Deep-Reef Fish Communities of the Great Barrier Reef Shelf-Break: Trophic Structure and Habitat Associations
Abstract
1. Introduction
2. Materials and Methods
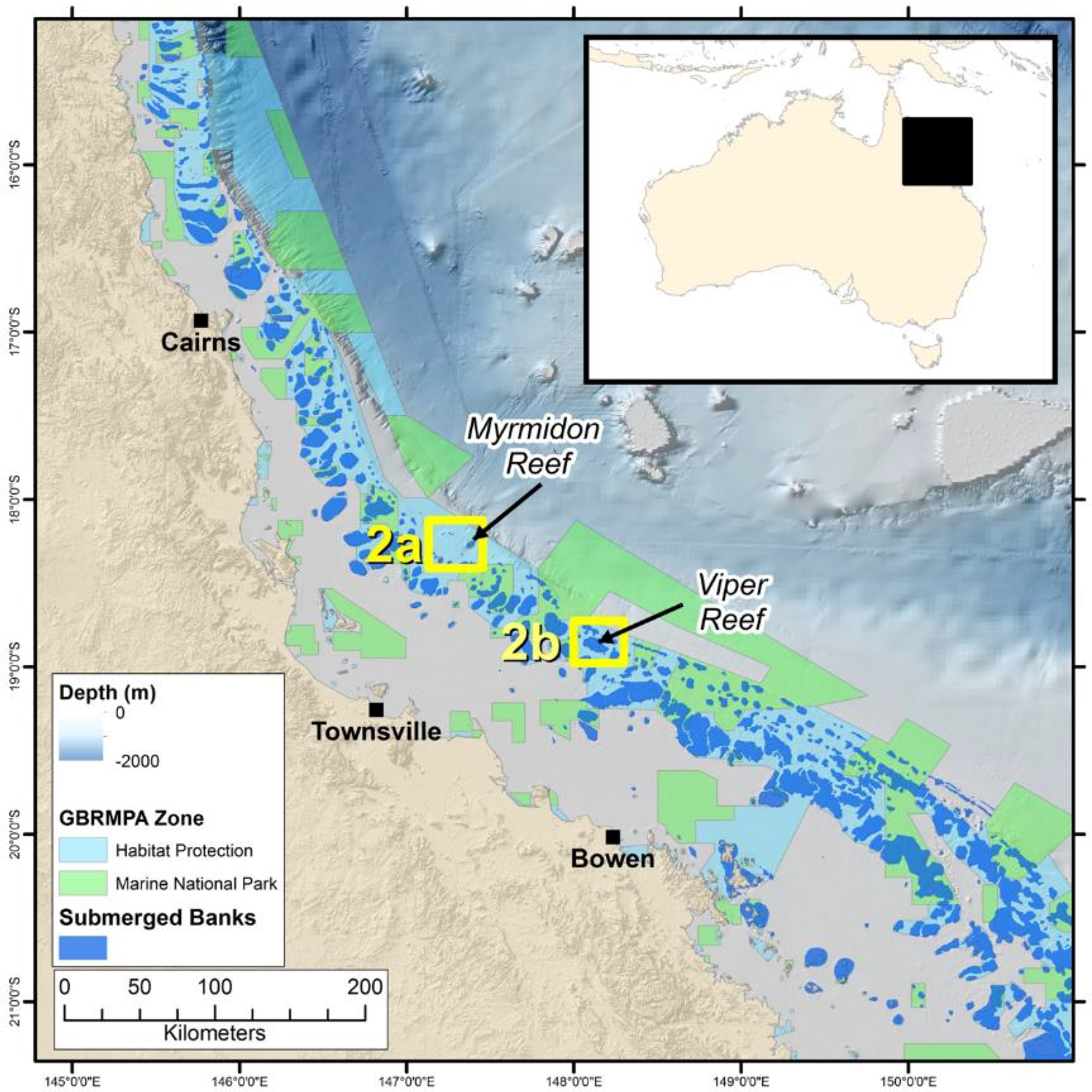
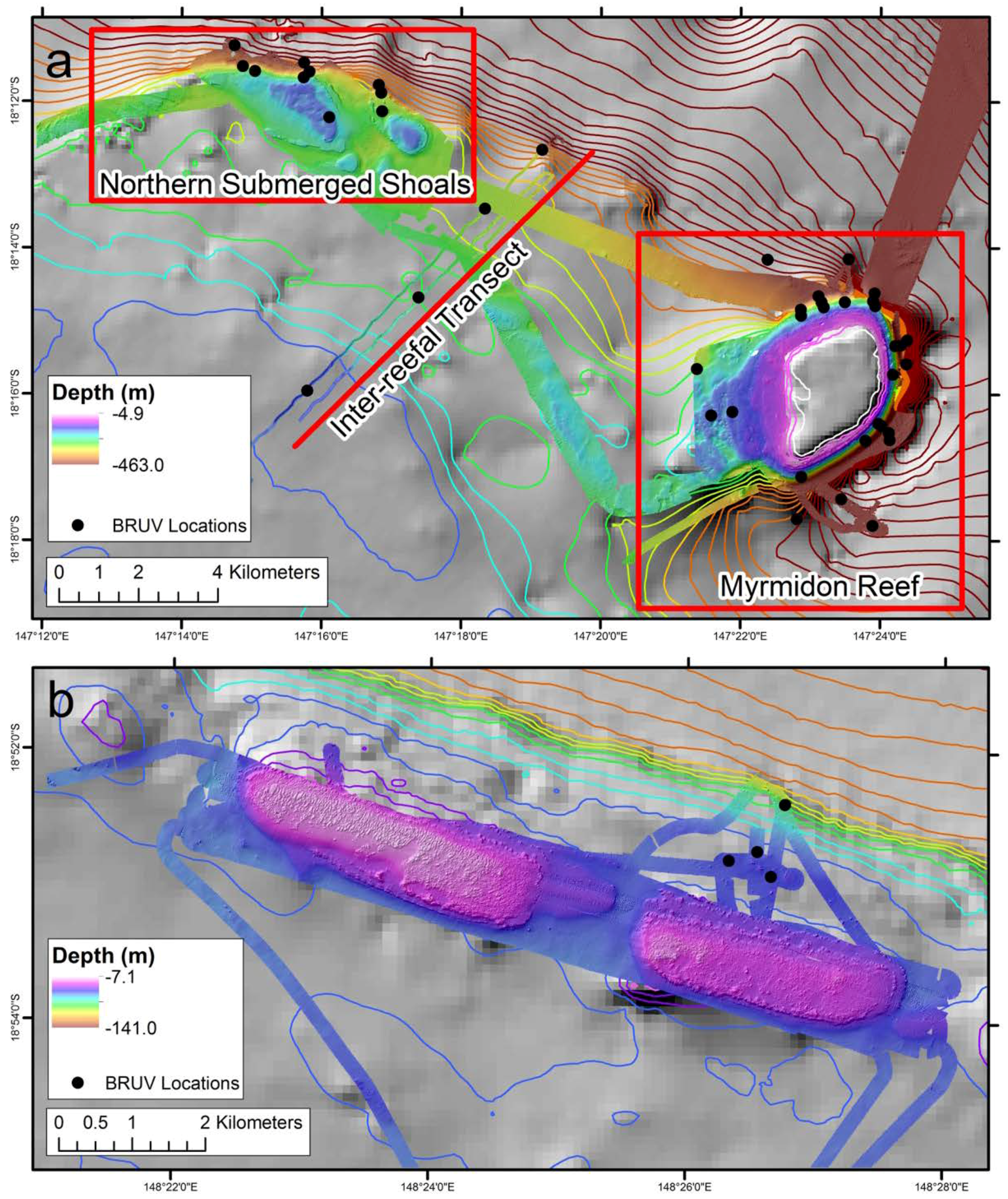
2.1. Study Locations
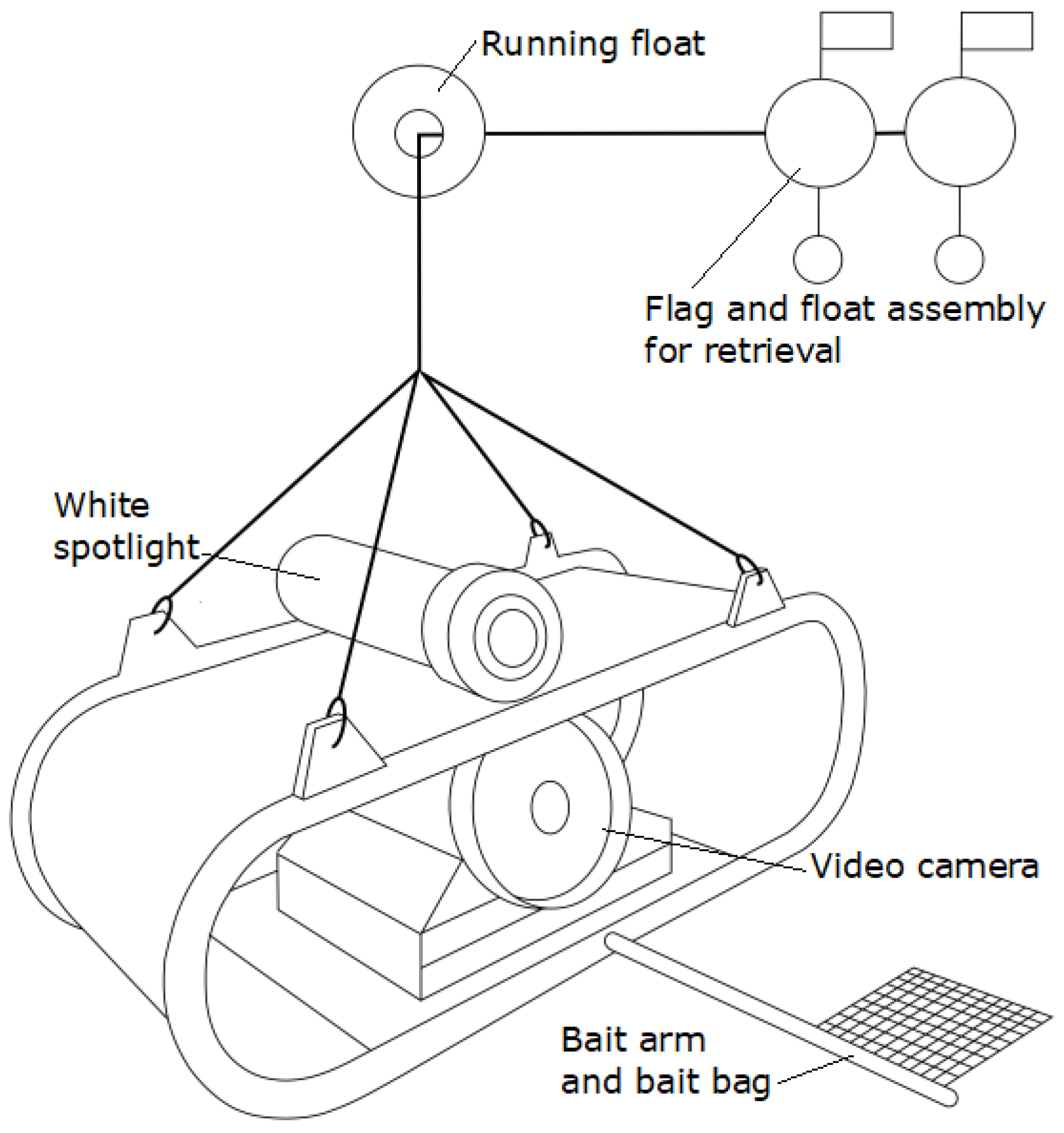
2.2. Baited Remote Underwater Video Stations (BRUVS)
2.3. Multibeam Sonar Acquisition
2.4. Secondary Datasets from Multibeam
2.5. Data Analysis
2.5.1. Habitats and Fish Communities Separated by Depth
2.5.2. Species-Species Associations
2.5.3. Trophic Communities
3. Results
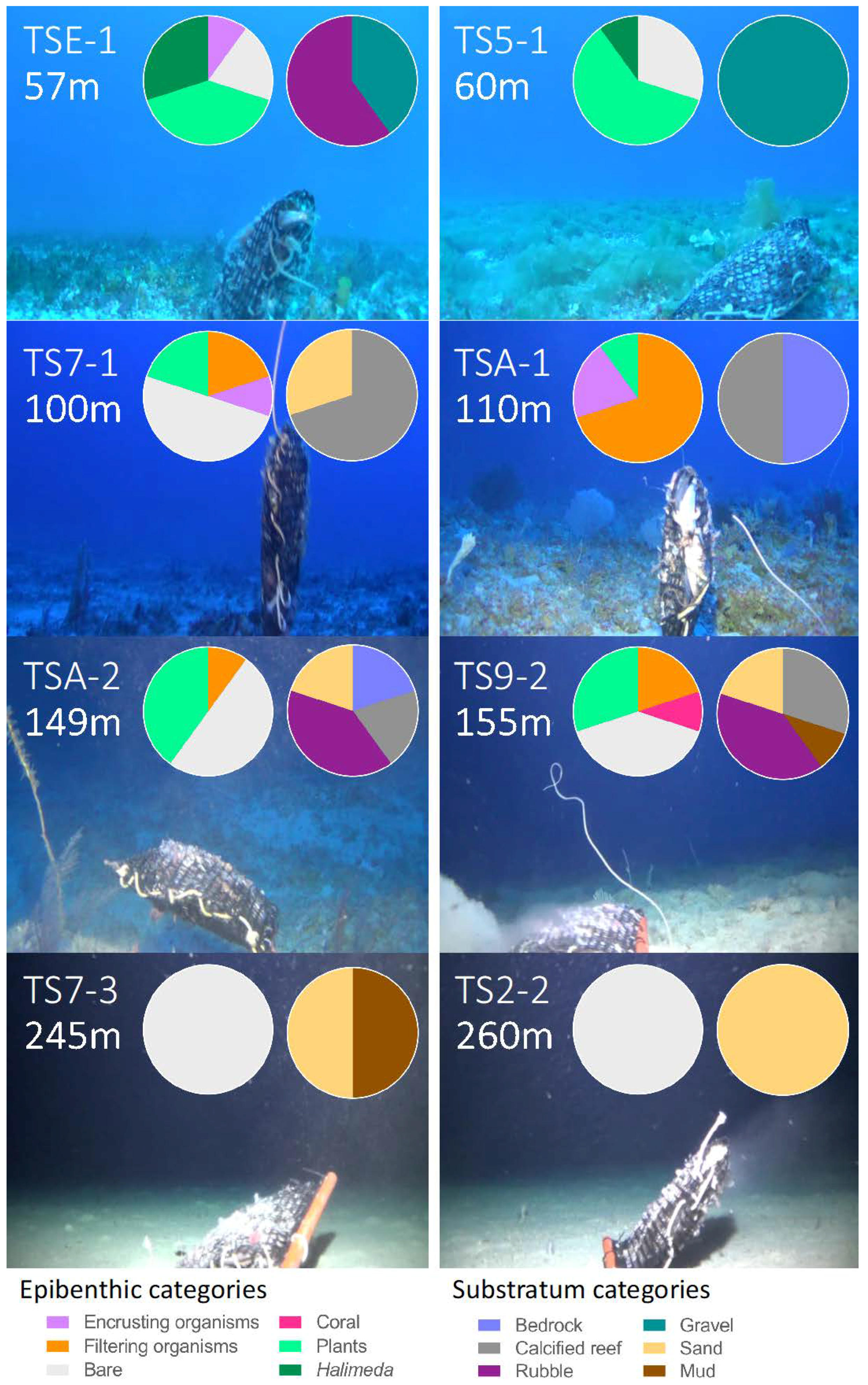
3.1. Description of Deep-Reef Benthic Shelf-Break Habitats
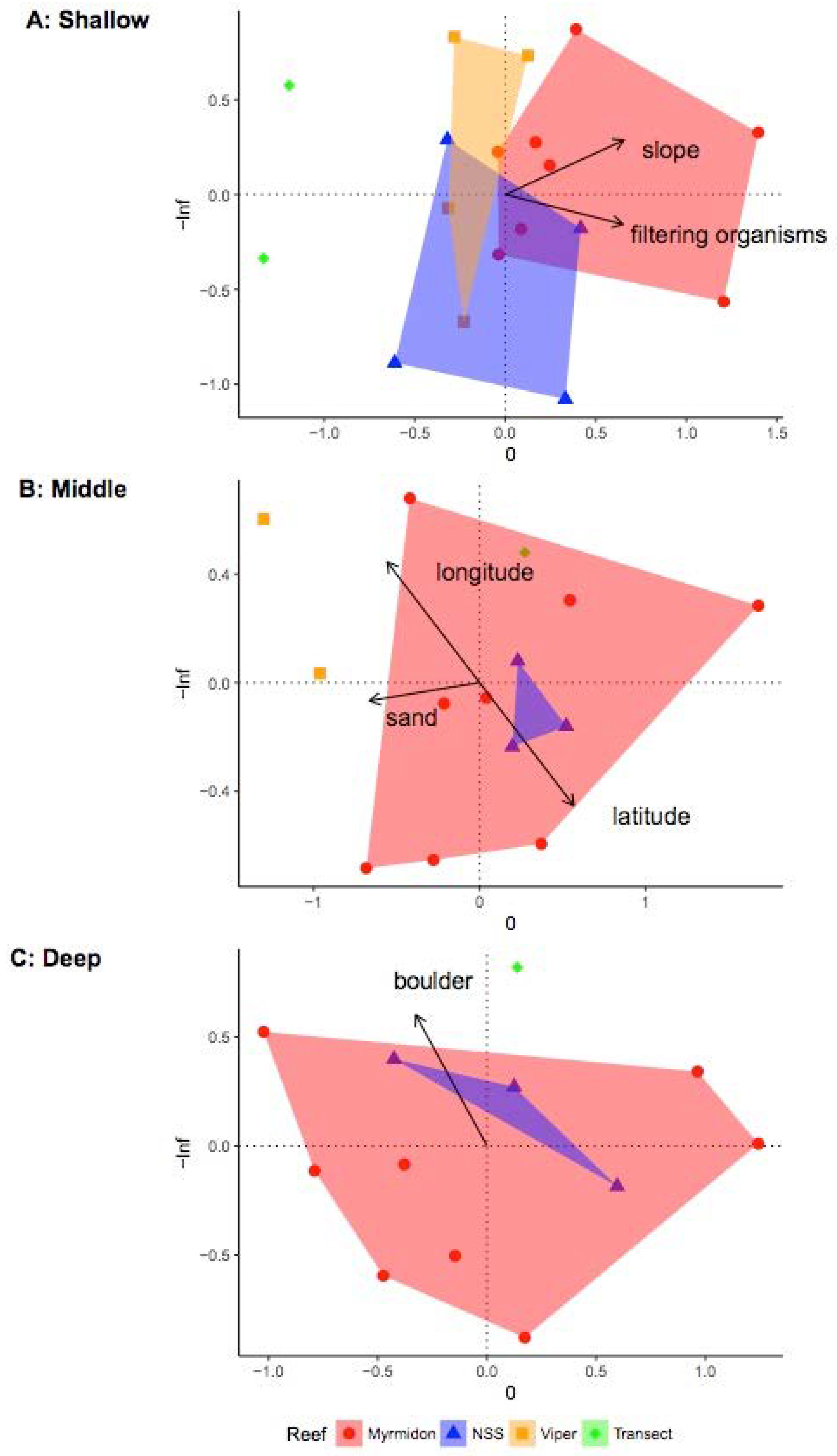
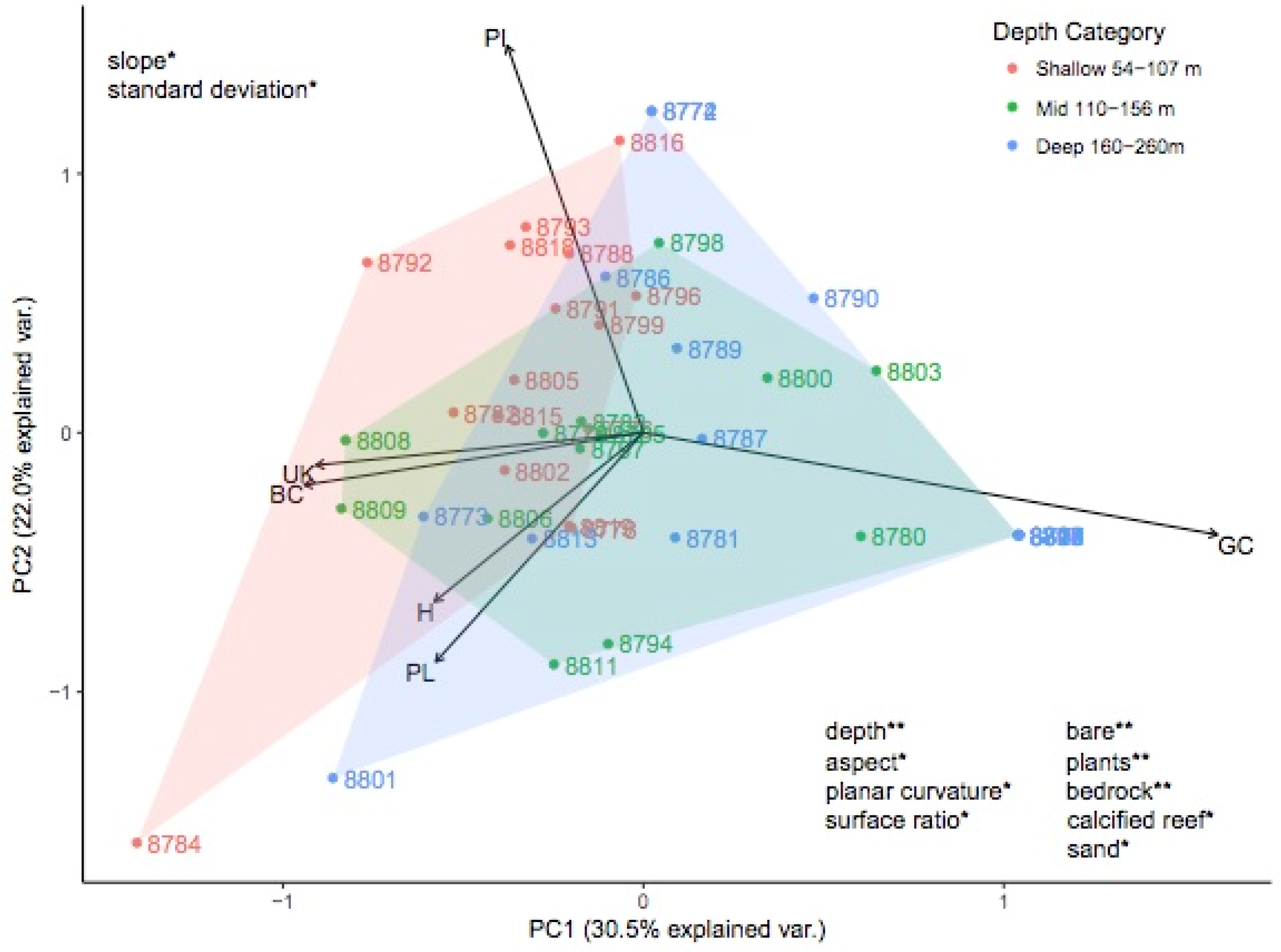
3.2. Investigating Habitats and Fish Communities within Depth Strata
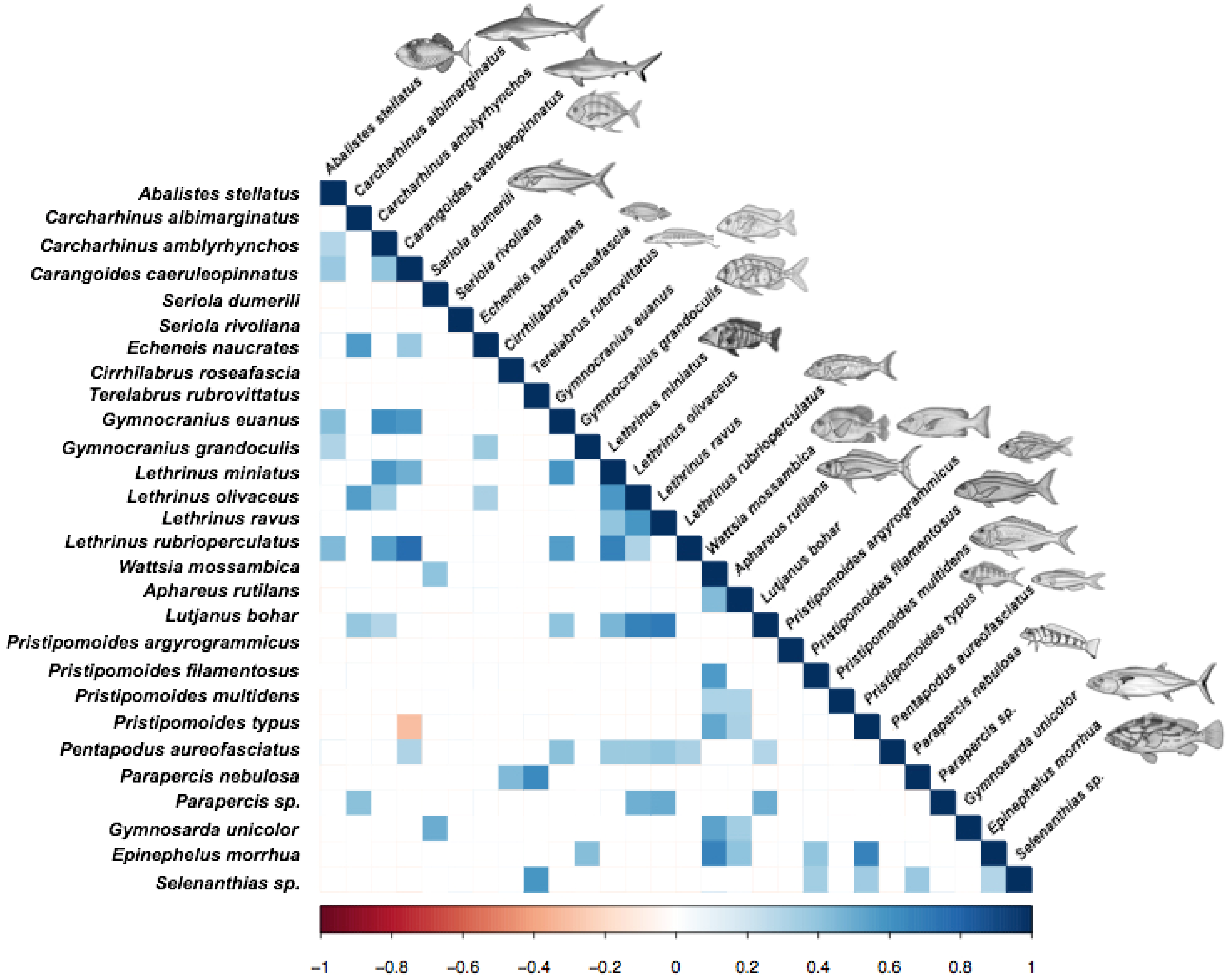
3.3. Relationships among Fish Species
3.4. Deep-Reef Fish Trophic Communities
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Lindfield, S.J.; Harvey, E.S.; Halford, A.R.; McIlwain, J.L. Mesophotic depths as refuge areas for fishery-targeted species on coral reefs. Coral Reefs 2016, 35, 125–137. [Google Scholar] [CrossRef]
- Garcia-Sais, J.R. Reef habitats and associated sessile-benthic and fish assemblages across a euphotic–mesophotic depth gradient in Isla Desecheo, Puerto Rico. Coral Reefs 2010, 29, 277–288. [Google Scholar] [CrossRef]
- Feitoza, B.M.; Rosa, R.S.; Rocha, L.A. Ecology and zoogeography of deep-reef fishes in northeastern Brazil. Bull. Mar. Sci. 2005, 76, 725–742. [Google Scholar]
- Jorgensen, S.J.; Klimley, A.P.; Muhlia-Melo, A.F. Scalloped hammerhead shark Sphyrna lewini, utilizes deep-water, hypoxic zone in the Gulf of California. J. Fish Biol. 2009, 74, 1682–1687. [Google Scholar] [CrossRef]
- Hoyos-Padilla, E.M.; Ketchum, J.T.; Klimley, A.P.; Galván-Magaña, F. Ontogenetic migration of a female scalloped hammerhead shark Sphyrna lewini in the Gulf of California. Anim. Biotelem. 2014, 2, 17. [Google Scholar] [CrossRef]
