An Alien Invader is the Cause of Homogenization in the Recipient Ecosystem: A Simulation-Like Approach
Abstract
1. Introduction
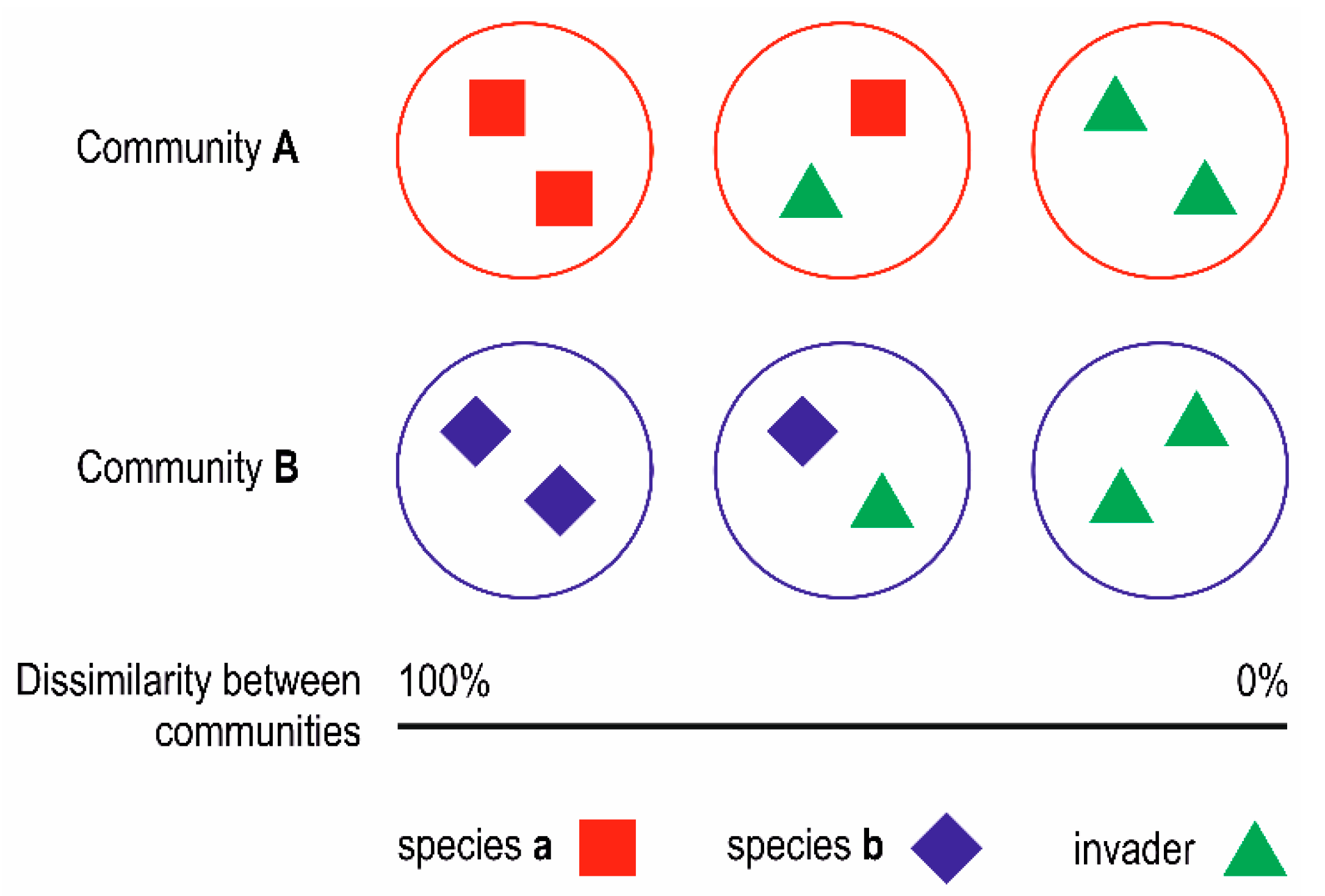
2. Materials and Methods
2.1. The Invader
2.2. Study Area
2.3. Data Sources and Management
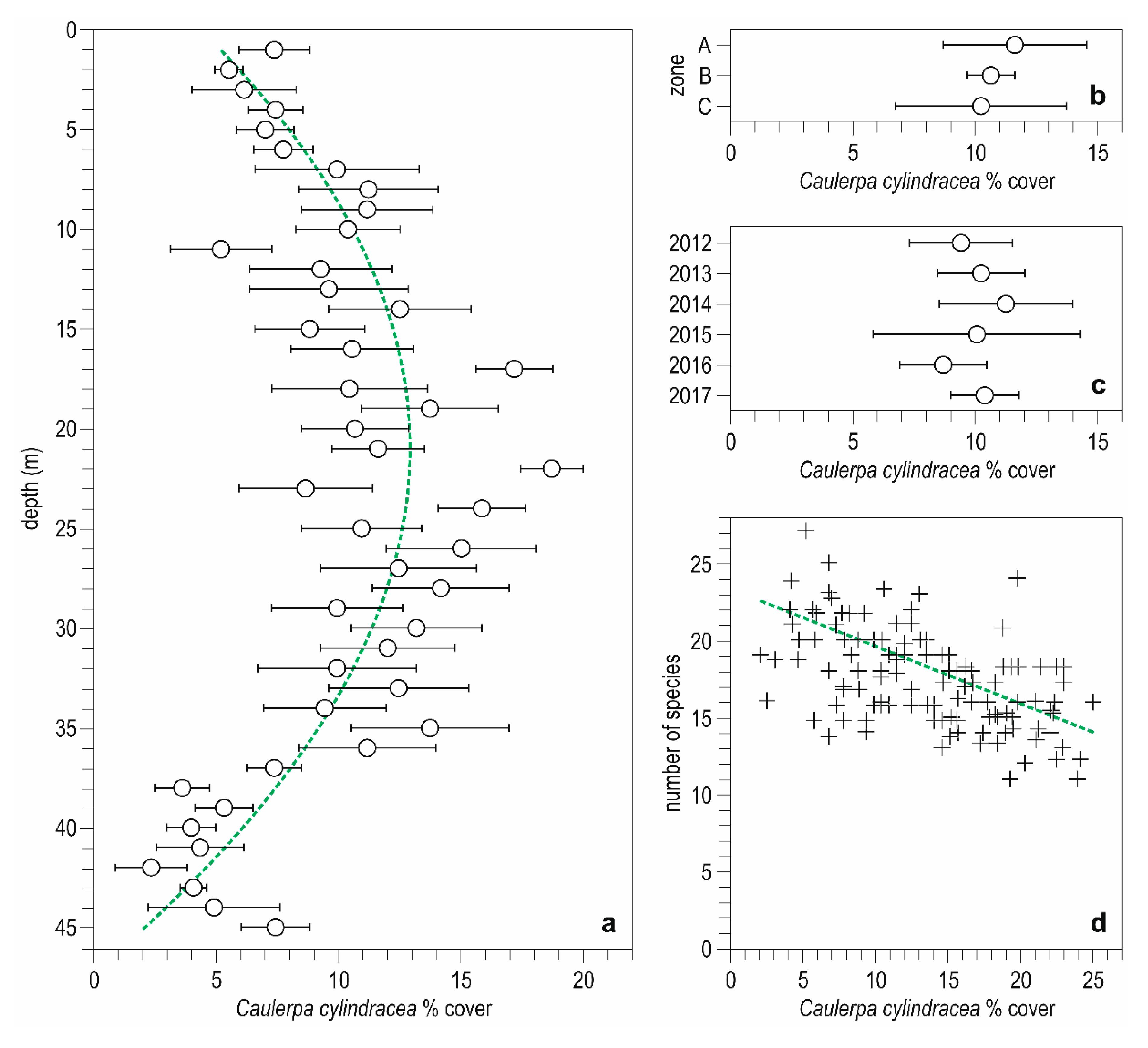
3. Results
3.1. Descriptive Analysis
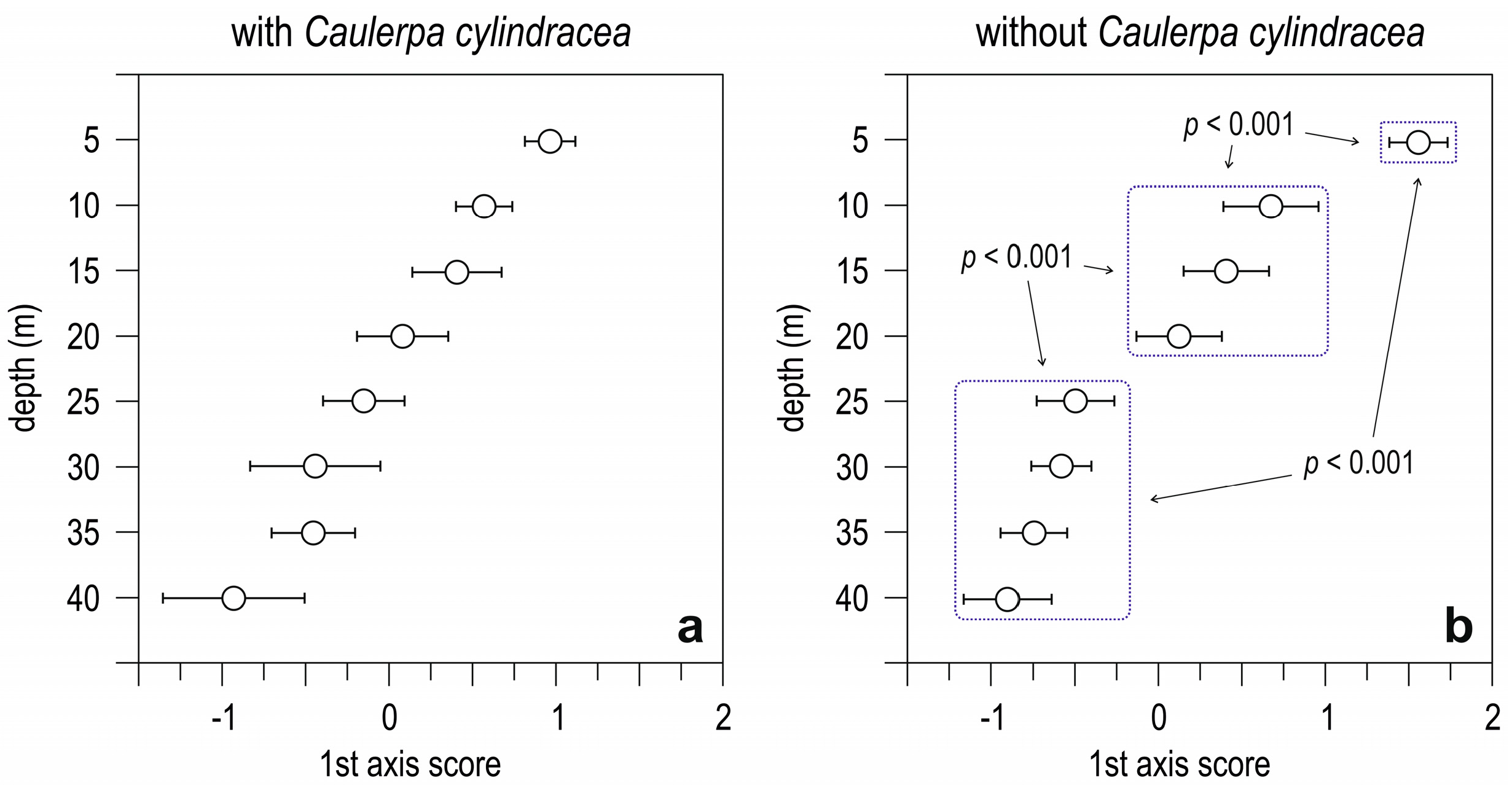
3.2. Simulation-Like Approach
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Drake, J.A.; di Castri, F.; Groves, R.H.; Kruger, F.J.; Rejmánek, M.; Williamson, M. Biological Invasions, a Global Perspective; John Wiley & Sons: Chichester, UK, 1989. [Google Scholar]
- Blackburn, T.M.; Pyšek, P.; Bacher, S.; Carlton, J.T.; Duncan, R.P.; Jarošík, V.; Wilson, J.R.U.; Richardson, D.M. A proposed unified framework for biological invasions. Trends Ecol. Evol. 2011, 26, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Occhipinti-Ambrogi, A.; Savini, D. Biological invasions as a component of global change in stressed marine ecosystems. Mar. Pollut. Bull. 2003, 46, 542–551. [Google Scholar] [CrossRef]
- Stock, A.; Crowder Larry, B.; Halpern, B.S.; Micheli, F. Uncertainty analysis and robust areas of high and low modeled human impact on the global oceans. Conserv. Biol. 2018, 32, 1368–1379. [Google Scholar] [CrossRef] [PubMed]
- Lowry, E.; Rollinson, E.J.; Laybourn, A.J.; Scott, T.E.; Aiello-Lammens, M.E.; Gray, S.M.; Mickley, J.; Gurevitch, J. Biological invasions: A field synopsis, systematic review, and database of the literature. Ecol. Evol. 2013, 3, 182–196. [Google Scholar] [CrossRef] [PubMed]
- Lewis, S.L.; Maslin, M.A. Defining the Anthropocene. Nature 2015, 519, 171–180. [Google Scholar] [CrossRef] [PubMed]
- Anthony, L. The Aliens among Us: How Invasive Species Are Transforming the Planet- and Ourselves; University Press: New Haven, CT, USA, 2017. [Google Scholar]
- Boudouresque, C.F.; Verlaque, M. Biological pollution in the Mediterranean Sea: Invasive versus introduced macrophytes. Mar. Pollut. Bull. 2002, 44, 32–38. [Google Scholar] [CrossRef]
- Kuhlenkamp, R.; Kind, B. Introduction of non-indigenous species. In Handbook on Marine Environment Protection; Salomon, M., Markus, T., Eds.; Springer: Cham, Switzerland, 2018; Chapter 25; pp. 487–516. [Google Scholar]
- Ehrenfeld, J.G. Ecosystem consequences of biological invasions. Annu. Rev. Ecol. Evol. Syst. 2010, 41, 59–80. [Google Scholar] [CrossRef]
- Fox, C.W. Towards a mechanistic understanding of global change ecology. Funct. Ecol. 2018, 32, 1648–1651. [Google Scholar] [CrossRef]
- Katsanevakis, S.; Wallentinus, I.; Zenetos, A.; Leppäkoski, E.; Çinar, M.E.; Oztürk, B.; Grabowski, M.; Golani, D.; Cardoso, A.C. Impacts of invasive alien marine species on ecosystem services and biodiversity: A pan-European review. Aquat. Invasions 2014, 9, 391–423. [Google Scholar] [CrossRef]
- Elton, C. The Ecology of Invasions by Animals and Plants; Chapman and Hall: London, UK, 1958. [Google Scholar]
- Richardson, D.M.; Pyšek, P.; Carlton, J. A compendium of essential concepts and terminology in invasion ecology. In Fifty Years of Invasion Ecology—The Legacy of Charles Elton; Richardson, D.M., Ed.; John Wiley & Sons Ltd.: Oxford, UK, 2011; pp. 409–420. [Google Scholar]
