A Guide to Using Compound-Specific Stable Isotope Analysis to Study the Fates of Molecules in Organisms and Ecosystems
Abstract
1. Stable Isotope Analysis of Bulk Tissues
2. Principles of Compound-Specific Isotope Analysis (CSIA)
3. CSIA Methods
3.1. Sample Collection and Storage
3.2. Chemical Preparation for AA Isotope Analysis
3.3. Chemical Preparation for FA Isotope Analysis
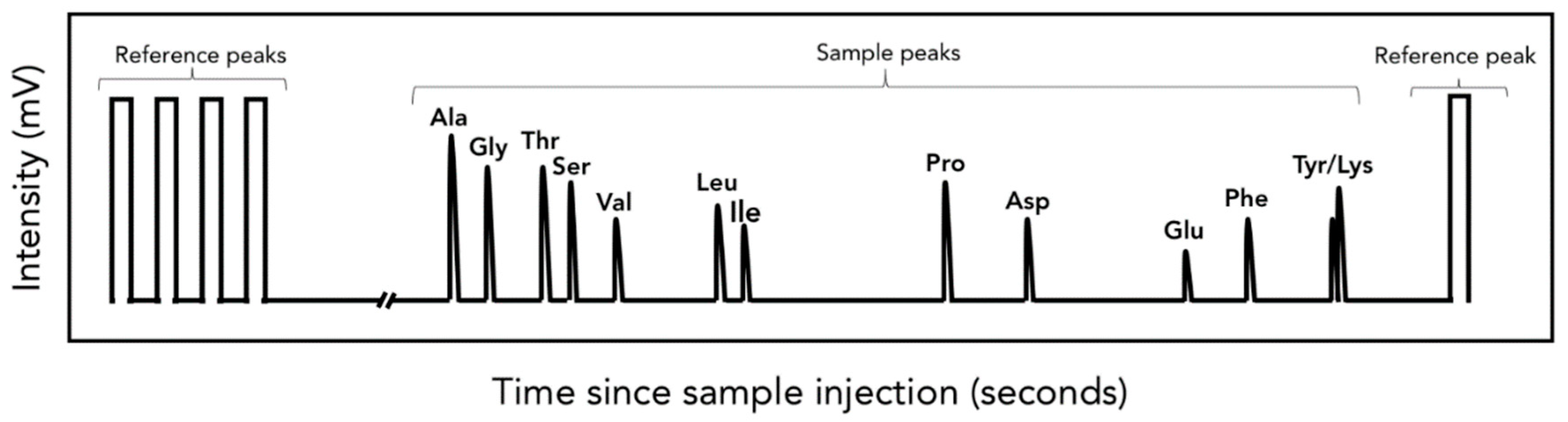
3.4. Isotopic Analysis of Individual AA and FA
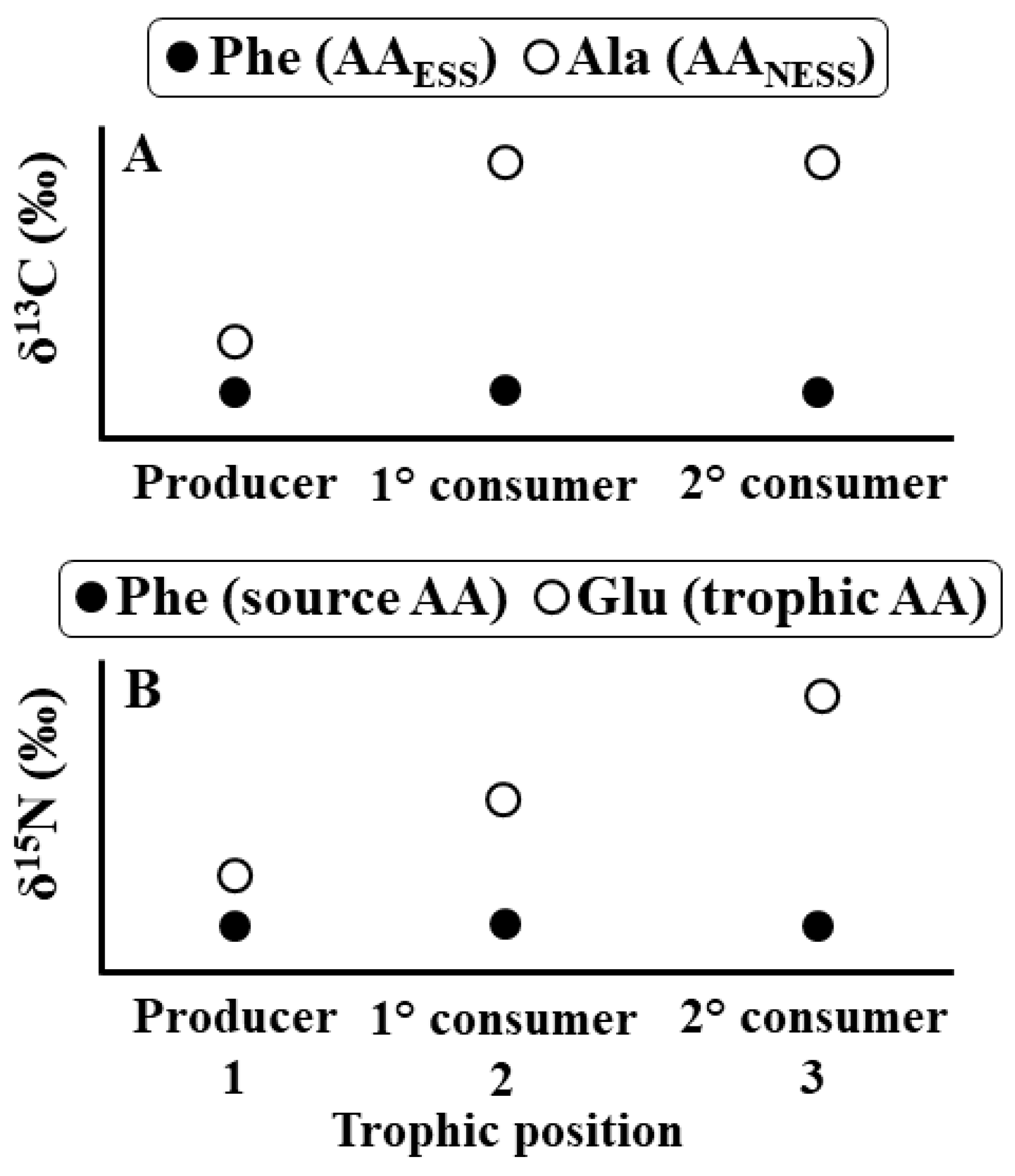
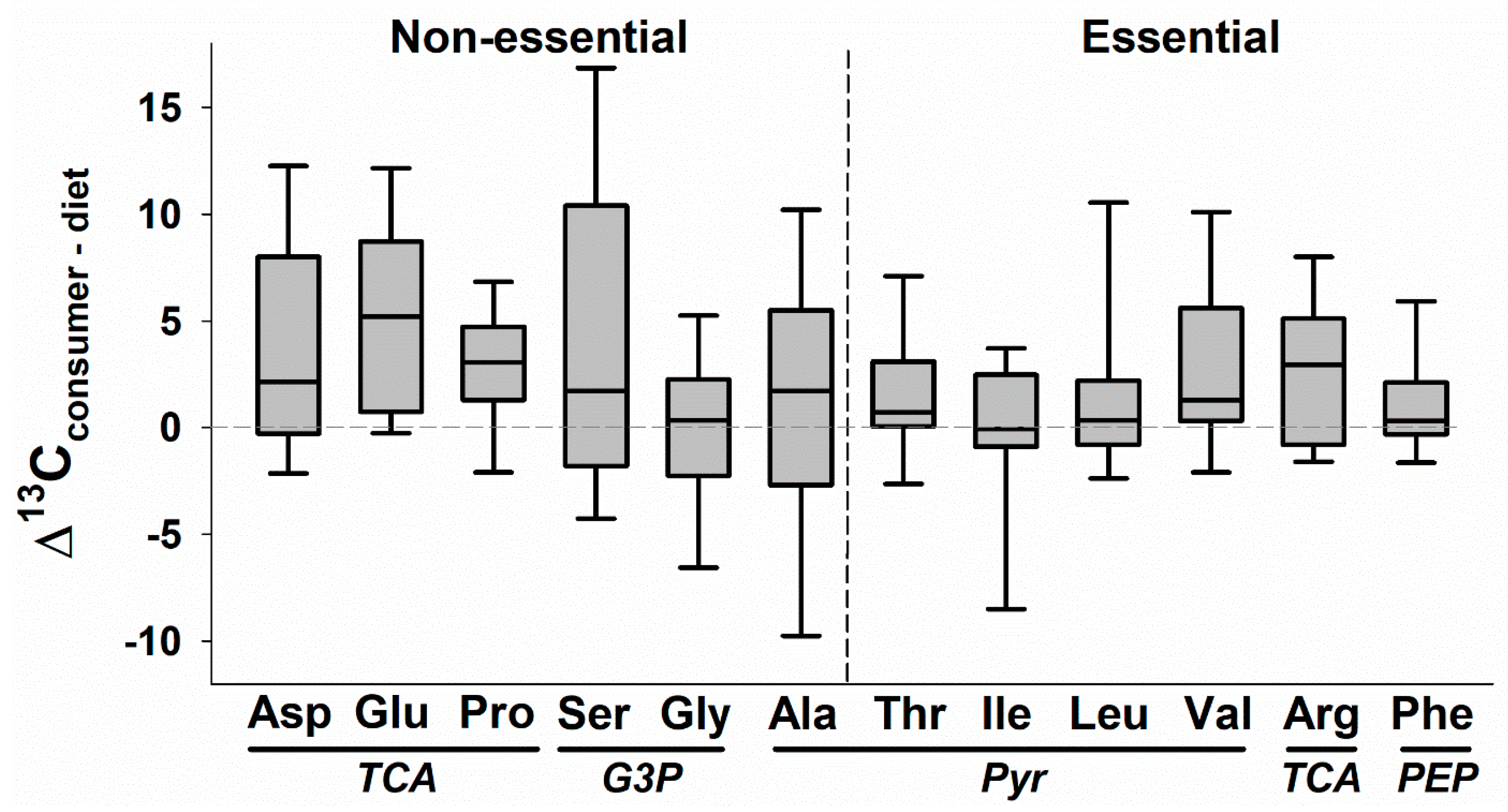
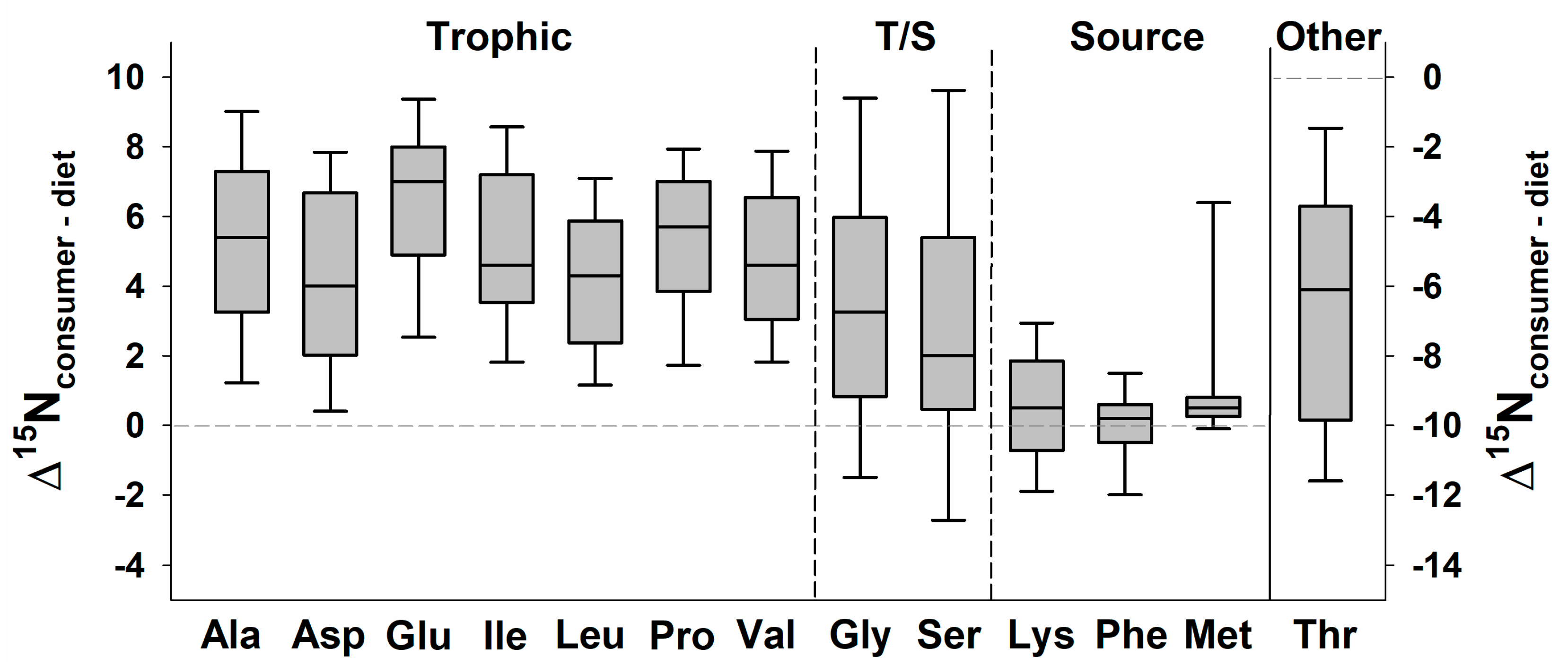
4. CSIA Applications to Date
5. CSIA Limitations
6. CSIA Future Directions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Michener, R.; Lajtha, K. (Eds.) Stable Isotopes in Ecology and Environmental Science, 2nd ed.; Wiley-Blackwell: Malden, MA, USA, 2007; ISBN 978-1-4051-2680-9. [Google Scholar]
- Kelly, J.F. Stable isotopes of carbon and nitrogen in the study of avian and mammalian trophic ecology. Can. J. Zool. 2000, 78, 1–27. [Google Scholar] [CrossRef]
- Vanderklift, M.A.; Ponsard, S. Sources of variation in consumer-diet 15N enrichment: A meta-analysis. Oecologia 2003, 136, 169–182. [Google Scholar] [CrossRef] [PubMed]
- Minagawa, M.; Wada, E. Stepwise enrichment of N-15 along food-chains—Further evidence and the relation between delta-N-15 and animal age. Geochim. Cosmochim. Acta 1984, 48, 1135–1140. [Google Scholar] [CrossRef]
- Hobson, K.A.; Atwell, L.; Wassenaar, L.I. Influence of drinking water and diet on the stable-hydrogen isotope ratios of animal tissues. Proc. Natl. Acad. Sci. USA 1999, 96, 8003–8006. [Google Scholar] [CrossRef] [PubMed]
- Bowen, G.J.; Wassenaar, L.I.; Hobson, K.A. Global application of stable hydrogen and oxygen isotopes to wildlife forensics. Oecologia 2005, 143, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Martínez del Rio, C.M.; Wolf, N.; Carleton, S.A.; Gannes, L.Z. Isotopic ecology ten years after a call for more laboratory experiments. Biol. Rev. 2009, 84, 91–111. [Google Scholar] [CrossRef] [PubMed]
- Layman, C.A.; Arrington, D.A.; Montana, C.G.; Post, D.M. Can stable isotope ratios provide for community-wide measures of trophic structure? Ecology 2007, 88, 42–48. [Google Scholar] [CrossRef]
- Newsome, S.D.; Martinez del Rio, C.; Bearhop, S.; Phillips, D.L. A niche for isotopic ecology. Front. Ecol. Environ. 2007, 5, 429–436. [Google Scholar] [CrossRef]
- Perkins, M.J.; McDonald, R.A.; van Veen, F.J.F.; Kelly, S.D.; Rees, G.; Bearhop, S. Application of nitrogen and carbon stable isotopes (δ15N and δ13C) to quantify food chain length and trophic structure. PLoS ONE 2014, 9, e93281. [Google Scholar] [CrossRef]
