Influence of a Large Lake on the Winter Range of a Small Mammal: Lake Michigan and the Silver-Haired Bat (Lasionycteris noctivagans)
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. Testing Izor’s Hypothesis
2.3. Modeling
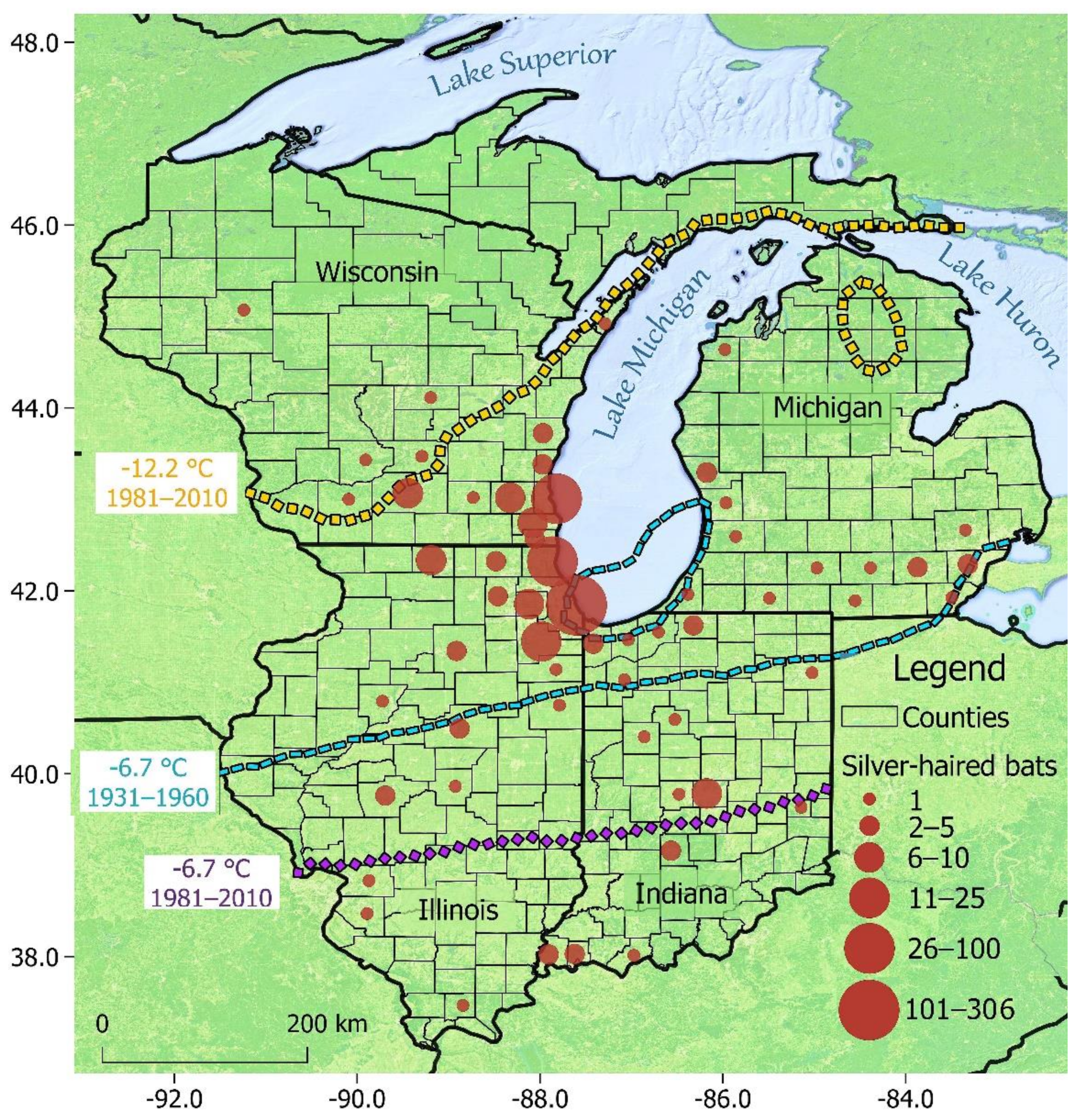
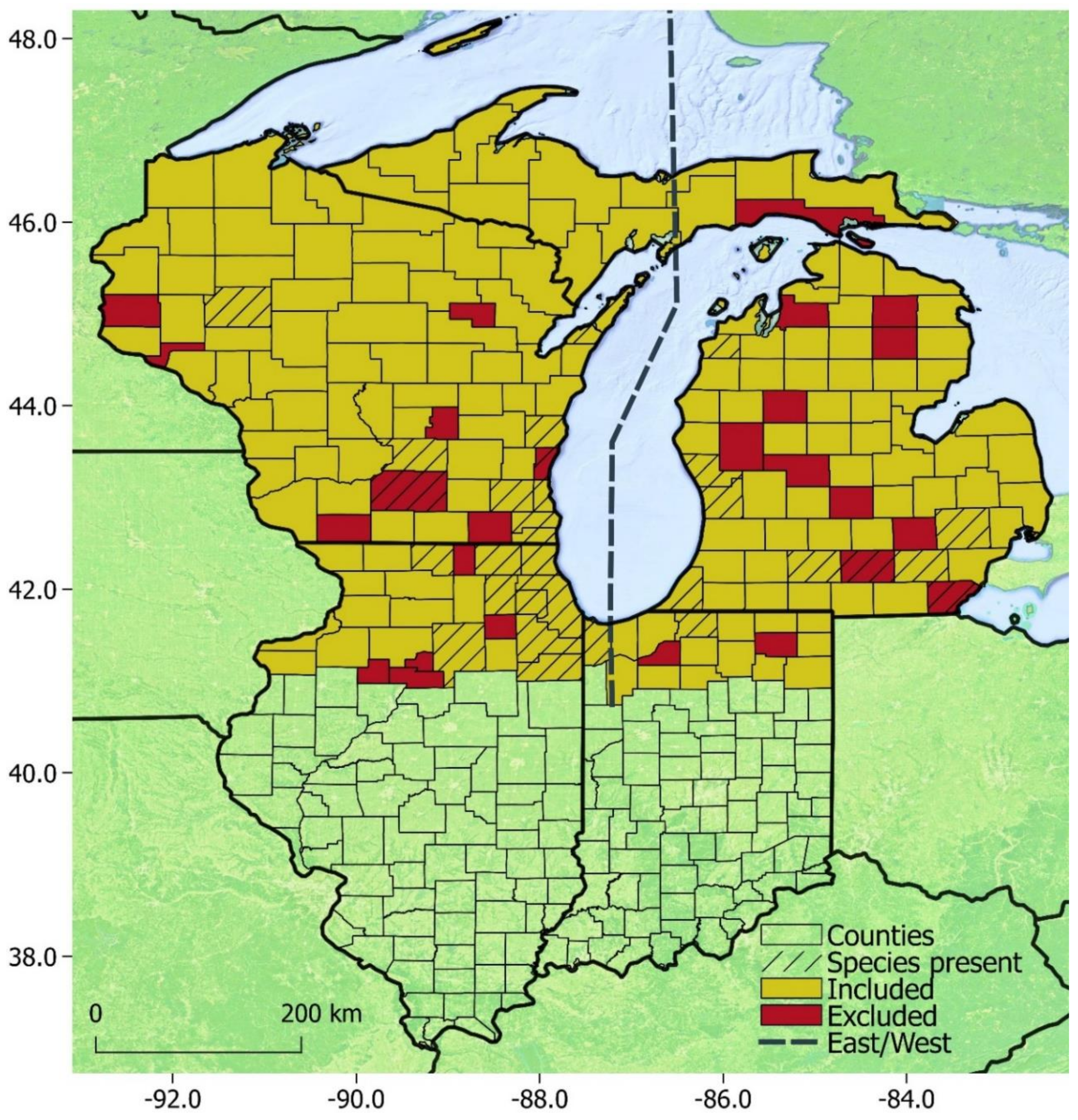
3. Results
3.1. Overall Patterns
3.2. Testing Izor’s Hypothesis
3.3. Modeling
4. Discussion
4.1. Overall Patterns
4.2. Testing Izor’s Hypothesis
4.3. Modeling
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cahill, A.E.; Aiello-Lammens, M.E.; Fisher-Reid, M.C.; Hua, X.; Karanewsky, C.J.; Ryu, H.Y.; Sbeglia, G.C.; Spagnolo, F.; Waldrom, J.B.; Wiens, J.J. Causes of warm-edge range limits: Systematic review, proximate factors and implications for climate change. J. Biogeogr. 2013, 41, 429–442. [Google Scholar] [CrossRef]
- Gaston, K.J. Geographic range limits: Achieving synthesis. Proc. R. Soc. B 2009, 276, 1395–1406. [Google Scholar] [CrossRef] [PubMed]
- Sexton, J.P.; McIntyre, P.J.; Angert, A.L.; Rice, K.J. Evolution and ecology of species range limits. Ann. Rev. Ecol. Syst. 2009, 40, 413–436. [Google Scholar] [CrossRef]
- Martin, T.G.; Chades, I.; Arcese, P.; Marra, P.P.; Possingham, H.P.; Norris, D.R. Optimal conservation of migratory species. PLoS ONE 2007, 2, e751. [Google Scholar] [CrossRef] [PubMed]
- McGuire, L.P.; Boyle, W.A. Altitudinal migration in bats: Evidence, patterns, and drivers. Biol. Rev. 2013, 8, 767–786. [Google Scholar] [CrossRef]
- Fleming, T.H.; Eby, P. Ecology of bat migration. In Bat Ecology; Kunz, T.H., Fenton, M.B., Eds.; University of Chicago Press: Chicago, IL, USA, 2003; pp. 156–208. ISBN 9780226462073. [Google Scholar]
