Spatial Patterns and Short-term Changes of Coral Assemblages Along a Cross-shelf Gradient in the Southwestern Lagoon of New Caledonia
Abstract
1. Introduction
2. Materials and Methods
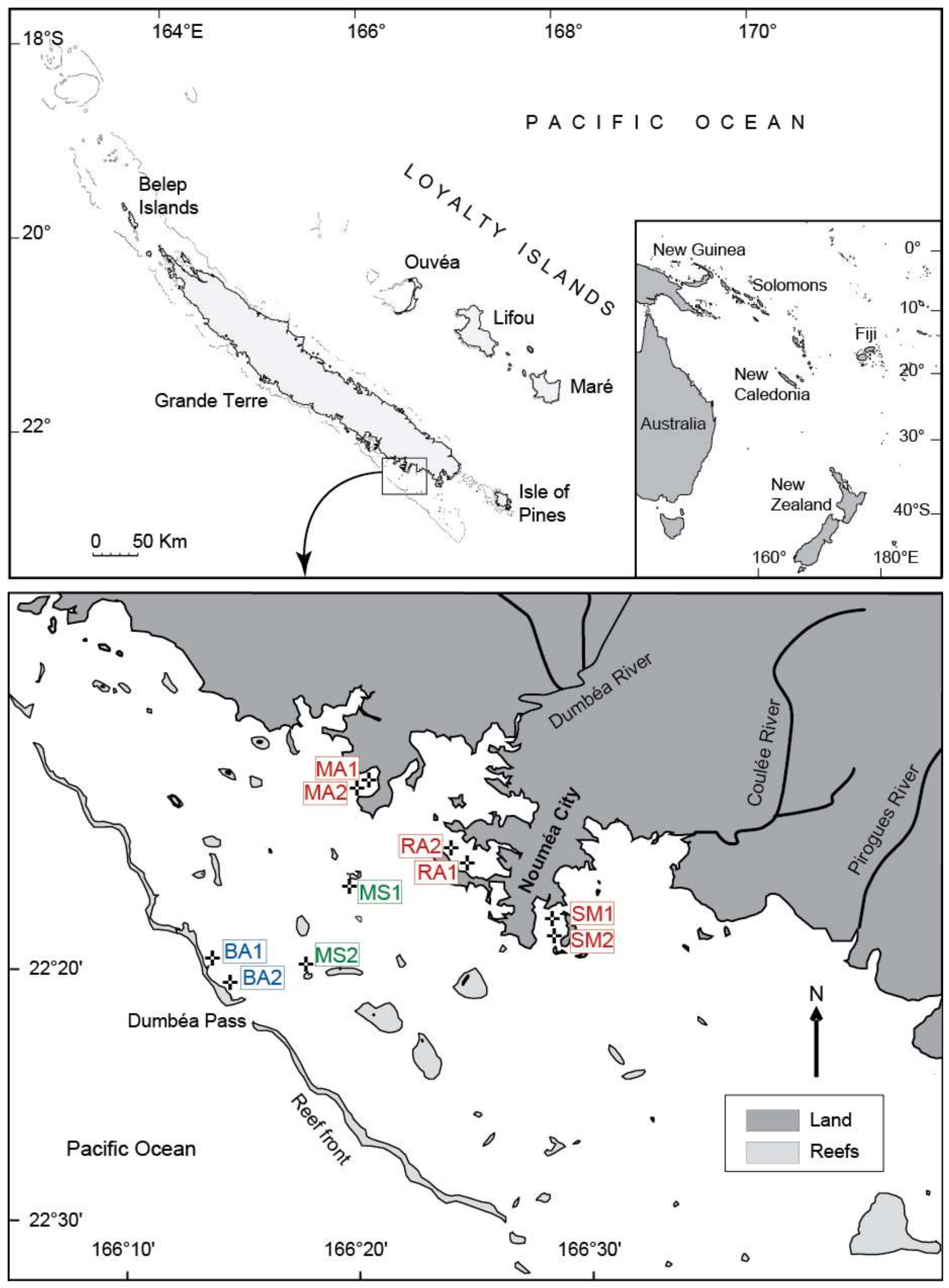
2.1. Study Area
2.2. Coral Sampling
2.3. Data Analysis
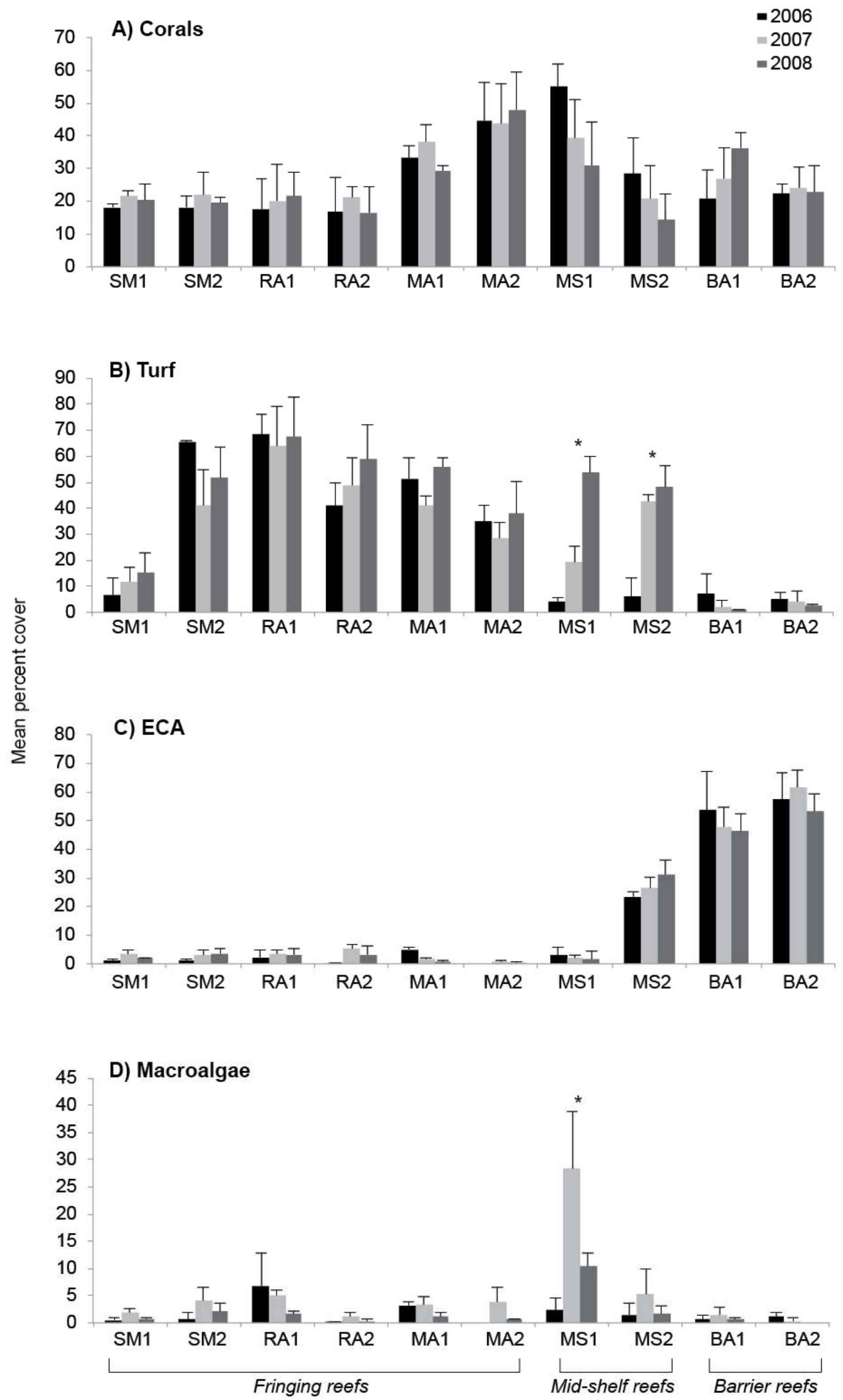
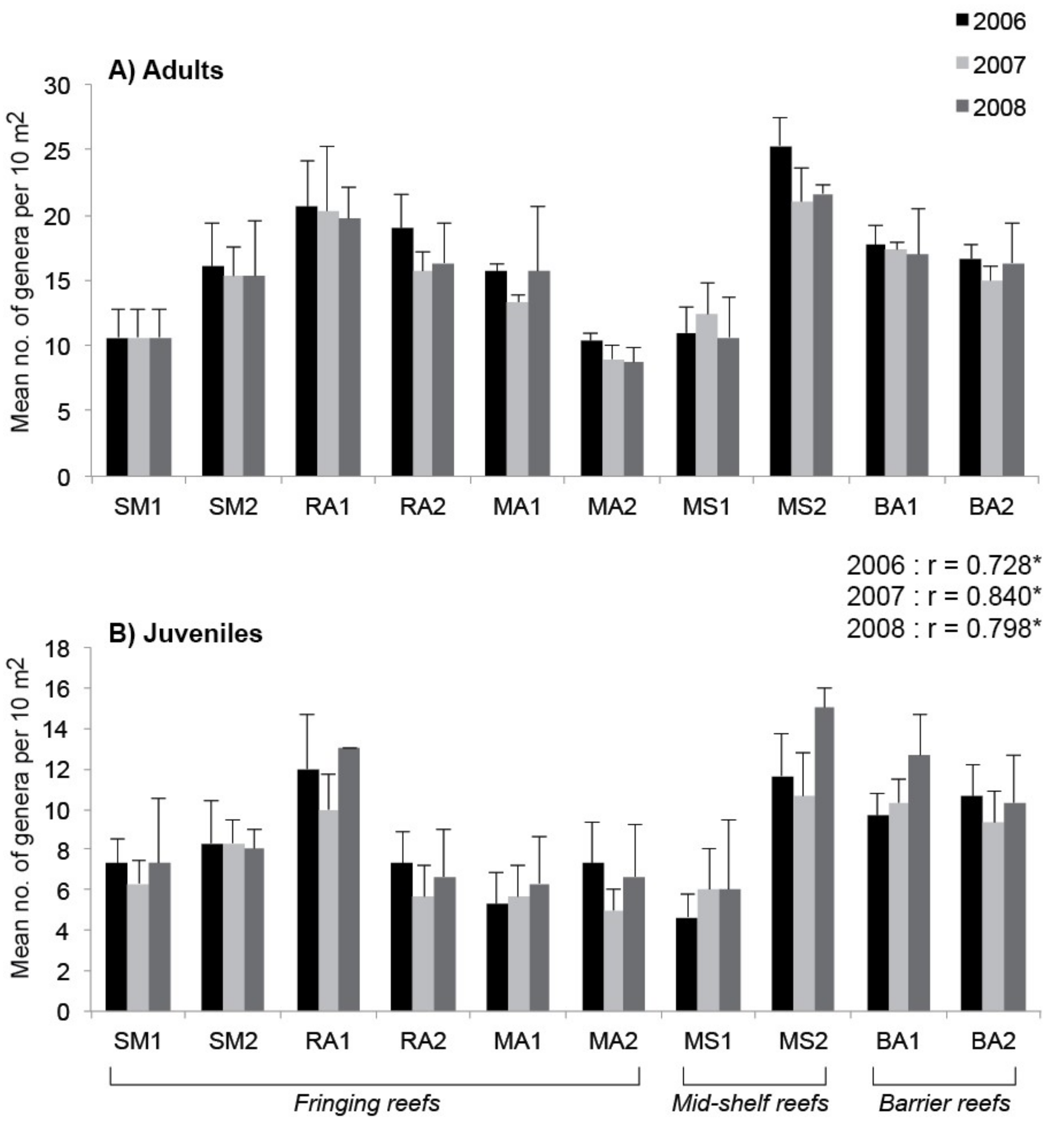
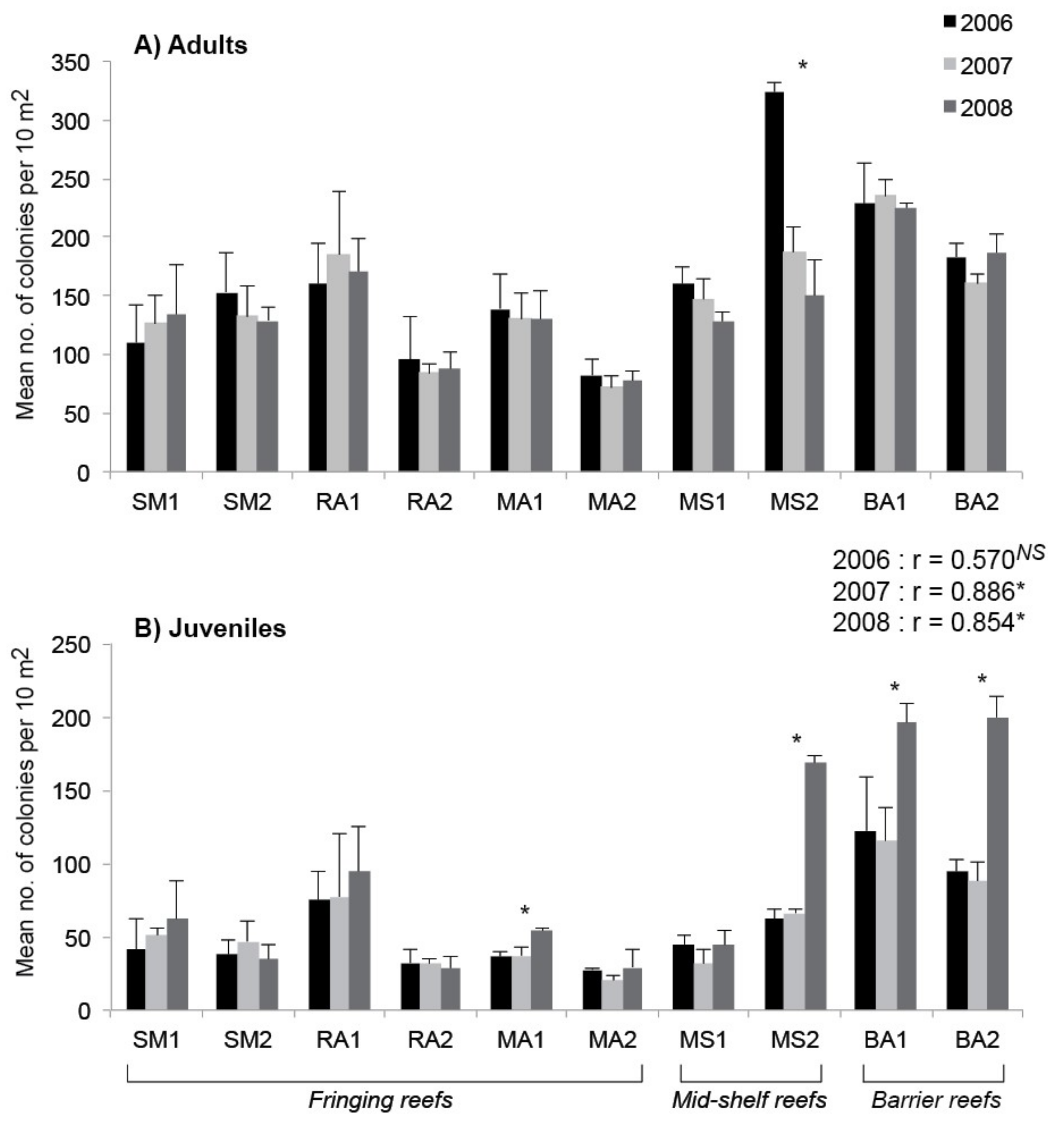
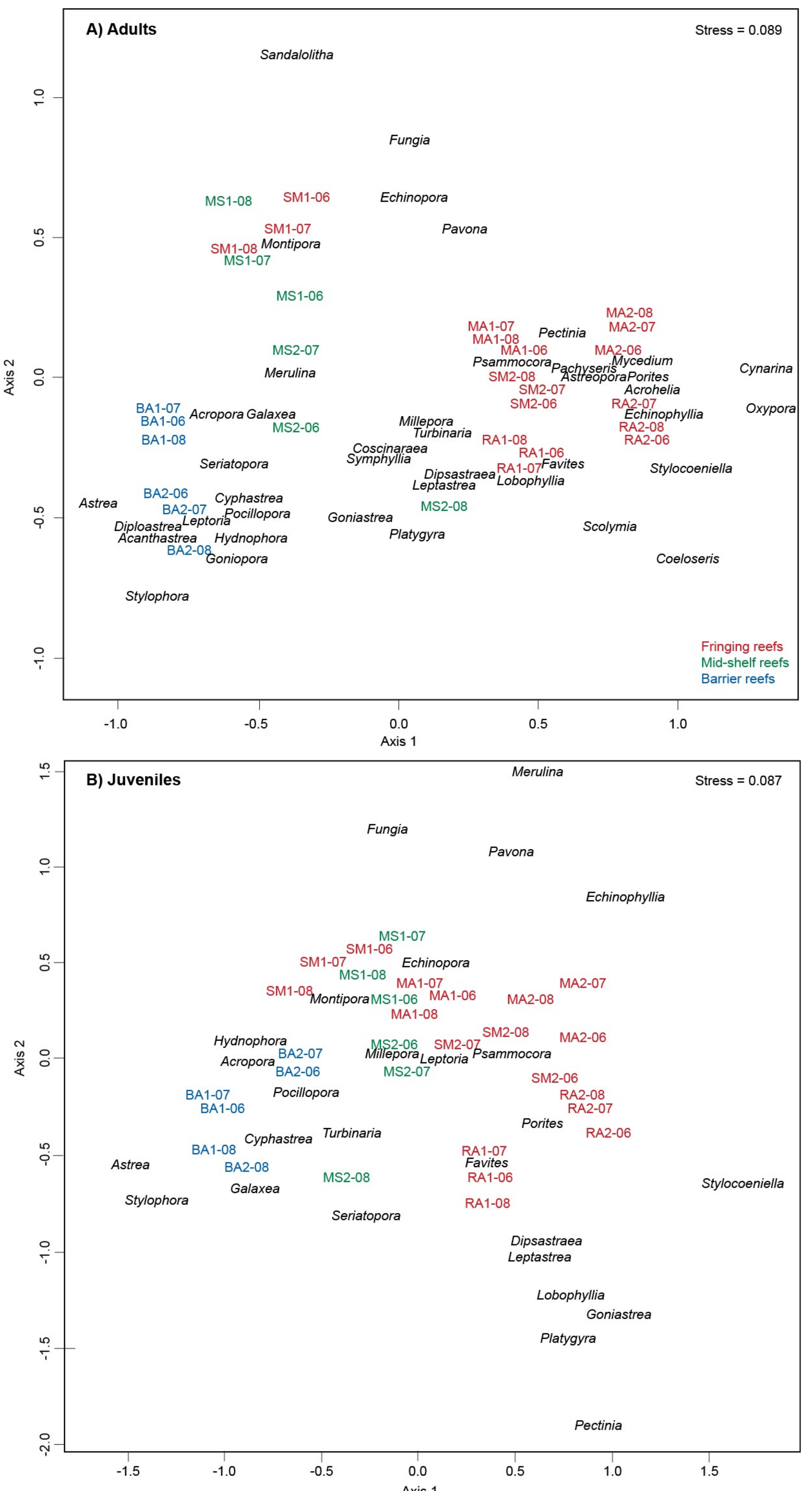
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Reaka-Kudla, M.L. The global biodiversity of coral reefs: A comparison with rain forests. In Biodiversity II: Understanding and Protecting Our Biological Resources; Reaka-Kudla, M.L., Wilson, D.E., Wilson, E.O., Eds.; The National Academy of Science, Joseph Henry Press: Washington, DC, USA, 1997; pp. 83–108. [Google Scholar]
- Fisher, R.; O’Leary, R.A.; Low-Choy, S.; Mengersen, K.; Knowlton, N.; Brainard, R.E.; Caley, M.J. Species richness on coral reefs and the pursuit of convergent global estimates. Curr. Biol. 2015, 25, 500–505. [Google Scholar] [CrossRef] [PubMed]
- Moberg, F.; Folke, C. Ecological goods and services of coral reef ecosystems. Ecol. Econ. 1999, 29, 215–233. [Google Scholar] [CrossRef]
- Kittinger, J.N.; Finkbeiner, E.M.; Glazier, E.W.; Crowder, L.B. Human dimensions of coral reef social-ecological systems. Ecol. Soc. 2012, 17, 17. [Google Scholar] [CrossRef]
- Connell, J.H. Diversity in a tropical rain forests and coral reefs. Science 1978, 199, 1302–1310. [Google Scholar] [CrossRef] [PubMed]
- Done, T.J. Coral zonation: Its nature and significance. In Perspectives on Coral Reefs; Barnes, D.J., Ed.; Australian Institute of Marine Science: Townsville, Australia, 1983; pp. 107–147. [Google Scholar]
- Huston, M.A. Patterns of species diversity on coral reefs. Annu. Rev. Ecol. Syst. 1985, 16, 149–177. [Google Scholar] [CrossRef]
- Cornell, H.V.; Karlson, R.H. Species richness of reef-building corals determined by local and regional processes. J. Anim. Ecol. 1996, 65, 233–241. [Google Scholar] [CrossRef]
- Connell, J.H.; Hughes, T.P.; Wallace, C.C. A 30-year study of coral abundance, recruitment, and disturbance at several scales in space and time. Ecol. Monogr. 1997, 67, 461–488. [Google Scholar] [CrossRef]
- Karlson, R.H.; Cornell, H.V. Species richness of coral assemblages: Detecting regional influences at local spatial scales. Ecology 2002, 83, 452–463. [Google Scholar] [CrossRef]
- Goreau, T.F. The ecology of Jamaican coral reefs. I. Species composition and zonation. Ecology 1959, 40, 67–90. [Google Scholar] [CrossRef]
