Cross-shelf Heterogeneity of Coral Assemblages in Northwest Australia
Abstract
1. Introduction
2. Materials and Methods
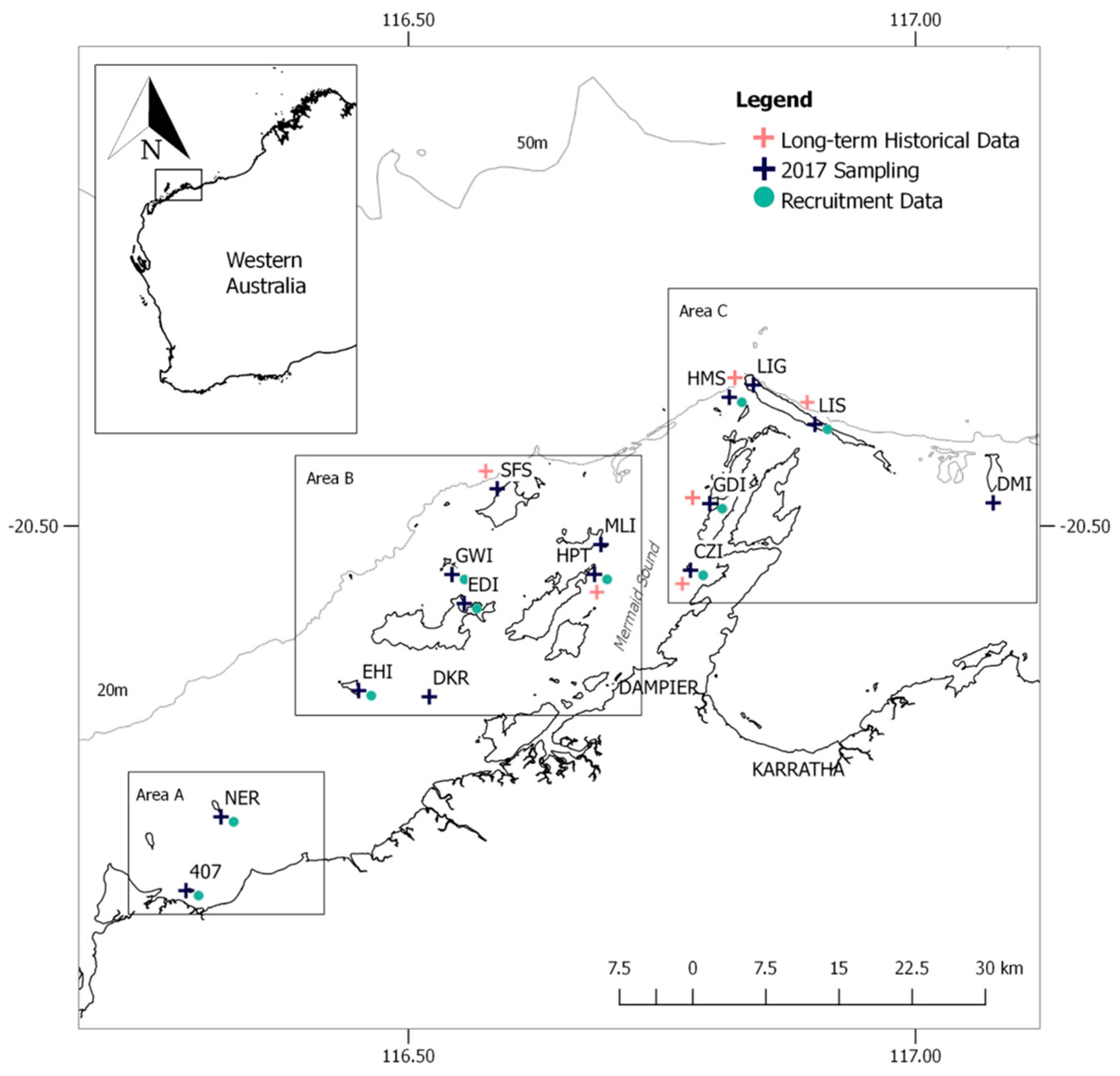
2.1. Study Location
2.2. Historical Data Collection
2.3. Benthic Data Collection
2.4. Coral Recruitment Data Collection
2.5. Environmental Variables
2.6. Data Analysis
3. Results
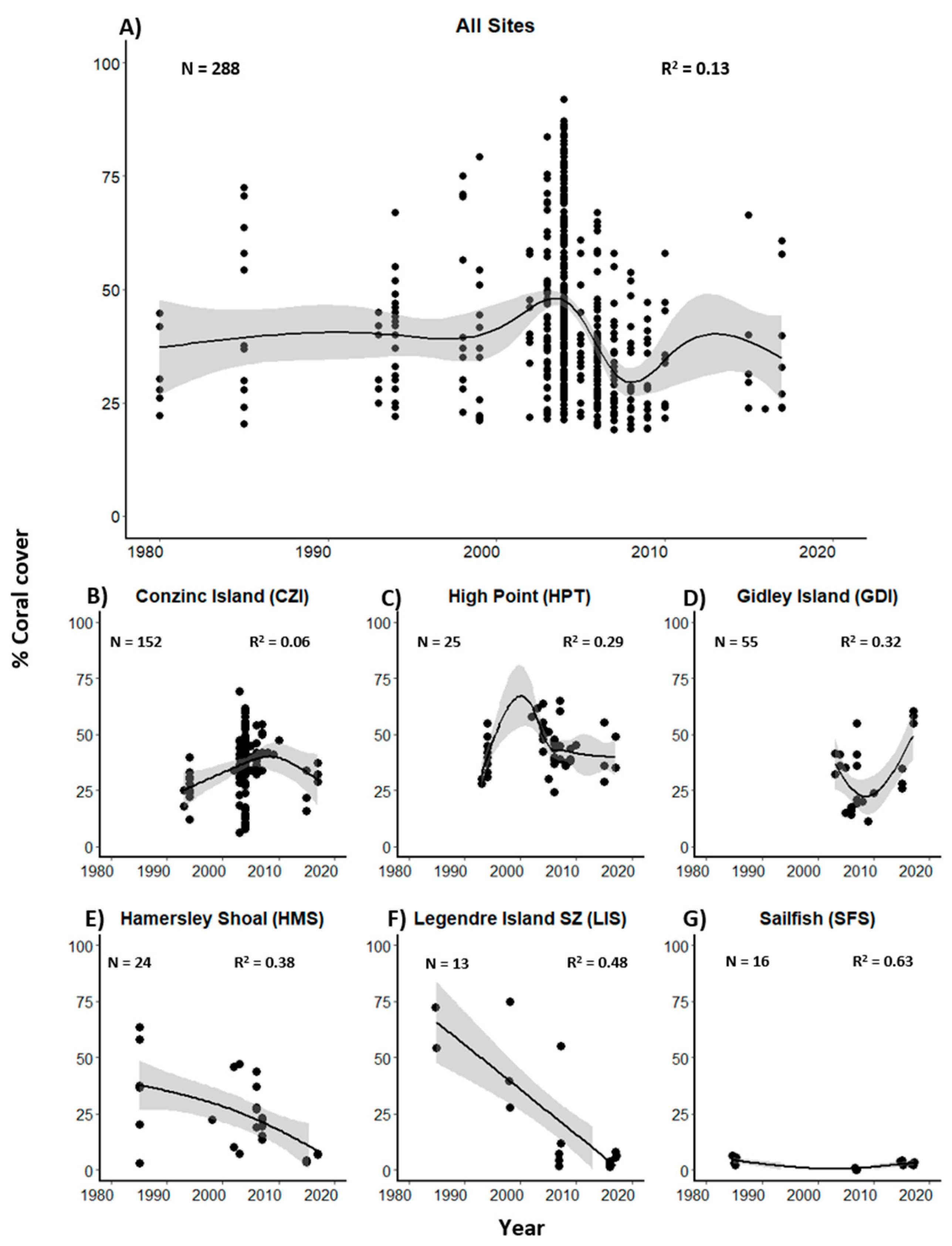
3.1. Historical Coral Cover and Composition in the Dampier Archipelago
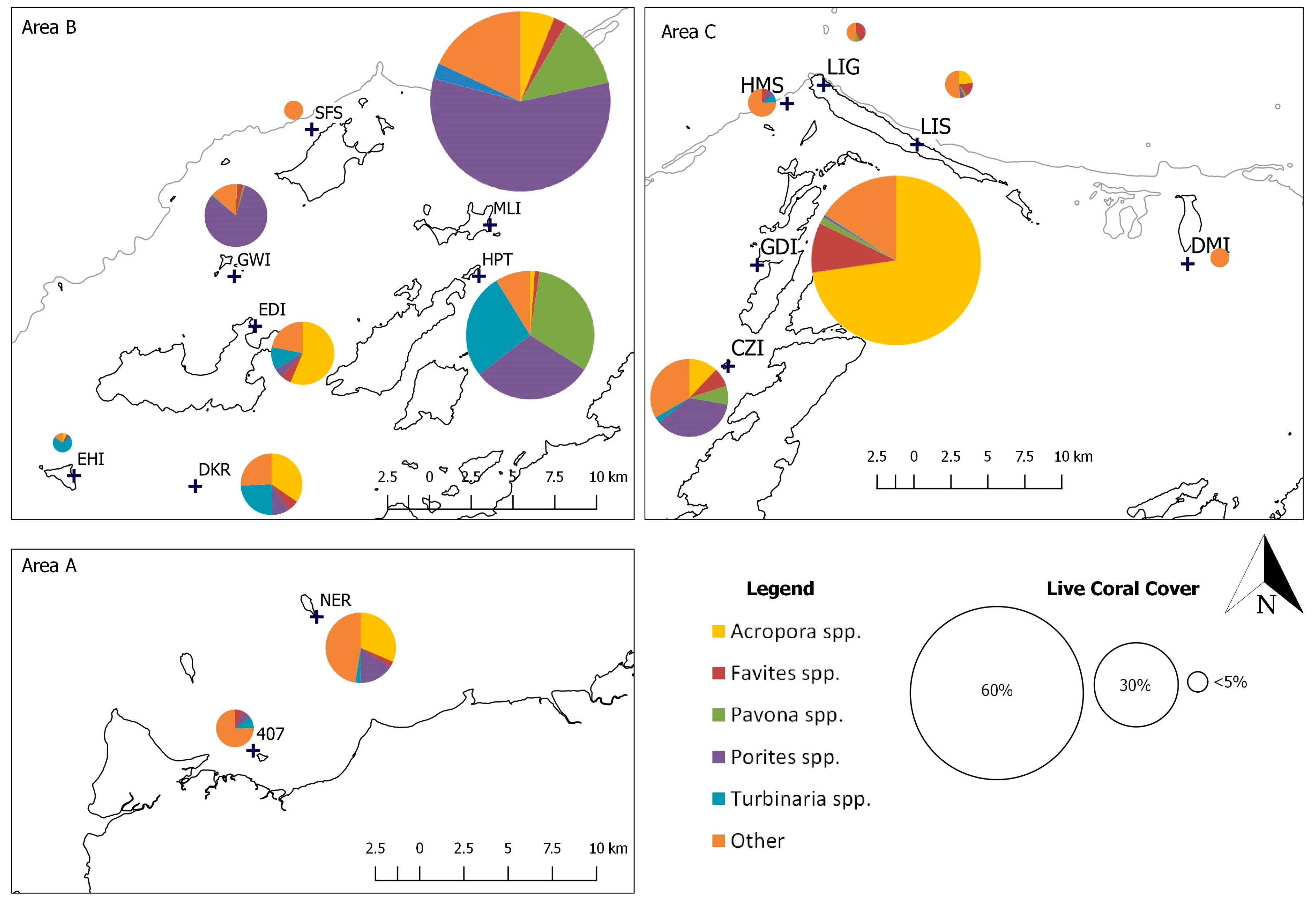
3.2. Present Day Coral Cover and Assemblage Composition in the Dampier Archipelago
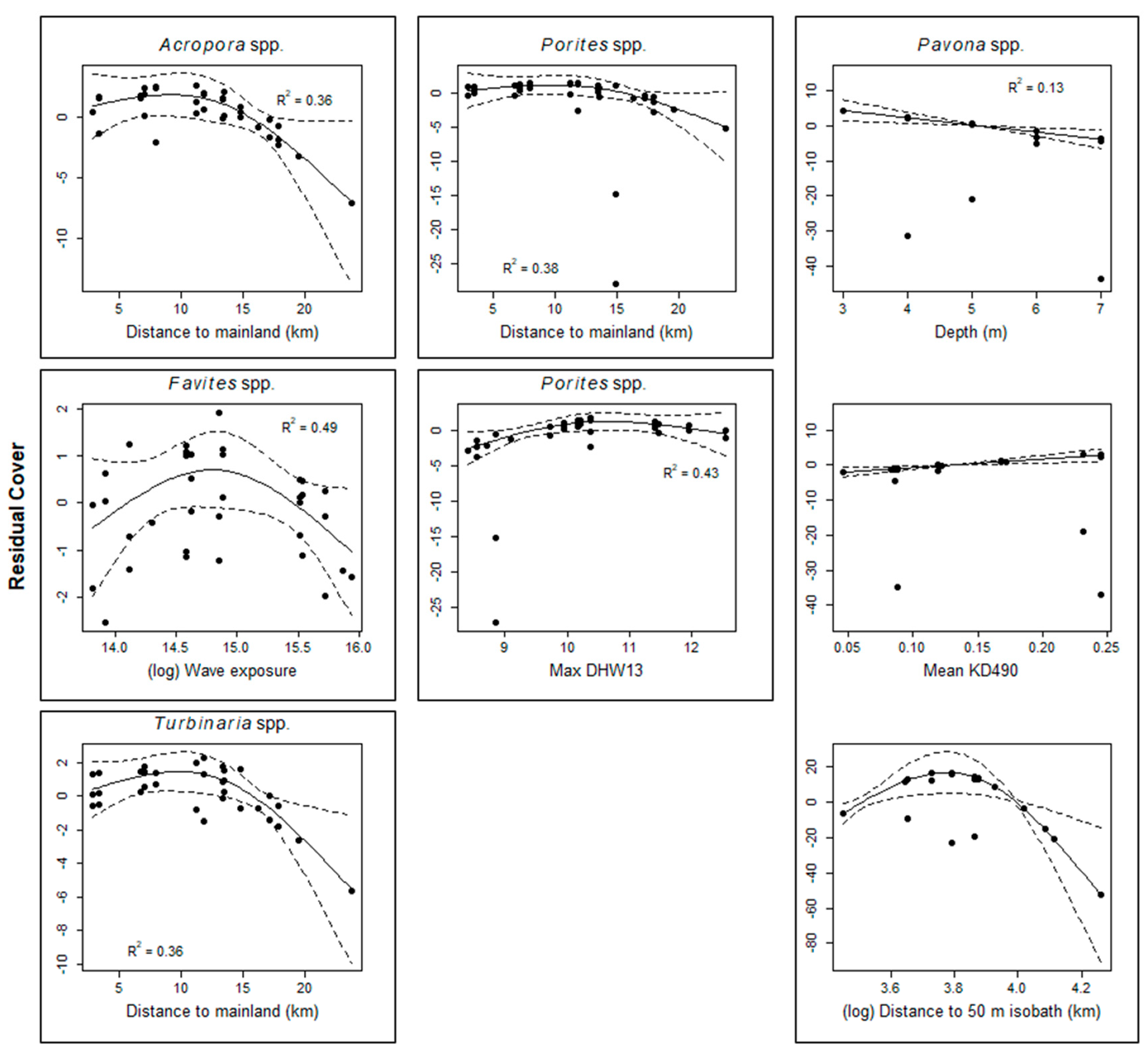
3.3. Predictors of Total and Genera Cover 2017 Data
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hatcher, B.G. Coral reef primary productivity: A beggar’s banquet. Trends Ecol. Evol. 1988, 3, 106–111. [Google Scholar] [CrossRef]
- Hoegh-Guldberg, O. Climate change, coral bleaching and the future of the world’s coral reefs. Mar. Freshw. Res. 1999, 50, 839–866. [Google Scholar] [CrossRef]
- Darling, E.S.; McClanahan, T.R.; Côté, I.M. Life histories predict coral community disassembly under multiple stressors. Glob. Chang. Biol. 2013, 19, 1930–1940. [Google Scholar] [CrossRef] [PubMed]
- Chabanet, P.; Ralambondrainy, H.; Amanieu, M.; Faure, G.; Galzin, R. Relationships between coral reef substrata and fish. Coral Reefs 1997, 16, 93–102. [Google Scholar] [CrossRef]
- Wilson, S.K.; Depczynski, M.; Fisher, R.; Holmes, T.H.; O’Leary, R.A.; Tinkler, P. Habitat associations of juvenile fish at Ningaloo Reef, Western Australia: The importance of coral and algae. PLoS ONE 2010, 5, e15185. [Google Scholar] [CrossRef] [PubMed]
- Kerry, J.; Bellwood, D. The effect of coral morphology on shelter selection by coral reef fishes. Coral Reefs 2012, 31, 415–424. [Google Scholar] [CrossRef]
- Coker, D.J.; Wilson, S.K.; Pratchett, M.S. Importance of live coral habitat for reef fishes. Rev. Fish Biol. Fish. 2014, 24, 89–126. [Google Scholar] [CrossRef]
