Trophic Trait Evolution Explains Variation in Nutrient Excretion Stoichiometry among Panamanian Armored Catfishes (Loricariidae)
Abstract
1. Introduction
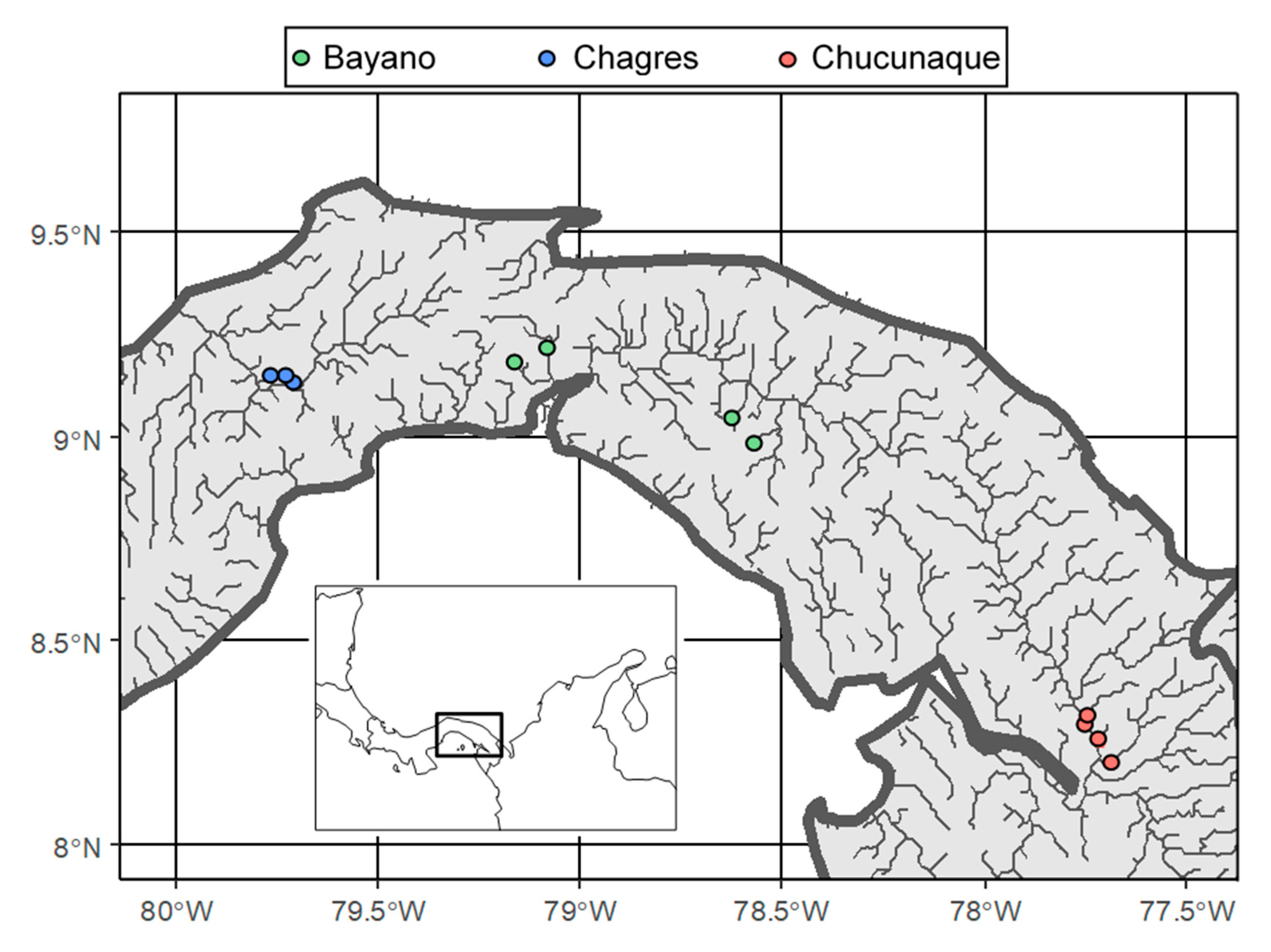
2. Methods
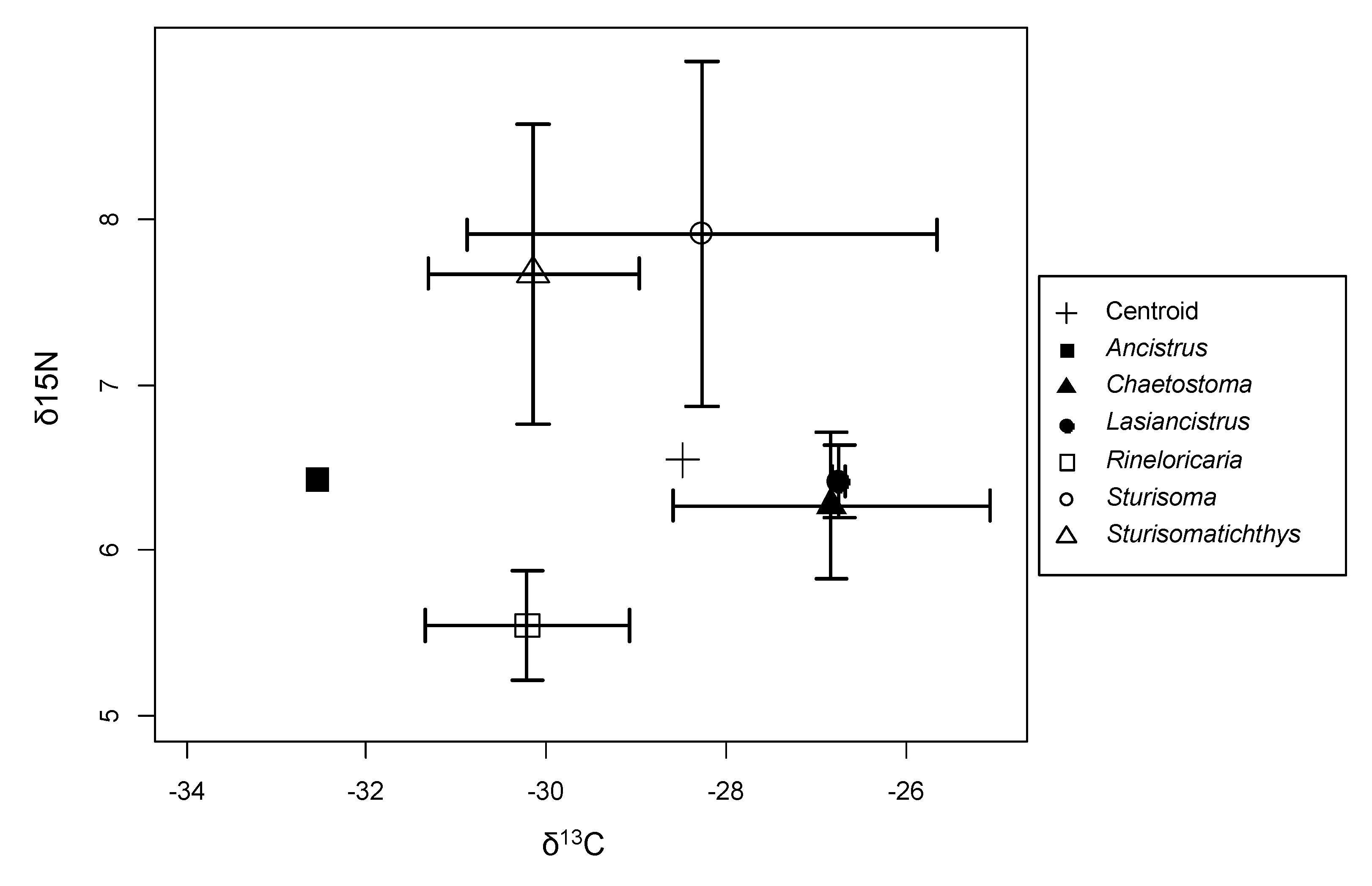
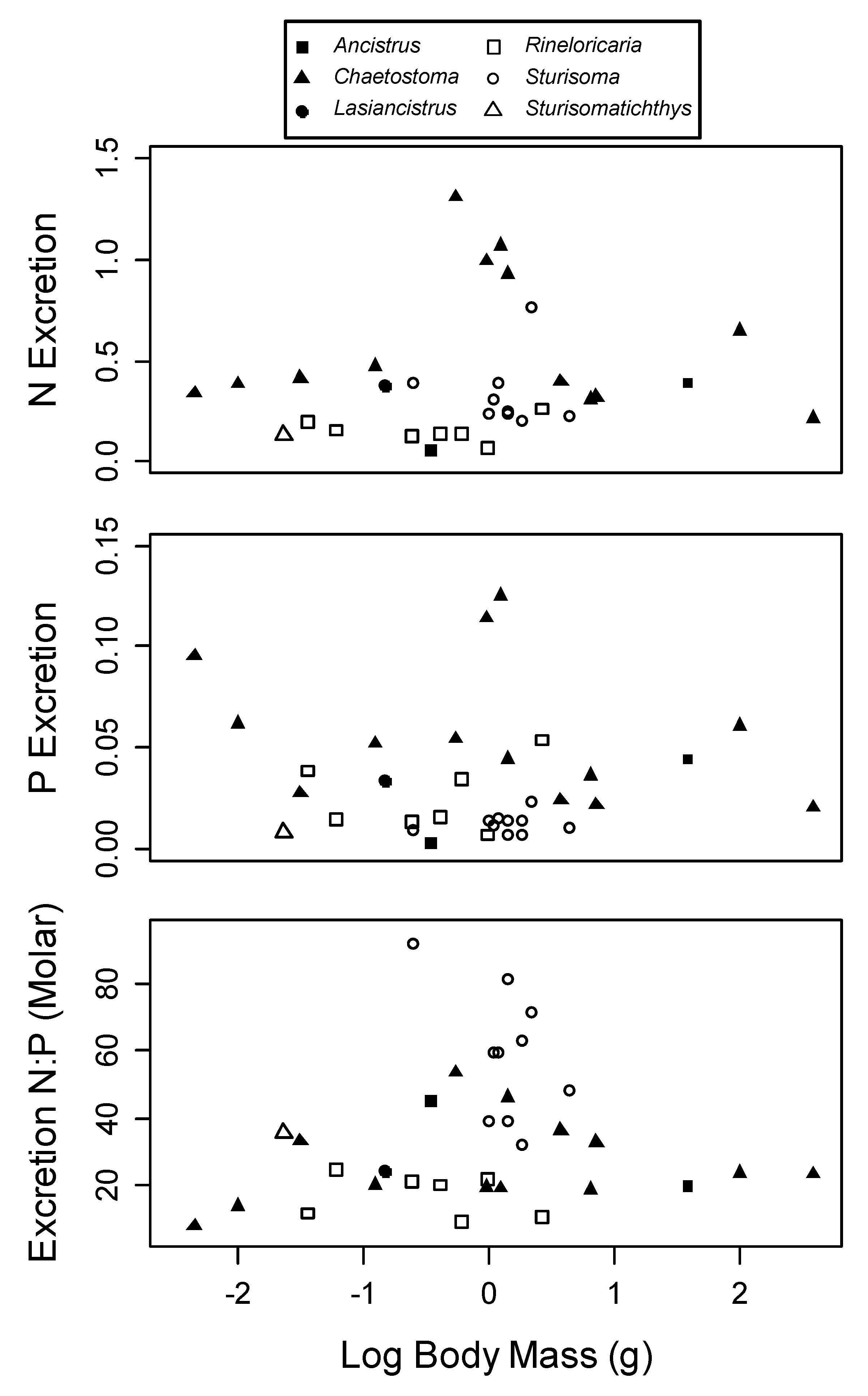
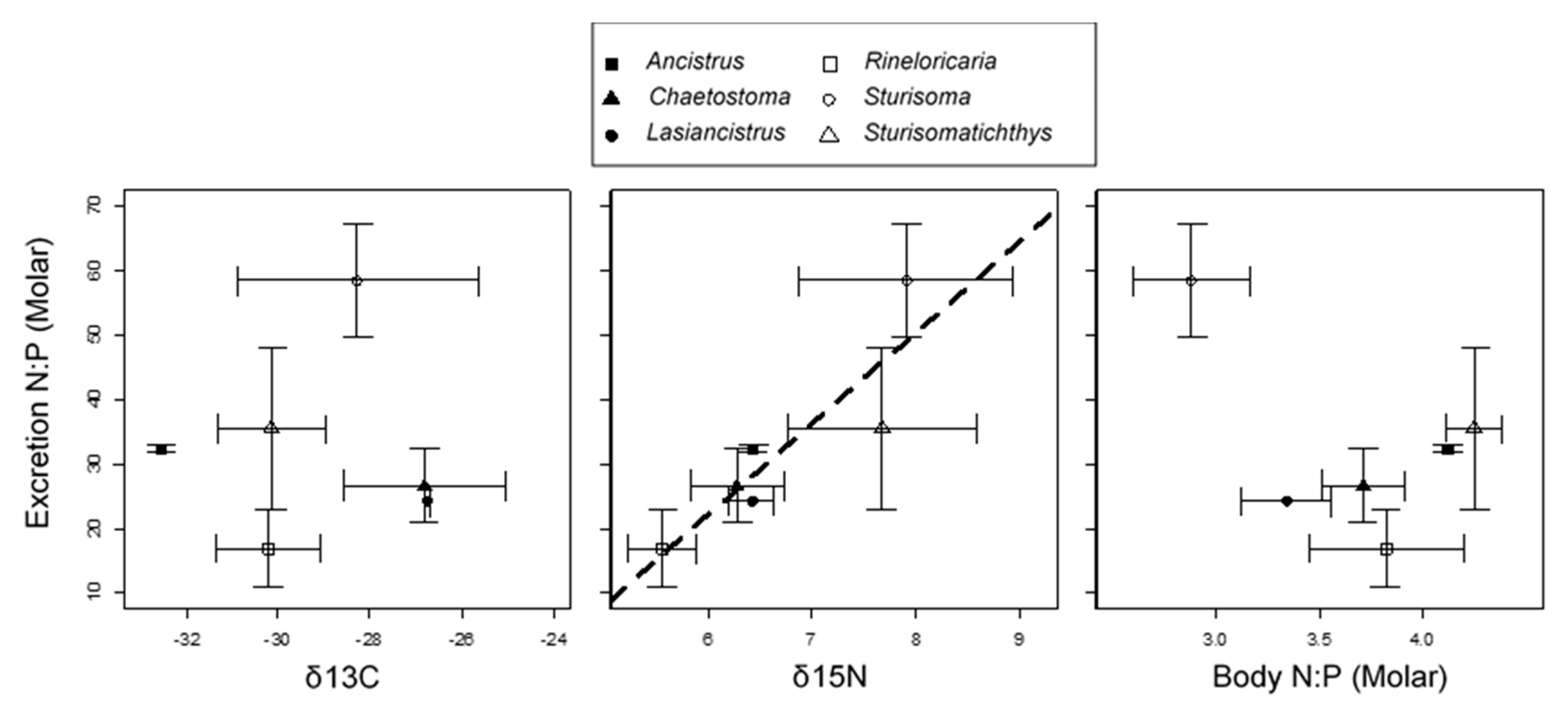
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vanni, M.J. Nutrient cycling by animals in freshwater ecosystems. Ann. Rev. Ecol. Syst. 2002, 33, 341–370. [Google Scholar] [CrossRef]
- McIntyre, P.B.; Flecker, A.S.; Vanni, M.J.; Hood, J.M.; Taylor, B.W.; Thomas, S.A. Fish distributions and nutrient cycling in streams: Can fish create biogeochemical hotspots. Ecology 2008, 89, 2335–2346. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, C.L.; Capps, K.A.; Rugenski, A.T.; Vanni, M.J. Consumer-driven nutrient dynamics in freshwater ecosystems: From individuals to ecosystems. Biol. Rev. 2017, 92, 2003–2023. [Google Scholar] [CrossRef] [PubMed]
- Sterner, R.W. The ratio of nitrogen to phosphorus resupplied by herbivores: Zooplankton and the algal competitive arena. Am. Nat. 1990, 136, 209–229. [Google Scholar] [CrossRef]
- Sterner, R.W.; Elser, J.J.; Hessen, D.O. Stoichiometric relationships among producers, consumers and nutrient cycling in pelagic ecosystems. Biogeochemistry 1992, 17, 49–67. [Google Scholar] [CrossRef]
- Vanni, M.J.; Flecker, A.S.; Hood, J.M.; Headworth, J.L. Stoichiometry of nutrient recycling by vertebrates in a tropical stream: Linking species identity and ecosystem processes. Ecol. Lett. 2002, 5, 285–293. [Google Scholar] [CrossRef]
- Evans-White, M.A.; Lamberti, G.A. Stoichiometry of consumer-driven nutrient recycling across nutrient regimes in streams. Ecol. Lett. 2006, 9, 1186–1197. [Google Scholar] [CrossRef] [PubMed]