- Newsome, S.D.; Tinker, M.T.; Monson, D.H.; Oftedal, O.T.; Ralls, K.; Staedler, M.M.; Fogel, M.L.; Estes, J.A. Using stable isotopes to investigate individual diet specialization in California sea otters (Enhydra lutris nereis). Ecology 2009, 90, 961–974. [Google Scholar] [CrossRef]
- Martínez del Rio, C.; Carleton, S.A. How fast and how faithful: The dynamics of isotopic incorporation into animal tissues. J. Mammal. 2012, 93, 353–359. [Google Scholar] [CrossRef]
- Busquets-Vass, G.; Newsome, S.D.; Calambokidis, J.; Serra-Valente, G.; Jacobsen, J.K.; Aguíñiga-García, S.; Gendron, D. Estimating blue whale skin isotopic incorporation rates and baleen growth rates: Implications for assessing diet and movement patterns in mysticetes. PLoS ONE 2017, 12, e0177880. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, G.D.; O’Leary, M.H.; Berry, J.A. On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Functional Plant Biol. 1982, 9, 121–137. [Google Scholar] [CrossRef]
- O’Leary, M.H. Carbon isotopes in photosynthesis. BioScience 1988, 38, 328–336. [Google Scholar] [CrossRef]
- Dawson, T.E.; Mambelli, S.; Plamboeck, A.H.; Templer, P.H.; Tu, K.P. Stable isotopes in plant ecology. Annu. Rev. Ecol. Syst. 2002, 33, 507–559. [Google Scholar] [CrossRef]
- Ambrose, S.H. Preparation and characterization of bone and tooth collagen for isotopic analysis. J. Archaeol. Sci. 1990, 17, 431–451. [Google Scholar] [CrossRef]
- Deniro, M.J.; Epstein, S. Mechanism of carbon isotope fractionation associated with lipid synthesis. Science 1977, 197, 261–264. [Google Scholar] [CrossRef]
- Cabana, G.; Rasmussen, J.B. Comparison of aquatic food chains using nitrogen isotopes. PNAS 1996, 93, 10844–10847. [Google Scholar] [CrossRef]
- Barnes, C.; Jennings, J.T.; Barry, S. Environmental correlates of large-scale spatial variation in the δ13C of marine animals. Estuar. Coast. Shelf Sci. 2009, 81, 368–374. [Google Scholar] [CrossRef]
- Finlay, J.C. Stable-carbon-isotope ratios of river biota: Implications for energy flow in lotic food webs. Ecology 2001, 82, 1052–1064. [Google Scholar] [CrossRef]
- Hershey, A.E.; Northington, R.M.; Finlay, J.C.; Peterson, B.J. Stable isotopes in stream food webs. In Methods in Stream Ecology; Elsevier: New York, NY, USA, 2017. [Google Scholar]
- Newsome, S.D.; Clementz, M.T.; Koch, P.L. Using stable isotope biogeochemistry to study marine mammal ecology. Mar. Mammal Sci. 2010, 26, 509–572. [Google Scholar] [CrossRef]