- Carlton, J.T. The scale and ecological consequences of biological invasions in the World’s oceans. In Invasive Species and Biodiversity Management; Sandlund, O.T., Schei, P.J., Viken, A., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1999; pp. 195–212. [Google Scholar]
- Occhipinti-Ambrogi, A.; Sheppard, C. Marine bioinvasions: A collection of reviews. Mar. Pollut. Bull. 2007, 55, 299–402. [Google Scholar] [CrossRef]
- Thomsen, M.S.; Wernberg, T.; Tuya, F.; Silliman, B.R. Evidence for impacts of nonindigenous macroalgae: A meta-analysis of experimental field studies. J. Phycol. 2009, 45, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, M.S.; Wernberg, T.; Schiel, D. Invasions by non-indigenous species. In Marine Ecosystems, Human Impacts on Biodiversity, Functioning and Services, 1st ed.; Crowe, T.P., Frid, C.L.J., Eds.; Cambridge University Press: Cambridge, UK, 2015; pp. 274–331. [Google Scholar]
- Thomsen, M.S.; Wernberg, T.; South, P.M.; Schiel, D.R. Non-native seaweeds drive changes in marine coastal communities around the world. In Seaweed Phylogeography; Hu, Z.-M., Fraser, C., Eds.; Springer: Dordrecht, The Netherlands, 2016; pp. 147–185. [Google Scholar]
- Galil, B.S.; Marchini, A.; Occhipinti-Ambrogi, A. Mare nostrum, mare quod invaditur: The history of bioinvasions in the Mediterranean Sea. In Histories of Bioinvasions in the Mediterranean. Environmental History; Queiroz, A., Pooley, S., Eds.; Springer: Cham, Switzerland, 2018; Chapter 2; Volume 8, pp. 21–49. [Google Scholar]
- Dar, P.A.; Reshi, Z.A. Do alien plant invasions cause biotic homogenization of terrestrial ecosystems in the Kashmir Valley, India? Trop. Ecol. 2015, 56, 111–123. [Google Scholar]
- Lososová, Z.; Chytrý, M.; Danihelka, J.; Tichý, L.; Ricotta, C. Biotic homogenization of urban floras by alien species: The role of species turnover and richness differences. J. Veg. Sci. 2016, 27, 452–459. [Google Scholar] [CrossRef]
- Price, E.P.F.; Spyreas, G.; Matthews, J.W. Biotic homogenization of regional wetland plant communities within short time-scales in the presence of an aggressive invader. J. Ecol. 2018, 106, 1180–1190. [Google Scholar] [CrossRef]
- Olden, J.D.; Rooney, T.P. On defining and quantifying biotic homogenization. Glob. Ecol. Biogeogr. 2006, 15, 113–120. [Google Scholar] [CrossRef]
- Smart, S.M.; Thompson, K.; Marrs, R.H.; Le Duc, M.G.; Maskell, L.C.; Firbank, L.G. Biotic homogenization and changes in species diversity across human-modified ecosystems. Proc. R. Soc. B 2006, 273, 2659–2665. [Google Scholar] [CrossRef]
- Vankleunen, M.; Dawson, W.; Schlaepfer, D.; Jeschke, J.M.; Fischer, M. Are invaders different? A conceptual framework of comparative approaches for assessing determinants of invasiveness. Ecol. Lett. 2010, 13, 947–958. [Google Scholar]
- Sol, D.; Maspons, J.; Vall-Llosera, M.; Bartomeus, I.; García-Peña, G.E.; Piñol, J.; Freckleton, R.P. Unraveling the life history of successful invaders. Science 2012, 337, 580–583. [Google Scholar] [CrossRef]
- Thomsen, M.S.; Olden, J.D.; Wernberg, T.; Griffin, J.N.; Silliman, B.R. A broad framework to organize and compare ecological invasion impacts. Environ. Res. 2011, 111, 899–908. [Google Scholar] [CrossRef]