- El-Sabaawi, R.W.; Marshall, M.C.; Bassar, R.D.; López-Sepulcre, A.; Palkovacs, E.P.; Dalton, C. Assessing the effects of guppy life history evolution on nutrient recycling: From experiments to the field. Freshw. Biol. 2015, 60, 590–601. [Google Scholar] [CrossRef]
- Allgeier, J.E.; Wenger, S.J.; Rosemond, A.D.; Schindler, D.E.; Layman, C.A. Metabolic theory and taxonomic identity predict nutrient recycling in a diverse food web. Proc. Natl. Acad. Sci. USA 2015, 112, E2640–E2647. [Google Scholar] [CrossRef]
- Mather, M.E.; Vanni, M.J.; Wissing, T.E.; Davis, S.A.; Schaus, M.H. Regeneration of nitrogen and phosphorus by bluegill and gizzard shad: Effect of feeding history. Can. J. Fish. Aquat. Sci. 1995, 52, 2327–2338. [Google Scholar] [CrossRef]
- Moody, E.K.; Corman, J.R.; Elser, J.J.; Sabo, J.L. Diet composition affects the rate and N:P ratio of fish excretion. Freshw. Biol. 2015, 60, 456–465. [Google Scholar] [CrossRef]
- Moody, E.K.; Carson, E.W.; Corman, J.R.; Espinosa-Pérez, H.; Ramos, J.; Sabo, J.L.; Elser, J.J. Consumption explains intraspecific variation in nutrient recycling stoichiometry in a desert fish. Ecology 2018, 99, 1552–1561. [Google Scholar] [CrossRef] [PubMed]
- McManamay, R.A.; Webster, J.R.; Valett, H.M.; Dolloff, C.A. Does diet influence consumer nutrient cycling? Macroinvertebrate and fish excretion in streams. J. N. Am. Benthol. Soc. 2011, 30, 84–102. [Google Scholar] [CrossRef]
- Böhning-Gaese, K.; Oberrath, R. Phylogenetic effects on morphological, life-history, behavioural and ecological traits of birds. Evol. Ecol. Res. 1999, 1, 347–364. [Google Scholar]
