Training for Translocation: Predator Conditioning Induces Behavioral Plasticity and Physiological Changes in Captive Eastern Hellbenders (Cryptobranchus alleganiensis alleganiensis) (Cryptobranchidae, Amphibia)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Animals
2.2. Phase 1. Predator Conditioning
2.3. Phase 2. Exposure Trials
2.4. Statistical Analyses
2.4.1. Phase 1: Predator Conditioning
2.4.2. Phase 2: Exposure Trials
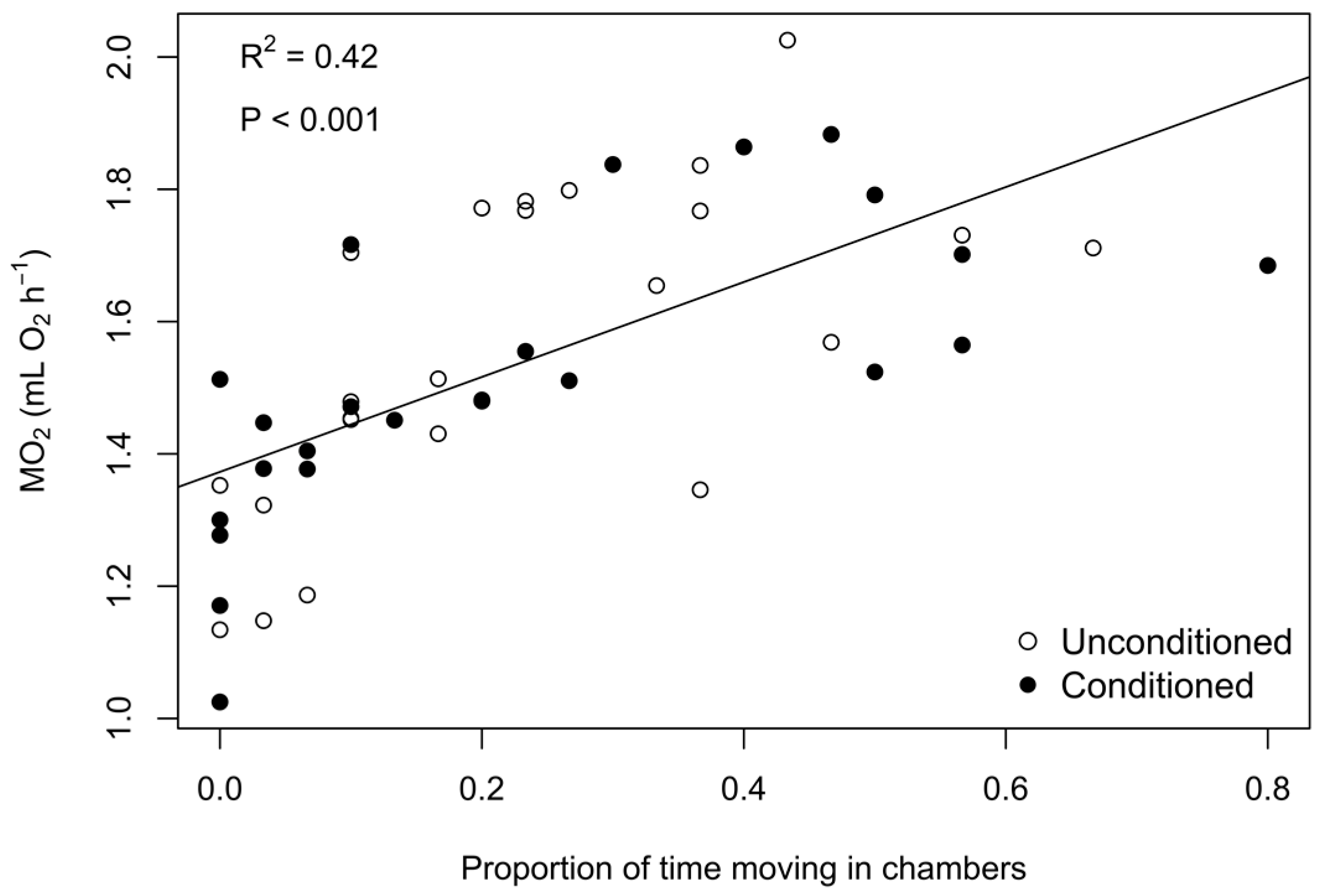
3. Results
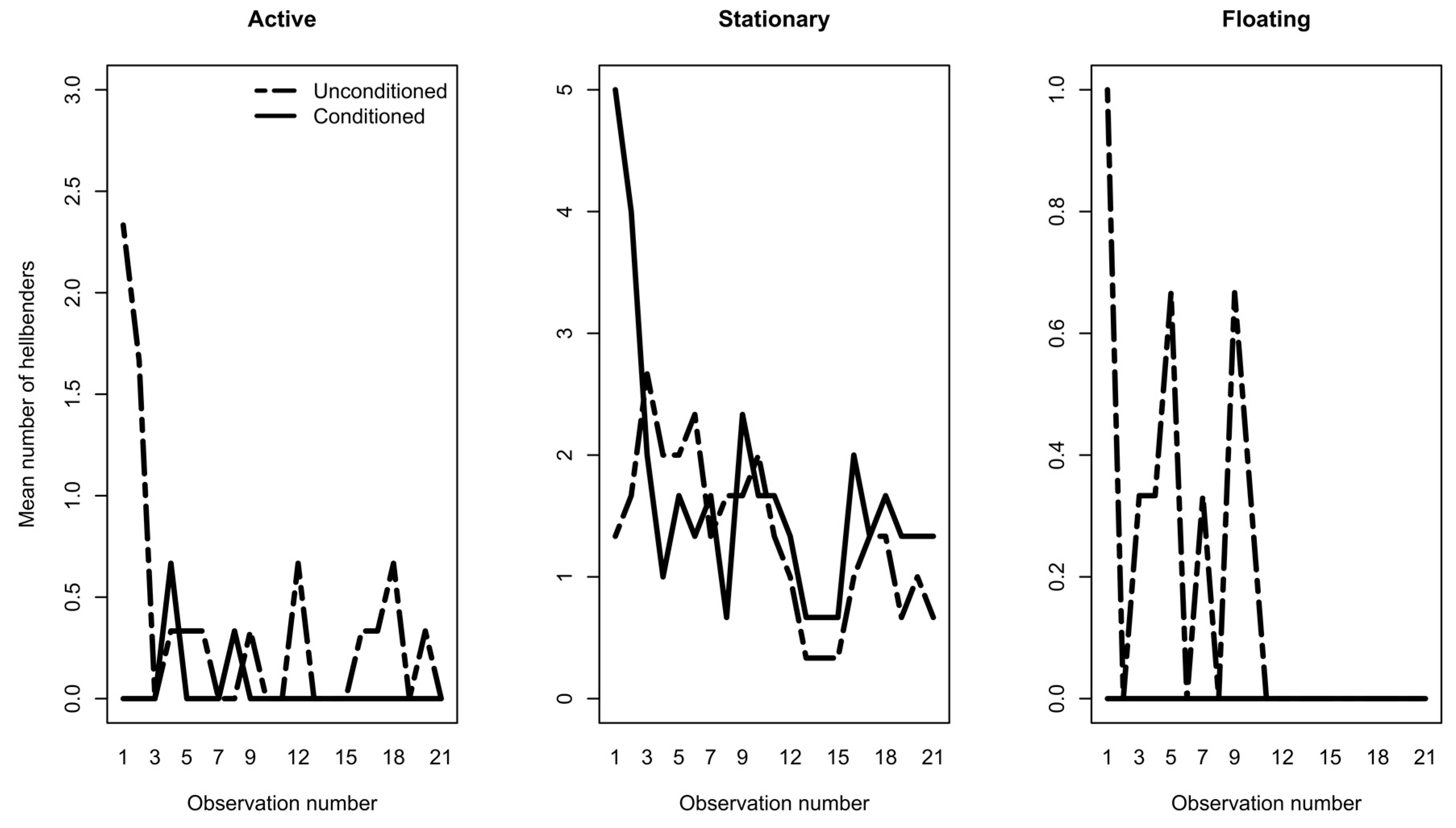
3.1. Phase 1: Predator Conditioning
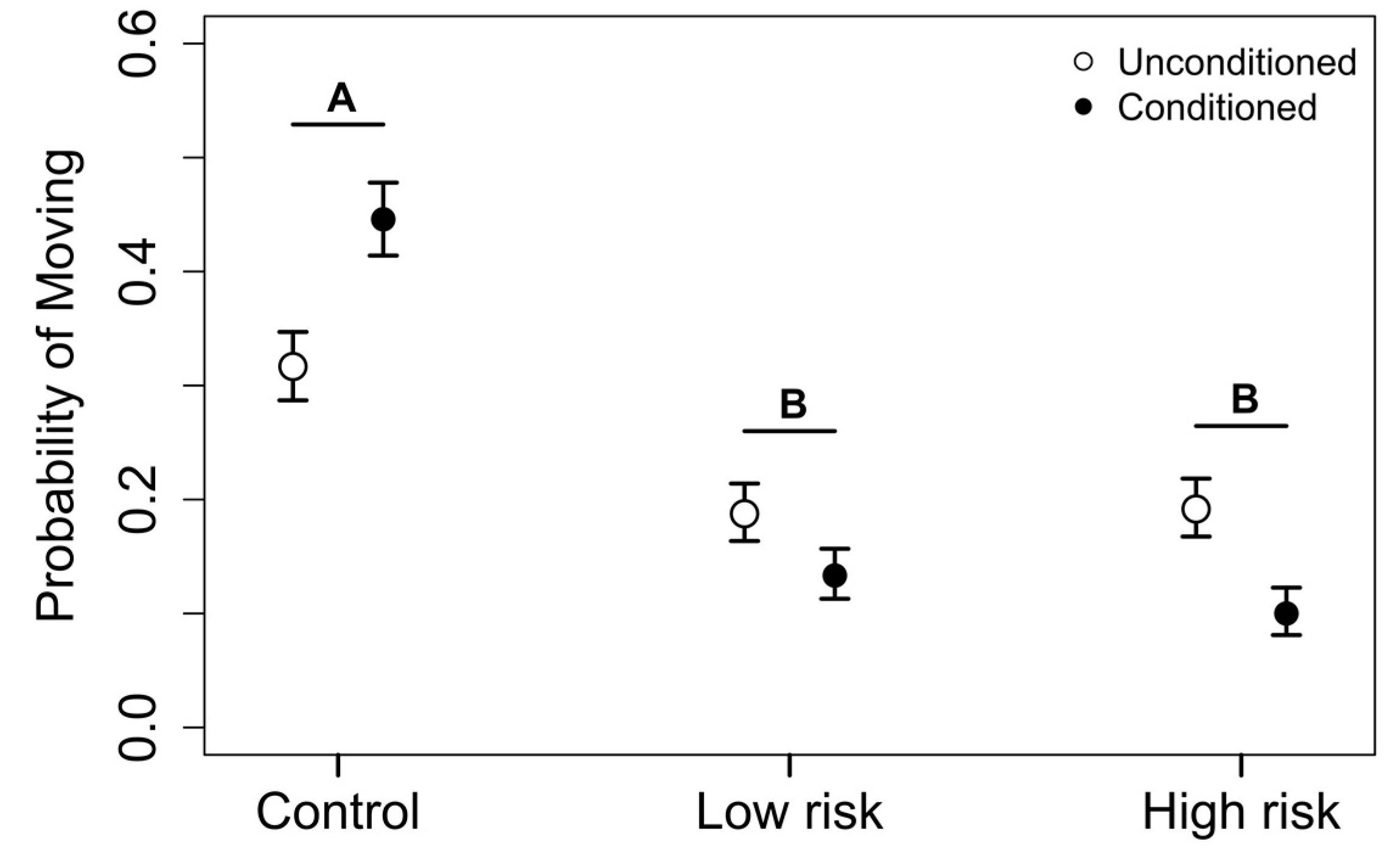
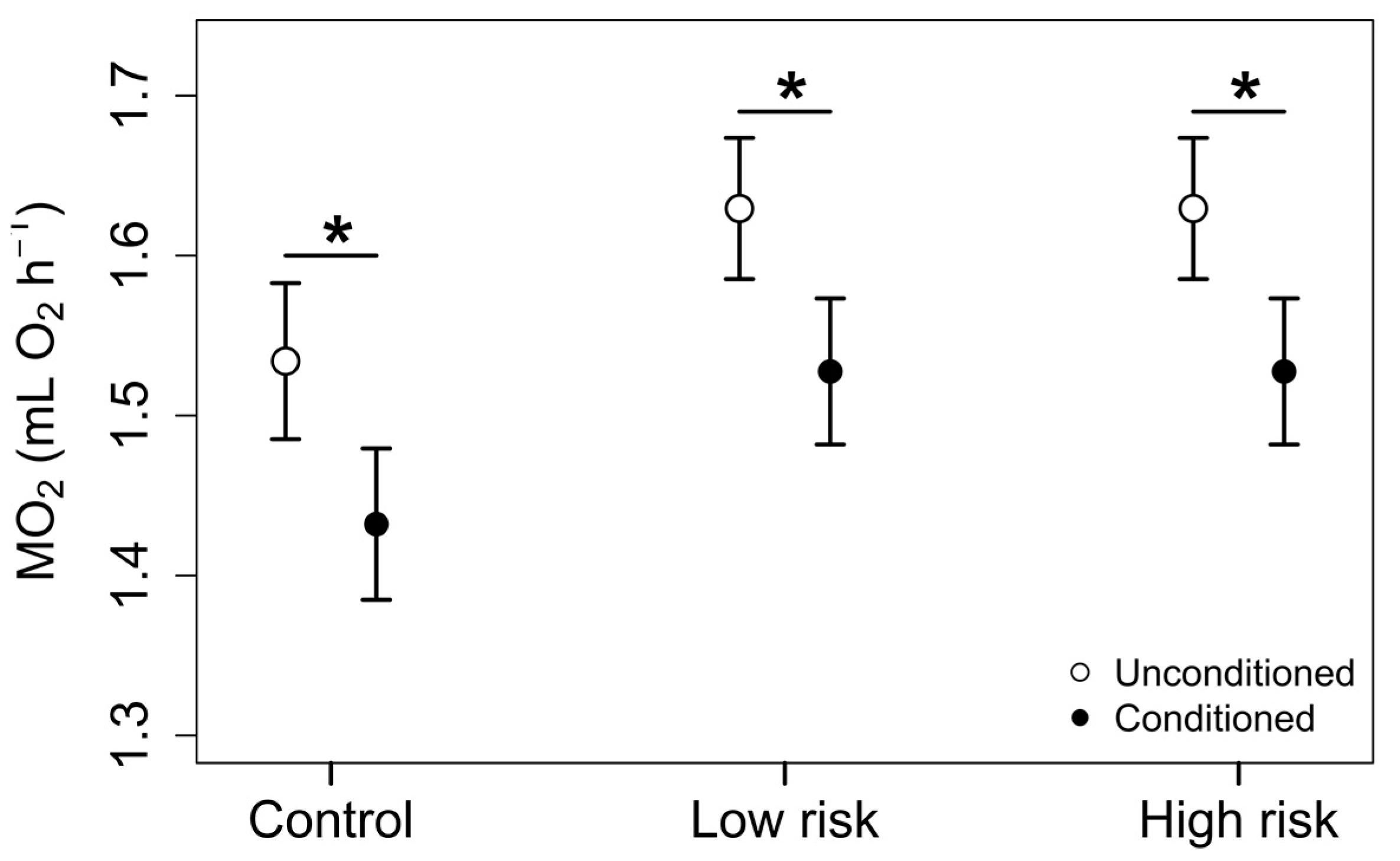
3.2. Phase 2: Exposure Trials
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Teixeira, C.P.; Schetini De Azevedo, C.; Mendl, M.; Cipreste, C.F.; Young, R.J. Revisiting translocation and reintroduction programmes : The importance of considering stress. Anim. Behav. 2007, 73, 1–13. [Google Scholar] [CrossRef]
- Dickens, M.J.; Delehanty, D.J.; Romero, L.M. Stress and translocation: Alterations in the stress physiology of translocated birds. Proc. R. Soc. Lond. B Biol. Sci. 2009, 276, 2051–2056. [Google Scholar] [CrossRef] [PubMed]
- Dickens, M.J.; Delehanty, D.J.; Michael Romero, L. Stress: An inevitable component of animal translocation. Biol. Conserv. 2010, 143, 1329–1341. [Google Scholar] [CrossRef]
- Engelsma, M.Y.; Hougee, S.; Nap, D.; Hofenk, M.; Rombout, J.H.W.M.; Van Muiswinkel, W.B.; Lidy Verburg-van Kemenade, B.M. Multiple acute temperature stress affects leucocyte populations and antibody responses in common carp, Cyprinus carpio L. Fish Shellfish Immunol. 2003, 15, 397–410. [Google Scholar] [CrossRef]