- Graham, N.A.J.; Cinner, J.E.; Norström, A.V.; Nyström, M. Coral reefs as novel ecosystems: Embracing new futures. Curr. Opin. Environ. Sustain. 2014, 7, 9–14. [Google Scholar] [CrossRef]
- Roberts, T.E.; Moloney, J.M.; Sweatman, H.P.A.; Bridge, T.C.L. Benthic community composition on submerged reefs in the central Great Barrier Reef. Coral Reefs 2015, 34, 569–580. [Google Scholar] [CrossRef]
- Fabricius, K.E.; De’ath, G.; McCook, L.; Turak, E.; Williams, D.M. Changes in algal, coral and fish assemblages along water quality gradients on the inshore Great Barrier Reef. Mar. Pollut. Bull. 2005, 51, 384–398. [Google Scholar] [CrossRef]
- Osborne, K.; Dolman, A.M.; Burgess, S.C.; Johns, K.A. Disturbance and the Dynamics of Coral Cover on the Great Barrier Reef (1995–2009). PLoS ONE 2011, 6, e17516. [Google Scholar] [CrossRef] [PubMed]
- Richards, Z.T.; Hobbs, J.P.A. Predicting coral species richness: The effect of input variables, diversity and scale. PLoS ONE 2014, 9, e83965. [Google Scholar] [CrossRef] [PubMed]
- Gil, M.; Goldenberg, S.; Ly Thai Bach, A.; Mills, S.; Claudet, J. Interactive effects of three pervasive marine stressors in a post-disturbance coral reef. Coral Reefs 2016, 35, 1281–1293. [Google Scholar] [CrossRef]
- Zinke, J.; Gilmour, J.P.; Fisher, R.; Puotinen, M.; Maina, J.; Darling, E.; Stat, M.; Richards, Z.T.; McLanahan, T.R.; Beger, M.; et al. Gradients of disturbance and environmental conditions shape coral community structure for south-eastern Indian Ocean reefs. Divers. Distrib. 2018, 24, 605–620. [Google Scholar] [CrossRef]
- Nash, K.L.; Graham, N.A.; Wilson, S.K.; Bellwood, D.R. Cross-scale habitat structure drives fish body size distributions on coral reefs. Ecosystems 2013, 16, 478–490. [Google Scholar] [CrossRef]
- Graham, N.A.J.; Jennings, S.; Macneil, M.A.; Mouillot, D.; Wilson, S.K. Predicting climate-driven regime shifts versus rebound potential in coral reefs. Nature 2015, 518, 94–97. [Google Scholar] [CrossRef] [PubMed]