- González, A.; Céréghino, R.; Dézerald, O.; Farjalla, V.F.; Leroy, C.; Richardson, B.A.; Richardson, M.J.; Romero, G.Q.; Srivastava, D.S. Ecological mechanisms and phylogeny shape invertebrate stoichiometry: A test using detritus-based communities across Central and South America. Funct. Ecol. 2018, 32, 2448–2463. [Google Scholar] [CrossRef]
- Losos, J.B. Phylogenetic niche conservatism, phylogenetic signal and the relationship between phylogenetic relatedness and ecological similarity among species. Ecol. Lett. 2008, 11, 995–1007. [Google Scholar] [CrossRef] [PubMed]
- Olalla-Tárraga, M.A.; González-Suárez, M.; Bernardo-Madrid, R.; Revilla, E.; Villalobos, F. Contrasting evidence of phylogenetic trophic niche conservatism in mammals worldwide. J. Biogeogr. 2017, 44, 99–110. [Google Scholar] [CrossRef]
- Lujan, N.K.; Winemiller, K.O.; Armbruster, J.W. Trophic diversity in the evolution and community assembly of loricariid catfishes. BMC Evol. Biol. 2012, 12, 124. [Google Scholar] [CrossRef]
- Lujan, N.K.; Armbruster, J.W.; Lovejoy, N.R.; López-Fernández, H. Multilocus molecular phylogeny of the suckermouth armored catfishes (Siluriformes: Loricariidae) with a focus on subfamily Hypostominae. Mol. Phylogenet. Evol. 2015, 82, 269–288. [Google Scholar] [CrossRef]
- Meek, S.E.; Hildebrand, S.F. The fishes of the fresh-waters of Panama. Field Mus. Nat. Hist. Zool. Ser. 1916, 10, 217–374. [Google Scholar]
- Hood, J.M.; Vanni, M.J.; Flecker, A.S. Nutrient recycling by two phosphorus-rich grazing catfish: The potential for phosphorus-limitation of fish growth. Oecologia 2005, 146, 247–257. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, P.B.; Flecker, A.S. Ecological stoichiometry as an integrative framework in stream fish ecology. Am. Fish. Soc. Symp. 2010, 73, 539–558. [Google Scholar]