- Breves, J.P.; Specker, J.L. Cortisol stress response of juvenile winter flounder (Pseudopleuronectes americanus, Walbaum) to predators. J. Exp. Mar. Biol. Ecol. 2005, 325, 1–7. [Google Scholar] [CrossRef]
- Poledník, L.; Řehulka, J.; Kranz, A.; Poledníková, K.; Hlaváč, V.; Kazihnitkoví, H. Physiological responses of over-wintering common carp (Cyprinus carpio) to disturbance by Eurasian otter (Lutra lutra). Fish Physiol. Biochem. 2008, 34, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Killen, S.S.; Brown, J.A. Energetic cost of reduced foraging under predation threat in newly hatched ocean pout. Mar. Ecol. Prog. Ser. 2006, 321, 255–266. [Google Scholar] [CrossRef]
- Van Dievel, M.; Janssens, L.; Stoks, R. Short- and long-term behavioural, physiological and stoichiometric responses to predation risk indicate chronic stress and compensatory mechanisms. Oecologia 2016, 181, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Aarts, L.; Van Schagen, I. Driving speed and the risk of road crashes: A review. Accid. Anal. Prev. 2006, 38, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Chipman, R.; Slate, D.; Rupprecht, C.; Mendoza, M. Downside risk of wildlife translocation. Dev. Biol. (Basel) 2008, 131, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Crane, A.L.; Mathis, A. Predator-recognition training: A conservation strategy to increase postrelease survival of hellbenders in head-starting programs. Zoo Biol. 2011, 30, 611–622. [Google Scholar] [CrossRef] [PubMed]
- Kellison, G.T.; Eggleston, D.B.; Burke, J.S. Comparative behaviour and survival of hatchery-reared versus wild summer flounder (Paralichthys dentatus). Can. J. Fish. Aquat. Sci. 2000, 57, 1870–1877. [Google Scholar] [CrossRef]