- Cappo, M.; Kelley, R. Connectivity in the Great Barrier Reef World Heritage Area—An overview of pathways and processes. In Oceanographic Processes of Coral Reefs; CRC Press LLC: Boca Raton, FL, USA, 2010; pp. 161–175. [Google Scholar]
- Kane, C.; Kosaki, R.K.; Wagner, D. High levels of mesophotic reef fish endemism in the Northwestern Hawaiian Islands. Bull. Mar. Sci. 2014, 90, 693–703. [Google Scholar] [CrossRef]
- Bejarano, I.; Appeldoorn, R.; Nemeth, M. Fishes associated with mesophotic coral ecosystems in La Parguera, Puerto Rico. Coral Reefs 2014, 33, 313–328. [Google Scholar] [CrossRef]
- Heyns, E.R.; Bernard, A.T.F.; Richoux, N.B.; Götz, A. Depth-related distribution patterns of subtidal macrobenthos in a well-established marine protected area. Mar. Biol. 2016, 163, 39. [Google Scholar] [CrossRef]
- Bridge, T.C.L.; Grech, A.M.; Pressey, R.L. Factors influencing incidental representation of previously unknown conservation features in marine protected areas. Conserv. Biol. 2016, 30, 154–165. [Google Scholar] [CrossRef]
- Harborne, A.R.; Mumby, P.J.; Kennedy, E.V.; Ferrari, R. Biotic and multi-scale abiotic controls of habitat quality and their effect on coral-reef fishes. Mar. Ecol. Prog. Ser. 2011, 437, 201–214. [Google Scholar] [CrossRef]
- Friedlander, A.M.; Parrish, J.D. Habitat characteristics affecting fish assemblages on a Hawaiian coral reef. J. Exp. Mar. Biol. Ecol. 1998, 224, 1–30. [Google Scholar] [CrossRef]
- Choat, J.; Ayling, A. The relationship between habitat structure and fish faunas on New Zealand reefs. J. Exp. Mar. Biol. Ecol. 1987, 110, 257–284. [Google Scholar] [CrossRef]
- Yoklavich, M.M.; Greene, H.G.; Cailliet, G.M.; Sullivan, D.E.; Lea, R.N.; Love, M.S. Habitat associations of deep-water rockfishes in a submarine canyon: An example of a natural refuge. Fish. Bull. 2000, 98, 625–641. [Google Scholar]
- Stein, D.L.; Tissot, B.N.; Hixon, M.A.; Barss, W. Fish-habitat associations on a deep reef at the edge of the Oregon continental-shelf. Fish. Bull. 1992, 90, 540–551. [Google Scholar]
- Majewski, A.; Atchison, S.; MacPhee, S.; Eert, J.; Niemi, A.; Michel, C.; Reist, J.D. Marine fish community structure and habitat associations on the Canadian Beaufort shelf and slope. Deep Sea Res. Part I Oceanogr. Res. Pap. 2017. [Google Scholar] [CrossRef]