- Cryan, P.M.; Bogan, M.A.; Rye, R.O.; Landis, G.P.; Kester, C.L. Stable hydrogen isotope analysis of bat hair as evidence of seasonal molt and long-distance migration. J. Mammal. 2004, 86, 995–1001. [Google Scholar] [CrossRef]
- Hutterer, R.; Ivanova, T.; Meyer-Cords, C.; Rodrigue, L. Bat Migrations in Europe: A Review of Banding Data and Literature; Federal Agency for Nature Conservation: Bonn, Germany, 2005; ISBN 9783784339283. [Google Scholar]
- Cryan, P.M. Seasonal distribution of migratory tree bats (Lasiurus and Lasionycteris) in North America. J. Mammal. 2003, 84, 579–593. [Google Scholar] [CrossRef]
- Messenger, S.L.; Smith, J.S.; Orciari, L.A.; Yager, P.A.; Rupprecht, C.E. Emerging pattern of rabies deaths and increased viral infectivity. Emerg. Infect. Dis. 2003, 9, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Hein, C.D.; Schirmacher, M.R. Impact of wind energy on bats: A summary of current knowledge. Hum.-Wildl. Interact. 2016, 10, 19–27. [Google Scholar]
- Fraser, E.; Brooks, D.; Longstaffe, F.J. Stable isotope investigation of the migratory behavior of silver-haired bats (Lasionycteris noctivagans) in eastern North America. J. Mammal. 2017, 98, 1225–1235. [Google Scholar] [CrossRef]
- McGuire, L.P.; Guglielmo, C.C.; Mackenzie, S.A.; Taylor, P.D. Migratory stopover in the long-distance migrant silver-haired bat, Lasionycteris noctivagans. J. Anim. Ecol. 2012, 81, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Jonasson, K.A.; Guglielmo, C.C. Sex differences in spring migration timing and body composition of silver-haired bats Lasionycteris noctivagans. J. Mammal. 2016, 97, 1535–1542. [Google Scholar] [CrossRef]
