Abstract
Keratinolytic and keratinophilic fungi, such as dermatophytes, are frequently a cause of infections in humans and animals. Underground ecosystems are inhabited by various animals and are of interest for tourists. Therefore, the main goal of our research was the first evaluation of sediment and soil samples taken inside and outside the Harmanecká Cave in Slovakia for the occurrence of keratinolytic and keratinophilic fungi. Tests with Vanbreuseghema bait, as well as phenotyping and molecular methods, showed that all of the sampling sites contained ten isolates, all of the same species of keratinophilic fungi, belonging to the Microsporum cookei clade and with close affinities to Paraphyton cookei (Ajello) Y. Gräser, Dukik & de Hoog. Our research showed that, dependent on the medium, its mycelium varied in color and showed different growth rates. It also produced metabolites alkalizing DTM (dermatophyte test medium) medium. It dissolved keratin in in vitro hair perforation tests and was able to utilize most substrates in the API® 20C AUX, except for MDG (α-methyl-D-glucoside). In addition, the vegetative structures of mycelium were viable after storage at temperatures from −72 to −5 °C for 56 days, and actively grew after 28 days at a temperature range from 15 to 37 °C, with 25 °C being optimal. It showed weak, but active, growth at 5 and 10 °C after 56 days. We can assume that due to the low temperature in the caves, this fungus will not be able to actively grow rapidly on keratin substrates, but the contact with mammals, along with other favorable factors, might lead to an infection.
1. Introduction
Keratinolytic and keratinophilic fungi are a group of organisms that are able to utilize keratin as their sole source of carbon and nitrogen. They are known mainly as hair inhabitants, and besides keratin, these species might accumulate another hair components [1]. The main difference between keratinolytic and keratinophilic fungi is that the former are able to dissolve keratin, while the latter use only substances bound to keratin, non-protein components, or keratin decomposition products. Both keratinolytic and keratinophilic fungi are pathogens or potential pathogens of humans and animals. Many of them are also toxin producers [2,3].
The degradation of keratin is dependent mainly on the fungal isolate, e.g., species of low keratinolytic activity decomposed keratin up to 40% in at least 8 weeks, whereas isolates of high keratinolytic activity degraded keratin up to 80% in the same time [4]. It is worth mentioning that keratinophilic fungi tend to use proteins or amino acids as a carbon source, even when sugars are available [5]. They can also utilize lipids, however, their lipolytic activity is rather limited. Keratin may be the main source of nitrogen for keratinophilic species, which remove nitrogen excess by intensive deamination and ammonia production, which leads to the alkalinization of the surrounding environment. Nevertheless, the optimal pH for keratinophilic fungi ranges from 6.5 to 7.0 [4].
The biodiversity of many specific groups of microscopic fungi (micromycetes) in underground ecosystems is still unknown, and some of these are keratinolytic and keratinophilic fungi [6]. This group is represented by fungi of high epidemiological relevance (dermatophytes). Their classification is as follows: division: Ascomycota, class: Eurotiomycetes, order: Onygenales, family: Arthrodermataceae [7]. These microscopic fungi cause dermatoses and may reside on epidermis, hair, and nails. Dermatophytes decompose keratin in vitro and may invade host tissues in vivo leading to mycosis [8]. They may be classified into different ecological types: geophilic, zoophilic, and antropophilic [9]. Geophilic isolates cause infections with intensified, but short, inflammation. They are commonly found in soil inhabited by mammals, which can facilitate spreading of these fungi. Geophilic species have the ability of intense sexual reproduction. The transmission of zoophilic dermatophytes to humans occurs through the environment or an animal host, and the infection entails moderate inflammation. These species are able to reproduce sexually, however, anamorphic forms are also known. Anthropophilic dermatophytes usually interact with the human host. Infections proceed with weak inflammation, but are chronic. A telomorphic form is not known for any species [7,9].
The taxonomy of dermatophytes used to be limited to only three genera: Trichophyton, Epidermophyton, and Microsporum. Nowadays, we are able to classify them more precisely. Advanced molecular tests (ITS rDNA and partial LSU sequencing, ribosomal 60S subunit, β-tubulin fragments, and translation elongation factor 3) allowed for the classification of dermatophytes into nine genera: Trichophyton, Epidermophyton, Nannizia, Paraphyton, Lophophyton, Microsporum, Arthroderma, Ctenomyces, and Guarromyces [9].
The data on dermatophytes inhabiting underground ecosystems are so far scarce, because it is a nutritionally specialized group of fungi that requires specialized techniques and media for isolation. Research focused on the presence of these fungi in soil, organic litter, fresh bat guano, as well as air-born dust in underground objects and the presence of Arthroderma curreyi, A. quadrifidum, Microsporum gypseum, Trichophyton mentagrophytes, T. rubrum, and T. terrestre has been documented [10,11,12,13,14]. It must be mentioned, however, that methodology and results obtained by Lurie and Way [11] were not precise enough, since the authors themselves stated that they had no knowledge if the animals were free from T. mentagrophytes or if the proper controls were included.
The main goal of our research was to investigate sediment and soil samples taken inside and outside the Harmanecká Cave in Slovakia for the occurrence of keratinolytic and keratinophilic fungi. To our knowledge, this is the first such analysis. Additionally, the chosen isolate from ten isolates (all the same species) was characterized genetically and phenotypically, and some of its features, like the assimilation of various carbon sources, hair perforation, growth rates, and the survival at different temperatures, were determined.
2. Materials and Methods
2.1. Study Area
Harmanecká Cave is located to the northwest of the Banská Bystrica area (Slovakia), in the south part of Vel’ka Fatra Mountains, on the north mountainside of Kotolnica, at the elevation of 821 m (coordinates: 8°81′39″N, 19°04′01″E) [15,16]. The limestone cave developed in the middle Triassic, mainly due to the effects of underground water. The cave’s chambers were created from the effects of stone crumbling and collapsing. The cave was discovered in 1932 by Michal Bacúrik and has been available for sightseeing since 1950. During the war, the cave served as the shelter for the local people. Today, it is one of the most frequently visited places in Slovakia. In 2014, it was visited by approximately 17,425 tourists [16,17]. The overall length of the cave is 3,123 m, with a tourist route 1029 m long [15,18]. The air temperature fluctuates from 5.8 to 6.4 °C. The cave is one the most significant sites of bat occurrence in Slovakia. The presence of eleven bat species has been documented, with the majority being Myotis myotis and M. blythii, repeatedly noticed in amounts of 1000–1500 specimens. Other frequently described species are Pipistrellus pipistrellus and M. daubentonii. The cave is also the location of many invertebrates, the most interesting being Allorhiscosoma sphinx and Bathynella natans [15,19].