- Lee, T.N.; Buck, C.L.; Barnes, B.M.; O’Brien, D.M. A test of alternative models for increased tissue nitrogen isotope ratios during fasting in hibernating arctic ground squirrels. J. Exp. Biol. 2012, 215, 3354–3361. [Google Scholar] [CrossRef] [PubMed]
- Hill, R.W.; Wyse, G.A.; Anderson, M. Animal Physiology, 2nd ed.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2008. [Google Scholar]
- Wu, G. Amino acids: Metabolism, functions, and nutrition. Amino Acids 2009, 37, 1–17. [Google Scholar] [CrossRef]
- Payne, S.H.; Loomis, W.F. Retention and loss of amino acid biosynthetic pathways based on analysis of whole-genome sequences. Eukaryot Cell 2006, 5, 272–276. [Google Scholar] [CrossRef]
- Austic, R.E. Nutritional and metabolic interrelationships of arginine, glutamic acid and proline in the chicken. Fed. Proc. 1976, 35, 1914–1916. [Google Scholar] [PubMed]
- Koutsos, E.A.; Matson, K.D.; Klasing, K.C. Nutrition of birds in the order Psittaciformes: A review. J. Avian Med. Surg. 2001, 15, 257–275. [Google Scholar] [CrossRef]
- Kanazawa, A.; Teshima, S. Essential amino acids of the prawn. Bull. Jpn. Soc. Sci. Fish. 1981, 47, 1375–1377. [Google Scholar] [CrossRef]
- McMahon, K.W.; McCarthy, M.D. Embracing variability in amino acid δ15N fractionation: Mechanisms, implications, and applications for trophic ecology. Ecosphere 2016, 7, e01511. [Google Scholar] [CrossRef]
- O’Connell, T.C. ‘Trophic’ and ‘source’ amino acids in trophic estimation: A likely metabolic explanation. Oecologia 2017, 184, 317–326. [Google Scholar] [CrossRef]
- Taylor, E.J.; Nott, H.M.R.; Earle, K.E. Dietary glycine: Its importance in growth and development of the budgerigar (Melopsittacus undulatus). J. Nutr. 1994, 124, 2555S–2558S. [Google Scholar] [CrossRef]
- McMahon, K.W.; Polito, M.J.; Abel, S.; McCarthy, M.D.; Thorrold, S.R. Carbon and nitrogen isotope fractionation of amino acids in an avian marine predator, the gentoo penguin (Pygoscelis papua). Ecol. Evol. 2015, 5, 1278–1290. [Google Scholar] [CrossRef] [PubMed]
- Newsome, S.D.; Fogel, M.L.; Kelly, L.; Martínez del Rio, C. Contributions of direct incorporation from diet and microbial amino acids to protein synthesis in Nile tilapia. Funct. Ecol. 2011, 25, 1051–1062. [Google Scholar] [CrossRef]
- Newsome, S.D.; Wolf, N.; Peters, J.; Fogel, M.L. Amino acid δ13C analysis shows flexibility in the routing of dietary protein and lipids to the tissue of an omnivore. Integr. Comp. Biol. 2014, 54, 890–902. [Google Scholar] [CrossRef] [PubMed]