- Nash, K.L.; Graham, N.A.J.; Jennings, S.; Wilson, S.K.; Bellwood, D.R. Herbivore cross-scale redundancy supports response diversity and promotes coral reef resilience. J. Appl. Ecol. 2016, 53, 646–655. [Google Scholar] [CrossRef]
- Richardson, L.E.; Graham, N.A.J.; Hoey, A.S. Cross-scale habitat structure driven by coral species composition on tropical reefs. Sci. Rep. 2017, 7, 7557. [Google Scholar] [CrossRef]
- Hughes, T.P.; Baird, A.H.; Bellwood, D.R.; Card, M.; Connolly, S.R.; Folke, C.; Grosberg, R.; Hoegh-Guldberg, O.; Jackson, J.B.C.; Kleypas, J.; et al. Climate Change, Human Impacts, and the Resilience of Coral Reefs. Science 2003, 301, 929–933. [Google Scholar] [CrossRef]
- Dornelas, M.; Gotelli, N.J.; McGill, B.; Shimadzu, H.; Moyes, F.; Sievers, C.; Magurran, A.E. Assemblage Time Series Reveal Biodiversity Change but Not Systematic Loss. Science 2014, 344, 296–299. [Google Scholar] [CrossRef]
- Bento, R.; Hoey, A.S.; Bauman, A.G.; Feary, D.A.; Burt, J.A. The implications of recurrent disturbances within the world’s hottest coral reef. Mar. Pollut. Bull. 2015, 105, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Pratchett, M.S.; Hoey, A.S.; Wilson, S.K. Reef degradation and the loss of critical ecosystem goods and services provided by coral reef fishes. Curr. Opin. Environ. Sustain. 2014, 7, 37–43. [Google Scholar] [CrossRef]
- Roff, G.; Zhao, J.; Mumby, P.J. Decadal-scale rates of reef erosion following El Niño related mass coral mortality. Glob. Chang. Biol. 2015, 21, 4415–4424. [Google Scholar] [CrossRef] [PubMed]
- Richardson, L.E.; Graham, N.A.J.; Pratchett, M.S.; Hoey, A.S. Structural complexity mediates functional structure of reef fish assemblages among coral habitats. Environ. Biol. Fishes 2017, 100, 193–207. [Google Scholar] [CrossRef]
- Australian and New Zealand Environment and Conservation Council: Task Force on Marine Protected Areas. Strategic Plan of Action for the National Representative System of Marine Protected Areas: A Guide for Action by Australian Government; Environment Australia: Canberra, Australia, 1999.