- Clark, M.K. A communal winter roost of silver-haired bats, Lasionycteris noctivagans. Brimleyana 1993, 19, 137–139. [Google Scholar]
- Kurta, A.; Stewart, M.E. Parturition in the silver-haired bat, Lasioncyteris noctivagans, with a description of the neonates. Can. Field-Nat. 1991, 104, 598–600. [Google Scholar]
- Parsons, H.J.; Smith, D.A.; Whittam, R.F. Maternity roosts of silver-haired bats, Lasionycteris noctivagans, in Ontario and Saskatchewan. J. Mammal. 1986, 67, 598–600. [Google Scholar] [CrossRef]
- Kurta, A. Mammals of the Great Lakes Region, 3rd ed.; University of Michigan Press: Ann Arbor, MI, USA, 2017; ISBN 978-0-472-05345-2. [Google Scholar]
- Archibald, K.M.; Buler, J.J.; Smolinsky, J.A.; Smith, R.J. Migrating birds reorient toward land at dawn over the Great Lakes, USA. Auk 2017, 134, 193–201. [Google Scholar] [CrossRef]
- Byre, V.J. A group of young peregrine falcons prey on migrating bats. Wilson Bull. 1990, 102, 728–730. [Google Scholar]
- Whitaker, J.O., Jr.; Mumford, R.E. Mammals of Indiana; Indiana Univ. Press: Bloomington, IN, USA, 2009; ISBN 978-0-253-34971-2. [Google Scholar]
- Feldhamer, G.A.; Hofmann, J.E.; Carter, T.C.; Kath, J.A. Bats of Illinois; Center for Bat Research, Outreach, and Conservation: Indiana State University, Terre Haute, IN, USA, 2015. [Google Scholar]
- Hofmann, J.E.; Merritt, J.F.; Mengelkoch, J.M.; Carpenter, S.K. A two-year mist-netting survey for bats in Cook, DuPage, Kane, Kankakee, Lake, McHenry, and Will counties in northeastern Illinois. Ill. Nat. Hist. Sur. Tech. Rep. 2008, 5, 1–41. [Google Scholar]