- Thomsen, M.S.; Wernberg, T.; Olden, J.D.; Griffin, J.N.; Silliman, B.R. A framework to study the context-dependent impacts of marine invasions. J. Exp. Mar. Biol. Ecol. 2011, 400, 322–327. [Google Scholar] [CrossRef]
- Streftaris, N.; Zenetos, A. Alien marine species in the Mediterranean-the 100 ‘worst invasives’ and their impact. Mediterr. Mar. Sci. 2006, 7, 87–118. [Google Scholar] [CrossRef]
- Katsanevakis, S.; Tempera, F.; Teixeira, H. Mapping the impact of alien species on marine ecosystems: The Mediterranean Sea case study. Divers. Distrib. 2016, 22, 694–707. [Google Scholar] [CrossRef]
- Gatti, G.; Bianchi, C.N.; Montefalcone, M.; Venturini, S.; Diviacco, G.; Morri, C. Observational information on a temperate reef community helps understanding the marine climate and ecosystem shift of the 1980–1990s. Mar. Pollut. Bull. 2017, 114, 528–538. [Google Scholar] [CrossRef]
- Thomsen, M.S.; Wernberg, T.; South, P.M.; Schiel, D.R. To include or not to include (the invader in community analyses)? That is the question. Biol. Invasions 2016, 18, 1515–1521. [Google Scholar] [CrossRef]
- Verlaque, M.; Durand, C.; Huisman, J.M.; Boudouresque, C.F.; Le Parco, Y. On the identity and origin of the Mediterranean invasive Caulerpa racemosa (Caulerpales, Chlorophyta). Eur. J. Phycol. 2003, 38, 325–329. [Google Scholar] [CrossRef]
- Belton, G.S.; Prud’homme van Reine, W.F.; Huisman, J.M.; Draisma, S.G.A.; Gurgel, C.F.D. Resolving phenotypic plasticity and species designation in the morphologically challenging Caulerpa racemosa-peltata complex (Chlorophyta, Caulerpaceae). J. Phycol. 2014, 50, 32–54. [Google Scholar] [CrossRef]
- Klein, J.; Verlaque, M. The Caulerpa racemosa invasion: A critical review. Mar. Pollut. Bull. 2008, 56, 205–225. [Google Scholar] [CrossRef]
- Verlaque, M.; Boudouresque, C.F.; Meinesz, A.; Gravez, V. The Caulerpa racemosa complex (Caulerpales, Ulvophyceae) in the Mediterranean Sea. Bot. Mar. 2000, 43, 49–68. [Google Scholar] [CrossRef]
- Piazzi, L.; Meinesz, A.; Verlaque, M.; Akçali, B.; Antolić, B.; Argyrou, M.; Balata, D.; Ballesteros, E.; Calvo, S.; Cinelli, F.; et al. Invasion of Caulerpa racemosa var. cylindracea (Caulerpales, Chlorophyta) in the Mediterranean Sea: An assessment of the spread. Cryptogam. Algol. 2005, 26, 189–202. [Google Scholar]
- Zenetos, A.; Gofas, S.; Verlaque, M.; Çinar, M.E.; García Raso, J.E.; Bianchi, C.N.; Morri, C.; Azzurro, E.; Bilecenoglu, M.; Froglia, C.; et al. Alien species in the Mediterranean Sea by 2010. A contribution to the application of European Union’s Marine Strategy Framework Directive (MSFD). Part 1. Spatial distribution. Mediterr. Mar. Sci. 2010, 11, 381–493. [Google Scholar] [CrossRef]
- Montefalcone, M.; Morri, C.; Parravicini, V.; Bianchi, C.N. A tale of two invaders: Divergent spreading kinetics of the alien green algae Caulerpa taxifolia and Caulerpa cylindracea. Biol. Invasions 2015, 17, 2717–2728. [Google Scholar] [CrossRef]
- Mannino, A.M.; Balistreri, P. An updated overview of invasive Caulerpa taxa in Sicily and circum-Sicilian Islands, strategic zones within the NW Mediterranean Sea. Flora Mediterr. 2017, 27, 221–240. [Google Scholar]