2.2. Sample Collection
The sediment and soil samples were taken on July 24th 2014 from one outdoor location (next to the entrance to the cave) and from four locations inside the cave, including one location in front of the airlock door (Vstupný dóm) and three locations behind the airlock door (near parts of the cave such as Gotický dóm, Dóm pagod, and Bludný dóm). The samples were collected using sterile plastic scoops and placed into sterile sampling bags. Approximately 1000 g of soil and sediment were collected from each tested site in triplicate. The samples were transported in cool conditions (ca. 10 ± 2.0 °C) to the laboratory and were stored in the cold room (ca. 7 ± 0.5 °C) until mycological analysis, which was carried out within 7 days.
2.3. Isolation of Fungi from Sediment and Soil Samples
The isolation of fungi was performed using Vanbreuseghem bait (hair bait) [20]. Samples of soil or sediment were placed on a sterile Petri dish. Then, using a sterile pincer, a clump of sterile human hair and distilled water were placed on the sample. Plates were then covered and incubated at 28 ± 0.5 °C for 4–8 weeks. Additionally, distilled water was supplemented if needed. The mycelium that appeared on the baits was transferred to Sabouraud’s glucose agar (SGA; 1000 mL distilled water, 10 g peptone, 40 g glucose, and 15 g agar), supplemented with ampiciline (50 mg · L−1), chloramphenicol (50 mg · L−1), and cycloheximide (100 mg · L−1) to inhibit bacterial and mold growth. The colonies of keratinophilic and keratinolytic fungi emerging on the Petri dishes were subcultured on PDA (potato dextrose agar, Biocorp, Poland) plates and incubated in the dark at room temperature (25 ± 0.5 °C) for four weeks. After incubation, fungi were purified by the single spore method and were subcultured on PDA slants for morphological and molecular identification.
2.4. Identification of Fungi
The preliminary identification of isolated fungi was based on macro- and microscopic evaluation of the culture (colony growth and color, alkaline metabolite production, and the presence of distinctive morphological structures, like spores) on the following media: Czapek-Dox agar (1.2% agar, Biocorp, Poland), DTM (dermatophyte test medium, Biocorp, Poland), malt extract agar (MEA, Biocorp, Poland), PDA, and SGA. Plates were incubated in the dark at 25 ± 0.5 °C for 7–49 days. The fungi were identified according to available monographs [7,9,21,22]. Microscopic slides were dyed with LPCB (lactophenol cotton blue, Sigma-Aldrich). Microscopic photographs were taken with Axio Image.M1 (Zeiss) and the macroscopic ones with Nikon Coolpix S3700. A total of 10 isolates of keratinophylic fungi were collected and preliminarily identified as one species, using classical methods. Next, one isolate with the phenotypic features characteristic for the group was used to confirm the species affiliation of the isolated fungi using molecular tests (ITS rDNA sequencing method). For this purpose, ITS1: TCCGTAGGTGAACCTGCGG, and ITS4: TCCTCCGCTTATTGATATGC primers were used [23]. DNA was extracted for a 21-day-old culture on PDA medium, according to the CTAB (cetyl trimethylammonium nromide) method [24] with minor modifications, such as the lack of tissue homogenization in liquid nitrogen in favor of the hyphae homogenization in lysis buffer (1% CTAB, 50 mM Tris-HCl pH = 8.0, 10 mM EDTA pH = 8.0, 0.7 M NaCl, 0.5% PVP40000, and 0.1% β-mercaptoethanol). The volume of the buffer was elevated from 400 to 700 μL and two extractions with chloroform: isoamyl alcohol (24:1; 500 μL) was performed twice, not once. The quality and quantity of DNA after extraction was checked using a spectrophotometer (NanoPhotometer® NP80, Inplent) at 230, 260, and 280 nm, and by electrophoresis in 1.0% agarose gel in Tris-borate in EDTA buffer (TBE). Amplification of fungal DNA was performed in the Bio-Rad T100 thermal cycler for 35 cycles in a 50 μL reaction mixture using the 2xPCR mixture: Taq polymerase (0.1 U µL−1), dNTP mix (2 mM), MgCl2 (4 mM), 0.25 μM of each primer, and 45 ng of genomic DNA [23]. After initial denaturation for 5 min at 94 °C, each cycle comprised 30 s denaturation at 94 °C, 30 s annealing at 55 °C, 45 s extension at 72 °C, and with a final extension for 7 min at 72 °C at the end of the last cycle. The amplification product was also verified using spectrophotometer and electrophoretic separation on 1.0% agarose gel in TBE. Then, the PCR product was purified using Clean-Up (A&A Biotechnology) and sequenced by the sequencing service at Macrogen Europe (Netherlands, http://dna.macrogen.com/eng/).
2.5. Bioinformatics Data Analysis
The PCR product sequences were analyzed using BioEdit Sequence Alignment Editor (http://www.mbio.ncsu.edu/bioedit/bioedit.html). The obtained fungal ITS sequences were compared with those deposited in the GenBank of the National Center for Biotechnology Information (NCBI, Bethesda, MD, USA) using the BLAST algorithm (http://www.ncbi.nlm.nih.gov/). The analysis of the genetic similarity of the studied fungal culture in terms of ITS sequences of other fungi deposited in the NCBI database was carried out using the MEGA program [25]. The genetic distance between analyzed sequences was evaluated using the likelihood method [26]. A phylogenetic tree was constructed using a neighbor-joining (NJ) technique and confirmed by bootstrap method with 1000 repetitions [27].
2.6. The Assimilation of Various Carbon Sources
For the evaluation of the assimilation of different carbon sources, commercially available API® 20C AUX (bioMérieux) was used and performed according to the manufacturer’s recommendations for filamentous fungi with high spore production, with some modifications. A mature, 4-week old colony was suspended in 10 mL of 0.85% NaCl with 0.025% Tween 80 and transferred to a sterile test tube, vortexed for 20 s, and set aside for 15–20 min. Homogenic supernatant was collected and the suspension density was set at 2.0 McF (McFarland). The suspension was transferred (150 µL) in triplicate to an API strip and incubated for 4 weeks at 24 ± 0.5 °C.
2.7. The in Vitro Hair Perforation Test
The experiment was performed according to Ogórek et al. [18]. Thirty fragments (2 cm) of blonde purified and sterile hair from a 2.5-year-old child were placed in a polystyrene Petri dish. Distilled water (25 mL) and 10% yeast extract (100 µL) was added and the mixture was inoculated with tested fungal culture. As the positive control, fF a clinical isolate of Trichophyton mentagrophytes (Department of Mycology and Genetics, Institute of Genetics and Microbiology, University of Wroclaw, Poland) was used. The negative control was not inoculated with any fungal culture. All samples were incubated at 25 ± 0.5 °C for 4 weeks. Microscopic slides were prepared and dyed with LPCB. Photographs were taken with Axio Image.M1 (Zeiss).