- Ninio, R.; Meekan, M.G. Spatial patterns in benthic communities and the dynamics of a mosaic ecosystem on the Great Barrier Reef, Australia. Coral Reefs 2002, 21, 95–103. [Google Scholar] [CrossRef]
- Karlson, R.H.; Cornell, H.V.; Hughes, T.P. Coral communities are regionnally enriched along an oceanic biodiversity gradient. Nature 2004, 429, 867–870. [Google Scholar] [CrossRef] [PubMed]
- Cleary, D.F.R.; Polonia, A.R.M.; Renema, W.; Hoeksema, B.W.; Rachello-Dolmen, P.G.; Moolenbeek, R.G.; Budiyanto, A.; Tufi, Y.; Draisma, S.G.A.; Rud’homme van Reine, W.F.; et al. Variation in the composition of corals, fishes, sponges, echinoderms, ascidians, molluscs, foraminifera and macroalgae across a pronounced in-to-offshore environmental gradient in the Jakarta Bay-Thousand Islands coral reef complex. Mar. Pollut. Bull. 2016, 110, 701–717. [Google Scholar] [CrossRef] [PubMed]
- Teichberg, M.; Wild, C.; Bednarz, V.N.; Kegler, H.F.; Lukman, M.; Gärdes, A.A.; Heiden, J.P.; Weiand, L.; Abu, N.; Nasir, A.; et al. Spatio-temporal patterns in coral reef communities of the Spermonde Archipelago, 2012–2014, I: Comprehensive reef monitoring of water and benthic indicators reflect changes in reef health. Front. Mar. Sci. 2018, 5, 33. [Google Scholar] [CrossRef]
- Adjeroud, M. Factors influencing spatial patterns on coral reefs around Moorea, French Polynesia. Mar. Ecol. Prog. Ser. 1997, 159, 105–119. [Google Scholar] [CrossRef]
- Koop, K.; Booth, D.; Broadbent, A.; Brodie, J.; Bucher, D.; Capone, D.; Coll, J.; Dennison, W.; Erdmann, M.; Harrison, P.; et al. ENCORE: The effects of nutrient enrichment on coral reefs. Synthesis of results and conclusions. Mar. Pollut. Bull. 2001, 42, 91–120. [Google Scholar] [CrossRef]
- McCook, L.; Jompa, J.; Diaz-Pullido, G. Competition between corals and algae on coral reefs: A review of evidence and mechanisms. Coral Reefs 2001, 19, 400–417. [Google Scholar] [CrossRef]
- Fabricius, K.; De’ath, G.; McCook, L.; Turak, E.; Williams, D.Mc.B. Changes in algal, coral and fish assemblages along water quality gradients on the inshore Great Barrier Reef. Mar. Pollut. Bull. 2005, 51, 384–398. [Google Scholar] [CrossRef]
- Caley, M.J.; Carr, M.H.; Hixon, M.A.; Hughes, T.P.; Jones, G.P.; Menge, B.A. Recruitment and the local dynamics of open marine populations. Annu. Rev. Ecol. Syst. 1996, 27, 477–500. [Google Scholar] [CrossRef]
- Hughes, T.P.; Baird, A.H.; Dinsdale, E.A.; Moltschaniwskyj, N.A.; Pratchett, M.S.; Tanner, J.E.; Willis, B.L. Supply-side ecology works both ways: The link between benthic adults, fecundity, and larval recruits. Ecology 2000, 81, 2241–2249. [Google Scholar] [CrossRef]