- Capps, K.A.; Flecker, A.S. Invasive aquarium fish transform ecosystem nutrient dynamics. Proc. R. Soc. B 2013, 280, 2013520. [Google Scholar] [CrossRef] [PubMed]
- Capps, K.A.; Flecker, A.S. Invasive fishes generate biogeochemical hotspots in a nutrient-limited system. PLoS ONE 2013, 8, e54093. [Google Scholar] [CrossRef] [PubMed]
- Moody, E.K.; Lujan, N.K.; Roach, K.A.; Winemiller, K.O. Threshold elemental ratios and the temperature-dependence of herbivory in fishes. Funct. Ecol. 2019, 33, 913–923. [Google Scholar] [CrossRef]
- Albert, J.S.; Lovejoy, N.R.; Crampton, W.G.R. Miocene tectonism and the separation of cis- and trans-Andean river basins: Evidence from Neotropical fishes. J. S. Am. Earth Sci. 2006, 21, 14–27. [Google Scholar] [CrossRef]
- Pineda, E. Aspectos de la Biología, Ecología, y Distribución de la Familia Loricariidae (Chupapiedras) en la Cuenca del Río Bayano. Ph.D. Thesis, Tésis de Licenciatura, Universidad de Panamá, Ciudad de Panamá, Panama, 1976. [Google Scholar]
- Angermeier, P.L.; Karr, J.R. Fish communities along environmental gradients in a system of tropical streams. Environ. Biol. Fishes 1983, 9, 117–135. [Google Scholar] [CrossRef]
- Power, M.E. Habitat quality and the distribution of algae-grazing catfish in a Panamanian stream. J. Anim. Ecol. 1984, 1984, 357–374. [Google Scholar] [CrossRef]
- Small, G.E.; Pringle, C.M.; Pyron, M.; Duff, J.H. Role of the fish Astyanax aeneus as a keystone nutrient recycler in low-nutrient Neotropical streams. Ecology 2011, 92, 386–397. [Google Scholar] [CrossRef]