- Parish, D.M.B.; Sotherton, N.W. The fate of released captive-reared grey partridges Perdix perdix: Implications for reintroduction programmes. Wildl. Biol. 2007, 13, 140–149. [Google Scholar] [CrossRef]
- Pfeiffer, W. The fright reaction of fish. Biol. Rev. 1962, 37, 495–511. [Google Scholar] [CrossRef] [PubMed]
- Werner, E.E.; Anholt, B.R. Predator-induced behavioral indirect effects: Consequences to competitive interactions in anuran larvae. Ecology 1996, 77, 157–169. [Google Scholar] [CrossRef]
- Fraser, D.F.; Gilliam, J.F. Nonlethal impacts of predator invasion: Facultative suppression of growth and reproduction. Ecology 1992, 73, 959–970. [Google Scholar] [CrossRef]
- Sapolsky, R.M. Endocrinology of the stress-response. In Behavioral Endocrinology; Becker, J.B., Breedlove, S.M., Crews, D., McCarthy, M.M., Eds.; MIT Press: Cambridge, UK, 2002; pp. 409–450. [Google Scholar]
- Slos, S.; Stoks, R. Predation risk induces stress proteins and reduces antioxidant defense. Funct. Ecol. 2008, 22, 637–642. [Google Scholar] [CrossRef]
- Steiner, U.K.; Van Buskirk, J. Predator-induced changes in metabolism cannot explain the growth/predation risk tradeoff. PLoS ONE 2009, 4, e6160. [Google Scholar] [CrossRef] [PubMed]
- Lankford, S.E.; Adams, T.E.; Miller, R.A.; Cech, J.J., Jr. The cost of chronic stress: Impacts of a nonhabituating stress response on metabolic variables and swimming performance in sturgeon. Physiol. Biochem. Zool. 2005, 78, 599–609. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, H.F.; Bodie, B.L.; Tauchi, M.; Dolgas, C.M.; Herman, J.P. Stress integration after acute and chronic predator stress: Differential activation of central stress circuitry and sensitization of the hypothalamo-pituitary-adrenocortical axis. Endocrinology 2003, 144, 5249–5258. [Google Scholar] [CrossRef] [PubMed]
- Petersson, E.; Valencia, A.C.; Järvi, T. Failure of predator conditioning: An experimental study of predator avoidance in brown trout (Salmo trutta). Ecol. Freshw. Fish 2015, 24, 329–337. [Google Scholar] [CrossRef]
- Chivers, D.P.; Mirza, R.S. Importance of predator diet cues in responses of larval wood frogs to fish and invertebrate predators. J. Chem. Ecol. 2001, 27, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.; Gardner, C.; Braithwaite, V.A. Differential stress responses in fish from areas of high- and low-predation pressure. J. Comp. Physiol. B Biochem. Syst. Environ. Physiol. 2005, 175, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Woodward, J.J.; Smith, L.S. Exercise training and the stress response in rainbow trout, Salmo gairdneri Richardson. J. Fish Biol. 1985, 26, 435–447. [Google Scholar] [CrossRef]
- Young, P.S.; Cech, J.J.J. Effects of exercise conditioning on stress response and recovery in cultured and wild young-of-the-year striped bass, Morone saxatilis. Can. J. Fish. Aquat. Sci. 1993, 50, 2094–2099. [Google Scholar] [CrossRef]
- Bodinof, C.M.; Briggler, J.T.; Junge, R.E.; Mong, T.; Beringer, J.; Wanner, M.D.; Schuette, C.D.; Ettling, J.; Millspaugh, J.J. Survival and body condition of captive-reared juvenile Ozark hellbenders (Cryptobranchus alleganiensis bishopi) following translocation to the wild. Copeia 2012, 2012, 150–159. [Google Scholar] [CrossRef]