- MacNeil, M.A.; Connolly, S.R. Multi-scale patterns and processes in reef fish abundance. In Ecology of Fishes on Coral Reefs; Mora, C., Ed.; Cambridge University Press: Cambridge, UK, 2015; pp. 116–124. [Google Scholar]
- Connell, S.; Jones, G. The influence of habitat complexity on postrecruitment processes in a temperate reef fish population. J. Exp. Mar. Biol. Ecol. 1991, 151, 271–294. [Google Scholar] [CrossRef]
- Heck, K.L.; Orth, R.J. Seagrass habitats: The roles of habitat complexity, competition and predation in structuring associated fish and motile macroinvertebrate assemblages. In Estuarine Perspectives; Elsevier: Amsterdam, The Netherlands, 1980; pp. 449–464. [Google Scholar]
- Johnson, D.W. Predation, habitat complexity, and variation in density-dependent mortality of temperate reef fishes. Ecology 2006, 87, 1179–1188. [Google Scholar] [CrossRef]
- Messmer, V.; Jones, G.P.; Munday, P.L.; Holbrook, S.J.; Schmitt, R.J.; Brooks, A.J. Habitat biodiversity as a determinant of fish community structure on coral reefs. Ecology 2011, 92, 2285–2298. [Google Scholar] [CrossRef]
- Alvarez-Filip, L.; Dulvy, N.K.; Gill, J.A.; Côté, I.M.; Watkinson, A.R. Flattening of Caribbean coral reefs: Region-wide declines in architectural complexity. Proc. R. Soc. B Biol. Sci. 2009, 276, 3019. [Google Scholar] [CrossRef]
- Graham, N.A. Habitat complexity: Coral structural loss leads to fisheries declines. Curr. Biol. 2014, 24, R359–R361. [Google Scholar] [CrossRef]
- Rocha, L.A.; Pinheiro, H.T.; Shepherd, B.; Papastamatiou, Y.P.; Luiz, O.J.; Pyle, R.L.; Bongaerts, P. Mesophotic coral ecosystems are threatened and ecologically distinct from shallow water reefs. Science 2018, 361, 281–284. [Google Scholar] [CrossRef]
- Andradi-Brown, D.; Laverick, J.; Bejarano, I.; Bridge, T.; Colin, P.; Eyal, G.; Jones, R.; Kahng, S.; Reed, J.; Smith, T. Mesophotic Coral Ecosystems—A Lifeboat for Coral Reefs? 2016. Available online: http://www.grida.no/publications/88 (accessed on 20 December 2016).