- Verlaque, M.; Afonso-Carrillo, J.; Gil-Rodriguez, M.C.; Durand, C.; Boudouresque, C.F.; Le Parco, Y. Blitzkrieg in a marine invasion: Caulerpa racemosa var. cylindracea (Bryopsidales, Chlorophyta) reaches the Canary Islands (NE Atlantic). Biol. Invasions 2004, 6, 269–281. [Google Scholar]
- Zenetos, A.; Gofas, S.; Morri, C.; Rosso, A.; Violanti, D.; García Raso, J.E.; Çinar, M.E.; Almogi-Labin, A.; Ates, A.S.; Azzurro, E.; et al. Alien species in the Mediterranean Sea by 2010. A contribution to the application of European Union’s Marine Strategy Framework Directive (MSFD). Part 2. Introduction trends and pathways. Mediterr. Mar. Sci. 2012, 13, 328–352. [Google Scholar] [CrossRef]
- Cantasano, N.; Pellicone, G.; Di Martino, V. The spread of Caulerpa cylindracea in Calabria (Italy) and the effects of shipping activities. Ocean Coast. Manag. 2017, 144, 51–58. [Google Scholar] [CrossRef]
- Raniello, R.; Lorenti, M.; Brunet, C.; Buia, M.C. Photoacclimation of the invasive alga Caulerpa racemosa var. cylindracea to depth and daylight patterns and a putative new role for siphonaxanthin. Mar. Ecol. 2006, 27, 20–30. [Google Scholar]
- Dumay, O.; Pergent, G.; Pergent-Martini, C.; Amade, P. Variations in caulerpenyne contents in Caulerpa taxifolia and Caulerpa racemosa. J. Chem. Ecol. 2002, 28, 343–352. [Google Scholar] [CrossRef]
- Argyrou, M.; Demetropoulos, A.; Hadjichristophorou, M. Expansion of the macroalga Caulerpa racemosa and changes in soft-bottom macrofaunal assemblages in Moni Bay, Cyprus. Oceanol. Acta 1999, 22, 517–528. [Google Scholar] [CrossRef]
- Piazzi, L.; Ceccherelli, G.; Balata, D.; Cinelli, F. Early patterns of Caulerpa racemosa recovery in the Mediterranean Sea: The influence of algal turfs. J. Mar. Biol. Assoc. UK 2003, 83, 27–29. [Google Scholar] [CrossRef]
- Piazzi, L.; Balata, D.; Cinelli, F. Invasions of alien macroalgae in Mediterranean coralligenous assemblages. Cryptogam. Algol. 2007, 28, 289–301. [Google Scholar]
- Capiomont, A.; Breugnot, E.; Den Haan, M.; Meinesz, A. Phenology of a deep water population of Caulerpa racemosa in the northwestern Mediterranean Sea. Bot. Mar. 2005, 48, 80–83. [Google Scholar] [CrossRef]
- Oprandi, A.; Montefalcone, M.; Ferrari, M.; Morri, C.; Bianchi, C.N. Invasion of the alien green alga Caulerpa racemosa and phase shift within the Posidonia oceanica seagrass meadow of Bergeggi. Biol. Mar. Mediterr. 2014, 21, 101–104. [Google Scholar]
- Balistreri, P.; Mannino, A.M. Preliminary data on the occurrence of alien macroalgae in the vermetid reef along the coasts of Favignana Island (Southern Tyrrhenian Sea). Biodivers. J. 2017, 8, 105–112. [Google Scholar]
- Katsanevakis, S.; Issaris, Y.; Poursanidis, D.; Thessalou-Legaki, M. Vulnerability of marine habitats to the invasive green alga Caulerpa racemosa var. cylindracea within a marine protected area. Mar. Environ. Res. 2010, 70, 210–218. [Google Scholar]
- Montefalcone, M.; Albertelli, G.; Morri, C.; Bianchi, C.N. Pattern of wide-scale substitution within Posidonia oceanica meadows of NW Mediterranean Sea: Invaders are stronger than natives. Aquat. Conserv. 2010, 20, 507–515. [Google Scholar] [CrossRef]