2.8. Survival at Different Temperatures
The experiment was carried out according to the method described by Ogórek et al. [18]. Fourteen-day-old fungal cultures on slants with PDA and YPG media (yeast extract peptone glucose: 10.0 mg · L−1 yeast extract, 20.0 mg · L−1 peptone, 20.0 mg · L−1 glucose, and 15.0 mg · L−1 agar) were placed at the following temperatures: -72, -25, -10, and 5 ± 0.5 °C. After storage (14, 28, 42, and 56 days) at each temperature, the fungal inocula (from three slants for each medium) were placed mycelium down in the center of PDA and YPG plates, in three replicates for each medium. The growth or lack of growth was evaluated after 10 days incubation in the dark at 25 ± 0.5 °C.
2.9. Growth at Different Temperatures
The first variant of the experiment using fungal spores was carried out according to Ogórek et al. [18]. Fungal spores (14-day-old cultures on PDA slant) were suspended in a solution of 0.85% NaCl with 0.025% Tween 80 (OD600 = 0.125, ~0.5 McFarland). Then, 5 µL of the spore suspension was spotted in the center of PDA and YPG plates, in three replicates for each medium. The growth or lack of growth was evaluated after 28 days of incubation in the dark at different temperatures: 5, 10, 15, 20, 25, 30, and 37 ± 0.5 °C. Additionally, the incubation at 5 and 10 °C was carried out for 56 days. In the second variant of the experiment, incubation time and temperature were the same, but mycelium was used instead of spores. The inocula of 6 mm diameter were taken from 14-day old fungal cultures on PDA medium and placed mycelium down in the center of the PDA and YPG plates, in three replicates for each medium.
3. Results
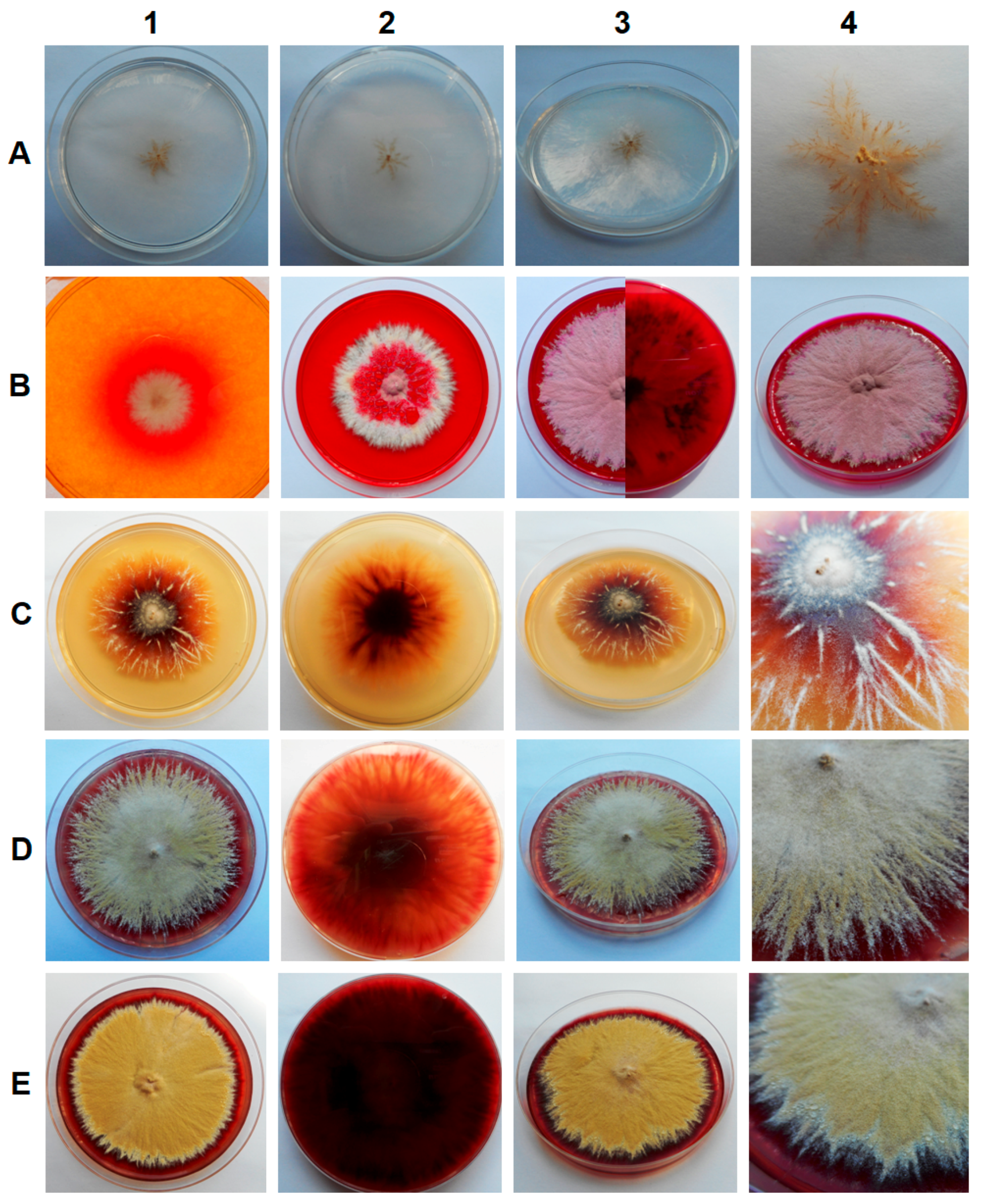
The samples of sediments and soil from all of the tested locations in the Harmanecká Cave contained keratinophilic fungi, which were isolated using hair bait. All isolates, a total of ten, varied phenotypically and showed different growth rates dependent on the medium, but exhibited similar phenotypic features on a given medium. The fastest growth was observed for the YPG medium and the slowest for Czapek-Dox agar medium. Colonies were granular and spreading, and their color ranged from deep grape-red to pale brown or suede-like, usually with a slightly raised and folded center and some radial grooves. The pigmentation on the reverse of the culture media varied from purple-red to dark reddish-brown, with the exception of the Czapek-Dox agar medium (Figure 1). Moreover, the secretion of alkaline metabolites on DTM medium was observed (change of color), which is typical for keratinophilic fungi (Figure 1B).