- Fitzpatrick, B.M.; Harvey, E.S.; Heyward, A.J.; Twiggs, E.J.; Colquhoun, J. Habitat specialization in tropical continental shelf demersal fish assemblages. PLoS ONE 2012, 7, e39634. [Google Scholar] [CrossRef]
- Sink, K.; Boshoff, W.; Samaai, T.; Timm, P.; Kerwath, S. Observations of the habitats and biodiversity of the submarine canyons at Sodwana Bay: Coelacanth research. S. Afr. J. Sci. 2006, 102, 466–474. [Google Scholar]
- Kelley, C.; Moffitt, R.; Smith, J.R. Mega- to micro-scale classification and description of bottomfish essential fish habitat on four banks in the Northwestern Hawaiian Islands. Atoll Res. Bull. 2006, 542, 319–332. [Google Scholar]
- Parker, R.; Mays, R. Southeastern US Deepwater Reef Fish Assemblages, Habitat Characteristics, Catches, and Life History Summaries; US Department of Commerce: Washington, DC, USA, 1998.
- Starr, R.M.; Green, K.; Sala, E. Deep-water fish assemblages at Isla del Coco National Park and Las Gemelas Seamounts, Costa Rica. Rev. Biol. Trop. 2012, 60, 347–362. [Google Scholar] [CrossRef]
- Heyns-Veale, E.R.; Bernard, A.T.F.; Richoux, N.B.; Parker, D.; Langlois, T.J.; Harvey, E.S.; Götz, A. Depth and habitat determine assemblage structure of South Africa’s warm-temperate reef fish. Mar. Biol. 2016, 163, 158. [Google Scholar] [CrossRef]
- Thresher, R.E.; Colin, P.L. Trophic structure, diversity and abundance of fishes of the deep reef (30–300 m) at Enewetak, Marshall Islands. Bull. Mar. Sci. 1986, 38, 253–272. [Google Scholar]
- Andradi-Brown, D.A.; Gress, E.; Wright, G.; Exton, D.A.; Rogers, A.D. Reef fish community biomass and trophic structure changes across shallow to upper-mesophotic reefs in the Mesoamerican Barrier Reef, Caribbean. PLoS ONE 2016, 11, e0156641. [Google Scholar] [CrossRef]
- Brokovich, E.; Einbinder, S.; Shashar, N.; Kiflawi, M.; Kark, S. Descending to the twilight-zone: Changes in coral reef fish assemblages along a depth gradient down to 65 m. Mar. Ecol. Prog. Ser. 2008, 371, 253–262. [Google Scholar] [CrossRef]
- Fukunaga, A.; Kosaki, R.K.; Wagner, D.; Kane, C. Structure of Mesophotic Reef Fish Assemblages in the Northwestern Hawaiian Islands. PLoS ONE 2016, 11, e0157861. [Google Scholar] [CrossRef]
- Kane, C.N.; Tissot, B.N. Trophic designation and live coral cover predict changes in reef-fish community structure along a shallow to mesophotic gradient in Hawaii. Coral Reefs 2017, 1–11. [Google Scholar] [CrossRef]
- Kahng, S.E.; Garcia-Sais, J.R.; Spalding, H.L.; Brokovich, E.; Wagner, D.; Weil, E.; Hinderstein, L.; Toonen, R.J. Community ecology of mesophotic coral reef ecosystems. Coral Reefs 2010, 29, 255–275. [Google Scholar] [CrossRef]
- Dennis, G.D.; Bright, T.J. Reef fish assemblages on hard banks in the northwestern Gulf of Mexico. Bull. Mar. Sci. 1988, 43, 280–307. [Google Scholar]
- Bridge, T.; Done, T.; Beaman, R.; Friedman, A.; Williams, S.; Pizarro, O.; Webster, J. Topography, substratum and benthic macrofaunal relationships on a tropical mesophotic shelf margin, central Great Barrier Reef, Australia. Coral Reefs 2011, 30, 143–153. [Google Scholar] [CrossRef]
- Pyle, R.L. Use of advanced mixed-gas diving technology to explore the coral reef “Twilight Zone”. Ocean Pulse Crit. Diagn. 1998, 71–88. [Google Scholar] [CrossRef]
- Cánovas-Molina, A.; Montefalcone, M.; Bavestrello, G.; Cau, A.; Bianchi, C.N.; Morri, C.; Canese, S.; Bo, M. A new ecological index for the status of mesophotic megabenthic assemblages in the Mediterranean based on ROV photography and video footage. Cont. Shelf Res. 2016, 121, 13–20. [Google Scholar] [CrossRef]
- McLean, D.L.; Partridge, J.C.; Bond, T.; Birt, M.J.; Bornt, K.R.; Langlois, T.J. Using industry ROV videos to assess fish associations with subsea pipelines. Cont. Shelf Res. 2017, 141, 76–97. [Google Scholar] [CrossRef]
- Williams, S.B.; Pizarro, O.; Webster, J.M.; Beaman, R.J.; Mahon, I.; Johnson-Roberson, M.; Bridge, T.C.L. Autonomous underwater vehicle–assisted surveying of drowned reefs on the shelf edge of the Great Barrier Reef, Australia. J. Field Robot. 2010, 27, 675–697. [Google Scholar] [CrossRef]
- Starr, R.M.; Fox, D.S.; Hixon, M.A.; Tissot, B.N.; Johnson, G.E.; Barss, W.H. Comparison of Submersible-Survey and Hydroacoustic-Survey Estimates of Fish Density on a Rocky Bank; US Dept. of Commerce, National Marine Fisheries Service: Washington, DC, USA, 1996.