- Cleary, D.F.R.; Polónia, A.R.M.; Renema, W.; Hoeksema, B.W.; Rachello-Dolmen, P.G.; Moolenbeek, R.G.; Budiyanto, A.; Yahmantoro; Tuti, Y.; Giyanto; et al. Variation in the composition of corals, fishes, sponges, echinoderms, ascidians, molluscs, foraminifera and macroalgae across a pronounced in-to-offshore environmental gradient in the Jakarta Bay-Thousand Islands coral reef complex. Mar. Pollut. Bull. 2016, 110, 701–717. [Google Scholar] [CrossRef]
- Done, T.J. Patterns in the distribution of coral communities across the central Great Barrier Reef. Coral Reefs 1982, 1, 95–107. [Google Scholar] [CrossRef]
- Blakeway, D.R. Patterns of mortality from natural and anthropogenic influences in Dampier corals: 2004 cyclone and dredging impacts. In Corals of the Dampier Harbour: Their Survival and Reproduction during the Dredging Programs of 2004; Stoddart, J.A., Stoddart, S.E., Eds.; MScience Pty Ltd.: Perth, Western Australia, 2005; pp. 65–76. [Google Scholar]
- Golbuu, Y.; van Woesik, R.; Richmond, R.H.; Harrison, P.; Fabricius, K.E. River discharge reduces coral reef diversity in Palau. Mar. Pollut. Bull. 2011, 62, 824–831. [Google Scholar] [CrossRef] [PubMed]
- Jupiter, S.; Roff, G.; Marion, G.; Henderson, M.; Schrameyer, V.; McCulloch, M.; Hoegh-Guldberg, O. Linkages between coral assemblages and coral proxies of terrestrial exposure along a cross-shelf gradient on the southern Great Barrier Reef. Coral Reefs 2008, 27, 887–903. [Google Scholar] [CrossRef]
- Thompson, A.A.; Dolman, A.M. Coral bleaching: One disturbance too many for near-shore reefs of the Great Barrier Reef. Coral Reefs 2010, 29, 637–648. [Google Scholar] [CrossRef]
- Baumann, J.H.; Townsend, J.E.; Courtney, T.A.; Aichelman, H.E.; Davies, S.W.; Lima, F.P.; Castillo, K.D. Temperature Regimes Impact Coral Assemblages along Environmental Gradients on Lagoonal Reefs in Belize. PLoS ONE 2016, 11, e0162098. [Google Scholar] [CrossRef]
- Ellis, J.; Anlauf, H.; Kürten, S.; Lozano-Cortés, D.; Alsaffar, Z.; Cúrdia, J.; Jones, B.; Carvalho, S. Cross shelf benthic biodiversity patterns in the Southern Red Sea. Sci. Rep. 2017, 7, 437. [Google Scholar] [CrossRef] [PubMed]
- van Woesik, R.; Tomascik, T.; Blake, S. Coral assemblages and physico-chemical characteristics of the Whitsunday Islands: Evidence of recent community changes. Mar. Fresh. Res. 1999, 50, 427–440. [Google Scholar] [CrossRef]
- Smith, T.B.; Nemeth, R.S.; Blondeau, J.; Calnan, J.M.; Kadison, E.; Herzlieb, S. Assessing coral reef health across onshore to offshore stress gradients in the US Virgin Islands. Mar. Pollut. Bull. 2008, 56, 1983–1991. [Google Scholar] [CrossRef] [PubMed]
- Aderjoud, M. Factors influencing spatial patterns on coral reefs around Moorea, French Polynesia. Mar. Ecol. Prog. Ser. 1997, 159, 105–119. [Google Scholar]
- Lirman, D.; Fong, P. Is proximity to land-based sources of coral stressors an appropriate measure of risk to coral reefs? An example from the Florida Reef Trace. Mar. Pollut. Bull. 2007, 54, 779–791. [Google Scholar] [CrossRef] [PubMed]
- Rippe, J.P.; Baumann, J.H.; De Leener, D.N.; Aichelman, H.E.; Friedlander, E.B.; Davies, S.W.; Castillo, K.D. Corals sustain growth but not skeletal density across the Florida Keys Reef Tract despite ongoing warming. Glob. Chang. Biol. 2018, 24, 5205–5217. [Google Scholar] [CrossRef] [PubMed]
- Mills, M.M.; Sebens, K.P. Ingestion and assimilation of nitrogen from benthic sediments by three species of coral. Mar. Biol. 2004, 145, 1097–1106. [Google Scholar] [CrossRef]
- Perry, C.T.; Larcombe, P. marginal and non-reef-building coral environments. Coral Reefs 2003, 22, 427–432. [Google Scholar] [CrossRef]
- Simpson, C.J. Ecology of Scleractinian Corals in the Dampier Archipelago; Technical Series No. 23; Environmental Protection Authority: Perth, Western Australia, 1988. [Google Scholar]
- Marine Parks and Reserves Selection Working Group. A representative marine reserve system for Western Australia: Report of the Marine Parks and Reserves Selection Working Group; Department of Conservation and Land Management: Perth, Western Australia, 1994.