Figure 1.
Macroscopic observations of 7- (B1), 14- (B2) and 28-day-old (A, B3, B4, C, D, E) fungal culture isolated from soil and sediment in the Harmanecká Cave, Slovakia, on various media: A. Czapek-Dox agar, B. DTM, C. MEA, D. PDA, and E. SGA.
All of the cultures formed identical characteristic microscopic structures. Numerous large, very thick-walled, echinulate (rough) elliptical macroconidia with predominantly 5–6 septa were observed. The size of macroconidia usually ranged from 12–28 to 31–70 μm. Spiral hyphae were sporadically visible. A moderate number of mainly slender clavate with some pyriform microconidia was observed, typically in sizes ranging from 2–4 to 3–8 μm (Figure 2).
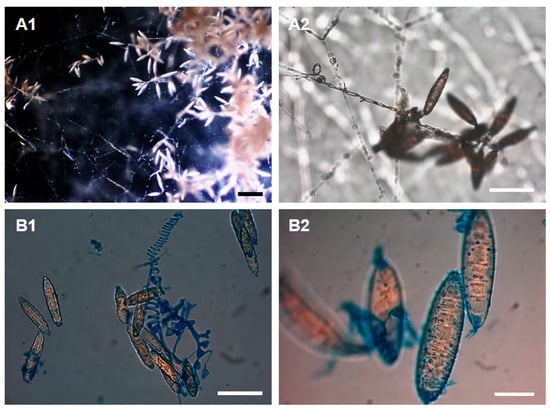
Figure 2.
Microscopic observations of morphological structures (A1. macroconidia, A2. macroconidia and spiral hyphae, B1. micro- and macroconidia with predominantly 5–6 septa and spiral hyphae, B2. thick-walled and rough elliptical macroconidia) of 4-week-old fungal culture isolated from soil and sediment in the Harmanecká Cave grown on PDA. A. Petri dish cultures under the optical microscope with (natural arrangement), B. microscopic slide dyed with lactophenol cotton blue. Scale bars: 100 μm (A1), 50 μm (A2, B1), 20 μm (B2).
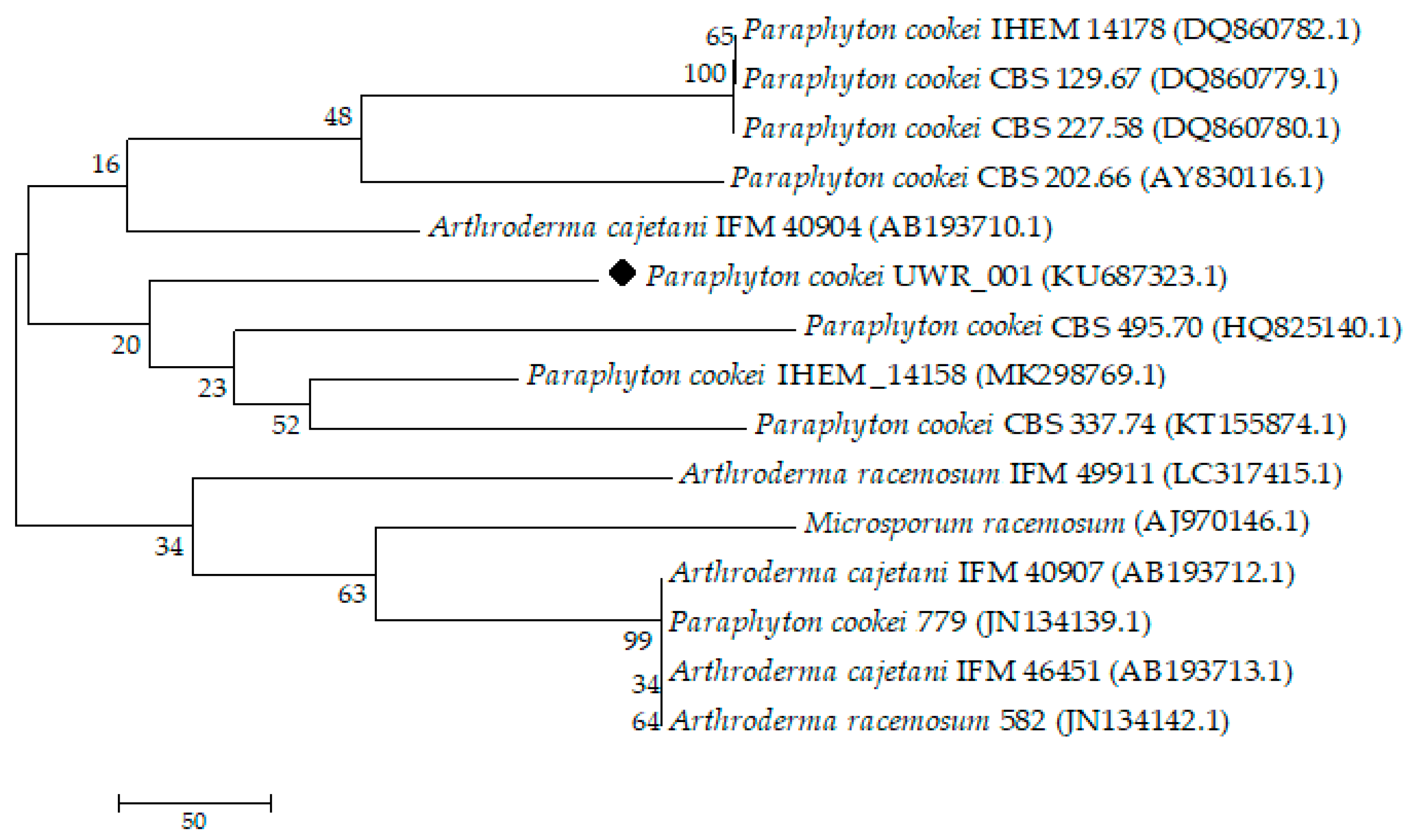
The comparison of those microscopic structures with the simultaneous macroscopic observation of the culture allowed for their probable affiliation to the Microsporum cookei clade and identification, with high probability, as Paraphyton cookei (Ajello) Y. Gräser, Dukik & de Hoog. Then, one isolate (UWR_001), out of ten collected during research was selected for further studies as a representative of a group with the same phenotypic features. The genetic studies were carried out in order to confirm preliminary classification. The ITS rDNA sequences (722 bp) were obtained from the isolate and they were compared to the sequences deposited in the GenBank of the National Center for Biotechnology Information by using the BLAST algorithm. This analysis confirmed that described dermatophyte had close affinities to P. cookei, and its ITS rDNA sequences were deposited in GenBank under the accession number KU687323.1. The propagation structures of the isolate were stored in the fungal collection of the Department of Mycology and Genetics, Institute of Genetics and Microbiology, University of Wroclaw, Poland. In addition, our research showed that the UWR_001 isolate belongs to one main group along with P. cookei CBS 495.70 (HQ825140.1), P. cookei IHEM_14158 (MK298769.1), and P. cookei CBS 337.74 (KT155874.1). It was phylogenetically related to the greatest extent to P. cookei CBS 495.70 (HQ825140.1), an isolate from the soil sample in the Netherlands (Figure 3).