- Kurta, A. Reproductive timing, distribution, and sex ratios of tree bats in Lower Michigan. J. Mammal. 2010, 91, 586–592. [Google Scholar] [CrossRef]
- Naughton, D. The Natural History of Canadian Mammals; University Toronto Press: Toronto, ON, Canada, 2012; ISBN 9781442644830. [Google Scholar]
- Perry, R.W.; Saugey, D.A.; Crump, B.G. Winter roosting ecology of silver-haired bats in an Arkansas forest. Southeast. Nat. 2010, 9, 563–572. [Google Scholar] [CrossRef]
- Izor, R.J. Winter range of the silver-haired bat. J. Mammal. 1979, 60, 641–643. [Google Scholar] [CrossRef]
- McNab, B.K. The Physiological Ecology of Vertebrates. A View from Energetics; Comstock Publishing Associates: Ithaca, New York, NY, USA, 2002; ISBN 978-0801439131. [Google Scholar]
- Keen, R.A. Michigan Weather; American & World Geographic Publishers: Helena, MT, USA, 1993; ISBN 978-0472034130. [Google Scholar]
- VertNet. Search VertNet. Available online: http://portal.vertnet.org/search (accessed on 5 April 2018).
- Gosling, N. Winter record of the silver-haired bat, Lasionycteris noctivagans Le Conte, in Michigan. J. Mamm. 1977, 58, 657–660. [Google Scholar] [CrossRef]
- Sherwood, D.E.; Kurta, A. Winter record of the silver-haired bat from Wayne Co. Mich. Birds Nat. Hist. 1999, 6, 153–155. [Google Scholar]
- Wisconsin Department of Natural Resources. Wildlife Rehabilitation Annual Report. Available online: http://dnr.wi.gov/files/PDF/forms/2300/2300-302.pdf (accessed on 1 March 2018).