- Boerner, J.A. Comparison of Movement Patterns in Captive-Released Eastern Hellbenders (Cryptobranchus alleganiensis alleganiensis) Using Three Different Release Methods; State University of New York Buffalo: Buffalo, NY, USA, 2014. [Google Scholar]
- Kraus, B.T.; McCallen, E.B.; Williams, R.N. Evaluating the survival of translocated adult and captive-reared, juvenile eastern hellbenders (Cryptobranchus alleganiensis alleganiensis). Herpetologica 2017, 73, 271–276. [Google Scholar] [CrossRef]
- Carnahan, D.P. Blue River Fisheries Survey and Game Fish Population Estimates in Crawford, Harrison, and Washington Counties; Indiana Department of Natural Resources: Indianapolis, Indiana, 2001. [Google Scholar]
- Gall, B.G. Predator-Prey Interactions between Hellbenders (Cryptobranchus alleganiensis alleganiensis) and (CA. Bishopi) and Native and Nonnative Fishes. Master’s Thesis, Missouri State University, Springfield, MO, USA, 2008. [Google Scholar]
- Gall, B.G.; Mathis, A. Innate predator recognition and the problem of introduced trout. Ethology 2010, 116, 47–58. [Google Scholar] [CrossRef]
- Burgmeier, N.G.; Unger, S.D.; Sutton, T.M.; Williams, R.N. Population status of the eastern hellbender (Cryptobranchus alleganiensis alleganiensis) in Indiana. J. Herpetol. 2011, 45, 195–201. [Google Scholar] [CrossRef]
- Kats, L.B.; Petranka, J.W.; Sih, A. Antipredator defenses and the persistence of amphibian larvae with fishes. Ecology 1988, 69, 1865–1870. [Google Scholar] [CrossRef]
- Barbosa, P.; Castellanos, I. Ecology of Predator-Prey Interactions; Oxford University Press: Oxford, UK, 2005; ISBN 9780195171204. [Google Scholar]