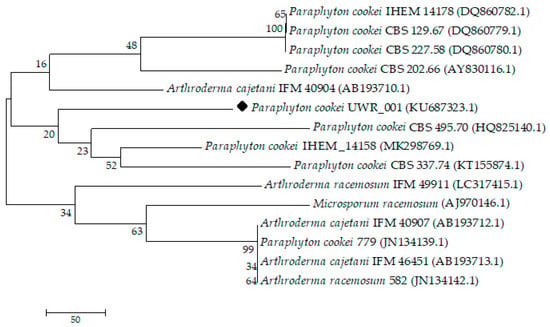
Figure 3.
The comparison of genetic similarity (neighbor-joining technique) of tested culture (P. cookei UWR_001 - KU687323.1, as black diamond) to other isolates deposited in the NCBI database based on ITS rDNA. For the analysis, the samples with at least 83.33% identicity were chosen. Isolate number (GenBank accession number).
As shown in the various temperature experiments, the isolate was able to grow on PDA and YPG after 28 days at 15, 20, 25, 30, and 37 °C but not at 5 and 10 °C, and with an optimum at 25 °C. However, after 56 days of incubation, it showed very weak growth also at 5 and 10 °C. On the other hand, its propagation structures were viable even after 56 days at the temperature range from −72 to −5 °C (Table 1).

Table 1.
Results of the growth and survival at different temperatures of Paraphyton cookei UWR_001 isolated from the sediment and soil in the Harmanecká Cave, Slovakia.
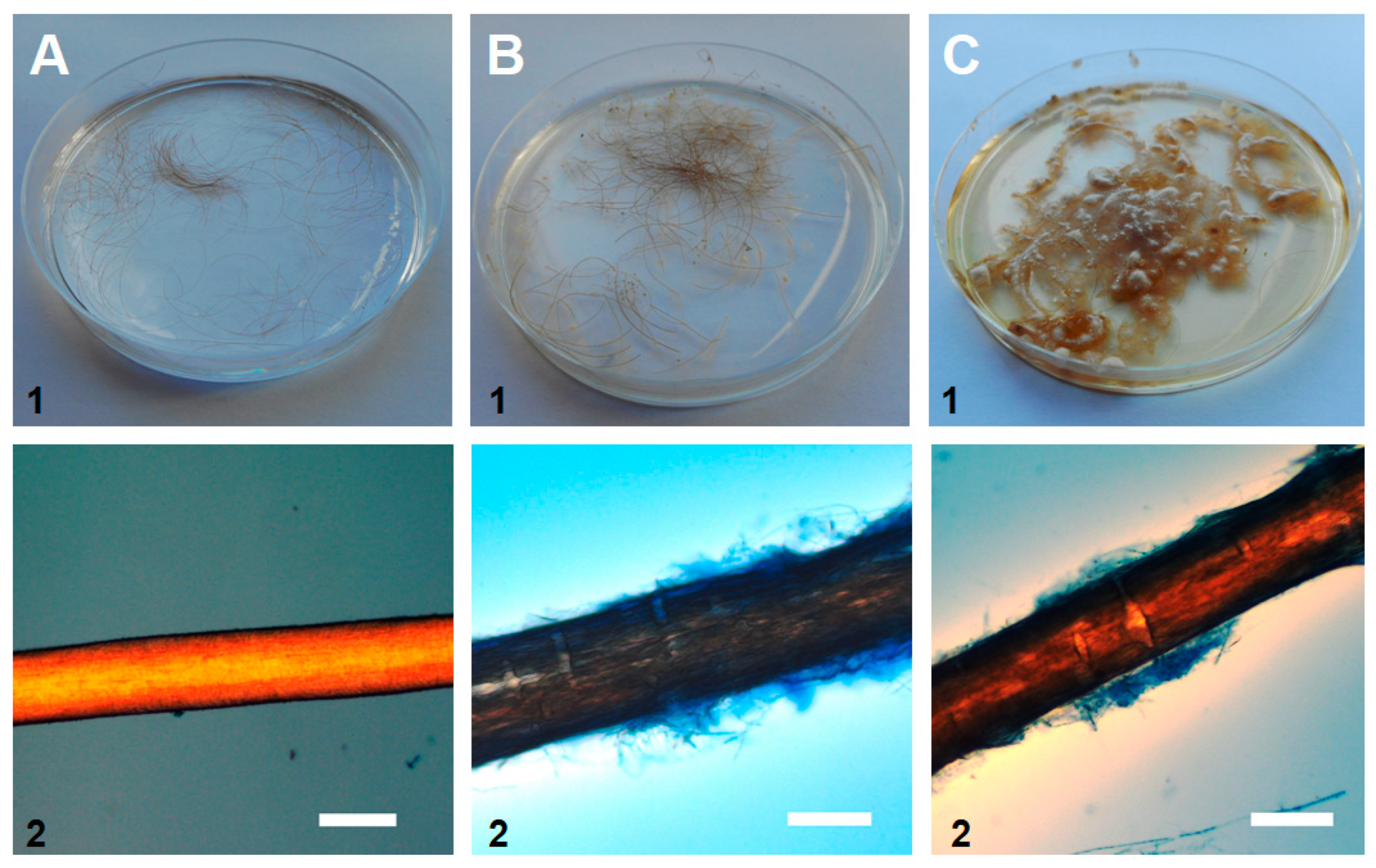
The hair perforation and the API® 20C AUX tests showed that our isolate was able to utilize keratin as a carbon source, as well as eighteen (out of nineteen) other tested substrates (the exception was α-methyl-D-glucoside, MDG). Moreover, hair pitting, caused by secreted fungal enzymes, appeared as perpendicular cotter, forming perforation organs, which is consistent with the positive control (T. mentagrophytes)—Figure 4.