- Gennaro, P.; Piazzi, L. The indirect role of nutrients in enhancing the invasion of Caulerpa racemosa var. cylindracea. Biol. Invasions 2013, 16, 1709–1717. [Google Scholar] [CrossRef]
- Piazzi, L.; Ceccherelli, G.; Cinelli, F. Threat to macroalgal diversity: Effects of the introduced green alga Caulerpa racemosa in the Mediterranean. Mar. Ecol. Progr. Ser. 2001, 210, 161–165. [Google Scholar] [CrossRef]
- Ceccherelli, G.; Campo, E.D. Different effects of Caulerpa racemosa on two co-occurring seagrasses. Bot. Mar. 2002, 45, 71–76. [Google Scholar] [CrossRef]
- Thomsen, M.S.; Wernberg, T.; Engelen, A.H.; Tuya, F.; Vanderklift, M.A.; Holmer, M.; McGlathery, K.J.; Arenas, F.; Kotta, J.; Silliman, B.R. A meta-analysis of seaweed impacts on seagrasses: Generalities and knowledge gaps. PLoS ONE 2012, 7, e28595. [Google Scholar] [CrossRef]
- Morri, C.; Bianchi, C.N.; Damiani, V.; Peirano, A.; Romeo, G.; Tunesi, L. The marine environment between Punta della Chiappa and Sestri Levante (Ligurian Sea): Ecotipological outline and proposal of a biocenotic map. Boll. Mus. Ist Biol. Univ. Genova 1986, 52, 213–231. [Google Scholar]
- Bussotti, S.; Conti, M.; Guidetti, P.; Martini, F.; Matricardi, G. First record of Caulerpa racemosa (Forsskål) J. Agardh along the coast of Genoa (North-Western Mediterranean). Doriana 1996, 6, 1–5. [Google Scholar]
- Modena, M.; Matricardi, G.; Vacchi, M.; Guidetti, P. Spreading of Caulerpa racemosa (Forsskål) J. Agardh (Bryopsidaceae, Chlorophyta) along the coasts of the Ligurian Sea. Cryptogam. Algol. 2000, 21, 301–304. [Google Scholar]
- Paoli, C.; Vassallo, P.; Dapueto, G.; Fanciulli, G.; Massa, F.; Venturini, S.; Povero, P. The economic revenues and the emergy costs of cruise tourism. J. Clean. Prod. 2017, 166, 1462–1478. [Google Scholar] [CrossRef]
- Bianchi, C.N.; Pronzato, R.; Cattaneo-Vietti, R.; Benedetti-Cecchi, L.; Morri, C.; Pansini, M.; Chemello, R.; Milazzo, M.; Fraschetti, S.; Terlizzi, A.; et al. Mediterranean marine benthos: A manual of methods for its sampling and study. 6: Hard bottoms. Biol. Mar. Mediterr. 2004, 11, 185–215. [Google Scholar]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PaSt: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 4. [Google Scholar]
- Peres-Neto, P.R.; Jackson, D.A.; Somers, K.M. Giving meaningful interpretation to ordination axes: Assessing loading significance in principal component analysis. Ecology 2003, 84, 2347–2363. [Google Scholar] [CrossRef]
- Jackson, D.A. Stopping rules in principal components analysis: A comparison of heuristical and statistical approaches. Ecology 1993, 74, 2204–2214. [Google Scholar] [CrossRef]
- Fresi, E.; Gambi, M.C. Some important aspects of the mathematical analysis of marine ecosystems. Nat. Sicil. 1982, 6 (Suppl. 3), 449–465. [Google Scholar]
- Legendre, P.; Legendre, L. Numerical Ecology, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 1998. [Google Scholar]
- Bianchi, C.N.; Cocito, S.; Diviacco, G.; Dondi, N.; Fratangeli, F.; Montefalcone, M.; Parravicini, V.; Rovere, A.; Sgorbini, S.; Vacchi, M.; et al. The park never born: Outcome of a quarter of a century of inaction on the sea-floor integrity of a proposed but not established Marine Protected Area. Aquat. Conserv. 2018, 28, 1209–1228. [Google Scholar] [CrossRef]