- Tissot, B.N.; Hixon, M.A.; Stein, D.L. Habitat-based submersible assessment of macro-invertebrate and groundfish assemblages at Heceta Bank, Oregon, from 1988 to 1990. J. Exp. Mar. Biol. Ecol. 2007, 352, 50–64. [Google Scholar] [CrossRef]
- Langlois, T.J.; Fitzpatrick, B.R.; Fairclough, D.V.; Wakefield, C.B.; Hesp, S.A.; McLean, D.L.; Harvey, E.S.; Meeuwig, J.J. Similarities between line fishing and baited stereo-video estimations of length-frequency: Novel application of kernel density estimates. PLoS ONE 2012, 7, e45973. [Google Scholar] [CrossRef]
- Johansson, C.; Stowar, M.; Cappo, M. The use of stereo BRUVS for measuring fish size. In Marine and Tropical Sciences Research Facility Report Series; Australian Institute of Marine Science: Cape Cleveland, Australia, 2008. [Google Scholar]
- Hannah, R.W.; Blume, M.T. The influence of bait and stereo video on the performance of a video lander as a survey tool for marine demersal reef fishes in Oregon waters. Mar. Coast. Fish. 2014, 6, 181–189. [Google Scholar] [CrossRef]
- Harvey, E.S.; Newman, S.J.; McLean, D.L.; Cappo, M.; Meeuwig, J.J.; Skepper, C.L. Comparison of the relative efficiencies of stereo-BRUVs and traps for sampling tropical continental shelf demersal fishes. Fish. Res. 2012, 125–126, 108–120. [Google Scholar] [CrossRef]
- Merritt, D.; Donovan, M.K.; Kelley, C.; Waterhouse, L.; Parke, M.; Wong, K.; Drazen, J.C. BotCam: A baited camera system for nonextractive monitoring of bottomfish species. Fish. Bull. 2011, 109, 56–67. [Google Scholar]
- Ellis, D.; DeMartini, E. Evaluation of a video camera technique for indexing abundances of juvenile pink snapper, Pristipomoides filamentosus, and other Hawaiian insular shelf fishes. Oceanogr. Lit. Rev. 1995, 9, 786. [Google Scholar]
- Whitmarsh, S.K.; Fairweather, P.G.; Huveneers, C. What is Big BRUVver up to? Methods and uses of baited underwater video. Rev. Fish. Biol. Fish. 2017, 27, 53–73. [Google Scholar] [CrossRef]
- Cappo, M.; De’ath, G.; Speare, P. Inter-reef vertebrate communities of the Great Barrier Reef Marine Park determined by baited remote underwater video stations. Mar. Ecol. Prog. Ser. 2007, 350, 209–221. [Google Scholar] [CrossRef]
- Langlois, T.; Harvey, E.; Fitzpatrick, B.; Meeuwig, J.; Shedrawi, G.; Watson, D. Cost-efficient sampling of fish assemblages: Comparison of baited video stations and diver video transects. Aquat. Biol. 2010, 9, 155–168. [Google Scholar] [CrossRef]
- Watson, D.L.; Harvey, E.S.; Fitzpatrick, B.M.; Langlois, T.J.; Shedrawi, G. Assessing reef fish assemblage structure: How do different stereo-video techniques compare? Mar. Biol. 2010, 157, 1237–1250. [Google Scholar] [CrossRef]
- Andradi-Brown, D.A.; Macaya-Solis, C.; Exton, D.A.; Gress, E.; Wright, G.; Rogers, A.D. Assessing Caribbean shallow and mesophotic reef fish communities using Baited-Remote Underwater Video (BRUV) and Diver-Operated Video (DOV) survey techniques. PLoS ONE 2016, 11, e0168235. [Google Scholar] [CrossRef]
- Cappo, M. Development of a Baited Video Technique and Spatial Models to Explain Patterns of Fish Biodiversity in Inter-Reef Waters. Ph.D. Thesis, James Cook University, Townsville, Australia, 2010. [Google Scholar]
- Espinoza, M.; Cappo, M.; Heupel, M.R.; Tobin, A.J.; Simpfendorfer, C.A. Quantifying shark distribution patterns and species-habitat associations: Implications of marine park zoning. PLoS ONE 2014, 9, e106885. [Google Scholar] [CrossRef]
- Harvey, E.; McLean, D.; Frusher, S.; Haywood, M.; Newman, S.; Williams, A. The Use of BRUVs as a Tool for Assessing Marine Fisheries and Ecosystems: A Review of the Hurdles and Potential; University of Western Australia: Perth, Australia, 2013. [Google Scholar]
- Mallet, D.; Pelletier, D. Underwater video techniques for observing coastal marine biodiversity: A review of sixty years of publications (1952–2012). Fish. Res. 2014, 154, 44–62. [Google Scholar] [CrossRef]
- Ierodiaconou, D.; Laurenson, L.; Burq, S.; Reston, M. Marine benthic habitat mapping using multibeam data, geo-referenced video and image classification techniques in Victoria, Australia. J. Spat. Sci. 2007, 52, 93–104. [Google Scholar] [CrossRef]
- Sih, T.L.; Cappo, M.; Kingsford, M. Deep-reef fish assemblages of the Great Barrier Reef shelf-break (Australia). Sci. Rep. 2017, 7, 10886. [Google Scholar] [CrossRef]
- Bridge, T.C.L.; Webster, J.M.; Sih, T.L.; Bongaerts, P. The Great Barrier Reef outer-shelf. In The Great Barrier Reef: Biology, Environment and Management, 2nd ed.; Hutchings, P., Kingsford, M., Hoegh-Guldberg, O., Eds.; CSIRO Publishing: Clayton South, Australia, 2019; pp. 73–84. [Google Scholar]
- Hopley, D.; Smithers, S.G.; Parnell, K. The Geomorphology of the Great Barrier Reef: Development, Diversity and Change; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Harris, P.; Davies, P. Submerged reefs and terraces on the shelf edge of the Great Barrier Reef, Australia. Coral Reefs 1989, 8, 87–98. [Google Scholar] [CrossRef]
- Symonds, P.; Davies, P.; Parisi, A. Structure and stratigraphy of the central Great Barrier Reef. BMR J. Aust. Geol. Geophys. 1983, 8, 277–291. [Google Scholar]
- Puga-Bernabéu, Á.; Webster, J.M.; Beaman, R.J.; Guilbaud, V. Variation in canyon morphology on the Great Barrier Reef margin, north-eastern Australia: The influence of slope and barrier reefs. Geomorphology 2013, 191, 35–50. [Google Scholar] [CrossRef]
- Andrews, J.C.; Gentien, P.P. Upwelling as a source of nutrients for the Great Barrier Reef ecosystems: A solution to Darwin’s question? Mar. Ecol. Prog. Ser. 1982, 8, 257–269. [Google Scholar] [CrossRef]
- Furnas, M.; Mitchell, A. Nutrient inputs into the central Great Barrier Reef (Australia) from subsurface intrusions of Coral Sea waters: A two-dimensional displacement model. Cont. Shelf Res. 1996, 16, 1127–1148. [Google Scholar] [CrossRef]
- Coles, R.; McKenzie, L.; De’ath, G.; Roelofs, A.; Long, W.L. Spatial distribution of deepwater seagrass in the inter-reef lagoon of the Great Barrier Reef World Heritage Area. Mar. Ecol. Prog. Ser. 2009, 392, 57–68. [Google Scholar] [CrossRef]
- Pitcher, C.R.; Doherty, P.; Arnold, P.; Hooper, J.; Gribble, N.; Bartlett, C.; Browne, M.; Campbell, N.; Cannard, T.; Cappo, M.; et al. Seabed Biodiversity on the Continental Shelf of the Great Barrier Reef World Heritage Area; AIMS/CSIRO/QM/QDPI: Cape Cleveland, Australia, 2007; p. 315. [Google Scholar]
- Bridge, T.C.; Done, T.J.; Friedman, A.; Beaman, R.J.; Williams, S.B.; Pizarro, O.; Webster, J.M. Variability in mesophotic coral reef communities along the Great Barrier Reef, Australia. Mar. Ecol. Prog. Ser. 2011, 428, 63–75. [Google Scholar] [CrossRef]
- Drew, E.A. Ocean nutrients to sediment banks via tidal jets and Halimeda meadows. In Oceanographic Processes of Coral Reefs: Physical and Biological Links in the Great Barrier Reef; CRC Press: Boca Raton, FL, USA, 2000; p. 255. [Google Scholar]
- Bridge, T.C.L.; Fabricius, K.E.; Bongaerts, P.; Wallace, C.C.; Muir, P.R.; Done, T.J.; Webster, J.M. Diversity of Scleractinia and Octocorallia in the mesophotic zone of the Great Barrier Reef, Australia. Coral Reefs 2012, 31, 179–189. [Google Scholar] [CrossRef]
- Fernandes, L.; Day, J.; Lewis, A.; Slegers, S.; Kerrigan, B.; Breen, D.; Cameron, D.; Jago, B.; Hall, J.; Lowe, D. Establishing representative no-take areas in the Great Barrier Reef: Large-scale implementation of theory on marine protected areas. Conserv. Biol. 2005, 19, 1733–1744. [Google Scholar] [CrossRef]
- Cappo, M.; Speare, P.; De’ath, G. Comparison of baited remote underwater video stations (BRUVS) and prawn (shrimp) trawls for assessments of fish biodiversity in inter-reefal areas of the Great Barrier Reef Marine Park. J. Exp. Mar. Biol. Ecol. 2004, 302, 123–152. [Google Scholar] [CrossRef]
- Cappo, M.; Harvey, E.; Malcolm, H.; Speare, P. Potential of video techniques to monitor diversity, abundance and size of fish in studies of marine protected areas. Aquatic Protected Areas—What Works Best and How Do We Know. 2003, pp. 455–464. Available online: https://pdfs.semanticscholar.org/2b99/d3f34a636dd59fce74fd87c6609a4ef0e5ed.pdf (accessed on 30 June 2013).
- Schobernd, Z.H.; Bacheler, N.M.; Conn, P.B. Examining the utility of alternative video monitoring metrics for indexing reef fish abundance. Can. J. Fish. Aquat. Sci. 2013, 71, 464–471. [Google Scholar] [CrossRef]
- Cappo, M.; Stowar, M.; Stieglitz, T.; Lawrey, E.; Johansson, C.; Macneil, A. Measuring and Communicating Effects of MPAs on Deep “Shoal” Fisheries. In Proceedings of the 12th International Coral Reef Symposium, Cairns, Australia, 9–13 July 2012. [Google Scholar]
- Hughes-Clarke, J.E.; Mayer, L.A.; Wells, D.E. Shallow-water imaging multibeam sonars: A new tool for investigating seafloor processes in the coastal zone and on the continental shelf. Mar. Geophys. Res. 1996, 18, 607–629. [Google Scholar] [CrossRef]
- Beaman, R.J.; Bridge, T.C.L.; Lüter, C.; Reitner, J.; Wörheide, G. Spatial patterns in the distribution of benthic assemblages across a large depth gradient in the Coral Sea, Australia. Mar. Biodivers. 2016, 1–14. [Google Scholar] [CrossRef]
- Webster, J.M.; Beaman, R.J.; Bridge, T.; Davies, P.J.; Byrne, M.; Williams, S.; Manning, P.; Pizarro, O.; Thornborough, K.; Woolsey, E.; et al. From corals to canyons: The Great Barrier Reef Margin. EOS Trans. Am. Geophys. Union 2008, 89, 217–218. [Google Scholar] [CrossRef]
- Stieglitz, T.C. The Yongala’s “Halo of Holes”—Systematic bioturbation close to a shipwreck. In Seafloor Geomorphology as Benthic Habitat: GeoHab Atlas of Seafloor Geomorphic Features and Benthic Habitats; Elsevier: London, UK, 2011; p. 277. [Google Scholar]
- Brown, C.J.; Smith, S.J.; Lawton, P.; Anderson, J.T. Benthic habitat mapping: A review of progress towards improved understanding of the spatial ecology of the seafloor using acoustic techniques. Estuar. Coast. Shelf Sci. 2011, 92, 502–520. [Google Scholar] [CrossRef]
- Flater, D. XTide. Available online: http://www.flaterco.com/xtide/ (accessed on 7 July 2016).
- Wilson, M.F.; O’Connell, B.; Brown, C.; Guinan, J.C.; Grehan, A.J. Multiscale terrain analysis of multibeam bathymetry data for habitat mapping on the continental slope. Mar. Geod. 2007, 30, 3–35. [Google Scholar] [CrossRef]
- Fisher, P.; Wood, J.; Cheng, T. Where is Helvellyn? Fuzziness of multi-scale landscape morphometry. Trans. Inst. Br. Geogr. 2004, 29, 106–128. [Google Scholar] [CrossRef]
- Heyward, A.; Jones, R.; Meeuwig, J.J.; Burns, K.; Radford, B.; Colquhoun, J.; Cappo, M.; Case, M.; O’Leary, R.A.; Fisher, R.; et al. Monitoring Study S5 Banks and Shoals, Montara 2011 Offshore Banks Assessment Survey, Report for PTTEP Australiasia (Ashmore Cartier) Pty. Ltd.; Australian Institute of Marine Science: Townsville, Australia, 2011; p. 253.