- U. S. Geological Survey. The National Atlas of the United States of America; U.S. Government Printing Office: Washington, DC, USA, 1970. Available online: https://www.loc.gov/resource/g3701gm.gct00013/?sp=2&st=gallery (accessed on 1 March 2018).
- National Centers for Environmental Information. 1981–2010 Climate Normals. Available online: https://www.ncdc.noaa.gov/data-access/land-based-station-data/land-based-datasets/climate-normals/1981-2010-normals-data (accessed on 1 March 2018).
- National Centers for Environmental Information. U.S. Climate Atlas. Available online: https://www.ncdc.noaa.gov/climateatlas/ (accessed on 1 March 2018).
- Arguez, A.; Durre, I.; Applequist, S.; Vose, R.S.; Squires, M.F.; Yin, X.; Heim, R.R., Jr.; Owen, T.W. NOAA’s 1981–2010 U.S. Climate Normals: An Overview. Bull. Am. Meteorol. Soc. 2012, 93, 1687–1697. [Google Scholar] [CrossRef]
- Hawkins, E.; Sutton, R. Connecting climate model projections of global temperature change with the real world. Bull. Am. Meterol. Soc. 2016, 97, 963–980. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. Available online: https://www.r-project.org/ (accessed on 1 March 2018).
- Patyk, K.; Turmelle, A.; Blanton, J.D.; Rupprecht, C.E. Trends in national surveillance data for bat rabies in the United States: 2001–2009. Vector-Borne Zoon. Dis. 2012, 12, 666–673. [Google Scholar] [CrossRef] [PubMed]
- U.S. Census Bureau. U.S. Gazetteer Files. Available online: https://www.census.gov/geo/maps-data/data/gazetteer2016.html (accessed on 1 March 2018).
- National Centers for Environmental Information. Data Tools: 1981–2010 Normals. Available online: https://www.ncdc.noaa.gov/cdo-web/datatools/normals (accessed on 1 March 2018).
- U.S. Census Bureau. County Population Totals Datasets: 2010–2016. Available online: https://www.census.gov/data/datasets/2016/demo/popest/counties-total.html (accessed on 1 March 2018).
- Homer, C.G.; Dewitz, J.A.; Yang, L.; Jin, S.; Danielson, P.; Xian, G.; Coulston, J.; Herold, N.D.; Wickham, J.D.; Megown, K. Completion of the 2011 National Land Cover Database for the conterminous United States––Representing a decade of land cover change information. Photogramm. Eng. Remote Sens. 2015, 81, 345–354. [Google Scholar] [CrossRef]
- Jung, M. LecoS—A python plugin for automated landscape ecology analysis. Ecol. Inf. 2016, 31, 18–21. [Google Scholar] [CrossRef]
- QGIS Development Team. QGIS Geographic Information System. Open Source Geospatial Foundation Project. Avaliable online: http://qgis.osgeo.org (accessed on 1 March 2018).
- George, K.G. (U.S.G.S. National Wildlife Health Center, Madison, WI, USA). Personal communication, 2018.