- Doropoulos, C.; Ward, S.; Roff, G.; Gonzales-Rivero, M.; Mumby, P.J. Linking demographic processes of juvenile corals to benthic recovery trajectories in two common reef habitats. PLoS ONE 2015, 10, e0128535. [Google Scholar] [CrossRef]
- Adjeroud, M.; Kayal, M.; Penin, L. The importance of recruitment processes in the dynamics and resilience of reef coral assemblages. In Marine Animal Forests; Rossi, S., Bramanti, L., Gori, A., Orejas, C., Eds.; Springer: Cham, Switzerland, 2017; pp. 549–569. [Google Scholar]
- Kayal, M.; Lenihan, H.S.; Brooks, A.J.; Holbrook, S.J.; Schmitt, R.J.; Kendall, B.E. Predicting coral community recovery using multi-species population dynamics models. Ecol. Lett. 2018, 21, 1790–1799. [Google Scholar] [CrossRef]
- Hughes, T.P.; Connell, J.H. Multiple stressors on coral reefs: A long-term perspective. Limnol. Oceanogr. 1999, 44, 932–940. [Google Scholar] [CrossRef]
- Bellwood, D.R.; Hughes, T.P.; Folke, C.; Nyström, M. Confronting the coral reef crisis. Nature 2004, 429, 827–833. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O.; Mumby, P.J.; Hooten, A.J.; Steneck, R.S.; Greenfield, P.; Gomez, E.; Harvell, C.D.; Sale, P.F.; Edwards, A.J.; Caldeira, K.; et al. Coral reefs under rapid climate change and ocean acidification. Science 2007, 318, 1737–1742. [Google Scholar] [CrossRef]
- Edmunds, P.J.; Adjeroud, M.; Baskett, M.L.; Baums, I.B.; Budd, A.F.; Carpenter, R.C.; Fabina, N.S.; Fan, T.Y.; Franklin, E.C.; Gross, K.; et al. Persistence and change in community composition of reef corals through present, past, and future climates. PLoS ONE 2014, 9, e107525. [Google Scholar] [CrossRef] [PubMed]
- Hughes, T.P.; Barnes, M.L.; Bellwood, D.R.; Cinner, J.E.; Cumming, G.S.; Jackson, J.B.C.; Kleypas, J.; van de Leemput, I.A.; Lough, J.M.; Morrison, T.H.; et al. Coral reefs in the Anthropocene. Nature 2017, 546, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Hughes, T.P.; Baird, A.H.; Bellwood, D.R.; Card, M.; Connolly, S.R.; Folke, C.; Grosberg, R.; Hoegh-Guldberg, O.; Jackson, J.B.C.; Kleypas, J.; et al. Climate change, human impacts, and the resilience of coral reefs. Science 2003, 301, 929–933. [Google Scholar] [CrossRef] [PubMed]
- Hughes, T.P.; Kerry, J.T.; Baird, A.H.; Connolly, S.R.; Dietzel, A.; Eakin, C.M.; Heron, S.F.; Hoey, A.S.; Hoogenboom, M.; Lui, G.; et al. Global warming transforms coral reef assemblages. Nature 2018, 556, 492–496. [Google Scholar] [CrossRef] [PubMed]