- Diesing, M.; Mitchell, P.; Stephens, D. Image-based seabed classification: What can we learn from terrestrial remote sensing? ICES J. Mar. Sci. 2016, 73, 2425–2441. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Hijmans, R.; van Etten, J. Raster: Geographic Analysis and Modeling with Raster Data; R Package Version 1.9-33; R Foundation for Statistical Computing: Vienna, Austria, 2011. [Google Scholar]
- Zevenbergen, L.W.; Thorne, C.R. Quantitative analysis of land surface topography. Earth Surf. Process. Landf. 1987, 12, 47–56. [Google Scholar] [CrossRef]
- Moore, C.H.; Harvey, E.S.; Van Niel, K.P. Spatial prediction of demersal fish distributions: Enhancing our understanding of species–environment relationships. ICES J. Mar. Sci. 2009, 66, 2068–2075. [Google Scholar] [CrossRef]
- Haywood, M.D.E.; Pitcher, C.R.; Ellis, N.; Wassenberg, T.J.; Smith, G.; Forcey, K.; McLeod, I.; Carter, A.; Strickland, C.; Coles, R. Mapping and characterisation of the inter-reefal benthic assemblages of the Torres Strait. Cont. Shelf Res. 2008, 28, 2304–2316. [Google Scholar] [CrossRef]
- Holmes, K.; Van Niel, K.; Radford, B.; Kendrick, G.; Grove, S. Modelling distribution of marine benthos from hydroacoustics and underwater video. Cont. Shelf Res. 2008, 28, 1800–1810. [Google Scholar] [CrossRef]
- Malcolm, H.A.; Jordan, A.; Schultz, A.L.; Smith, S.D.A.; Ingleton, T.; Foulsham, E.; Linklater, M.; Davies, P.; Ferrari, R.; Hill, N.; et al. Integrating seafloor habitat mapping and fish assemblage patterns improves spatial management planning in a marine park. J. Coast. Res. 2016, 75, 1292–1296. [Google Scholar] [CrossRef]
- Costa, B.; Taylor, J.C.; Kracker, L.; Battista, T.; Pittman, S. Mapping reef fish and the seascape: Using acoustics and spatial modeling to guide coastal management. PLoS ONE 2014, 9, e85555. [Google Scholar] [CrossRef]
- Oyafuso, Z.S.; Drazen, J.C.; Moore, C.H.; Franklin, E.C. Habitat-based species distribution modelling of the Hawaiian deepwater snapper-grouper complex. Fish. Res. 2017, 195, 19–27. [Google Scholar] [CrossRef]
- Moore, C.H.; Van Niel, K.; Harvey, E.S. The effect of landscape composition and configuration on the spatial distribution of temperate demersal fish. Ecography 2011, 34, 425–435. [Google Scholar] [CrossRef]
- Hirzel, A.H.; Hausser, J.; Chessel, D.; Perrin, N. Ecological-niche factor analysis: How to compute habitat-suitability maps without absence data? Ecology 2002, 83, 2027–2036. [Google Scholar] [CrossRef]
- Dartnell, P.; Gardner, J.V. Predicting seafloor facies from multibeam bathymetry and backscatter data. Photogramm. Eng. Remote Sens. 2004, 70, 1081–1091. [Google Scholar] [CrossRef]
- Misa, W. Establishing species-habitat associations for four eteline snappers using a baited stereo-video camera system. Fish. Bull. 2013, 111, 293–308. [Google Scholar] [CrossRef]
- Jenness, J.; Brost, B.; Beier, P. Land Facet Corridor Designer—Manual. 2011. Available online: http://www.jennessent.com/downloads/land_facet_tools.pdf (accessed on 1 June 2016).
- Weiss, A. Topographic position and landforms analysis. In Proceedings of the ESRI User Conference 2001, San Diego, CA, USA, 9–13 July 2001; Volume 200. [Google Scholar]
- Iampietro, P.J.; Kvitek, R.G.; Morris, E. Recent advances in automated genus-specific marine habitat mapping enabled by high-resolution multibeam bathymetry. Mar. Technol. Soc. J. 2005, 39, 83–93. [Google Scholar] [CrossRef]
- Riley, S.J.; DeGloria, S.D.; Elliot, R. A terrain ruggedness index that quantifies topographic heterogeneity. Intermt. J. Sci. 1999, 5, 23–27. [Google Scholar]
- Dartnell, P. Applying Remote Sensing Techniques to Map Seafloor Geology/Habitat Relationships. Ph.D. Thesis, San Francisco State University, San Francisco, CA, USA, 2000. [Google Scholar]
- Yates, K.L.; Mellin, C.; Caley, M.J.; Radford, B.T.; Meeuwig, J.J. Models of marine fish biodiversity: Assessing predictors from three habitat classification schemes. PLoS ONE 2016, 11, e0155634. [Google Scholar] [CrossRef]
- Jenness, J.S. Calculating landscape surface area from digital elevation models. Wildl. Soc. Bull. 2004, 32, 829–839. [Google Scholar] [CrossRef]
- Oksanen, J. Vegan: An Introduction to Ordination. Available online: http://cran.r-project.org/web/packages/vegan/vignettes/introvegan.pdf (accessed on 10 June 2015).
- Wei, T.; Simko, V. R Package ‘Corrplot’: Visualization of a Correlation Matrix (Version 0.84). 2017. Available online: https://cran.r-project.org/web/packages/corrplot/corrplot.pdf (accessed on 1 December 2017).
- Harrell, F.E., Jr. Package ‘Hmisc’: Harrell Miscellaneous. R Foundation for Statistical Computing. 2017. Available online: https://cran.r-project.org/web/packages/Hmisc/Hmisc.pdf (accessed on 1 December 2017).
- Wilson, S.K.; Burgess, S.C.; Cheal, A.J.; Emslie, M.; Fisher, R.; Miller, I.; Polunin, N.V.; Sweatman, H. Habitat utilization by coral reef fish: Implications for specialists vs. generalists in a changing environment. J. Anim. Ecol. 2008, 77, 220–228. [Google Scholar] [CrossRef]
- Heupel, M.R.; Williams, A.J.; Welch, D.J.; Davies, C.R.; Penny, A.; Kritzer, J.P.; Marriott, R.J.; Mapstone, B.D. Demographic characteristics of exploited tropical lutjanids: A comparative analysis. Fish. Bull. 2010, 108, 420–432. [Google Scholar]
- Froese, R.; Pauly, D. FishBase. World Wide Web Electronic Publication. 2018. Available online: www.fishbase.org (accessed on 10 July 2015).
- Bray, J.R.; Curtis, J.T. An ordination of the upland forest communities of southern Wisconsin. Ecol. Monogr. 1957, 27, 325–349. [Google Scholar] [CrossRef]
- Oksanen, J. Multivariate Analysis of Ecological Communities in R: Vegan Tutorial. 2006. Available online: http://cc.oulu.fi/~jarioksa/opetus/metodi/vegantutor.pdf (accessed on 1 November 2015).
- Guyon, I.; Elisseeff, A. An introduction to variable and feature selection. J. Mach. Learn. Res. 2003, 3, 1157–1182. [Google Scholar]
- Rees, A.; Yearsley, G.; Gowlett-Holmes, K.; Pogonoski, J. Codes for Australian Aquatic Biota (Online Version); CSIRO Marine and Atmospheric Research; World Wide Web Electronic Publication: Hobart, Australia, 1999; Available online: http://www.cmar.csiro.au/caab/ (accessed on 10 June 2016).
- Newman, S.J.; Williams, D.M.; Russ, G.R. Patterns of zonation of assemblages of the Lutjanidae, Lethrinidae and Serranidae (Epinephelinae) within and among mid-shelf and outer-shelf reefs in the central Great Barrier Reef. Mar. Freshw. Res. 1997, 48, 119–128. [Google Scholar] [CrossRef]
- Brokovich, E.; Ayalon, I.; Einbinder, S.; Segev, N.; Shaked, Y.; Genin, A.; Kark, S.; Kiflawi, M. Grazing pressure on coral reefs decreases across a wide depth gradient in the Gulf of Aqaba, Red Sea. Mar. Ecol. Prog. Ser. 2010, 399, 69–80. [Google Scholar] [CrossRef]
- Asher, J.; Williams, I.D.; Harvey, E.S. Mesophotic depth gradients impact reef fish assemblage composition and functional group partitioning in the Main Hawaiian Islands. Front. Mar. Sci. 2017, 4. [Google Scholar] [CrossRef]
- Pinheiro, H.T.; Goodbody-Gringley, G.; Jessup, M.E.; Shepherd, B.; Chequer, A.D.; Rocha, L.A. Upper and lower mesophotic coral reef fish communities evaluated by underwater visual censuses in two Caribbean locations. Coral Reefs 2016, 35, 139–151. [Google Scholar] [CrossRef]
- Pyle, R.L.; Boland, R.; Bolick, H.; Bowen, B.W.; Bradley, C.J.; Kane, C.; Kosaki, R.K.; Langston, R.; Longenecker, K.; Montgomery, A.; et al. A comprehensive investigation of mesophotic coral ecosystems in the Hawaiian Archipelago. PeerJ 2016, 4, e2475. [Google Scholar] [CrossRef]
- Bridge, T.C.L.; Luiz, O.J.; Coleman, R.R.; Kane, C.N.; Kosaki, R.K. Ecological and morphological traits predict depth-generalist fishes on coral reefs. Proc. R. Soc. B Biol. Sci. 2016, 283. [Google Scholar] [CrossRef]
- Weaver, D.; Dennis, G.; Sulak, K. Northeastern Gulf of Mexico and Marine Ecosystem Program: Community Structure and Trophic Ecology of Demersal Fishes on the Pinnacles Reef Tract: Final Synthesis Report; Department of the Interior and Minerals Management Service: New Orleans, LA, USA, 2001.