- Catford, J.A.; Vesk, P.A.; Richardson, D.M.; Pyšek, P. Quantifying levels of biological invasion: Towards the objective classification of invaded and invasible ecosystems. Glob. Chang. Biol. 2012, 18, 44–62. [Google Scholar] [CrossRef]
- Bulleri, F.; Benedetti-Cecchi, L.; Ceccherelli, G.; Tamburello, L. A few is enough: A low cover of a non-native seaweed reduces the resilience of Mediterranean macroalgal stands to disturbances of varying extent. Biol. Invasions 2017, 19, 2291–2305. [Google Scholar] [CrossRef]
- Guidetti, P.; Baiata, P.; Ballesteros, E.; Di Franco, A.; Hereu, B.; Macpherson, E.; Micheli, F.; Pais, A.; Panzalis, P.; Rosenberg, A.A.; et al. Large-scale assessment of Mediterranean marine protected areas effects on fish assemblages. PLoS ONE 2014, 9, e91841. [Google Scholar] [CrossRef]
- Guidetti, P.; Sala, E. Community-wide effects of marine reserves in the Mediterranean Sea. Mar. Ecol. Progr. Ser. 2007, 335, 43–56. [Google Scholar] [CrossRef]
- Giakoumi, S.; Pey, A.; Di Franco, A.; Francour, P.; Kizilkaya, Z.; Arda, Y.; Raybaud, V.; Guidetti, P. Exploring the relationships between marine protected areas and invasive fish in the world’s most invaded sea. Ecol. Appl. 2019, 29, e01809. [Google Scholar] [CrossRef]
- Spiridonov, V.A. Introduced species challenges and opportunities for marine conservation ecology and management practices: Notes inspired by a recent MSC certification. Aquat. Conserv. 2018, 28, 522–526. [Google Scholar] [CrossRef]
- Boudouresque, C.F.; Verlaque, M. An overview of species introduction and invasion processes in marine and coastal lagoon habitats. Cah. Biol. Mar. 2012, 53, 309–317. [Google Scholar]
- Geburzi, J.C.; McCarthy, M.L. How do they do it?—Understanding the success of marine invasive species. In YOUMARES 8—Oceans across Boundaries: Learning from Each Other; Jungblut, S., Liebich, V., Bode, M., Eds.; Springer: Cham, Switzerland, 2018; pp. 109–144. [Google Scholar]
- Pyšek, P.; Jarošík, V.; Hulme, P.E.; Pergl, J.; Hejda, M.; Schaffner, U.; Vilà, M. A global assessment of invasive plant impacts on resident species, communities and ecosystems: The interaction of impact measures, invading species’ traits and environment. Glob. Chang. Biol. 2012, 18, 1725–1737. [Google Scholar] [CrossRef]
- Kumschick, S.; Gaertner, M.; Vilà, M.; Essl, F.; Jeschke, J.M.; Pyšek, P.; Ricciardi, A.; Bacher, S.; Blackburn, T.M.; Dick, J.T.A.; et al. Ecological impacts of alien species: Quantification, scope, caveats, and recommendations. BioScience 2015, 65, 55–63. [Google Scholar] [CrossRef]
- Davis, K.T.; Callaway, R.M.; Fajardo, A.; Pauchard, A.; Nuñez, M.A.; Brooker, R.W.; Maxwell, B.D.; Dimarco, R.D.; Peltzer, D.A.; Mason, B.; et al. Severity of impacts of an introduced species corresponds with regional eco-evolutionary experience. Ecography 2019, 42, 12–22. [Google Scholar] [CrossRef]
- Fridley, J.D.; Stachowicz, J.J.; Naeem, S.; Sax, D.F.; Seabloom, E.W.; Smith, M.D.; Stohlgren, T.J.; Tilman, D.; Von Holle, B. The invasion paradox: Reconciling pattern and process in species invasions. Ecology 2007, 88, 3–17. [Google Scholar] [CrossRef]