- Chatterjee, S.; Price, B. Regression Analysis by Example; John Wiley and Sons: New York, NY, USA, 1977; ISBN 0471015210. [Google Scholar]
- Durbin, J.; Watson, G.S. Testing for serial correlation in least squares regression, I. Biometrika 1950, 37, 409–428. [Google Scholar] [CrossRef] [PubMed]
- Pregibon, D. Logistic regression diagnostics. Ann. Stat. 1981, 9, 705–724. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference: A Practical Information-theoretic Approach, 2nd ed.; Springer: New York, NY, USA, 2002; ISBN 0-387-95364-7. [Google Scholar]
- Akaike, H. A new look at the statistical model identification. IEEE Trans. Autom. Control 1974, 19, 716–723. [Google Scholar] [CrossRef]
- Osius, G.; Rojek, D. Normal goodness-of-fit tests for multinomial models with large degrees of freedom. J. Am. Stat. Assoc. 1992, 87, 1145–1152. [Google Scholar] [CrossRef]
- Roberts, D.R.; Bahn, V.; Ciuti, S.; Boyce, M.S.; Elith, J.; Guillera-Arroita, G.; Hauenstein, S.; Lahoz-Monfort, J.J.; Schrӧder, B.; Thuiller, W.; et al. Cross-validation strategies for data with temporal, spatial, hierarchical, or phylogenetic structure. Ecography 2017, 40, 913–929. [Google Scholar] [CrossRef]
- Hosmer, D.W.; Lemeshow, S. Applied Logistic Regression, 2nd ed.; John Wiley & Sons: New York, NY, USA, 2005; ISBN 9780471356325. [Google Scholar]
- Metz, C.E. Basic principles of ROC Analysis. Semin. Nucl. Med. 1978, 8, 283–289. [Google Scholar] [CrossRef]
- Cook, R.D.; Weisberg, S. Residuals and Influence in Regression; Chapman and Hall: New York, NY, USA, 1982; ISBN 0-412-24280-0. [Google Scholar]
- Streicker, D.G.; Turmelle, A.S.; Vonhof, M.J.; Kuzmin, I.V.; McCracken, G.F.; Rupprecht, C.E. Host phylogeny constrains cross-species emergence and establishment of rabies virus in bats. Science 2010, 329, 676–679. [Google Scholar] [CrossRef] [PubMed]
- Pearson, E.W. Bats hibernating in silica mines in southern Illinois. J. Mammal. 1962, 43, 27–33. [Google Scholar] [CrossRef]
- Kurta, A.; Smith, S.M.; DePue, J.E. A new northern record for a hibernating silver-haired bat (Lasionycteris noctivagans) in interior North America. Bat Res. News 2017, 58, 23–24. [Google Scholar]
- Dorr, J.A., Jr.; Eschman, D.F. Geology of Michigan; University of Michigan Press: Ann Arbor, MI, USA, 1970; ISBN 9780472082803. [Google Scholar]
- Dott, R.H.; Attig, J.W. Roadside Geology of Wisconsin; Mountain Press Publishing Company: Missoula, MT, USA, 2004; ISBN 9780878424924. [Google Scholar]
- Hall, R.D. Geology of Indiana; Kendall Hunt Publishing: Dubuque, IA, USA, 1989; ISBN 9780840356369. [Google Scholar]
- Kolata, D.R.; Nimz, C.K. Geology of Illinois; University of Illinois at Urbana-Champaign: Champaign, IL, USA, 2010; ISBN 9780615417394. [Google Scholar]
- Eichenlaub, V.L. Weather and Climate of the Great Lakes Region; University of Notre Dame Press: Notre Dame, IN, USA, 1979; ISBN 9780268019297. [Google Scholar]
- Sluiter, J.W.; Voûte, A.M.; van Heerdt, P.F. Hibernation of Nyctalus noctula. Period. Biol. 1973, 75, 181–188. [Google Scholar]
- Muul, I. Behavioral and Physiological Influences on the Distribution of the Flying Squirrel, Glaucomys volans; Miscellaneous Publications Museum of Zoology, University of Michigan: Ann Arbor, MI, USA, 1968; Volume 134, pp. 1–66. [Google Scholar]
- Voigt, C.C.; Phelps, K.L.; Aguirre, L.F.; Schoeman, M.C.; Vanitharani, J.; Zubaid, A. Bats and buildings: The conservation of synanthropic bats. In Bats in the Anthropocene: Conservation of Bats in a Changing World; Voigt, C.C., Kingston, T., Eds.; Springer: New York, NY, USA, 2016; pp. 427–462. ISBN 9783319252209. [Google Scholar]