- Bryan, D.R.; Kilfoyle, K.; Gilmore, R.G.; Spieler, R.E. Characterization of the mesophotic reef fish community in south Florida, USA. J. Appl. Ichthyol. 2013, 29, 108–117. [Google Scholar] [CrossRef]
- Goldstein, E.D.; D’Alessandro, E.K.; Sponaugle, S. Fitness consequences of habitat variability, trophic position, and energy allocation across the depth distribution of a coral-reef fish. Coral Reefs 2017, 36, 957–968. [Google Scholar] [CrossRef]
- Papastamatiou, Y.P.; Meyer, C.G.; Kosaki, R.K.; Wallsgrove, N.J.; Popp, B.N. Movements and foraging of predators associated with mesophotic coral reefs and their potential for linking ecological habitats. Mar. Ecol. Prog. Ser. 2015, 521, 155–170. [Google Scholar] [CrossRef]
- Weaver, D.C.; Sedberry, G.R. Trophic subsidies at the Charleston Bump: Food web structure of reef fishes on the continental slope of the southeastern United States. In Island in the Stream: Oceanography and Fisheries of the Charleton Bump; Sedberry, G.R., Ed.; American Fisheries Society Symposium 25: Bethesda, MD, USA, 2001; pp. 137–152. [Google Scholar]
- Pope, J. Stock Assessment in Multistock Fisheries, with Special Reference to the Trawl Fishery in the Gulf of Thailand; South China Seas Development and Coordinating Programme: Manila, Philippines, 1979. [Google Scholar]
- Tulloch, A.I.T.; Chadès, I.; Lindenmayer, D.B. Species co-occurrence analysis predicts management outcomes for multiple threats. Nat. Ecol. Evol. 2018, 2, 465–474. [Google Scholar] [CrossRef]
- Hill, N.A.; Barrett, N.; Lawrence, E.; Hulls, J.; Dambacher, J.M.; Nichol, S.; Williams, A.; Hayes, K.R. Quantifying fish assemblages in large, offshore marine protected areas: An Australian case study. PLoS ONE 2014, 9, e110831. [Google Scholar] [CrossRef]
- Bacheler, N.M.; Schobernd, Z.H.; Berrane, D.J.; Schobernd, C.M.; Mitchell, W.A.; Teer, B.Z.; Gregalis, K.C.; Glasgow, D.M. Spatial distribution of reef fish species along the Southeast US Atlantic Coast inferred from underwater video survey data. PLoS ONE 2016, 11, e0162653. [Google Scholar] [CrossRef] [PubMed]
- Kosaki, R.K.; Pyle, R.L.; Leonard, J.C.; Hauk, B.B.; Whitton, R.K.; Wagner, D. 100% endemism in mesophotic reef fish assemblages at Kure Atoll, Hawaiian Islands. Mar. Biodivers. 2016, 1–2. [Google Scholar] [CrossRef]
- Baldwin, C.C.; Tornabene, L.; Robertson, D.R. Below the Mesophotic. Sci. Rep. 2018, 8, 4920. [Google Scholar] [CrossRef]
- Wahab, M.A.A.; Radford, B.; Cappo, M.; Colquhoun, J.; Stowar, M.; Depczynski, M.; Miller, K.; Heyward, A. Biodiversity and spatial patterns of benthic habitat and associated demersal fish communities at two tropical submerged reef ecosystems. Coral Reefs 2018, 37, 327–343. [Google Scholar] [CrossRef]
- Kahng, S.E.; Copus, J.M.; Wagner, D. Recent advances in the ecology of mesophotic coral ecosystems (MCEs). Curr. Opin. Environ. Sustain. 2014, 7, 72–81. [Google Scholar] [CrossRef]
- Kahng, S.; Copus, J.M.; Wagner, D. Mesophotic Coral Ecosystems. In Marine Animal Forests: The Ecology of Benthic Biodiversity Hotspots; Rossi, S., Bramanti, L., Gori, A., Orejas, C., Eds.; Springer International Publishing: Berlin, Germany, 2017; pp. 185–206. [Google Scholar]
- Bridge, T.; Guinotte, J. Mesophotic Coral Reef Ecosystems in the Great Barrier Reef World Heritage Area: Their Potential Distribution and Possible Role as Refugia from Disturbance; Great Barrier Reef Marine Park Authority: Queensland, Australia, 2012. [Google Scholar]
- Bongaerts, P.; Frade, P.R.; Hay, K.B.; Englebert, N.; Latijnhouwers, K.R.W.; Bak, R.P.M.; Vermeij, M.J.A.; Hoegh-Guldberg, O. Deep down on a Caribbean reef: Lower mesophotic depths harbor a specialized coral-endosymbiont community. Sci. Rep. 2015, 5, 7652. [Google Scholar] [CrossRef]
- Englebert, N.; Bongaerts, P.; Muir, P.; Hoegh-Guldberg, O.; Hay, K.B. Deepest Zooxanthellate Corals of the Great Barrier Reef and Coral Sea. Mar. Biodivers. 2014. [Google Scholar] [CrossRef]
- Spalding, H.L. Ecology of Mesophotic Macroalgae and Halimeda kanaloana Meadows in the Main Hawaiian Islands. Ph.D. Thesis, University of Hawaii at Manoa, Honolulu, HI, USA, 2012. [Google Scholar]
- Wagner, D.; Barkman, A.; Spalding, H.; Calcinai, B.; Godwin, S. A Photographic Guide to the Benthic Flora and Fauna from Mesophotic Coral Ecosystems in the Papahānaumokuākea Marine National Monument; U.S. Department of Commerce, National Oceanic and Atmospheric Administration, Office of National Marine Sanctuaries: Silver Spring, MD, USA, 2016; 86p.
- Olavo, G.; Costa, P.A.; Martins, A.S.; Ferreira, B.P. Shelf-edge reefs as priority areas for conservation of reef fish diversity in the tropical Atlantic. Aquat. Conserv. Mar. Freshw. Ecosyst. 2011, 21, 199–209. [Google Scholar] [CrossRef]
- Moffitt, R.; Parrish, F.A. Habitat and life history of juvenile Hawaiian pick snapper, Pristipomoides filamentosus. Pac. Sci. 1996, 50, 371–381. [Google Scholar]
- Smith, C.L. A spawning aggregation of Nassau grouper, Epinephelus striatus (Bloch). Trans. Am. Fish. Soc. 1972, 101, 257–261. [Google Scholar] [CrossRef]
- Heyman, W.D.; Kjerfve, B. Characterization of transient multi-species reef fish spawning aggregations at Gladden Spit, Belize. Bull. Mar. Sci. 2008, 83, 531–551. [Google Scholar]
- Mourier, J.; Maynard, J.; Parravicini, V.; Ballesta, L.; Clua, E.; Domeier, M.L.; Planes, S. Extreme inverted trophic pyramid of reef sharks supported by spawning groupers. Curr. Biol. 2016, 26, 2011–2016. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.T.; Bridge, T.C.L.; Beaman, R.J.; Webster, J.M.; Nichol, S.L.; Brooke, B.P. Submerged banks in the Great Barrier Reef, Australia, greatly increase available coral reef habitat. ICES J. Mar. Sci. J. Cons. 2013, 70, 284–293. [Google Scholar] [CrossRef]
- Moore, C.H.; Radford, B.T.; Possingham, H.P.; Heyward, A.J.; Stewart, R.R.; Watts, M.E.; Prescott, J.; Newman, S.J.; Harvey, E.S.; Fisher, R.; et al. Improving spatial prioritisation for remote marine regions: Optimising biodiversity conservation and sustainable development trade-offs. Sci. Rep. 2016, 6, 32029. [Google Scholar] [CrossRef]
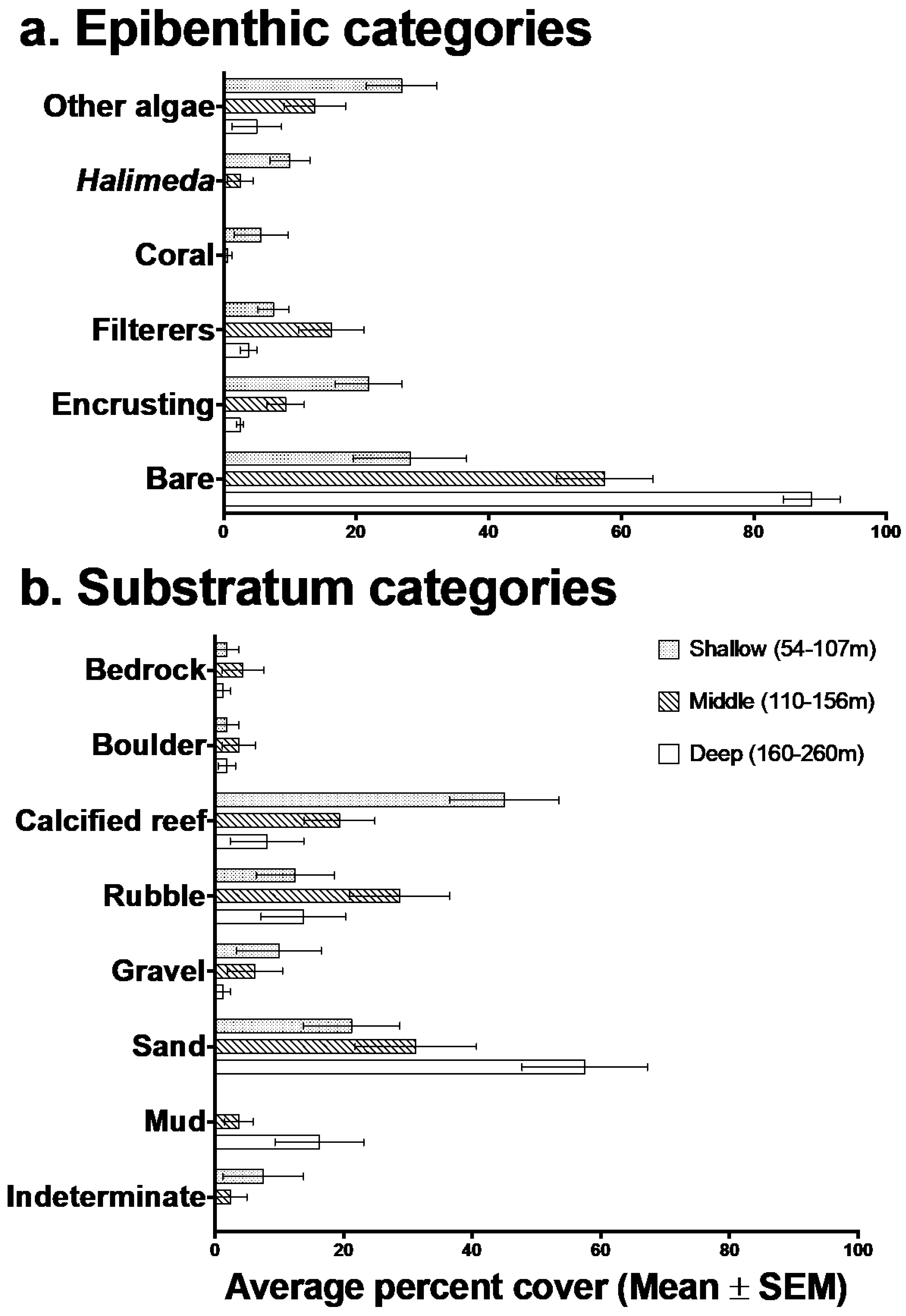


| Covariate Name (Abbreviation) | Covariate Type | Definition | Reference | |
|---|---|---|---|---|
| Bedrock | % composition of seafloor by substratum categories | FOV estimated % Bedrock | ||
| Boulder | % composition of seafloor by substratum categories | FOV estimated % Boulder | Moore et al., 2009 [93] | |
| Calcified reef | % composition of seafloor by substratum categories | FOV estimated % Calcareous reef | Moore et al., 2009 | |
| Gravel | % composition of seafloor by substratum categories | FOV estimated % Gravel (2–64mm) | Haywood et al., 2008 [94] Holmes et al., 2008 [95] Malcolm et al., 2016 [96] | |
| Indeterminate | % composition of seafloor by substratum categories | FOV estimated % Indeterminate | ||
| Mud | % composition of seafloor by substratum categories | FOV estimated % Mud/silt | Haywood et al., 2008 | |
| Rubble | % composition of seafloor by substratum categories | FOV estimated % Rubble | ||
| Sand | % composition of seafloor by substratum categories | FOV estimated % Sand | Malcolm et al., 2016 Kane & Tissot 2017 [36] | |
| Filtering organisms | % composition of seafloor by epibenthic categories | % combined Fans, Hydroids, Sponges, Whips | Holmes et al., 2008 | |
| Encrusting organisms | % composition of seafloor by epibenthic categories | FOV estimated % combined Bryozoans/encrusting animals, coralline algae | ||
| Coral | % composition of seafloor by epibenthic categories | FOV estimated % combined Hard coral and Soft coral | Garcia-Sais 2010 [2] Kane & Tissot 2017 | |
| Bare | % composition of seafloor by epibenthic categories | FOV estimated % no epibenthic cover | ||
| Plants | % composition of seafloor by epibenthic categories | FOV estimated % combined Macro-algae and Seagrass | Holmes et al., 2008 | |