- Lujan, N.K.; German, D.P.; Winemiller, K.O. Do wood-grazing fishes partition their niche? Morphological and isotopic evidence for trophic segregation in Neotropical Loricariidae. Funct. Ecol. 2011, 25, 1327–1338. [Google Scholar] [CrossRef]
- Post, D.M.; Layman, C.A.; Arrington, D.A.; Takimoto, G.; Quattrochi, J.; Montaña, C. Getting to the fat of the matter: Models, methods and assumptions for dealing with lipids in stable isotope analyses. Oecologia 2007, 152, 179–189. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: http://www.R-project.org/ (accessed on 30 March 2019).
- Bassar, R.D.; Marshall, M.C.; López-Sepulcre, A.; Zandonà, E.; Auer, S.K.; Travis, J.; Pringle, C.M.; Flecker, A.S.; Thomas, S.A.; Fraser, D.F.; et al. Local adaptation in Trinidadian guppies alters ecosystem processes. Proc. Natl. Acad. Sci. USA 2010, 107, 616–3621. [Google Scholar] [CrossRef] [PubMed]
- Lujan, N.K.; Armbruster, J.W. Morphological and functional diversity of the mandible in suckermouth armored catfishes (Siluriformes, Loricarioidea). J. Morphol. 2012, 273, 24–39. [Google Scholar] [CrossRef] [PubMed]
- Covain, R.; Fisch-Muller, S.; Oliveira, C.; Mol, J.H.; Montoya-Burgos, J.I.; Dray, S. Molecular phylogeny of the highly diversified catfish subfamily Loricariinae (Siluriformes, Loricariidae) reveals incongruences with morphological classification. Mol. Phylogenet. Evol. 2016, 94, 492–517. [Google Scholar] [CrossRef] [PubMed]
- Raubenheimer, D.; Simpson, S.J. The geometry of compensatory feeding in the locust. Anim. Behav. 1993, 45, 953–964. [Google Scholar] [CrossRef]
- Jochum, M.; Barnes, A.D.; Ott, D.; Lang, B.; Klarner, B.; Farajallah, A.; Scheu, S.; Brose, U. Decreasing stoichiometric resource quality drives compensatory feeding across trophic levels in tropical litter invertebrate communities. Am. Nat. 2017, 190, 131–143. [Google Scholar] [CrossRef]