- Anthony, K.R.; Marshall, P.A.; Abdulla, A.; Beeden, R.; Bergh, C.; Black, R.; Eakin, C.M.; Game, E.T.; Gooch, M.; Graham, N.A.; et al. Operationalizing resilience for adaptive coral reef management under global environmental change. Glob. Chang. Biol. 2015, 21, 48–61. [Google Scholar] [CrossRef] [PubMed]
- Adjeroud, M.; Kayal, M.; Iborra-Cantonnet, C.; Vercelloni, J.; Bosserelle, P.; Liao, V.; Chancerelle, Y.; Claudet, J.; Penin, L. Recovery of coral assemblages despite acute and recurrent disturbances on a South Central Pacific reef. Sci. Rep. 2018, 8, 9680. [Google Scholar] [CrossRef] [PubMed]
- Done, T.J. Patterns in the distribution of coral communities across the central Great Barrier Reef. Coral Reefs 1982, 1, 95–107. [Google Scholar] [CrossRef]
- Adjeroud, M. Zonation of coral assemblages in the Ryukyu islands (southern Japan): The importance of land-ocean gradients in reef habitats. Proceedings of 10th International Coral Reef Symposium, Okinawa, Japan, 28 June–2 July 2004; pp. 310–318. [Google Scholar]
- Cleary, D.F.R.; Becking, L.E.; de Voogd, N.J.; Renema, W.; de Beer, M.; van Soest, R.W.M.; Hoeksema, B.W. Variation in the diversity and composition of benthic taxa as a function of distance offshore, depth and exposure in the Spermonde Archipelago, Indonesia. Estuar. Coast. Shelf Sci. 2005, 65, 557–570. [Google Scholar] [CrossRef]
- Smith, T.B.; Nemeth, R.S.; Blondeau, J.; Calnan, J.M.; Kadison, E.; Herzlieb, S. Assessing coral reef health across onshore to offshore stress gradients in the US Virgin Islands. Mar. Pollut. Bull. 2008, 56, 1983–1991. [Google Scholar] [CrossRef] [PubMed]
- Ellis, J.; Anlauf, H.; Kürten, S.; Lozano-Cortés, D.; Alsaffar, Z.; Curdia, J.; Jones, B.; Carvalho, S. Cross shelf benthic biodiversity patterns in the Southern Red Sea. Sci. Rep. 2017, 7, 437. [Google Scholar] [CrossRef] [PubMed]
- Adjeroud, M.; Salvat, B. Spatial patterns in biodiversity of a fringing reef community along Opunohu Bay, Moorea, French Polynesia. Bull. Mar. Sci. 1996, 59, 175–187. [Google Scholar]
- Adjeroud, M. Zonation des communautés macrobenthiques le long de deux baies d’un écosystème corallien insulaire (Moorea, Polynésie française). Comptes Rendus Acad. Sci. Ser. III-Sci. Vie-Life Sci. 2000, 323, 305–313. [Google Scholar] [CrossRef]
- Adjeroud, M.; Fernandez, J.M.; Carroll, A.G.; Harrison, P.L.; Penin, L. Spatial patterns and recruitment processes of coral assemblages among contrasting environmental conditions in the southwestern lagoon of New Caledonia. Mar. Pollut. Bull. 2010, 61, 375–386. [Google Scholar] [CrossRef]