- Halsall, A.L.; Boyles, J.G.; Whitaker, J.O., Jr. Body temperature patterns of big brown bats during winter in a building hibernaculum. J. Mammal. 2012, 93, 497–503. [Google Scholar] [CrossRef]
- Whitaker, J.O., Jr.; Gummer, S.L. Hibernation of the big brown bat, Eptesicus fuscus, in buildings. J. Mamm. 1992, 73, 312–316. [Google Scholar] [CrossRef]
- Brack, V., Jr.; Carter, J.C. Use of an underground burrow by Lasionycteris. Bat Res. News 1985, 26, 28–29. [Google Scholar]
- Kurta, A.; Slider, R. The Bat Community at Pictured Rocks National Lakeshore, with Emphasis on the Northern Long-Eared Bat (Myotis septentrionalis); U.S. National Park Service: Pictured Rocks National Lakeshore, Munising, MI, USA, 2016. [Google Scholar]
- Kurta, A.; Schaetz, B.; Slider, R. A Mist-Netting Survey for the Northern Long-Eared Bat, near White Pine, Ontonagon County, Michigan; White Water and Associates: Amasa, MI, USA, 2015. [Google Scholar]
- McNab, B.K. Energetics and distribution of vampire bats. J. Mammal. 1973, 54, 131–144. [Google Scholar] [CrossRef]
- Fenton, M.B. Distribution and Overwintering of Myotis leibii and Eptesicus fuscus (Chiroptera: Vespertilionidae) in Ontario; Life Sciences Occasional Papers; Royal Ontario Museum: Toronto, ON, Canada, 1972; Volume 21, pp. 1–8. [Google Scholar] [CrossRef]
- Davis, A.D.; Morgan, S.M.D.; Dupuis, M.; Poulliott, C.E.; Davis, J.A.; Franchini, R.; Clobridge, A.; Rudd, R.J. Overwintering of rabies virus in silver-haired bats (Lasionycteris noctivagans). PLoS ONE 2016, 11, e015554. [Google Scholar] [CrossRef] [PubMed]
- Dunbar, M.B.; Brigham, R.M. Thermoregulatory variation among populations of bats along a latitudinal gradient. J. Comp. Physiol. B 2010, 180, 885–893. [Google Scholar] [CrossRef] [PubMed]
- Notaro, M.; Holman, K.; Zarrin, A.; Fluck, E.; Vavrus, S.; Bennington, V. Influence of the Laurentian Great Lakes on regional climate. J. Clim. 2013, 26, 789–804. [Google Scholar] [CrossRef]
- Chagnon, S.A., Jr.; Jones, D.M.A. Review of the influences of the Great Lakes on weather. Water Resour. Res. 1972, 8, 360–371. [Google Scholar] [CrossRef]
- Kurta, A. The misuse of relative humidity in ecological studies of hibernating bats. Acta Chiropterol. 2014, 16, 249–254. [Google Scholar] [CrossRef]
- Ewert, D.N.; Hamas, M.J.; Smith, R.J.; Dallman, M.E.; Jorgensen, S.W. Distribution of migratory landbirds along the northern Lake Huron shoreline. J. Ornithol. 2011, 123, 536–547. [Google Scholar] [CrossRef]
- Kurta, A.; Whitaker, J.O., Jr. Diet of the endangered Indiana bat (Myotis sodalis) on the northern edge of its range. Am. Midl. Nat. 1998, 140, 280–286. [Google Scholar] [CrossRef]
- Münzer, O.M.; Schaetz, B.A.; Kurta, A. Dietary ecology of a Nearctic species, the evening bat (Nycticeius humeralis), at the northern edge of its range. Acta Chiropterol. 2016, 18, 499–508. [Google Scholar] [CrossRef]
- Ijäs, A.; Kahilainen, A.; Vasko, V.V.; Lilley, T.M. Evidence of the migratory bat, Pipistrellus nathusii, aggregating to the coastlines in the northern Baltic Sea. Acta Chiropterol. 2017, 19, 127–139. [Google Scholar] [CrossRef]
- Hatch, S.K. Behavior of Migratory Tree Bats in the Western Basin of Lake Erie Using Telemetry and Stable Isotope Analysis. Master’s Thesis, University of Akron, Akron, OH, USA, 2015. Available online: https://etd.ohiolink.edu/!etd.send_file?accession=akron1449157127&disposition=inline (accessed on 1 March 2018).