- Ferland-Raymond, B.; Murray, D.L. Predator diet and prey adaptive responses: Can tadpoles distinguish between predators feeding on congeneric vs. conspecific prey? Can. J. Zool. 2008, 86, 1329–1336. [Google Scholar] [CrossRef]
- Schoeppner, N.M.; Relyea, R.A. When should prey respond to consumed heterospecifics? Testing hypotheses of perceived risk. Copeia 2009, 2009, 190–194. [Google Scholar] [CrossRef]
- Altmann, J. Observational study of behavior: Sampling methods. Behaviour 1974, 49, 227–267. [Google Scholar] [CrossRef] [PubMed]
- Maher, J.M.; Werner, E.E.; Denver, R.J. Stress hormones mediate predator-induced phenotypic plasticity in amphibian tadpoles. Proc. R. Soc. B Biol. Sci. 2013, 280, 1–9. [Google Scholar] [CrossRef]
- Hall, A.E.; Clark, T.D. Seeing is believing: Metabolism provides insight into threat perception for a prey species of coral reef fish. Anim. Behav. 2016, 115, 117–126. [Google Scholar] [CrossRef]
- Burraco, P.; Duarte, L.J.; Gomez-Mestre, I. Predator-induced physiological responses in tadpoles challenged with herbicide pollution. Curr. Zool. 2013, 59, 475–484. [Google Scholar] [CrossRef]
- Alvarez, D.; Cano, J.M.; Nicieza, A.G. Microgeographic variation in metabolic rate and energy storage of brown trout: Countergradient selection or thermal sensitivity? Evol. Ecol. 2006, 20, 345–363. [Google Scholar] [CrossRef]
- Relyea, R.A. Fine-tuned phenotypes: Tadpole plasticity under 16 combinations of predators and competitors. Ecology 2004, 85, 172–179. [Google Scholar] [CrossRef]