- Chikaraishi, Y.; Steffan, S.A.; Ogawa, N.O.; Ishikawa, N.F.; Sasaki, Y.; Tsuchiya, M.; Ohkouchi, N. High-resolution food webs based on nitrogen isotopic composition of amino acids. Ecol. Evol. 2014, 4, 2423–2449. [Google Scholar] [CrossRef] [PubMed]
- Fuller, B.T.; Petzke, K.J. The dietary protein paradox and threonine 15N-depletion: Pyridoxal-5′-phosphate enzyme activity as a mechanism for the δ15N trophic level effect. Rapid Commun. Mass Spectrom. 2017, 31, 705–718. [Google Scholar] [CrossRef]
- Budge, S.M.; Wang, S.W.; Hollmén, T.E.; Wooller, M.J. Carbon isotopic fractionation in eider adipose tissue varies with fatty acid structure: Implications for trophic studies. J. Exp. Biol. 2011, 214, 3790–3800. [Google Scholar] [CrossRef]
- Carter, W.A.; Whiteman, J.P.; Cooper-Mullin, C.; Newsome, S.D.; McWilliams, S.R. The dynamics of individual fatty acids in muscle fat stores and membranes of a songbird are affected by exercise and have functional and ecological importance. Physiol. Biochem. Zool. 2019, in press. [Google Scholar]
- Chamberlain, P.M.; Bull, I.D.; Black, H.I.J.; Ineson, P.; Evershed, R.P. Lipid content and carbon assimilation in Collembola: Implications for the use of compound-specific carbon isotope analysis in animal dietary studies. Oecologia 2004, 139, 325–335. [Google Scholar] [CrossRef]
- Ruess, L.; Chamberlain, P.M. The fat that matters: Soil food web analysis using fatty acids and their carbon stable isotope signature. Soil Biol. Biochem. 2010, 42, 1898–1910. [Google Scholar] [CrossRef]
- Budge, S.M.; Iverson, S.J.; Koopman, H.N. Studying trophic ecology in marine ecosystems using fatty acids: A primer on analysis and interpretation. Mar. Mammal Sci. 2006, 22, 759–801. [Google Scholar] [CrossRef]
- Newsome, S.D.; Chivers, S.J.; Berman Kowalewski, M. The influence of lipid-extraction and long-term DMSO preservation on carbon (δ13 C) and nitrogen (δ15 N) isotope values in cetacean skin. Mar. Mammal Sci. 2018, 34, 277–293. [Google Scholar] [CrossRef]
- Kim, S.L.; Koch, P.L. Methods to collect, preserve, and prepare elasmobranch tissues for stable isotope analysis. Environ. Biol. Fish. 2012, 95, 53–63. [Google Scholar] [CrossRef]
- Kelly, B.; Dempson, J.B.; Power, M. The effects of preservation on fish tissue stable isotope signatures. J. Fish Biol. 2006, 69, 1595–1611. [Google Scholar] [CrossRef]
- Hobson, K.A.; Gloutney, M.L.; Gibbs, H.L. Preservation of blood and tissue samples for stable-carbon and stable-nitrogen isotope analysis. Can. J. Zool. 1997, 75, 1720–1723. [Google Scholar] [CrossRef]