- Pichon, M. Scleractinia of New Caledonia: Check list of reef dwelling species. In Compendium of Marine Species from New Caledonia, second edition; Payri, C., Richer de Forges, B., Eds.; Doc. Sci. Tech. II7; IRD Editions: Nouméa, New Caledonia, 2007; pp. 149–157. [Google Scholar]
- Chevalier, J.P. Coral reefs of New Caledonia. In Biology and Geology of Coral Reefs; Jones, O.A., Endean, R., Eds.; Academic Press: New York, NY, USA; London, UK, 1973; pp. 143–167. [Google Scholar]
- Andréfouët, S.; Cabioch, G.; Flamand, B.; Pelletier, B. A reappraisal of the diversity of geomorphological and genetic processes of New Caledonian coral reefs: A synthesis from optical remote sensing, coring and acoustic multibeam observations. Coral Reefs 2009, 28, 691–707. [Google Scholar] [CrossRef]
- Wantiez, L. Les récifs coralliens de Nouvelle-Calédonie en 2006: États des lieux et réseau de suivi. Rev. Ecol. Terre Vie 2008, 63, 117–132. [Google Scholar]
- Wantiez, L.; Château, O.; Le Mouellic, S. Initial and mid-term impacts of cyclone Erica on coral reef fish communities and habitat in the South Lagoon Marine Park of New Caledonia. J. Mar. Biol. Assoc. 2006, 86, 1229–1236. [Google Scholar] [CrossRef]
- Guillemot, N.; Chabanet, P.; Le Pape, O. Cyclone effects on coral reef habitats in New Caledonia (South Pacific). Coral Reefs 2010, 29, 445–453. [Google Scholar] [CrossRef]
- Adjeroud, M.; Kayal, M.; Peignon, C.; Juncker, M.; Mills, S.C.; Beldade, R.; Dumas, P. Ephemeral and localized outbreaks of the coral predator Acanthaster cf. solaris in the southwestern lagoon of New Caledonia. Zool. Stud. 2018, 57, 4. [Google Scholar]
- Payri, C.; Benzoni, F.; André, L.V.; Houlbrèque, F. Le blanchissement corallien de 2016. In Nouvelle-Calédonie, Archipel de Corail; Payri, C.E., Ed.; IRD Editions/Solaris: Marseille, France, 2018; pp. 161–166. [Google Scholar]
- Jouon, A.; Douillet, P.; Ouillon, S.; Fraunié, P. Calculations of hydrodynamic time parameters in a semi-opened coastal zone using a 3D hydrodynamic model. Cont. Shelf Res. 2006, 26, 1395–1415. [Google Scholar] [CrossRef]
- Jacquet, S.; Delesalle, B.; Torréton, J.P.; Blanchot, J. Response of phytoplankton communities to increased anthropogenic influences (southwestern lagoon, New Caledonia). Mar. Ecol. Prog. Ser. 2006, 320, 65–78. [Google Scholar] [CrossRef]
- Mari, X.; Rochelle-Newall, E.; Torréton, J.P.; Pringault, O.; Jouon, A.; Migon, C. Water residence time: A regulatory factor of the DOM to TOM transfer efficiency. Limnol. Oceanogr. 2007, 52, 808–819. [Google Scholar] [CrossRef]
- Migon, C.; Ouillon, S.; Mari, X.; Nicolas, Z. Geochemical and hydrodynamic constraints on the distribution of trace metal concentrations in the lagoon of Nouméa, New Caledonia. Estuar. Coast. Shelf Sci. 2007, 74, 756–765. [Google Scholar] [CrossRef]
