 8 | |
| Myotis horsfieldii | Miho | 35.0–38.0 1 | 5.5–7.0 1 | 47.8–59.5 9 | 2.3–6.4 9 | 12.7–37.1 9 | |
| Myotis rufopictus | Myru | 50.3 5 | 14.0 5 | 46.8–53.7 5 | 0.7–3.8 5 | 13.7–35.2 5 | |
| Rhinolophus arcuatus | Rhar | 40.0–49.0 1 | 6.0–9.0 1 | 46.8–50.0 8 | 23.0–97.5 8 | 0.0–3.9 8 | |
| Rhinolophus macrotis | Rhma | 44.0–50.0 1 | 6.0–8.0 1 | 46.8–50.0 8 | 23.0–97.5 8 | 0.0–3.9 8 | |
| Guild/Taxon | Total Passes | Relative Bat Activity (Relative Feeding Activity) | ||||||
|---|---|---|---|---|---|---|---|---|
| 2015 | 2016 | |||||||
| Water | Early | Late | Water | Early | Late | Grass | ||
| Edge-space | 34,301 | 72.88 (2.13) | 74.40 (4.39) | 37.86 (0.62) | 82.17 (2.33) | 94.93 (4.37) | 33.44 (0.98) | 10.00 (0.08) |
| Miau | 400 | 1.52 (0.02) | 0.82 (0.03) | 0.52 | 1.25 (0.08) | 0.78 (0.02) | 0.71 | 0.25 |
| Narrow-space | 606 | 3.23 (0.23) | 0.84 (0.03) | 1.35 (0.04) | 0.25 | 0.84 (0.10) | 0.58 (0.01) | 0.63 |
| Myru/Myho | 426 | 2.19 (0.31) | 0.66 (0.03) | 1.10 (0.04) | 0.17 | 0.66 (0.10) | 0.29 | 0.33 |
| Hidi | 84 | 0.60 | 0.10 | 0.18 | 0.08 | 0.10 | 0.09 | 0.04 |
| Rhar | 11 | -- | 0.03 | 0.02 | -- | 0.03 | 0.03 | -- |
| Rhma | 81 | 0.02 | 0.18 | 0.23 | -- | 0.08 | 0.20 | 0.25 |
| Rhsp | 4 | -- | 0.04 | 0.01 | 0.08 | -- | -- | -- |
| Open-space | 2840 | 1.54 | 5.06 (0.01) | 1.48 | 0.92 | 4.11 (0.13) | 2.79 | 0.25 (0.04) |
| Response Variable | Independent Variable | 2015 | 2016 | ||||
|---|---|---|---|---|---|---|---|
| Estimate (± SE) | Test statistic § | P | Estimate (± SE) | Test statistic § | P | ||
| Edge−space occurrence | |||||||
| Stage | −0.36 ± 0.04 | 65.70 | *** | −0.37 ± 0.06 | 77.29 | *** | |
| Hour | −0.03 ± 0.01 | 58.11 | *** | −0.03 ± 0.01 | 19.55 | *** | |
| Stage*Hour | 0.03 ±0.01 | 15.99 | *** | 0.01 ± 0.01 | 3.67 | n.s. | |
| Edge−space activity | |||||||
| Stage | −0.70 ± 0.11 | 44.97 | *** | −0.86 ± 0.12 | 98.20 | *** | |
| Hour | −0.08 ± 0.01 | 49.96 | *** | −0.02 ± 0.01 | 1.31 | n.s. | |
| Stage*Hour | 0.05 ± 0.02 | 11.20 | *** | 0.04 ± 0.02 | 6.78 | ** | |
| Edge−space foraging | |||||||
| Stage | −2.90 ± 0.70 | −4.12 | *** | −3.37 ± 0.78 | −4.33 | *** | |
| Hour | −0.20 ± 0.06 | −3.11 | ** | −0.19 ± 0.07 | −2.72 | ** | |
| Stage*Hour | 0.21 ± 0.09 | 2.30 | * | 0.19 ± 0.10 | 1.92 | n.s. | |
| Narrow−space presence | |||||||
| Stage | 0.29 ± 0.59 | 0.48 | n.s. | −0.04 ± 0.68 | −0.07 | n.s. | |
| Hour | 0.02 ± 0.05 | 0.49 | n.s. | −0.04 ± 0.06 | −0.61 | n.s. | |
| Stage*Hour | −0.09 ± 0.08 | −1.14 | n.s. | −0.07 ± 0.09 | −0.72 | n.s. | |
| Open−space presence | |||||||
| Stage | −1.98 ± 0.72 | −2.77 | ** | −1.79 ± 0.85 | −2.10 | * | |
| Hour | 0.09 ± 0.07 | 1.28 | n.s. | 0.06 ± 0.07 | 0.93 | n.s. | |
| Stage*Hour | 0.10 ± 0.10 | 1.01 | n.s. | 0.04 ± 0.11 | 0.45 | n.s. | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sedlock, J.L.; Stuart, A.M.; Horgan, F.G.; Hadi, B.; Como Jacobson, A.; Alviola, P.A.; Alvarez, J.D.V. Local-Scale Bat Guild Activity Differs with Rice Growth Stage at Ground Level in the Philippines. Diversity 2019, 11, 148. https://doi.org/10.3390/d11090148
Sedlock JL, Stuart AM, Horgan FG, Hadi B, Como Jacobson A, Alviola PA, Alvarez JDV. Local-Scale Bat Guild Activity Differs with Rice Growth Stage at Ground Level in the Philippines. Diversity. 2019; 11(9):148. https://doi.org/10.3390/d11090148
Chicago/Turabian StyleSedlock, Jodi L., Alexander M. Stuart, Finbarr G. Horgan, Buyung Hadi, Angela Como Jacobson, Phillip A. Alviola, and James D. V. Alvarez. 2019. "Local-Scale Bat Guild Activity Differs with Rice Growth Stage at Ground Level in the Philippines" Diversity 11, no. 9: 148. https://doi.org/10.3390/d11090148
APA StyleSedlock, J. L., Stuart, A. M., Horgan, F. G., Hadi, B., Como Jacobson, A., Alviola, P. A., & Alvarez, J. D. V. (2019). Local-Scale Bat Guild Activity Differs with Rice Growth Stage at Ground Level in the Philippines. Diversity, 11(9), 148. https://doi.org/10.3390/d11090148