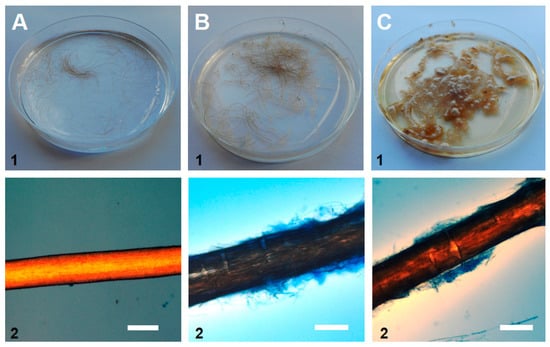
Figure 4.
Macroscopic observations of 28-day-old fungal culture on (1) a Petri dish in a hair perforation test in vitro and (2) microscopic observations of the hair (dyed with lactophenol cotton blue). A. negative control without fungi (no mycelium or hair pitting), B. tested sample of P. cookei KU687323.1 - UWR_001 (fungal growth and hair pitting), and C. positive control with T. mentagrophytes (observations identical to the tested sample). Scale bars: 100 μm.
4. Discussion
Fungi are cosmopolitan organisms, but underground ecosystems, both natural and anthropogenic, are not favorable to these microorganisms [28,29], due to the relatively low temperature and modest resources of nutrients [30]. As a consequence, fungi occur mainly in the form of spores and other propagation structures, dispersed in the air as the biological air fraction [31,32,33,34]. Active growth as a mycelium is relatively rare [18]. Underground objects are inhabited mainly by ascomycetous fungi (69% of all occurring fungi), with the most frequent isolations being of Alternaria, Aspergillus, Cladosporium, and Penicillium. On the other hand, the data on the presence of the Arthrodermataceae family in underground ecosystems is rather scarce [6]. Slovakian caves are in particular rarely studied, thus the present research was conducted.
The phenotypic studies showed that our UWR_001 isolate exhibits phenotype characteristic for the Microsporum cookei clade and probably belongs or is closely related to the P. cookie species. According to the literature, fungi belonging to this clade have very specific macro- and microscopic appearances [21]. Generally, the surface of the colony is very granular and its color is usually from deep grape-red to pale brown, whereas reverse pigment is from purple-red to dark reddish-brown, which is comparable to the isolate. A very specific feature of this clade, that was also observed, are macroconidia that are large (from 12–28 to 31–70 μm), rough, have very thick walls, and have 2 to 9 septa (predominantly 5–6 septa). Less specific and difficult to observe are microconidia with sizes of 2–4 to 3–8 μm, however, we were able to detect both microconidia and spiral hyphae, also characteristic for P. cookei [7,21,35]. The completion of UWR_001 isolate identification was phylogenetic studies, which showed that the isolated fungus is most closely related to P. cookei CBS 495.70 (HQ825140.1), an isolate from soil in the Netherlands. The isolate collected in our research was also found in soil samples. Thus, according to genotype-phenotype studies, we may conclude that the fungus isolated from soil and sediment in the Harmanecká Cave belongs to Microsporum cookei clade and probably to P. cookei (Ajello) Y. Gräser, Dukik & de Hoog (basionym: Microsporum cookei Ajello, Fac. syn.: Nannizzia cajetana Ajello ≡ Arthroderma cajetani (Ajello) Ajello, Weitzman, McGinnis & A.A. Padhye, and Microsporum racemosum Borelli ≡ Nannizzia racemosa Rush-Munro, J.M.B. Sm. & Borelli ≡ Arthroderma racemosum (Rush-Munro, J.M.B. Sm. & Borelli) Weitzman, McGinnis, A.A. Padhye & Ajello) species [9,35].
The composition of the medium is of great importance in culture methods, which was also confirmed in the present research. Depending on the medium, the isolated fungus showed different phenotypic features, like growth rate or the colony color, which may cause some difficulties in the proper classification of the species (affiliation to various phylogenetic groups). On the other hand, however, competent usage of those phenotypic differences on various media may be applied in classical identification methods [18]. Since our research showed the appearance of colony and fungal structures on various media, it may in the future facilitate identification of fungi belonging to the M. cookei clade and with close affinities to P. cookei in other dermatophyte research centers.
The ability of the assimilation of various carbon sources by the isolate UWR_001 points at the production of multiple enzymes and, as a consequence, at a good ability to respond to changes in environmental conditions. The only carbon source that was not utilized in API® 20C AUX test was MDG. Our isolate was also able to degrade keratin in hair perforation experiments. Such features are associated with the biosynthesis of intra- and extracellular hydrolytic and redox enzymes [36]. Keratin degradation, along with the color change of DTM medium, classify this fungus as a dermatophyte with an infective potential [37]. On the other hand, P. cookei could be used as a bioremediation and composting agent for soil contaminated with keratin waste. Enzymes secreted by P. cookei could be also used in feed production during fermentation. The application of viable, metabolically active cells for soil bioremediation is a common technique, however, it has some limitations, like the risk of introduction of applied microorganisms to the environment and infection of humans and animals [36].
Another important factor influencing microbial growth rate is the temperature of incubation. It can also have an effect on fungal structure formation, e.g., P. glandicola at 8 °C forms coremia [18]. The preferences of microorganisms for various temperatures classify them into psychrophilic, psychrotrophic, mesopjilic, or thermophilic groups. Our studies in the Harmanecká Cave focused mainly on the presence of cold-adapted fungi. According to Morita [38], those fungi may be classified as to psychrophilic fungi, which grow well at a temperature of 15 °C or lower, and psychrotrophic fungi, which require temperatures above 20 °C for maximum growth. Our isolate UWR_001 belongs to psychrotrophic fungi, since its growth was observed at 15–37 °C after 28 days on PDA and YPG medium, and the optimal temperature for the growth was 25 °C on both media. The growth at 5 and 10 °C, a temperature prevailing in underground ecosystems like the Harmanecká Cave, was observed only after 56 days. This feature, along with keratin degradation, points at the possible ability to infect internal and superficial tissues. According to Al-Fakih [39], these are two main attributes of such pathogens.
The propagation structures of the tested isolate closely related to P. cookei were viable after 56 days at low temperatures from −72 to −5 °C. Although this fungus was not capable of active and fast growth at 5 and 10 °C, it confirms that underground objects are ecological niches for highly specialized microorganisms and are probably inhabited by extremophilic fungi [18,40]. Such organisms are capable of survival or even actively grow in harsh conditions because they have developed mechanisms of tolerance to different stressors [41]. The survival strategy of the dermatophyte inhabiting the Harmanecká Cave is most likely connected to the preservation of propagation ability in unfavorable environments for a long time. Contact with the host (e.g., a rodent) may lead to infection and spread to other locations.