- Walsh, R.G.; He, S.; Yarnes, C.T. Compound-specific δ13C and δ15N analysis of amino acids: A rapid, chloroformate-based method for ecological studies. Rapid Commun. Mass Spectrom. 2014, 28, 96–108. [Google Scholar] [CrossRef] [PubMed]
- McCullagh, J.S.O.; Juchelka, D.; Hedges, R.E.M. Analysis of amino acid 13C abundance from human and faunal bone collagen using liquid chromatography/isotope ratio mass spectrometry. Rapid Commun. Mass Spectrom. 2006, 20, 2761–2768. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Lees, M.; Stanley, G.H.S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 497–509. [Google Scholar]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Sotiropoulos, M.A.; Tonn, W.M.; Wassenaar, L.I. Effects of lipid extraction on stable carbon and nitrogen isotope analyses of fish tissues: Potential consequences for food web studies. Ecol. Freshw. Fish 2004, 13, 155–160. [Google Scholar] [CrossRef]
- Silfer, J.A.; Engel, M.H.; Macko, S.A.; Jumeau, E.J. Stable carbon isotope analysis of amino acid enantiomers by conventional isotope ratio mass spectrometry and combined gas chromatography/isotope ratio mass spectrometry. Anal. Chem. 1991, 63, 370–374. [Google Scholar] [CrossRef]
- Amelung, W.; Zhang, X. Determination of amino acid enantiomers in soils. Soil Biol. Biochem. 2001, 33, 553–562. [Google Scholar] [CrossRef]
- McMahon, K.W.; Fogel, M.L.; Johnson, B.J.; Houghton, L.A.; Thorrold, S.R. A new method to reconstruct fish diet and movement patterns from δ13C values in otolith amino acids. Can. J. Fish. Aquat. Sci. 2011, 68, 1330–1340. [Google Scholar] [CrossRef]
- Metges, C.C.; Petzke, K.J.; Hennig, U. Gas chromatography/combustion/isotope ratio mass spectrometric comparison of N-acetyl- and N-pivaloyl amino acid esters to measure 15N isotopic abundances in physiological samples: A pilot study on amino acid synthesis in the upper gastro-intestinal tract of minipigs. J. Mass Spectrom. 1996, 31, 367–376. [Google Scholar] [PubMed]
- Chikaraishi, Y.; Kashiyama, Y.; Ogawa, N.O.; Kitazato, H.; Ohkouchi, N. Metabolic control of nitrogen isotope composition of amino acids in macroalgae and gastropods: Implications for aquatic food web studies. Mar. Ecol. Prog. Ser. 2007, 342, 85–90. [Google Scholar] [CrossRef]
- Hušek, P. Rapid derivatization and gas chromatographic determination of amino acids. J. Chromatogr. A 1991, 552, 289–299. [Google Scholar] [CrossRef]
- O’Brien, D.M.; Fogel, M.L.; Boggs, C.L. Renewable and nonrenewable resources: Amino acid turnover and allocation to reproduction in Lepidoptera. Proc. Natl. Acad. Sci. USA 2002, 99, 4413–4418. [Google Scholar] [CrossRef] [PubMed]