- Dalto, A.G.; Grémare, A.; Dinet, A.; Fichet, D. Muddy-bottom meiofauna responses to metal concentrations and organic enrichment in New Caledonia South-West Lagoon. Estuar. Coast. Shelf Sci. 2006, 67, 629–644. [Google Scholar] [CrossRef]
- Fernandez, J.M.; Ouillon, S.; Chevillon, C.; Douillet, P.; Fichez, R.; Le Gendre, R. A combined modelling and geochemical study of the fate of terrigenous inputs from mixed natural and mining sources in a coral reef lagoon (New Caledonia). Mar. Pollut. Bull. 2006, 52, 320–331. [Google Scholar] [CrossRef]
- Dumas, P.; Kulbicki, M.; Chifflet, S.; Fichez, R.; Ferraris, J. Environmental factors influencing urchin spatial distributions on disturbed coral reefs (New Caledonia, South Pacific). J. Exp. Mar. Biol. Ecol. 2007, 344, 88–100. [Google Scholar] [CrossRef]
- Breau, L. Etude de la Bioaccumulation des Métaux dans Quelques Espèces Marines Tropicales: Recherche de Bioindicateurs de Contamination et Application à la Surveillance de L’environnement Côtier dans le Lagon Sud-Ouest de la Nouvelle-Calédonie. Ph.D. Thesis, Université de La Rochelle, La Rochelle, France, 2003. [Google Scholar]
- Harrison, P.L.; Wallace, C.C. Reproduction, dispersal and recruitment of scleractinian corals. In Coral Reef Ecosystems, Ecosystems of the World; Dubinsky, Z., Ed.; Elsevier Science Publishers: Amsterdam, The Netherlands, 1990; Volume 25, Chapter 7; pp. 133–207. [Google Scholar]
- Loya, Y. Plotless and transect methods. In Coral Reefs: Research Methods; Stoddart, D.R., Johannes, R.E., Eds.; UNESCO: Paris, France, 1978; pp. 197–217. [Google Scholar]
- Cleary, D.F.R.; Suharsono; Hoeksema, B.W. Coral diversity across a disturbance gradient in the Pulau Seribu reef complex off Jakarta, Indonesia. Biodivers. Conserv. 2006, 15, 3653–3674. [Google Scholar] [CrossRef]
- Grimaud, J.; Kulbicki, M. Influence de la distance à l’océan sur les peuplements ichtyologiques des récifs frangeants de Nouvelle-Calédonie. Comptes Rendus Acad. Sci. Ser. III-Sci. Vie-Life Sci. 1998, 321, 923–931. [Google Scholar] [CrossRef]
- Moustaka, M.; Mohring, M.B.; Holmes, T.; Evans, R.D.; Thomson, D.; Nutt, C.; Stoddart, J.; Wilson, S.K. Cross-shelf heterogeneity of coral assemblages in Northwest Australia. Diversity 2019, 11, 15. [Google Scholar] [CrossRef]
- Edmunds, P.J. Patterns in the distribution of juvenile corals and coral reef community structure in St John, US Virgin Islands. Mar. Ecol. Prog. Ser. 2000, 202, 113–124. [Google Scholar] [CrossRef]