Paraphyton cookei is a geophilic fungus, isolated from hair of small mammals, with no clinical lesions. Infections have been reported in rodents, dogs and, rarely, in humans. It is unclear how this species invades hair in vivo, but it causes hair perforation in vitro, which was also confirmed by our research [42]. Since small mammals, like bats or rodents, are present in the Harmanecká Cave, the isolation of this fungus from this location is not surprising, however, so far it has not been reported from underground environments. In humans, this species mainly infects farmers, miners, and people involved in breeding (e.g., bird breeders). Thus, the presence of this species in the cave is a potential source of infection for visitors, especially immunocompromised ones [7].
Speleomycological studies of caves and other underground objects enable better understanding of relations in mycobiota of those peculiar ecosystems, as well as observations of the factors influencing microbiological balance [18,32,43,44,45,46]. Such studies are especially relevant in the case of objects inhabited by bats and open for tourists, since it may be the source of infections and allergies [47,48]. The discovery of P. cookei, an etiological agent of dermatoses, in soil and sediment is very important for aspects of ecology, as well as biological safety of the Harmanecká Cave.
5. Conclusions
We believe that this study contributes to a better understanding of the role of keratinolytic and keratinophilic fungi in underground ecosystems. The soil and sediments inside and outside Harmanecká Cave contained one keratinophilic species, which had close affinities (based on phenotypic and molecular studies) to P. cookei. The isolate changed its phenotypic features (colony color, growth rate) dependent on the medium. Moreover, it secreted metabolites alkalizing DTM medium, which is a characteristic for keratinophilic fungi. The tested fungus utilized a wide range of substrates as a carbon source, which was shown in an API® 20C AUX test, and degraded keratin in vitro in a hair perforation test. In addition, the vegetative structures of mycelium were viable in vitro after storage at temperatures from −72 to −5 °C for 56 days, and actively grew at temperatures from 15 to 37 °C, with an optimum at 25 °C after 28 days of incubation, and at 5 and 10 °C after 56 days of incubation. Thus, it can be assumed that underground ecosystems are probable reservoirs for this geophilic species, which was introduced there due to the activity of rodents, bats, and other small mammals. Low temperature in the cave is disadvantageous for the fungus to grow actively on keratin substrates left in the cave and to form numerous propagation structures, but the contact with mammals, along with other favorable factors (immunosupression, epidermal lesions), might lead to infection. Moreover, physiological and phenotypic profiling will in the future facilitate identification of this species.
Author Contributions
R.O. conceptualization, data curation, formal analysis, investigation, methodology, supervision, validation, visualization, writing—original draft; A.P. writing—original draft; Z.V. investigation; M.C. writing—original draft; K.N. writing—original draft. All the authors revised the paper and approved the final version.
Funding
This research received no external funding.
Acknowledgments
We thank Mariusz Dyląg (Department of Mycology and Genetics, Institute of Genetics and Microbiology, University of Wroclaw, Poland) for technical support and materials used for experiments in the study.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sharma, R.; Rajak, R.C. Keratinophilic fungi: nature’s keratin degrading machines! Resonance 2003, 8, 28–40. [Google Scholar] [CrossRef]
- Gherbawy, Y.A.M.H. Keratinolytic and keratinophilic fungi of mangrove’s soil and air in the city of Qena and their response to garlic extract and onion oil treatments. Acta Mycol. 1996, 31, 87–89. [Google Scholar] [CrossRef][Green Version]
- Ulfig, K.; Płaza, G.; Terakowskip, M.; Janda-Ulfig, K. Sewage sludge open-air drying affects on keratinolytic, keratinophilic and actidione-resistant fungi. Roczniki PZH 2006, 57, 371–379. [Google Scholar]
- Kunert, J. Physiology of keratinophilic fungi. Rev. Iberoam Micol. 2000, 669, 77–85. [Google Scholar]
- Philpot, C.M. The use of nutritional tests for the differentiation of dermatophytes. Sabouraudia 1977, 15, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Vanderwolf, K.J.; Malloch, D.; McAlpine, D.F.; Forbes, G.J. A world review of fungi, yeasts, and slime molds in caves. Int. J. Speleol. 2013, 42, 77–96. [Google Scholar] [CrossRef]
- Weitzman, I.; Summerbell, R.C. The Dermatophytes. CMR 1995, 8, 240–259. [Google Scholar] [CrossRef]
- Simpanya, M.F. Dermatophytes: Their taxonomy, ecology and pathogenicity. Rev. Iberoam Micol. 2000, 669, 1–12. [Google Scholar]
- de Hoog, G.S.; Dukik, K.; Monod, M.; Packeu, A.; Stubbe, D.; Hendrickx, M.; Kupsch, C.; Stielow, J.B.; Freeke, J.; Göker, M.; et al. Toward a novel multilocus phylogenetic taxonomy for the dermatophytes. Mycopathologia 2017, 182, 5–31. [Google Scholar] [CrossRef]
- Lurie, H.I.; Borok, R. Trichophyton mentagrophytes isolated from the soil of caves. Mycologia 1955, 47, 506–510. [Google Scholar] [CrossRef]
- Lurie, H.I.; Way, M. The isolation of dermatophytes from the atmosphere of caves. Mycologia 1957, 49, 178–180. [Google Scholar] [CrossRef]
- Kajihiro, E.S. Occurrence of dermatophytes in fresh bat guano. Appl. Microbiol. 1965, 13, 720–724. [Google Scholar] [PubMed]
- Balabanoff, V.A. Comparative studies of dermatophytes isolated from caves and stables in Bulgaria. Mycopathol. Mycol. Appl. 1967, 32, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.F.; Liu, F.; Zhou, X.; Liu, X.Z.; Liu, S.J.; Cai, L. Culturable mycobiota from Karst caves in China, with descriptions of 20 new species. Persoonia 2017, 39, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Bella, P.; Hlavac, J.; Gazik, P. Protection and Management of Show Caves in Slovakia. In Proceedings of the 13th International Congress of Speleology 4th Speleological Congress of Latin América and Caribbean 26th Brazilian Congress of Speleology Brasília, Brasília, Brazil, 15–22 July 2001; pp. 287–290. [Google Scholar]