- Post, D.M. Using stable isotopes to estimate trophic position: Models, methods, and assumptions. Ecology 2002, 83, 703–718. [Google Scholar] [CrossRef]
- Moodie, G.E.E.; Power, M.E. The reproductive biology of an armoured catfish, Loricaria uracantha, from Central America. Environ. Biol. Fishes 1982, 7, 143–148. [Google Scholar]
- El-Sabaawi, R.W.; Zandonà, E.; Kohler, T.J.; Marshall, M.C.; Moslemi, J.M.; Travis, J.; López-Sepulcre, A.; Ferriére, R.; Pringle, C.M.; Thomas, S.A.; et al. Widespread intraspecific organismal stoichiometry among populations of the Trinidadian guppy. Funct. Ecol. 2012, 26, 666–676. [Google Scholar] [CrossRef]
- Ambus, N.; Moody, E.K. Size-based sexual dimorphism of nitrogen excretion in livebearing fishes. Ecol. Freshw. Fish 2019, 28, 222–228. [Google Scholar] [CrossRef]
- German, D.P.; Nagle, B.C.; Villeda, J.M.; Ruiz, A.M.; Thomson, A.W.; Contreras Balderas, S.; Evans, D.H. Evolution of herbivory in a carnivorous clade of minnows (Teleostei: Cyprinidae): Effects on gut size and digestive physiology. Physiol. Biochem. Zool. 2009, 83, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.H.; Wainwright, P.C. Trophic novelty is linked to exceptional rates of morphological diversification in two adaptive radiations of Cyprinodon pupfishes. Evolution 2011, 65, 2197–2212. [Google Scholar] [CrossRef] [PubMed]
| Site | Watershed | DIN (μg/L) | DIP (μg/L) | N:P (molar) |
|---|---|---|---|---|
| Quebrada Juan Grande | Chagres | 154.6 | 22.8 | 15.0 |
| Río Frijoles | Chagres | 262.7 | 20.8 | 28.0 |
| Río Frijolito | Chagres | 319.2 | 46.8 | 15.1 |
| Río Chechebre | Bayano | 118.3 | 21.5 | 12.2 |
| Río Curtí | Bayano | 118.9 | 54.7 | 4.8 |