- Vilà, M.; Espinar, J.L.; Hejda, M.; Hulme, P.E.; Jarošík, V.; Maron, J.L.; Pergl, J.; Schaffner, U.; Sun, Y.; Pyšek, P. Ecological impacts of invasive alien plants: A meta-analysis of their effects on species, communities and ecosystems. Ecol. Lett. 2011, 14, 702–708. [Google Scholar] [CrossRef]
- Castro-Díez, P.; Pauchard, A.; Traveset, A.; Vilà, M. Linking the impacts of plant invasion on community functional structure and ecosystem properties. J. Veg. Sci. 2016, 27, 1233–1242. [Google Scholar] [CrossRef]
- Morri, C.; Bellan-Santini, D.; Giaccone, G.; Bianchi, C.N. Principles of bionomy: Definition of assemblages and use of taxonomic descriptors (macrobenthos). Biol. Mar. Mediterr. 2004, 11 (Suppl. 1), 573–600. [Google Scholar]
- Thrush, S.F.; Gray, J.S.; Hewitt, J.E.; Ugland, K.I. Predicting the effects of habitat homogenization on marine biodiversity. Ecol. Appl. 2006, 16, 1636–1642. [Google Scholar] [CrossRef]
- Anderson, M.J.; Ellingsen, K.E.; McArdle, B.H. Multivariate dispersion as a measure of beta diversity. Ecol. Lett. 2006, 9, 683–693. [Google Scholar] [CrossRef]
- Piazzi, L.; Balata, D. The spread of Caulerpa racemosa var. cylindracea in the Mediterranean Sea: An example of how biological invasions can influence beta diversity. Mar. Environ. Res. 2008, 65, 50–61. [Google Scholar]
- Macdougall, A.S.; Turkington, R. Are invasive species the drivers or passengers of change in degraded ecosystems? Ecology 2005, 86, 42–55. [Google Scholar] [CrossRef]
- Cucherousset, J.; Fried, G.; Cote, J.; Renault, D.; Cote, J.; Renault, D. Biological invasions and ecosystem functioning: Assessment of the ecological impacts driven by invasive species. Rev. Ecologie-Terre Vie 2015, 70, 49–52. [Google Scholar]
- South, P.M.; Thomsen, M.S. The ecological role of invading Undaria pinnatifida: An experimental test of the driver–passenger models. Mar. Biol. 2016, 163, 175. [Google Scholar] [CrossRef]
- Bulleri, F.; Balata, D.; Bertocci, I.; Tamburello, L.; Benedetti-Cecchi, L. The seaweed Caulerpa racemosa on Mediterranean rocky reefs: From passenger to driver of ecological change. Ecology 2010, 91, 2205–2212. [Google Scholar] [CrossRef]
- Boudouresque, C.F.; Blanfuné, A.; Fernandez, C.; Lejeusne, C.; Pérez, T.; Ruitton, S.; Thibault, D.; Thibaut, T.; Verlaque, M. Marine biodiversity-warming vs. biological invasions and overfishing in the Mediterranean Sea: Take care, ‘one train can hide another’. MOJ Ecol. Environ. Sci. 2017, 2, 1–13. [Google Scholar] [CrossRef]
- Piazzi, L.; Balata, D.; Bulleri, F.; Gennaro, P.; Ceccherelli, G. The invasion of Caulerpa cylindracea in the Mediterranean: The known, the unknown and the knowable. Mar. Biol. 2016, 163, 161. [Google Scholar] [CrossRef]
- Vassallo, P.; Bianchi, C.N.; Paoli, C.; Holon, F.; Navone, A.; Bavestrello, G.; CattaneoVietti, R.; Morri, C. A predictive approach to benthic marine habitat mapping: Efficacy and management implications. Mar. Pollut. Bull. 2018, 131, 218–232. [Google Scholar] [CrossRef]
- Galil, B.S.; Marchini, A.; Occhipinti-Ambrogi, A.; Minchin, D.; Narščius, A.; Ojaveer, H.; Olenin, S. International arrivals: Widespread bioinvasions in European Seas. Ethol. Ecol. Evol. 2014, 26, 152–171. [Google Scholar] [CrossRef]
- Blackburn, T.M. Macroecology and invasion biology. Glob. Ecol. Biogeogr. 2019, 28, 28–32. [Google Scholar] [CrossRef]