- Bella, P. Slovakia Show Caves; Grafon: Liptovský Mikuláš, Slovakia, 2003; pp. 21–55. [Google Scholar]
- Nudziková, Ľ. Course of show caves attendance in Slovakia since 2009. Aragonit 2014, 19, 35–38. (In Slovak) [Google Scholar]
- Ogórek, R.; Dyląg, M.; Kozak, B. Dark stains on rock surfaces in Driny Cave (Little Carpathian Mountains, Slovakia). Extremophiles 2016, 20, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Lehotská, B.; Lehotský, R. 15 years of winter monitoring of bats in the Driny Cave. Aragonit 2009, 14, 171–172. (In Slovak) [Google Scholar]
- Vanbreuseghem, R. Technique biologique pour I’ isolement des dermat ophytes dusol. Ann. Soc. Belge. Med. Trop. 1952, 32, 173–178. [Google Scholar]
- Choi, J.S.; Gräser, Y.; Walther, G.; Peano, A.; Symoens, F.; de Hoog, S. Microsporum mirabile and its teleomorph Arthroderma mirabile, a new dermatophyte species in the M. cookei clade. Med. Mycol. 2012, 50, 161–169. [Google Scholar] [CrossRef][Green Version]
- Metin, B.; Heitman, J. Sexual reproduction in dermatophytes. Mycopathol 2017, 182, 45–55. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Doyle, J.J.; Doyle, J.L. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytoch. Bull. 1987, 19, 11–15. [Google Scholar]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Nei, M.; Kumar, S. Prospects for inferring very large phylogenies by using the neighbor-joining method. Proc. Natl. Acad. Sci. USA 2004, 101, 11030–11035. [Google Scholar] [CrossRef] [PubMed]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [PubMed]
- Ogórek, R.; Lejman, A.; Matkowski, K. Fungi isolated from the Niedźwiedzia Cave in Kletno (Lower Silesia, Poland). Int. J. Speleol. 2013, 42, 161–166. [Google Scholar] [CrossRef]
- Nováková, A.; Hubka, V.; Valinová, Š.; Kolařík, M.; Hillebrand-Voiculescu, A.M. Cultivable microscopic fungi from an underground chemosynthesis-based ecosystem: A preliminary study. Folia Microbiol. 2018, 63, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Poulson, T.L.; White, W.B. The cave environment. Science 1969, 165, 971–981. [Google Scholar] [CrossRef]
- Nováková, A. Microscopic fungi isolated from the Domica Cave system (Slovak Karst National Park, Slovakia). A review. Int. J. Speleol. 2009, 38, 71–82. [Google Scholar] [CrossRef]
- Pusz, W.; Ogórek, R.; Uklańska-Pusz, C.; Zagożdżon, P. Speleomycological research in underground Osówka Complex in Sowie Mountains (Lower Silesia, Poland). Int. J. Speleol. 2014, 43, 27–34. [Google Scholar] [CrossRef]
- Mulec, J.; Vaupotic, J.; Walochnik, J. Prokaryotic and eukaryotic airborne microorganisms as tracers of microclimatic changes in the underground (Postojna Cave, Slovenia). Environ. Microbiol. 2012, 64, 654–667. [Google Scholar] [CrossRef]
- Wang, W.; Ma, X.; Ma, Y.; Mao, L.; Wu, F.; Ma, X.; An, L.; Feng, H. Seasonal dynamics of airborne fungi in different caves of the Mogao Grottoes, Dunhuang, China. Int. Biodeter. Biodeger. 2010, 64, 461–466. [Google Scholar] [CrossRef]
- Borelli, D. Microsporum racemosum nova species. Acta Med. Venez. 1965, 12, 148–151. [Google Scholar]
- Gianfreda, L.; Rao, M. Potential of extra cellular enzymes in remediation of polluted soils: A review. Enzyme Microb. Technol. 2004, 35, 339–354. [Google Scholar] [CrossRef]
- Deshmukh, S.K.; Agrawal, S.C. In vitro degradation of human hair by some keratinophilic fungi. Mykosen 1982, 25, 454–458. [Google Scholar] [CrossRef] [PubMed]
- Morita, R.Y. Psychrophilic bacteria. Bacteriol. Rev. 1975, 39, 144–167. [Google Scholar] [PubMed]
- Al-Fakih, A.A. Overview on the fungal metabolites involved in mycopathy. Open J. Med. Microbiol. 2014, 4, 38–63. [Google Scholar] [CrossRef]
- Schabereiter-Gurtner, C.; Saiz-Jimenez, C.; Piñar, G.; Lubitz, W.; Rolleke, S. Phylogenetic diversity of bacteria associated with Paleolithic paintings and surrounding rock walls in two Spanish caves (Llonin and La Garma). FEMS Microb. Ecol. 2004, 47, 1–13. [Google Scholar] [CrossRef]
- McKenzie, R.L.; Bjorn, L.O.; Bais, A.; Iayis, M. Changes in biologically active ultraviolet radiation reaching the Earth’s Surface. Photochem. Photobiol. Sci. 2003, 2, 5–15. [Google Scholar] [CrossRef]
- Kidd, S.; Halliday, C.; Alexiou, H.; Ellis, D. Descriptions of Medical Fungi, 3rd ed.; Newstyle Printing: Adelaide, South Australia, 2006; p. 149. [Google Scholar]
- Bastian, F.; Jurado, V.; Nováková, A.; Alabouvette, C.; Saiz-Jimenez, C. The microbiology of Lascaux Cave. Microbiology 2010, 156, 644–652. [Google Scholar] [CrossRef]
- Griffin, D.W.; Gray, M.A.; Lyles, M.B.; Northup, D.E. The transport of nonindigenous microorganisms into caves by human visitation: A case study at Carlsbad Caverns National Park. Geomicrob. J. 2014, 31, 175–185. [Google Scholar] [CrossRef]
- Martin-Sanchez, P.M.; Nováková, A.; Bastian, F.; Alabouvette, C.; Saiz-Jimenez, C. Two new species of the genus Ochroconis, O. lascauxensis and O. anomala isolated from black stains in Lascaux Cave, France. Fungal Biol. 2012, 116, 574–589. [Google Scholar] [CrossRef] [PubMed]
- Ogórek, R.; Višňovská, Z.; Tančinová, D. Mycobiota of underground habitats: Case study of Harmanecká Cave in Slovakia. Microb. Ecol. 2016, 71, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Kokurewicz, T.; Ogórek, R.; Pusz, W.; Matkowski, K. Bats increase the number of cultivable airborne fungi in the “Nietoperek’’ bat reserve in Western Poland. Microb. Ecol. 2016, 72, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Victor, T.R.; Rajkumar, S.S.; Li, X.; Okoniewski, J.C.; Hicks, A.C.; Davis, A.D.; Broussard, K.; LaDeau, S.L.; Chaturvedi, S.; et al. Mycobiome of the bat White Nose Syndrome affected caves and mines reveals diversity of fungi and local adaptation by the fungal pathogen Pseudogymnoascus (Geomyces) destructans. PLoS ONE 2014, 9, e116149. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).