| Río Mamoní | Bayano | 98.3 | 13.1 | 16.6 |
| Río Piriatí | Bayano | 135.1 | 35.8 | 8.4 |
| Quebrada La Hoya | Chucunaque | 139.2 | 8.1 | 38.1 |
| Río Chucunaque | Chucunaque | 144.4 | 14.4 | 22.2 |
| Río Icuanatí | Chucunaque | 172.8 | 29.0 | 13.2 |
| Río Tupisa | Chucunaque | 173.5 | 25.6 | 15.0 |
| Species | Bayano | Chagres | Chucunaque |
|---|---|---|---|
| Ancistrus chagresi | N/A | 2 | N/A |
| Chaetostoma fischeri | 16 | X | 1 |
| Lasiancistrus caucanus | 4 | N/A | X |
| Rineloricaria uracantha | X | 12 | N/A |
| Sturisoma dariense | X | N/A | 10 |
| Sturisomatichthys citurensis | X | N/A | 5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moody, E.K.; Alda, F.; Capps, K.A.; Puebla, O.; Turner, B.L. Trophic Trait Evolution Explains Variation in Nutrient Excretion Stoichiometry among Panamanian Armored Catfishes (Loricariidae). Diversity 2019, 11, 88. https://doi.org/10.3390/d11060088
Moody EK, Alda F, Capps KA, Puebla O, Turner BL. Trophic Trait Evolution Explains Variation in Nutrient Excretion Stoichiometry among Panamanian Armored Catfishes (Loricariidae). Diversity. 2019; 11(6):88. https://doi.org/10.3390/d11060088
Chicago/Turabian StyleMoody, Eric K., Fernando Alda, Krista A. Capps, Oscar Puebla, and Benjamin L. Turner. 2019. "Trophic Trait Evolution Explains Variation in Nutrient Excretion Stoichiometry among Panamanian Armored Catfishes (Loricariidae)" Diversity 11, no. 6: 88. https://doi.org/10.3390/d11060088
APA StyleMoody, E. K., Alda, F., Capps, K. A., Puebla, O., & Turner, B. L. (2019). Trophic Trait Evolution Explains Variation in Nutrient Excretion Stoichiometry among Panamanian Armored Catfishes (Loricariidae). Diversity, 11(6), 88. https://doi.org/10.3390/d11060088