- Griffith, J.K. Scleractinian corals collected during 1998 from the Dampier Archipelago, Western Australia. In Report on the Results of the Western Australian Museum/Woodside Energy Ltd. Partnership to Explore the Marine Biodiversity of the Dampier Archipelago Western Australia 1998–2000; Jones, D.S., Ed.; Records of the Western Australian Museum, Supplement 66; Western Australian Museum: Perth, Western Australia, 2004; pp. 101–120. [Google Scholar]
- Roberts, C.M.; McClean, C.J.; Veron, J.E.N.; Hawkins, J.P.; Allen, G.R.; McAllister, D.E.; Mittermeier, C.G.; Schueler, F.W.; Spalding, M.; Wells, F.; et al. Marine Biodiversity Hotspots and Conservation Priorities for Tropical Reefs. Science 2002, 295, 1280–1284. [Google Scholar] [CrossRef]
- Blakeway, D.; Byers, M.; Stoddart, J.; Rossendell, J. Coral Colonisation of an Artificial Reef in a Turbid Nearshore Environment, Dampier Harbour, Western Australia. PLoS ONE 2013, 8, e75281. [Google Scholar] [CrossRef]
- Moustaka, M.; Langlois, T.J.; McLean, D.; Bond, T.; Fisher, R.; Fearns, P.; Dorji, P.; Evans, R.D. The effects of suspended sediment on coral reef fish assemblages and feeding guilds of north-west Australia. Coral Reefs 2018, 37, 659–673. [Google Scholar] [CrossRef]
- Moore, J.A.Y.; Bellchambers, L.M.; Depczynski, M.R.; Evans, R.D.; Evans, S.N.; Field, S.N.; Friedman, K.J.; Gilmour, J.P.; Holmes, T.H.; Middlebrook, R.; et al. Unprecedented Mass Bleaching and Loss of Coral across 12° of Latitude in Western Australia in 2010–11. PLoS ONE 2012, 7, e51807. [Google Scholar] [CrossRef] [PubMed]
- Dampier Port Authority. Dredging & Spoil Management Plan—Dampie; Dampier Port Authority: Dampier, Western Australia; Available online: https://www.pilbaraports.com.au/PilbaraPortsAuthority/media/Documents/DAMPIER/Environment%20and%20Heritage/Dredging-and-Spoil-Management-Plan-DAMPIER.pdf (accessed on 10 March 2018).
- Ryan, K.L.; Hall, N.G.; Lai, E.K.; Smallwood, C.B.; Taylor, S.M.; Wise, B.S. Statewide survey of boat-based recreational fishing in Western Australia 2015/16. In Fisheries Research Report No. 287; Department of Primary Industries and Regional Development: Perth, Western Australia, 2017. [Google Scholar]
- Wilson, S.; Kendrick, A.; Wilson, B. The North-Western Margin of Australia. In World Seas: An Environmental Evaluation, 2nd ed.; Sheppard, C., Ed.; Academic Press: London, UK, 2019; pp. 303–331. [Google Scholar]
- Stoddart, J.A.; Blakeway, D.R.; Grey, K.A.; Stoddart, S.E. Rapid high-precision monitoring of coral communities to support reactive management of dredging in Mermaid Sound, Dampier, Western Australia. In Corals of the Dampier Harbour: Their survival and Reproduction during the Dredging Programs of 2004; Stoddart, J.A., Stoddart, S.E., Eds.; MScience Pty Ltd.: Perth, Western Australia, 2005; pp. 35–51. [Google Scholar]
- Commonwealth of Australia. A Guide to the Integrated Marine and Coastal Regionalisation of Australia Version 4.0; Department of the Environment and Heritage: Canberra, Australia, 2006. [Google Scholar]
- Pearce, A.F.; Buchan, S.; Chiffings, T.; D’Adamo, N.; Fandry, C.; Fearns, P.R.C.S.; Mills, D.J.; Phillips, R.C.; Simpson, C. A review of the oceanography of the Dampier Archipelago. In The Marine Flora and Fauna of Dampier; Wells, F.E., Walker, D.I., Jones, D.S., Eds.; Western Australian Museum: Perth, Western Australia, 2003; Volume 1, pp. 13–50. [Google Scholar]
- Environmental Protection Authority. Bulletin 1259: Pluto LNG Development, Burrup Peninsula, Woodside Energy Ltd.; Environmental Protection Authority: Perth, Western Australia, 2007. [Google Scholar]
- Pilbara Ports Authority, Port of Dampier. Available online: https://www.pilbaraports.com.au/Port-of-Dampier (accessed on 19 November 2018).
- Mohring, M.B.; Nutt, C.D.; Bancroft, K.P.; Friedman, K.J.; Severin, C.R.; Douglas, R.A.; Fetahovic, E. Report for Pluto Offset ‘D’ Project ‘i’—‘Learn from the Past to Manage the Future’; Department of Parks and Wildlife: Perth, Western Australia, 2015. [Google Scholar]
- QGIS Development Team. QGIS Geographic Information System (Version 2.18.16). Open Source Geospatial Foundation Project. Available online: http://qgis.osgeo.org (accessed on 15 June 2018).
- Hastie, T. Gam: Generalized Additive Models, R Package Version 1.16; Available online: https://CRAN.R-project.org/package=gam (accessed on 1 March 2018).
- R Core Team. R: A Language and Environment for Statistical Computing. 2018. Available online: https://www.r-project.org (accessed on 1 March 2018).
- Akaike, H. Information theory and an extension of the maximum likelihood principle. In Proceedings of the Second International Symposium on Information Theory, Tsahkadsor, Armenia, 2–8 September 1971; pp. 267–281. [Google Scholar]
- Shedrawi, G.; Falter, J.L.; Friedman, K.J.; Lowe, R.J.; Pratchett, M.S.; Simpson, C.J.; Speed, C.W.; Wilson, S.K.; Zhang, Z. Localised hydrodynamics influence vulnerability of coral communities to environmental disturbances. Coral Reefs 2017, 36, 861–872. [Google Scholar] [CrossRef]
- EcoPAAS. EcoPAAS Desktop (Version 2.0.10.0); Ocean Vision Environmental Research, 2013. Available online: https://oceanvision.com.au/2014/07/introducing-ecopaas/ (accessed on 1 June 2017).
- Jonker, M.; Johns, K.; Osborne, K. Surveys of Benthic reef Communities Using Underwater Digital Photography and Counts of Juvenile Corals—Long-Term Monitoring of the Great Barrier Reef—Standard Operation Procedure Number 10; Australian Institute of Marine Science: Townsville, Australia, 2008. [Google Scholar]
- Thomson, D.T.; Babcock, R.; Rule, M.; Feng, M.; Trapon, M.; Evans, R.D.; Wilson, S.K. Coral recruitment patterns in the Dampier Archipelago. Status (unpublished work; manuscript in preparation).
- Mundy, C.; Babcock, R. Are vertical distribution patterns of scleractinian corals maintained by pre-or post-settlement processes? A case study of three contrasting species. Mar. Ecol. Prog. Ser. 2000, 198, 109–119. [Google Scholar] [CrossRef]
- Mundy, C.N. An appraisal of methods used in coral recruitment studies. Coral Reefs 2000, 19, 124–131. [Google Scholar] [CrossRef]
- Google Earth. Google Earth V7.1.8.3036. Available online: https://www.google.com/earth/ (accessed on 17 March 2017).
- Pante, E.; Simon-Bouhet, B. marmap: A Package for Importing, Plotting and Analyzing Bathymetric and Topographic Data in R. PLoS ONE 2013, 8, 9. [Google Scholar] [CrossRef] [PubMed]
- Burrows, M.T.; Harvey, R.; Robb, L. Wave exposure indicies from digital coastlines and the prediction of rocky shore community structure. Mar. Ecol. Prog. Ser. 2008, 1–12. [Google Scholar] [CrossRef]
- Hill, N.; Pepper, A.; Puotinen, M.; Hughes, M.; Edgar, G.; Barrett, N.; Stuart-Smith, R.; Leaper, R. Quantifying wave exposure in shallow temperate reef systems: Applicability of fetch models for predicting algal biodiversity. Mar. Ecol. Prog. Ser. 2010, 417, 83–95. [Google Scholar] [CrossRef]
- Australian Government Bureau of Meteorology Climate Data Online. Available online: http://www.bom.gov.au/climate/data/ (accessed on 1 May 2018).