| Halimeda | % composition of seafloor by epibenthic categories | FOV estimated % Halimeda | ||
| Name | Source | Description | Possible Ecological Context | Reference |
| Depth * (m) | Vessel depth sounder | Depth below sea-level | Location relative to Photic Zone Potential impact by waves and storms Location relative to thermoclines/haloclines | Costa et al., 2014 [97] Oyafuso et al., 2017 [98] Kane & Tissot 2017 Moore et al., 2009 Moore et al., 2011 [99] |
| Latitude | Handheld GPS unit | Position of the deployment | Location relative to latitudinal gradients | Cappo et al., 2007 |
| Longitude | Handheld GPS unit | Position of the deployment | Location relative to longitudinal gradients | Cappo et al., 2007 |
| Easting ** | Bathymetry derivative | Easterly component of the kernel azimuth | Level of exposure or protection from oceanographic processes | Hirzel et al., 2002 [100] |
| Northing ** | Bathymetry derivative | Northerly component of the kernel azimuth | Level of exposure or protection from oceanographic processes | Hirzel et al., 2002 |
| Slope ** (Degree) | Bathymetry derivative | Change in elevation as a function of distance within the kernel | Indicate activity of gravity driven processes Indication of hard substratum | Dartnell and Gardner 2004 [101] Misa et al., 2013 [102] Moore et al., 2009 |
| Topographic Position Index ** (TPI) | Bathymetry derivative | Difference between center kernel value and the average of all kernel values. Example of TPI interpretation as defined in Weiss 2001 (SD is standard deviation of bathymetry): Ridge: z0 > SD Upper slope: SD ≥ z0 > 0.5 SD Middle slope: 0.5 SD ≥ z0 ≥ −0.5 SD, slope > 5° Flat area: 0.5 SD ≥ z0 ≥ −0.5 SD, slope ≤ 5° Lower slope: −0.5 SD > z0 > -SD Valley: z0 < -SD | Relative topographic position in the neighborhood: Positive TPI values are higher than their surroundings (i.e., ridges) and negative TPI values are lower than their surroundings (i.e., valleys). TPI values near zero are flat areas. 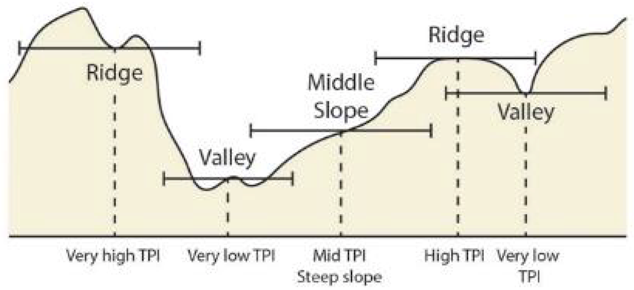 (re-drawn from [103]) | Weiss 2001 [104] Iampietro et al., 2005 [105] Moore et al., 2009 |
| Terrain Ruggedness Index ** | Bathymetry derivative | Average of the absolute difference between the center kernel values and each of the other kernel values | Index of surface roughness indicating degree of structural complexity | Riley et al., 1999 [106] |
| Range*** | Bathymetry derivative | Difference between the maximum and minimum values within the kernel | Index of surface roughness indicating degree of structural complexity | Dartnell 2000 [107] Yates et al., 2016 [108] Moore et al., 2009 Holmes et al., 2008 |
| Surface Ratio ** | Bathymetry derivative | Ratio of the kernel surface area and planimetric area | Relative vertical relief indicating degree of structural complexity | Jenness 2004 [109] Moore et al., 2011 |
| Standard Deviation *** (m) | Bathymetry derivative | Standard deviation of values within the kernel | Index of surface roughness | Costa et al., 2014 |
| Curvature ** (Degrees/m) | Bathymetry derivative | Index of concavity/convexity measured within the kernel | Measure of overall curvature within kernel (planform left to right + −, 0; profile top to bottom, −, +, 0)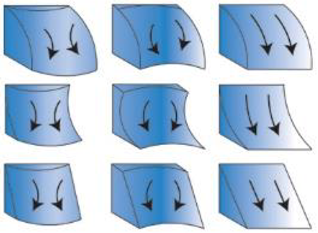 (re-drawn from “Curvature type” ArcGIS help files) | Zevenbergen and Thorne 1987 [92] |
| Planar Curvature ** (Degrees/m) | Bathymetry derivative | Index of concavity/convexity measured perpendicular to slope within the kernel | Identifies ridges, valleys, and flat slopes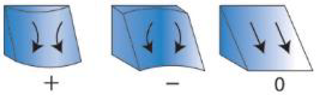 (re-drawn from “Curvature type” ArcGIS help files) | Zevenbergen and Thorne 1987 |
| Profile Curvature ** (Degrees/m) | Bathymetry derivative | Index of concavity/convexity measured parallel to the slope within the kernel | Concave or convex slopes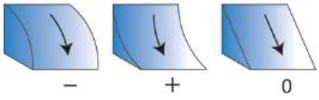 (re-drawn from “Curvature type” ArcGIS help files) | Zevenbergen and Thorne 1987 Moore et al., 2009 |
| Acoustic Backscatter * (Decibels) | Backscatter derivative | Acoustic backscatter | Proxy for seabed substratum | Hughes-Clarke et al., 1996 [80] |
| Ave Backscatter *** (Decibels) | Backscatter derivative | Average backscatter within the kernel | Proxy for seabed substratum | Brown et al., 2011 [84] |
| StdDev Backscatter *** (Decibels) | Backscatter derivative | Standard deviation of values within the kernel | Variation in substratum within the kernel | Brown et al., 2011 |
| Location | Myrmidon Reef | Northern Submerged Shoals | Viper Reef | Inter-Reefal Transect | |
|---|---|---|---|---|---|
| Depth strata | Shallow (54–115 m) | n sites = 8 Average similarity: 28.0% Individual species contributions: Carangoides caeruleopinnatus, (15.3%) Lutjanus bohar (13.6%) Carcharhinus amblyrhynchos (9.9%) Aphareus rutilans (8.9%) Gymnocranius euanus (8.9%) Cirrhilabrus roseafascia (6.0%) Pristipomoides filamentosus (5.3%) Lethrinus miniatus (5.0%). | n sites = 4 Average similarity: 15.9% Individual species contributions: Carangoides caeruleopinnatus (21.7%) Gymnocranius grandoculis (13.1%) Carcharhinus albimarginatus (10.0%) Lethrinus rubrioperculatus (9.1%) Carcharhinus amblyrhynchos (7.2%) Pomacanthus imperator (7.2%) Plectropomus leopardus (7.2%) | n sites = 4 Average similarity: 25.6% Individual species contributions: Carangoides dinema (23.6%) Echeneis naucrates (11.4%) Lethrinus olivaceus (9.5%) Aphareus rutilans (4.7%) Carcharhinus albimarginatus (4.7%) Carangoides fulvoguttatus (4.7%) Lutjanus bohar (4.7%) Parapercis sp. (4.7%) Epinephelus cyanopodus (4.7%) | n sites = 2 Individual species contributions: All similarities are zero |
| Middle (128–160 m) | n sites = 8 Average similarity: 29.5% Individual species contributions: Aphareus rutilans (31.2%) Pristipomoides typus (14.3%) Pristipomoides filamentosus (13.1%) Parapercis nebulosa (10.3%) Pristipomoides multidens (9.4%) | n sites = 3 Average similarity: 58.3% Individual species contributions: Bodianus sp. (10.4%) Wattsia mossambica (10.4%) Aphareus rutilans (10.4%) Pristipomoides filamentosus (10.4%) Pristipomoides multidens (10.4%) Pristipomoides typus (10.4%) Gymnosarda unicolor (10.4%) | n sites = 2 Average similarity: 28.57 Individual species contributions: Carcharhinus albimarginatus (100%) | n sites = 1 | |
| Deep (179–260 m) | n sites = 8 Average similarity: 17.0% Individual species contributions: Pristipomoides argyrogrammicus (39.0%) Pristipomoides multidens (31.2%) | n sites = 3 Average similarity: 31.7% Individual species contributions: Gymnosarda unicolor (48.9%) Seriola dumerili (13.2%) Pristipomoides argyrogrammicus (13.2%) | n sites = 0 | n sites = 1 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sih, T.L.; Daniell, J.J.; Bridge, T.C.L.; Beaman, R.J.; Cappo, M.; Kingsford, M.J. Deep-Reef Fish Communities of the Great Barrier Reef Shelf-Break: Trophic Structure and Habitat Associations. Diversity 2019, 11, 26. https://doi.org/10.3390/d11020026
Sih TL, Daniell JJ, Bridge TCL, Beaman RJ, Cappo M, Kingsford MJ. Deep-Reef Fish Communities of the Great Barrier Reef Shelf-Break: Trophic Structure and Habitat Associations. Diversity. 2019; 11(2):26. https://doi.org/10.3390/d11020026
Chicago/Turabian StyleSih, Tiffany L., James J. Daniell, Thomas C.L. Bridge, Robin J. Beaman, Mike Cappo, and Michael J. Kingsford. 2019. "Deep-Reef Fish Communities of the Great Barrier Reef Shelf-Break: Trophic Structure and Habitat Associations" Diversity 11, no. 2: 26. https://doi.org/10.3390/d11020026
APA StyleSih, T. L., Daniell, J. J., Bridge, T. C. L., Beaman, R. J., Cappo, M., & Kingsford, M. J. (2019). Deep-Reef Fish Communities of the Great Barrier Reef Shelf-Break: Trophic Structure and Habitat Associations. Diversity, 11(2), 26. https://doi.org/10.3390/d11020026