- Yarnes, C.T.; Herszage, J. The relative influence of derivatization and normalization procedures on the compound-specific stable isotope analysis of nitrogen in amino acids. Rapid Commun. Mass Spectrom. 2017, 31, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Chikaraishi, Y.; Steffan, S.A.; Takano, Y.; Ohkouchi, N. Diet quality influences isotopic discrimination among amino acids in an aquatic vertebrate. Ecol. Evol. 2015, 5, 2048–2059. [Google Scholar] [CrossRef]
- McMahon, K.W.; Thorrold, S.R.; Elsdon, T.S.; McCarthy, M.D. Trophic discrimination of nitrogen stable isotopes in amino acids varies with diet quality in a marine fish: Trophic discrimination of amino acids. Limnol. Oceanogr. 2015, 60, 1076–1087. [Google Scholar] [CrossRef]
- Howland, M.R.; Corr, L.T.; Young, S.M.M.; Jones, V.; Jim, S.; Van Der Merwe, N.J.; Mitchell, A.D.; Evershed, R.P. Expression of the dietary isotope signal in the compound-specific δ13C values of pig bone lipids and amino acids. Int. J. Osteoarchaeol. 2003, 13, 54–65. [Google Scholar] [CrossRef]
- Jim, S.; Jones, V.; Ambrose, S.H.; Evershed, R.P. Quantifying dietary macronutrient sources of carbon for bone collagen biosynthesis using natural abundance stable carbon isotope analysis. Br. J. Nutr. 2006, 95, 1055–1062. [Google Scholar] [CrossRef] [PubMed]
- Tarroux, A.; Ehrich, D.; Lecomte, N.; Jardine, T.D.; Bety, J.; Berteaux, D. Sensitivity of stable isotope mixing models to variation in isotopic ratios: Evaluating consequences of lipid extraction. Methods Ecol. Evol. 2010, 1, 231–241. [Google Scholar] [CrossRef]
- Ayayee, P.A.; Jones, S.C.; Sabree, Z.L. Can 13C stable isotope analysis uncover essential amino acid provisioning by termite-associated gut microbes? PeerJ 2015, 3, e1218. [Google Scholar] [CrossRef] [PubMed]
- Ayayee, P.A.; Larsen, T.; Sabree, Z. Symbiotic essential amino acids provisioning in the American cockroach, Periplaneta americana (Linnaeus) under various dietary conditions. PeerJ 2016, 4, e2046. [Google Scholar] [CrossRef] [PubMed]
- Larsen, T.; Ventura, M.; Maraldo, K.; Triadó-Margarit, X.; Casamayor, E.O.; Wang, Y.V.; Andersen, N.; O’Brien, D.M. The dominant detritus-feeding invertebrate in Arctic peat soils derives its essential amino acids from gut symbionts. J. Anim. Ecol. 2016, 85, 1275–1285. [Google Scholar] [CrossRef] [PubMed]
- Newsome, S.D.; Feeser, K.L.; Bradley, C.J.; Wolf, C.; Takacs-Vesbach, C.; Fogel, M.L. Quantifying the role of the gut microbiome in host essential amino acid metabolism. ISME J. in review.