- Stuart-Smith, R.D.; Barrett, N.S.; Crawford, C.M.; Frusher, S.D.; Stevenson, D.G.; Edgar, G.J. Spatial patterns in impacts of fishing on temperate rocky reefs: Are fish abundance and mean size related to proximity to fisher access points? J. Exp. Mar. Biol. Ecol. 2008, 365, 116–125. [Google Scholar] [CrossRef]
- Wenger, A.S.; Williamson, D.H.; da Silva, E.T.; Ceccarelli, D.M.; Browne, N.K.; Petus, C.; Devlin, M.J. Effects of reduced water quality on coral reefs in and out of no-take marine reserves. Cons. Biol. 2015, 30, 142–153. [Google Scholar] [CrossRef]
- McLean, D.L.; Langlois, T.J.; Newman, S.J.; Holmes, T.H.; Birt, M.J.; Bornt, K.R.; Bond, T.; Collins, D.L.; Evans, S.N.; Travers, M.J.; et al. Distribution, abundance, diversity and habitat associations of fishes across a bioregion experiencing rapid coastal development. Estuar. Coast. Shelf Sci. 2016, 178, 36–47. [Google Scholar] [CrossRef]
- Sheppard, C.R.C. Coral Populations on Reef Slopes and their Major Controls. Mar. Ecol. Prog. Ser. 1982, 7, 83–115. [Google Scholar] [CrossRef]
- Chollett, I.; Mumby, P.J. Predicting the distribution of Montastrea reefs using wave exposure. Coral Reefs 2012, 31, 493–503. [Google Scholar] [CrossRef]
- Jones, R.; Bessell-Browne, P.; Fisher, R.; Klonowski, W.; Slivkoff, M. Assessing the impacts of sediments from dredging on corals. Mar. Pollut. Bull. 2016, 2012, 9–29. [Google Scholar] [CrossRef] [PubMed]
- Bessell-Browne, P.; Negri, A.P.; Fisher, R.; Clode, P.L.; Duckworth, A.; Jones, R. Impacts of turbidity on corals: The relative importance of light limitation and suspended sediments. Mar. Pollut. Bull. 2017, 117, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Muir, P.R.; Marshall, P.A.; Abdulla, A.; Aguirre, J.D. Species identity and depth predict bleaching severity in reef-building corals: Shall the deep inherit the reef? Proc. Biol. Sci. 2017, 284. [Google Scholar] [CrossRef]
- Williams, G.J.; Sandin, S.A.; Zgliczynski, B.J.; Fox, M.D.; Gove, J.M.; Rogers, J.S.; Furby, K.A.; Hartmann, A.C.; Caldwell, Z.R.; Price, N.N.; et al. Biophysical drivers of coral trophic depth zonation. Mar. Biol. 2018, 165. [Google Scholar] [CrossRef]
- Hajime, K. Validation of degree heating weeks as a coral bleaching index in the northwestern Pacific. Coral Reefs 2017, 36, 63–70. [Google Scholar] [CrossRef]
- Ridgeway, T.; Inostroza, K.; Synnot, L.; Trapon, M.; Twomey, L.; Westera, M. Temporal patterns of coral cover in the offshore Pilbara, Western Australia. Mar. Biol. 2016, 163. [Google Scholar] [CrossRef]
- Hughes, T.P.; Baird, A.H.; Dinsdale, E.A.; Moltschaniwskyj, N.A.; Pratchett, M.S.; Tanner, J.E.; Willis, B.L. Supply-Side Ecology Works Both Ways: The link between Benthic Adults, Fecundity, and Larval Recruits. Ecology 2000, 81, 2241–2249. [Google Scholar] [CrossRef]
- Babcock, R.; Gilmour, J.; Thomson, D. Measurement and modeling of key demographic processes in corals of the Dampier Archipelago. In WAMSI Dredging Science Node, Theme 4 Report; Western Australia Marine Science Institute: Perth, Western Australia, 2017. [Google Scholar]
- Kleiber, C.; Zeileis, A. Applied Econometrics with R; Springer: New York, NY, USA, 2008. [Google Scholar]
- Hothorn, T.; Bretz, F.; Westfall, P. Simultaneous Inference in General Parametric Models. Biom. J. 2008, 50, 346–363. [Google Scholar] [CrossRef] [PubMed]
- Martinez Arbizu, P. pairwiseAdonis: Pairwise Multilevel Comparison Using Adonis, R Package Version 0.0.1; 2017. Available online: https://github.com/pmartinezarbizu/pairwiseAdonis (accessed on 1 March 2018).
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlinn, D.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; et al. Vegan: Community Ecology Package, R Package Version 2.4-6; 2018. Available online: https://CRAN.R-project.org/package=vegan (accessed on 1 March 2018).
- Wood, S.; Scheipl, F. Package ‘gamm4’ (Version 0.2-5). Generalized Additive Mixed Models Using mgcv and lme4. 2015. Available online: https://cran.r-project.org/web/packages/ gamm4/gamm4.pdf (accessed on 1 April 2018).
- Harrison, X.A. Using observation-level random effects to model overdispersion in count data in ecology and evolution. PeerJ 2014, 2, e616. [Google Scholar] [CrossRef] [PubMed]
- Fisher, R.; Wilson, S.K.; Sin, T.M.; Lee, A.C.; Langlois, T.J. A simple function for full-subsets multiple regression in ecology with R. Ecol. Evol. 2018, 8, 6104–6113. [Google Scholar] [CrossRef]
- Graham, M.H. Confronting multicollinearity in ecological multiple regression. Ecology 2003, 84, 2809–2815. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach, 2nd ed.; Springer: New York, NY, USA, 2002. [Google Scholar]
- Wood, S. Fast stable restricted maximum likelihood and marginal likelihood estimation of semiparametric generalized linear models. J. R. Stat. Soc. Ser. B Stat. Methodol. 2011, 73, 3–36. [Google Scholar] [CrossRef]
- Perry, C.T.; Smithers, S.G.; Gulliver, P.; Browne, N. Evidence of very rapid reef accretion and reef growth under high turbidity and terrigenous sedimentation. Geology 2012, 40, 719–722. [Google Scholar] [CrossRef]
- Cacciapaglia, C.; van Woesik, R. Climate-change refugia: Shading reef corals by turbidity. Glob. Chang. Biol. 2015, 22, 1145–1154. [Google Scholar] [CrossRef]
- Morgan, K.M.; Perry, C.T.; Johnson, J.A.; Smithers, S.G. Nearshore Turbid-Zone Corals Exhibit High Bleaching Tolerance on the Great Barrier Reef Following the 2016 Ocean Warming Event. Front. Mar. Sci. 2017, 4. [Google Scholar] [CrossRef]
- Marsh, L.M. Report on the Marine Fauna and Flora of the Dampier Archipelago; Western Australian Museum: Perth, Western Australia, 1978.