- Beebe, R. Influence of the Great Lakes on the migration of birds. Wilson Bull. 1933, 45, 118–121. [Google Scholar]
- Coleman, J.L.; Barclay, R.M.R. Urbanization and the abundance and diversity of prairie bats. Urban Ecosyst. 2012, 15, 87–102. [Google Scholar] [CrossRef]
| Predictor | Frequency or Mean (Range); Median |
|---|---|
| Area of land (km2) | 1805 (626–4717); 1570 |
| Distance to lake (km) | 130.2 (4–386); 117 |
| Latitude (degrees) | 43.70 (41.03–47.59); 43.68 |
| Side of lake | E: 77 counties W: 95 counties |
| Number of bats submitted/county | 198.9 (0–4895); 76.5 |
| Mean daily minimum temperature for January (°C) | −11.6 (−18.4–−5.6); −11.4 |
| Urban index | 0.25 (−0.53–19.93); −0.24 |
| Model | d.f. | Log Likelihood | AICc | Deviance |
|---|---|---|---|---|
| Distance from lake + side of lake + number submitted + urbanization index + temperature | 6 | −39.79 | 92.09 | 79.58 |
| Area + distance from lake + side of lake + number submitted + urbanization index + temperature | 7 | −39.76 | 94.21 | 79.52 |
| Area + latitude + distance from lake + side of lake + number submitted + urbanization index + temperature | 8 | −39.39 | 95.65 | 78.77 |
| Predictor | Estimated Coefficient | Estimated Standard Error | Estimated Odds Ratio |
|---|---|---|---|
| Distance to lake (km) | −0.014 | 0.005 | 0.986 |
| Side of lake (west = 1) | 1.914 | 0.794 | 6.783 |
| Number of bats submitted | 0.004 | 0.001 | 1.004 |
| Urbanization index | 0.425 | 0.225 | 1.529 |
| Temperature (°C) | 0.181 | 0.101 | 1.198 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kurta, A.; Auteri, G.G.; Hofmann, J.E.; Mengelkoch, J.M.; White, J.P.; Whitaker, J.O., Jr.; Cooley, T.; Melotti, J. Influence of a Large Lake on the Winter Range of a Small Mammal: Lake Michigan and the Silver-Haired Bat (Lasionycteris noctivagans). Diversity 2018, 10, 24. https://doi.org/10.3390/d10020024
Kurta A, Auteri GG, Hofmann JE, Mengelkoch JM, White JP, Whitaker JO Jr., Cooley T, Melotti J. Influence of a Large Lake on the Winter Range of a Small Mammal: Lake Michigan and the Silver-Haired Bat (Lasionycteris noctivagans). Diversity. 2018; 10(2):24. https://doi.org/10.3390/d10020024
Chicago/Turabian StyleKurta, Allen, Giorgia G. Auteri, Joyce E. Hofmann, Jean M. Mengelkoch, J. Paul White, John O. Whitaker, Jr., Thomas Cooley, and Julie Melotti. 2018. "Influence of a Large Lake on the Winter Range of a Small Mammal: Lake Michigan and the Silver-Haired Bat (Lasionycteris noctivagans)" Diversity 10, no. 2: 24. https://doi.org/10.3390/d10020024
APA StyleKurta, A., Auteri, G. G., Hofmann, J. E., Mengelkoch, J. M., White, J. P., Whitaker, J. O., Jr., Cooley, T., & Melotti, J. (2018). Influence of a Large Lake on the Winter Range of a Small Mammal: Lake Michigan and the Silver-Haired Bat (Lasionycteris noctivagans). Diversity, 10(2), 24. https://doi.org/10.3390/d10020024