- Rogers, C.S.; Fitz, H.C.; Gilnack, M.; Beets, J.; Hardin, J. Scleractinian recruitment patterns at Salt River Canyon, St. Croix, U.S. Virgin Islands. Coral Reefs 1984, 3, 69–76. [Google Scholar] [CrossRef]
- Gleason, M.G. Coral recruitment in Moorea, French Polynesia: The importance of patch type and temporal variation. J. Exp. Mar. Biol. Ecol. 1996, 207, 79–101. [Google Scholar] [CrossRef]
- Carlon, D.B. Depth-related patterns of coral recruitment and cryptic suspention-feeding invertebrates on Guana Island, British Virgin Islands. Bull. Mar. Sci. 2001, 68, 525–541. [Google Scholar]
- Miller, M.W.; Weil, E.; Szmant, A.M. Coral recruitment and juvenile mortality as structuring factors for reef benthic communities in Biscayne National Park, USA. Coral Reefs 2000, 19, 115–123. [Google Scholar] [CrossRef]
- Mundy, C.N.; Babcock, R.C. Are vertical distribution patterns of scleractinian corals maintained by pre- or post-settlement processes? A case study of three contrasting species. Mar. Ecol. Prog. Ser. 2000, 198, 109–119. [Google Scholar] [CrossRef]
- Penin, L.; Adjeroud, M. Relative importance of recruitment and post-settlement processes in the maintenance of coral assemblages in an insular, fragmented reef system. Mar. Ecol. Prog. Ser. 2013, 473, 149–162. [Google Scholar] [CrossRef]
- Baird, A.H.; Babcock, R.C.; Mundy, C.P. Habitat selection by larvae influences the depth distribution of six common coral species. Mar. Ecol. Prog. Ser. 2003, 252, 289–293. [Google Scholar] [CrossRef]
- Penin, L.; Adjeroud, M.; Pratchett, M.S.; Hughes, T.P. Spatial distribution of juvenile and adult corals around Moorea (French Polynesia): Implications for population regulation. Bull. Mar. Sci. 2007, 80, 379–389. [Google Scholar]
- Pichon, M.; Morrissey, J. Benthic zonation and community structure of South Island Reef, Lizard Island (Great Barrier Reef). Bull. Mar. Sci. 1981, 31, 581–593. [Google Scholar]
- Jupiter, S.; Roff, G.; Marion, G.; Henderson, M.; Schrameyer, V.; McCulloch, M.; Hoegh-Guldberg, O. Linkages between coral assemblages and coral proxies of terrestrial exposure along a cross-shelf gradient on the southern Great Barrier Reef. Coral Reefs 2008, 27, 887–903. [Google Scholar] [CrossRef]
- Baum, G.; Januar, H.I.; Ferse, S.C.A.; Kunzmann, A. Local and regional impacts of pollution on coral reefs along the Thousand Islands North of the megacity Jakarta, Indonesia. PLoS ONE 2015, 10, e0138271. [Google Scholar] [CrossRef] [PubMed]