- Gilmour, J.P.; Cook, K.L.; Ryan, N.M.; Puotinen, M.L.; Green, R.H.; Shedrawi, G.; Hobbs, J.A.; Thomson, D.P.; Babcock, R.C.; Buckee, J.; et al. The state of Western Australia’s coral reefs. Coral Reefs, in review.
- Green, D.H.; Edmunds, P.J.; Pochon, X.; Gates, R.D. The effects of substratum type on the growth, mortality, and photophysiologu of juvenile corals in St. John, US Virgin Islands. J. Exp. Mar. Biol. Ecol. 2010, 384, 18–29. [Google Scholar] [CrossRef]
- Boschetti, F.; Babcock, R.C.; Doropoulos, C.; Thomson, D.P.; Feng, M.; Slawinski, D.; Berry, O.; Vanderklift, M.A. Setting priorities for conservation at the interface between ocean circulation, connectivity, and population dynamics. Ecol. Appl. in press.
- Hoey, A.; Howells, E.; Johansen, J.; Hobbs, J.-P.; Messmer, V.; McCowan, D.; Wilson, S.K.; Pratchett, M. Recent Advances in Understanding the Effects of Climate Change on Coral Reefs. Diversity 2016, 8, 12. [Google Scholar] [CrossRef]
- Blakeway, D.R.; Radford, B.T.M. Scleractinian corals of the Dampier Port and inner Mermaid Sound: Species list, community composition and distributional data. In Corals of the Dampier Harbour: Their Survival and Reproduction During the Dredging Programs of 2004; Stoddart, J.A., Stoddart, S.E., Eds.; Mscience Pty. Ltd.: Perth, Western Australia, 2004; pp. 1–11. [Google Scholar]
- Sofonia, J.J.; Anthony, K.R.N. High-sediment tolerance in the reef coral Turbinaria mesenterina from the inner Great Barrier Reef lagoon (Australia). Estuar. Coast. Shelf. Sci. 2008, 78, 748–752. [Google Scholar] [CrossRef]
- Lafratta, A.; Fromont, J.; Speare, P.; Schönberg, C.H.L. Coral bleaching in turbid water of north-western Australia. Mar. Freshw. Res. 2016, 68, 65–75. [Google Scholar] [CrossRef]
- Hughes, T.P.; Kerry, J.T.; Álvarez-Noriega, M.; Álvarez-Romero, J.G.; Anderson, K.D.; Baird, A.; Babcock, R.C.; Beger, M.; Bellwood, D.R.; Berkelmans, R.; et al. Global warming and recurrent mass bleaching of corals. Nature 2017, 543, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Feng, M.; Colberg, F.; Slawinski, D.; Berry, O.; Babcock, R. Ocean circulation drives heterogeneous recruitments and connectivity among coral populations on the North West Shelf of Australia. J. Mar. Syst. 2016, 164, 1–12. [Google Scholar] [CrossRef]
- Gilmour, J.; Speed, C.W.; Babcock, R. Coral reproduction in Western Australia. PeerJ 2016, 4. [Google Scholar] [CrossRef]
- Evans, R.D.; Ryan, N.M.; Travers, M.J.; Feng, M.; Hitchen, Y.; Kennington, W.J. A seascape genetic analysis of a stress-tolerant coral species along the Western Australian coast. Coral Reefs 2018. [Google Scholar] [CrossRef]
- Adam, T.C.; Schmitt, R.J.; Holbrook, S.J.; Brooks, A.J.; Edmunds, P.J.; Carpenter, R.C.; Bernardi, G. Herbivory, Connectivity, and Ecosystem Resilience: Response of a Coral Reef to a Large-Scale Perturbation. PLoS ONE 2011, 6, e23717. [Google Scholar] [CrossRef]
- Fabricius, K. Effects of terrestrial runoff on the ecology of corals and coral reefs: Review and synthesis. Mar. Pollut. Bull. 2005, 50, 125–146. [Google Scholar] [CrossRef]
- Graham, N.A.J.; Wilson, S.K.; Carr, P.; Hoey, A.S.; Jennings, S.; Macneil, M.A. Seabirds enhance coral reef productivity and functioning in the absence of invasive rats. Nature 2018, 559, 250–253. [Google Scholar] [CrossRef] [PubMed]
- Game, E.T.; McDonald-Madden, E.; Puotinen, M.L.; Possingham, H.P. Should we protect the strong or the weak? Risk, resilience, and the selection of marine protected areas. Cons. Biol. 2008, 22, 1619–1629. [Google Scholar] [CrossRef] [PubMed]
- Cvitanovic, C.; Fulton, C.J.; Wilson, S.K.; van Kerkhoff, L.; Cripps, I.L.; Muthiga, N. Utility of primary scientific literature to environmental managers: An international case study on coral-dominated marine protected areas. Ocean. Coast. Manag. 2014, 102, 72–78. [Google Scholar] [CrossRef]
- Blackhall, K. Finding studies for inclusion in systematic reviews of interventions for injury prevention- the importance of grey and unpublished literature. Injury Prevention 2007, 13, 359. [Google Scholar] [CrossRef] [PubMed]
- Pappas, C.; Williams, I. Grey Literature: Its Emerging Importance. J. Hosp. Librariansh. 2011, 11, 228–234. [Google Scholar] [CrossRef]
| Variable | Description | Justification | References |
|---|---|---|---|