- Metges, C.C. Contribution of microbial amino acids to amino acid homeostasis of the host. J. Nutr. 2000, 130, 1857S–1864S. [Google Scholar] [CrossRef]
- Wu, G. Intestinal mucosal amino acid catabolism. J. Nutr. 1998, 128, 4. [Google Scholar] [CrossRef]
- Whiteman, J.P.; Kim, S.L.; McMahon, K.W.; Koch, P.L.; Newsome, S.D. Amino acid isotope discrimination factors for a carnivore: Physiological insights from leopard sharks and their diet. Oecologia 2018, 188, 977–989. [Google Scholar] [CrossRef]
- Nuche-Pascual, M.T.; Lazo, J.P.; Ruiz-Cooley, R.I.; Herzka, S.Z. Amino acid-specific δ5N trophic enrichment factors in fish fed with formulated diets varying in protein quantity and quality. Ecol. Evol. 2018, 8, 9192–9217. [Google Scholar] [CrossRef]
- Nielsen, J.M.; Popp, B.N.; Winder, M. Meta-analysis of amino acid stable nitrogen isotope ratios for estimating trophic position in marine organisms. Oecologia 2015, 178, 631–642. [Google Scholar] [CrossRef] [PubMed]
- Randall, D.J.; Tsui, T.K.N. Ammonia toxicity in fish. Mar. Pollut. Bull. 2002, 45, 17–23. [Google Scholar] [CrossRef]
- Germain, L.R.; Koch, P.L.; Harvey, J.; McCarthy, M.D. Nitrogen isotope fractionation in amino acids from harbor seals: Implications for compound-specific trophic position calculations. Mar. Ecol. Prog. Series 2013, 482, 265–277. [Google Scholar] [CrossRef]
- Barreto-Curiel, F.; Focken, U.; D’Abramo, L.R.; Viana, M.T. Metabolism of Seriola lalandi during starvation as revealed by fatty acid analysis and compound-specific analysis of stable isotopes within amino acids. PLoS ONE 2017, 12, e0170124. [Google Scholar] [CrossRef] [PubMed]
- Steffan, S.A.; Chikaraishi, Y.; Currie, C.R.; Horn, H.; Gaines-Day, H.R.; Pauli, J.N.; Zalapa, J.E.; Ohkouchi, N. Microbes are trophic analogs of animals. PNAS 2015, 112, 15119–15124. [Google Scholar] [CrossRef]
- McClelland, J.W.; Montoya, J.P. Trophic relationships and the nitrogen isotopic composition of amino acids in plankton. Ecology 2002, 83, 2173. [Google Scholar] [CrossRef]
- Steffan, S.A.; Chikaraishi, Y.; Horton, D.R.; Ohkouchi, N.; Singleton, M.E.; Miliczky, E.; Hogg, D.B.; Jones, V.P. Trophic hierarchies illuminated via amino acid isotopic analysis. PLoS ONE 2013, 8, e76152. [Google Scholar] [CrossRef] [PubMed]
- Chikaraishi, Y.; Ogawa, N.O.; Ohkouchi, N. Further evaluation of the trophic level estimation based on nitrogen isotopic composition of amino acids. In Earth, Life, and Isotopes; Kyoto University Press: Kyoto, Janpan, 2010; Volume 415, pp. 37–51. [Google Scholar]
- Dale, J.; Wallsgrove, N.; Popp, B.; Holland, K. Nursery habitat use and foraging ecology of the brown stingray Dasyatis lata determined from stomach contents, bulk and amino acid stable isotopes. Mar. Ecol. Prog. Series 2011, 433, 221–236. [Google Scholar] [CrossRef]
- Seminoff, J.A.; Benson, S.R.; Arthur, K.E.; Eguchi, T.; Dutton, P.H.; Tapilatu, R.F.; Popp, B.N. Stable isotope tracking of endangered sea turtles: Validation with satellite telemetry and δ15N analysis of amino acids. PLoS ONE 2012, 7, e37403. [Google Scholar] [CrossRef]
- Ruiz-Cooley, R.I.; Koch, P.L.; Fiedler, P.C.; McCarthy, M.D. Carbon and nitrogen isotopes from top predator amino acids reveal rapidly shifting ocean biochemistry in the outer California Current. PLoS ONE 2014, 9, e110355. [Google Scholar] [CrossRef]
- Scott, J.H.; O’Brien, D.M.; Emerson, D.; Sun, H.; McDonald, G.D.; Salgado, A.; Fogel, M.L. An examination of the carbon isotope effects associated with amino acid biosynthesis. Astrobiology 2006, 6, 867–880. [Google Scholar] [CrossRef] [PubMed]
- Larsen, T.; Taylor, D.L.; Leigh, M.B.; O’Brien, D.M. Stable isotope fingerprinting: A novel method for identifying plant, fungal, or bacterial origins of amino acids. Ecology 2009, 90, 3526–3535. [Google Scholar] [CrossRef]
- Larsen, T.; Ventura, M.; Andersen, N.; O’Brien, D.M.; Piatkowski, U.; McCarthy, M.D. Tracing carbon sources through aquatic and terrestrial food webs using amino acid stable isotope fingerprinting. PLoS ONE 2013, 8, e73441. [Google Scholar] [CrossRef] [PubMed]
- Elliott Smith, E.A.; Harrod, C.; Newsome, S.D. The importance of kelp to an intertidal ecosystem varies by trophic level: Insights from amino acid δ13C analysis. Ecosphere 2018, 9, e02516. [Google Scholar] [CrossRef]
- Larsen, T.; Wooller, M.J.; Fogel, M.L.; O’Brien, D.M. Can amino acid carbon isotope ratios distinguish primary producers in a mangrove ecosystem? Rapid Commun. Mass Spectrom. 2012, 26, 1541–1548. [Google Scholar] [CrossRef]