- Kayal, M.; Vercelloni, J.; Lison de Loma, T.; Bosserelle, P.; Chancerelle, Y.; Geoffroy, S.; Stievenart, C.; Michonneau, F.; Penin, L.; Planes, S.; et al. Predator Crown-of-Thorns Starfish (Acanthaster planci) outbreak, mass mortality of corals, and cascading effects on reef fish and benthic communities. PLoS ONE 2012, 7, e47363. [Google Scholar] [CrossRef] [PubMed]
- Darling, E.S.; Alvarez-Filip, L.; Oliver, T.A.; McClanahan, T.R.; Côté, I.M. Evaluating life-history strategies of reef corals from species traits. Ecol. Lett. 2012, 15, 1378–1386. [Google Scholar] [CrossRef]
- Kayal, M.; Vercelloni, J.; Wand, M.P.; Adjeroud, M. Searching for the best bet in life-strategy: A quantitative approach to individual performance and population dynamics in reef-building corals. Ecol. Complex. 2015, 23, 73–84. [Google Scholar] [CrossRef]
- Dollar, S.J.; Grigg, R.W. Anthropogenic and natural stresses on selected coral reefs in Hawai’i: A multidecade synthesis of impact and recovery. Pac. Sci. 2004, 58, 281–304. [Google Scholar] [CrossRef]
- Dumas, P.; Jimenez, H.; Peignon, C.; Wantiez, L.; Adjeroud, M. Small-scale habitat structure modulates the effects of no-take marine reserves for coral reef macroinvertebrates. PLoS ONE 2013, 8, e58998. [Google Scholar] [CrossRef]
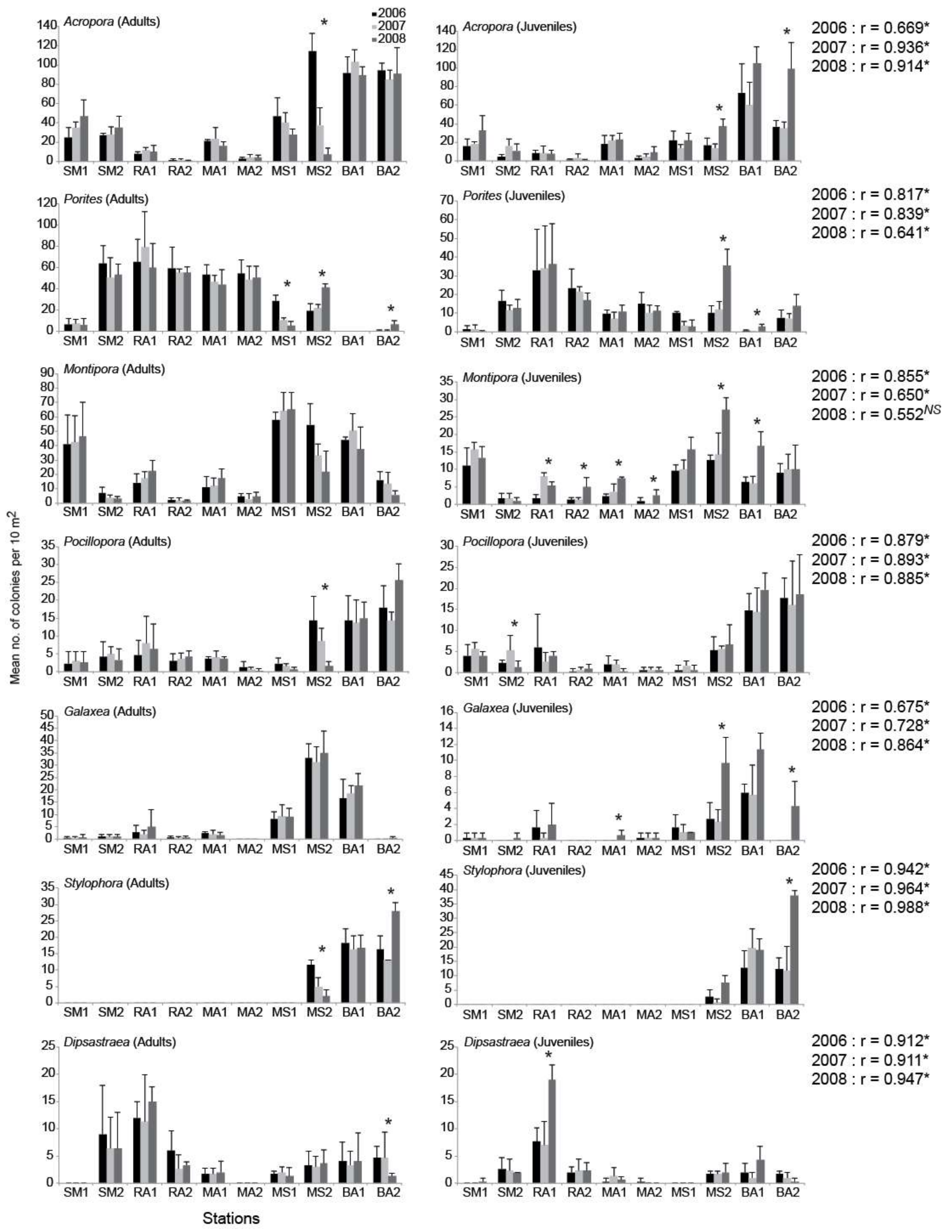
| Descriptor | 2006 | 2007 | 2008 |
|---|---|---|---|
| Percent cover | 0.043 NS | 0.099 NS | 0.048 NS |
| Generic Richness | |||
| Adults | 0.289 NS | 0.343 NS | 0.321 NS |
| Juveniles | 0.387 NS | 0.518 NS | 0.548 NS |
| Abundance | |||
| Adults | 0.701 * | 0.696 * | 0.733 * |
| Juveniles | 0.815 * | 0.725 * | 0.886 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adjeroud, M.; Poisson, E.; Peignon, C.; Penin, L.; Kayal, M. Spatial Patterns and Short-term Changes of Coral Assemblages Along a Cross-shelf Gradient in the Southwestern Lagoon of New Caledonia. Diversity 2019, 11, 21. https://doi.org/10.3390/d11020021
Adjeroud M, Poisson E, Peignon C, Penin L, Kayal M. Spatial Patterns and Short-term Changes of Coral Assemblages Along a Cross-shelf Gradient in the Southwestern Lagoon of New Caledonia. Diversity. 2019; 11(2):21. https://doi.org/10.3390/d11020021
Chicago/Turabian StyleAdjeroud, Mehdi, Emeline Poisson, Christophe Peignon, Lucie Penin, and Mohsen Kayal. 2019. "Spatial Patterns and Short-term Changes of Coral Assemblages Along a Cross-shelf Gradient in the Southwestern Lagoon of New Caledonia" Diversity 11, no. 2: 21. https://doi.org/10.3390/d11020021
APA StyleAdjeroud, M., Poisson, E., Peignon, C., Penin, L., & Kayal, M. (2019). Spatial Patterns and Short-term Changes of Coral Assemblages Along a Cross-shelf Gradient in the Southwestern Lagoon of New Caledonia. Diversity, 11(2), 21. https://doi.org/10.3390/d11020021