| Distance to Mainland | Distance to the mainland (km) Range 3.14–24.57 km | Nearshore marine communities are subjected to higher turbidity due to terrigenous runoff and river plumes containing sediment, nutrients and pesticides. Coral reefs closer to shore also experience higher levels of recreational fishing pressure. | [72,73] |
| Distance to Land | Distance to the nearest land mass (km) Range 0.04–4.35 km | Distance to the nearest land mass influences how exposed a site is to wind and wave energy. Additionally, sites closer to land have increased terrestrial runoff. | [70,73] |
| Distance to 50 m Isobath | Distance to the 50 m depth isobath (km) Range 30.5–69.79 km | Sites closer to the 50 m isobath are influenced more by oceanic waters and generally have lower levels of turbidity. | [74] |
| Wave exposure | Exposure of the site based on average wave fetch and wind energy for across 32 angular sectors. Range 1005288–83322391 wave exposure index | Wave and wind energy act as a mechanical stress on corals and indirectly affect corals by influencing temperature, sediment flux, nutrient intake and productivity. | [69,75,76] |
| Turbidity (Mean KD490) | MODIS diffuse attenuation coefficient at 490 nm (KD2 algorithm). Higher KD490 value reflects a smaller attenuation depth, and lower clarity of ocean water. Range 0.074–0.259 KD490 index | Water turbidity influences the amount of available light for photosynthesis of coral symbionts whilst settling sediments can smother or abrade corals. | [27,77,78] |
| Depth | Depth of site (m) Range 3–7 m | Increasing depth reduces light availability, decreasing the autotrophic capacity of coral endosymbionts. Corals at shallower depths are also exposed to greater incidental light and subsequently increase the likelihood of bleaching. | [79,80] |
| Heat stress 2011 (Max DHW 11) | Maximum degree heating weeks between 01/12/2010–31/05/2011 (°C-weeks) Range 0–2.52 °C-weeks | Degree Heating Weeks represents accumulated thermal stress over the past 12 weeks. Coral bleaching is likely when DHW reaches 4°C-weeks. Severe bleaching was recorded in northwestern Australia in the summer of 2010–2011. | [47,81] |
| Heat stress 2013 (Max DHW 13) | Maximum degree heating weeks between 01/12/2012–31/05/2013 (°C-weeks) Range 8.4–12.56 °C-weeks | Severe bleaching was recorded in northwestern Australia in the summer of 2012–2013. | [81,82] |
| Mean Coral Recruits | Average density of coral recruits (2015–2017) for the 9 sites where data were available (recruits cm−2) Range 0.215–13.652 recruits cm−2 | Successful coral recruitment is essential for the maintenance of coral communities and recovery of reefs after disturbance events. | [64,83,84] |
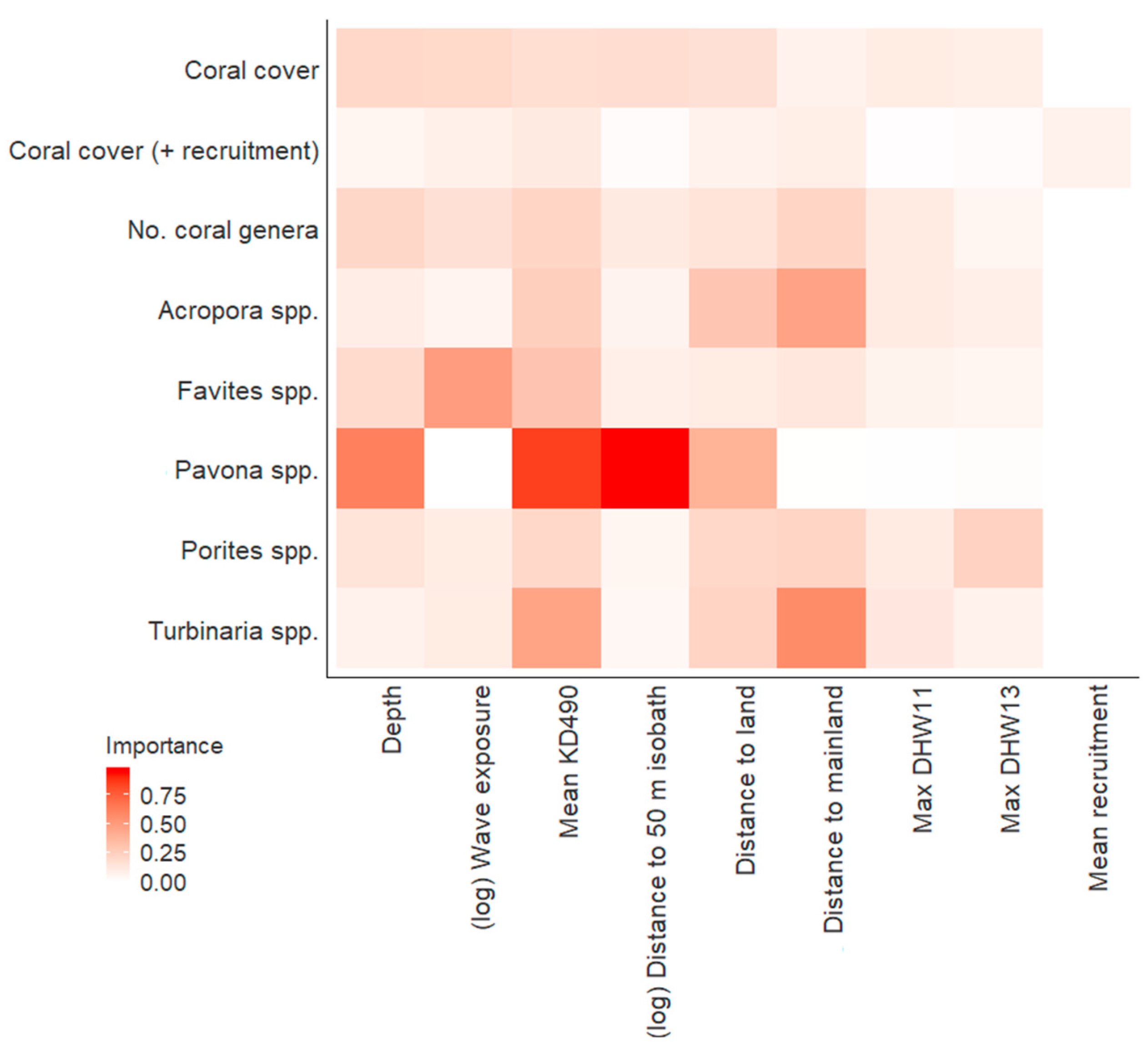
| Response Variable | Model | ∆AICc | AICc Weight | R2 | edf |
|---|---|---|---|---|---|
| Coral cover | null | 1.61 | 0.03 | 0.55 | 14.38 |
| Coral cover (including recruitment data) | null | 0.00 | 0.50 | 0.00 | 2.00 |
| No. coral genera | null | 0.41 | 0.06 | 0.83 | 13.17 |
| Acropora spp. | Distance to mainland | 0.11 | 0.13 | 0.36 | 12.84 |
| Favites spp. | Wave exposure | 0.00 | 0.19 | 0.49 | 11.56 |
| Pavona spp. | Depth + Mean KD490 + Distance to 50 m isobath | 0.00 | 0.57 | 0.13 | 5.92 |
| Porites spp. | Distance to mainland | 0.00 | 0.10 | 0.38 | 13.19 |
| Max DHW13 | 1.01 | 0.06 | 0.43 | 13.29 | |
| Turbinaria spp. | Distance to mainland | 1.39 | 0.14 | 0.36 | 12.04 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moustaka, M.; Mohring, M.B.; Holmes, T.; Evans, R.D.; Thomson, D.; Nutt, C.; Stoddart, J.; Wilson, S.K. Cross-shelf Heterogeneity of Coral Assemblages in Northwest Australia. Diversity 2019, 11, 15. https://doi.org/10.3390/d11020015
Moustaka M, Mohring MB, Holmes T, Evans RD, Thomson D, Nutt C, Stoddart J, Wilson SK. Cross-shelf Heterogeneity of Coral Assemblages in Northwest Australia. Diversity. 2019; 11(2):15. https://doi.org/10.3390/d11020015
Chicago/Turabian StyleMoustaka, Molly, Margaret B Mohring, Thomas Holmes, Richard D Evans, Damian Thomson, Christopher Nutt, Jim Stoddart, and Shaun K Wilson. 2019. "Cross-shelf Heterogeneity of Coral Assemblages in Northwest Australia" Diversity 11, no. 2: 15. https://doi.org/10.3390/d11020015
APA StyleMoustaka, M., Mohring, M. B., Holmes, T., Evans, R. D., Thomson, D., Nutt, C., Stoddart, J., & Wilson, S. K. (2019). Cross-shelf Heterogeneity of Coral Assemblages in Northwest Australia. Diversity, 11(2), 15. https://doi.org/10.3390/d11020015