- Larsen, T.; Bach, L.T.; Salvatteci, R.; Wang, Y.V.; Andersen, N.; Ventura, M.; McCarthy, M.D. Assessing the potential of amino acid 13C patterns as a carbon source tracer in marine sediments: Effects of algal growth conditions and sedimentary diagenesis. Biogeosciences 2015, 12, 4979–4992. [Google Scholar] [CrossRef]
- McMahon, K.W.; Thorrold, S.R.; Houghton, L.A.; Berumen, M.L. Tracing carbon flow through coral reef food webs using a compound-specific stable isotope approach. Oecologia 2016, 180, 809–821. [Google Scholar] [CrossRef]
- Thorp, J.H.C.; Bowes, R.E. Carbon sources in riverine food webs: New evidence from amino acid isotope techniques. Ecosystems 2016, 20, 1029–1041. [Google Scholar] [CrossRef]
- Larsen, T.; Pollierer, M.M.; Holmstrup, M.; D’Annibale, A.; Maraldo, K.; Andersen, N.; Eriksen, J. Substantial nutritional contribution of bacterial amino acids to earthworms and enchytraeids: A case study from organic grasslands. Soil Biol. Biochem. 2016, 99, 21–27. [Google Scholar] [CrossRef]
- Bradley, C.J.; Wallsgrove, N.J.; Choy, C.A.; Drazen, J.C.; Hetherington, E.D.; Hoen, D.K.; Popp, B.N. Trophic position estimates of marine teleosts using amino acid compound specific isotopic analysis. Limnol. Oceanogr. Methods 2015, 13, 476–493. [Google Scholar] [CrossRef]
- Tieszen, L.L.; Boutton, T.W.; Tesdahl, K.G.; Slade, N.A. Fractionation and turnover of stable carbon isotopes in animal tissues: Implications for 13C analysis of diet. Oecologia 1983, 57, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Cerling, T.E.; Ayliffe, L.K.; Dearing, M.D.; Ehleringer, J.R.; Passey, B.H.; Podlesak, D.W.; Torregrossa, A.-M.; West, A.G. Determining biological tissue turnover using stable isotopes: The reaction progress variable. Oecologia 2007, 151, 175–189. [Google Scholar] [CrossRef] [PubMed]
- Bradley, C.J.; Madigan, D.J.; Block, B.A.; Popp, B.N. Amino acid isotope incorporation and enrichment factors in Pacific Bluefin Tuna, Thunnus orientalis. PLoS ONE 2014, 9, e85818. [Google Scholar] [CrossRef] [PubMed]
- Downs, E.; Popp, B.; Holl, C. Nitrogen isotope fractionation and amino acid turnover rates in the Pacific white shrimp Litopenaeus vannamei. Mar. Ecol. Prog. Ser. 2014, 516, 239–250. [Google Scholar] [CrossRef]
- Fogel, M.L.; Griffin, P.L.; Newsome, S.D. Hydrogen isotopes in individual amino acids reflect differentiated pools of hydrogen from food and water in Escherichia coli. PNAS 2016, 113, E4648–E4653. [Google Scholar] [CrossRef] [PubMed]
- Fry, B.; Carter, J.F.; Yamada, K.; Yoshida, N.; Juchelka, D. Position-specific 13C/12C analysis of amino acid carboxyl groups—Automated flow-injection analysis based on reaction with ninhydrin. Rapid Commun. Mass Spectrom. 2018, 32, 992–1000. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.L.; Cox, M.M. Lehninger Principles of Biochemistry, 5th ed.; W. H. Freeman: New York, NY, USA, 2008; ISBN 978-0-7167-7108-1. [Google Scholar]
- Gannes, L.Z.; O’Brien, D.M.; Martinez del Rio, C.M. Stable isotopes in animal ecology: Assumptions, caveats, and a call for more laboratory experiments. Ecology 1997, 78, 6. [Google Scholar] [CrossRef]
| 13C Classification | 15N Classification | ||
|---|---|---|---|
| Trophic AA | Source AA | Other AA | |
| AAESS | Isoleucine Leucine Valine | Lysine Methionine Phenylalanine Tyrosine 1 | Arginine 2 Threonine 3 |
| AANESS | Aspartic acid Alanine Glutamic acid Proline | Glycine 4 Serine 5 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Whiteman, J.P.; Elliott Smith, E.A.; Besser, A.C.; Newsome, S.D. A Guide to Using Compound-Specific Stable Isotope Analysis to Study the Fates of Molecules in Organisms and Ecosystems. Diversity 2019, 11, 8. https://doi.org/10.3390/d11010008
Whiteman JP, Elliott Smith EA, Besser AC, Newsome SD. A Guide to Using Compound-Specific Stable Isotope Analysis to Study the Fates of Molecules in Organisms and Ecosystems. Diversity. 2019; 11(1):8. https://doi.org/10.3390/d11010008
Chicago/Turabian StyleWhiteman, John P., Emma A. Elliott Smith, Alexi C. Besser, and Seth D. Newsome. 2019. "A Guide to Using Compound-Specific Stable Isotope Analysis to Study the Fates of Molecules in Organisms and Ecosystems" Diversity 11, no. 1: 8. https://doi.org/10.3390/d11010008
APA StyleWhiteman, J. P., Elliott Smith, E. A., Besser, A. C., & Newsome, S. D. (2019). A Guide to Using Compound-Specific Stable Isotope Analysis to Study the Fates of Molecules in Organisms and Ecosystems. Diversity, 11(1), 8. https://doi.org/10.3390/d11010008