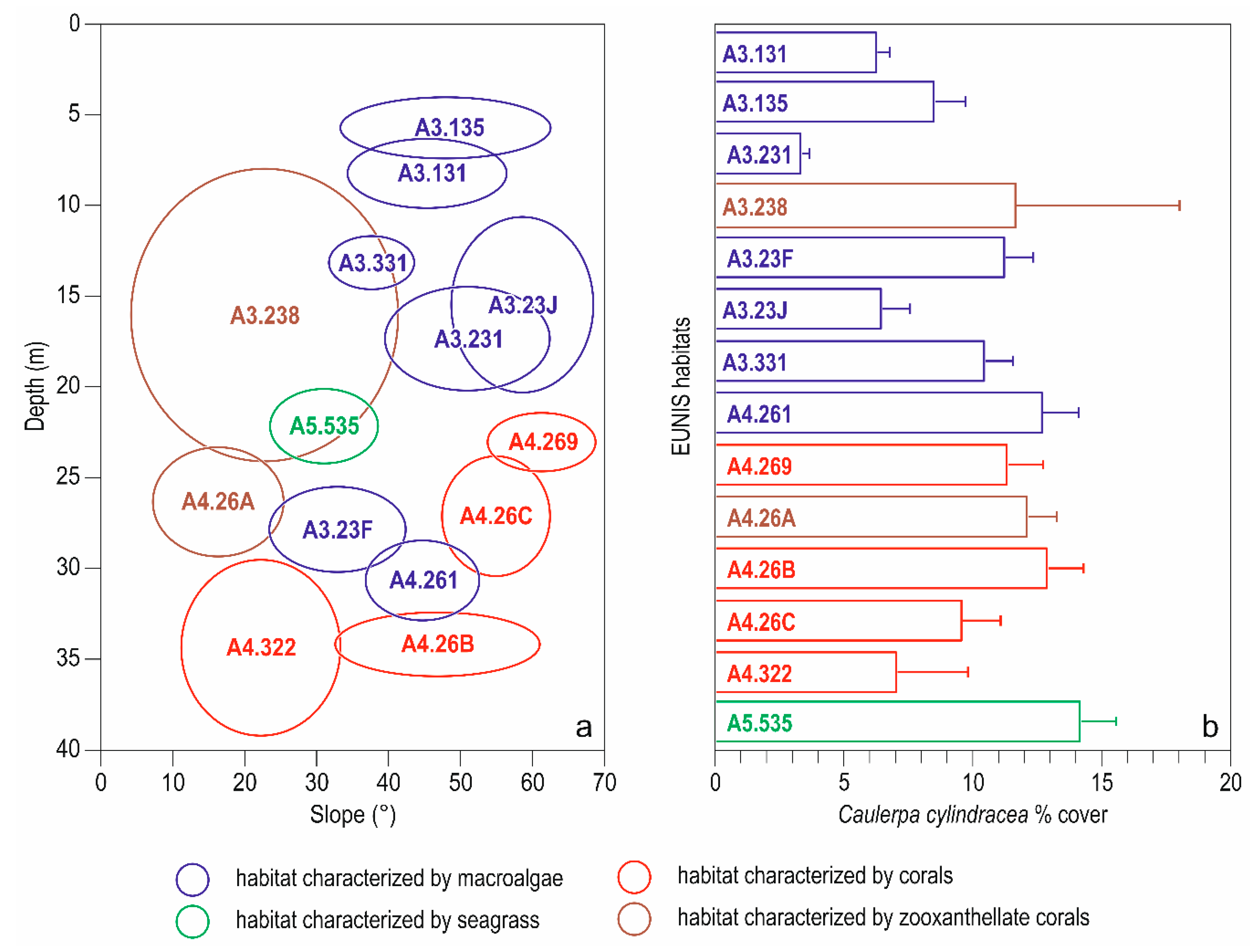

| Code | Extended Name | n |
|---|---|---|
| habitats characterized by macroalgae | ||
| A3.131 | Facies with encrusting algae (Lithophyllum incrustans) and sea urchins | 23 |
| A3.135 | Association with Ellisolandia elongata | 17 |
| A3.231 | Association with Codium vermilara | 5 |
| A3.23F | Association with Dictyopteris polypodioides | 43 |
| A3.23J | Association with Flabellia petiolata and Peyssonnelia squamaria | 17 |
| A3.331 | Association with Halopteris scoparia | 109 |
| A4.261 | Association with Cystoseira zosteroides | 32 |
| habitats characterized by seagrass | ||
| A5.535 | Posidonia oceanica beds | 31 |
| habitats characterized by zooxanthellate corals | ||
| A3.238 | Facies with Cladocora caespitosa | 16 |
| A4.26A | Facies with Eunicella singularis | 43 |
| habitats characterized by corals | ||
| A4.269 | Facies with Eunicella cavolini | 82 |
| A4.26B | Facies with Paramuricea clavata | 29 |
| A4.26C | Facies with Parazoanthus axinellae | 26 |
| A4.322 | Facies with Leptogorgia sarmentosa | 9 |
| Source of Variation | SS | df | s2 | F13,468 | p |
|---|---|---|---|---|---|
| Among habitats | 2077.91 | 13 | 159.84 | 2.45 | 0.003 |
| Within habitats | 30478.26 | 468 | 65.12 | ||
| Total | 32556.17 | 481 |
| Source of Variation | SS | df | s2 | F7,32 | p |
|---|---|---|---|---|---|
| “Observed” Dataset | |||||
| Among depths | 3.73 | 7 | 0.53 | 83.4 | 9.96 × 10−19 |
| Within depths | 0.20 | 32 | 0.01 | ||
| Total | 3.93 | 39 | |||
| “Simulated” Dataset | |||||
| Among depths | 6.42 | 7 | 0.92 | 111.1 | 1.29 × 10−20 |
| Within depths | 0.26 | 32 | 0.01 | ||
| Total | 6.68 | 39 |
| Source of Variation | SS | df | s2 | F2,37 | p |
|---|---|---|---|---|---|
| Among depth groups | 23.2559 | 2 | 11.6279 | 225.6 | 1.87 × 10−21 |
| Within depth groups | 1.9068 | 37 | 0.0515351 | ||
| Total | 25.1627 | 39 |
| Indices | “Observed” Dataset | “Simulated” Dataset |
|---|---|---|
| Average dissimilarity | 0.51 | 0.61 |
| Standard deviation | 0.16 | 0.19 |
| Coefficient of variation | 0.32 | 0.49 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morri, C.; Montefalcone, M.; Gatti, G.; Vassallo, P.; Paoli, C.; Bianchi, C.N. An Alien Invader is the Cause of Homogenization in the Recipient Ecosystem: A Simulation-Like Approach. Diversity 2019, 11, 146. https://doi.org/10.3390/d11090146
Morri C, Montefalcone M, Gatti G, Vassallo P, Paoli C, Bianchi CN. An Alien Invader is the Cause of Homogenization in the Recipient Ecosystem: A Simulation-Like Approach. Diversity. 2019; 11(9):146. https://doi.org/10.3390/d11090146
Chicago/Turabian StyleMorri, Carla, Monica Montefalcone, Giulia Gatti, Paolo Vassallo, Chiara Paoli, and Carlo Nike Bianchi. 2019. "An Alien Invader is the Cause of Homogenization in the Recipient Ecosystem: A Simulation-Like Approach" Diversity 11, no. 9: 146. https://doi.org/10.3390/d11090146
APA StyleMorri, C., Montefalcone, M., Gatti, G., Vassallo, P., Paoli, C., & Bianchi, C. N. (2019). An Alien Invader is the Cause of Homogenization in the Recipient Ecosystem: A Simulation-Like Approach. Diversity, 11(9), 146. https://doi.org/10.3390/d11090146