- Orlofske, S.A.; Hopkins, W.A. Energetics of metamorphic climax in the pickerel frog (Lithobates palustris). Comp. Biochem. Physiol. Part A 2009, 154, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Kearney, B.D.; Byrne, P.G.; Reina, R.D. Short-and long-term consequences of developmental saline stress: Impacts on anuran respiration and behaviour. R. Soc. Open Sci. 2016, 3, 150640. [Google Scholar] [CrossRef] [PubMed]
- Srean, P.; Almeida, D.; Rubio-Gracia, F.; Luo, Y.; Garcia-Berthou, E. Effects of size and sex on swimming performance and metabolism of invasive mosquitofish Gambusia holbrooki. Ecol. Freshw. Fish 2017, 26, 424–433. [Google Scholar] [CrossRef]
- Guimond, R.W.; Hutchison, V.H. Aquatic respiration: An unusual strategy in the hellbender Cryptobranchus alleganiensis alleganiensis (Daudin). Science 1973, 182, 1263–1265. [Google Scholar] [CrossRef] [PubMed]
- Conover, W.J. Practical Nonparametric Statistics; Wiley: Hoboken, NJ, USA, 1999; ISBN 0471160687. [Google Scholar]
- Team, R.C. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Norrdahl, K.; Korpimäki, E. Fear in farmlands: How much does predator avoidance affect bird community structure? J. Avian Biol. 1998, 29, 79–85. [Google Scholar] [CrossRef]
- Kenison, E.K.E.K.; Weldy, P.Y.P.Y.; Williams, R.N.R.N. There must be something in the water: Assessing the behavioral responses of rusty crayfish (Orconectes rusticus) to fish and amphibian predator kairomones. J. Ethol. 2017, 1–8. [Google Scholar] [CrossRef]
- Bleckmann, H.; Zelick, R. Lateral line system of fish. Integr. Zool. 2009, 4, 13–25. [Google Scholar] [CrossRef] [PubMed]
- McPeek, M.A. The growth/predation risk trade-off: So what is the mechanism? Am. Nat. 2004, 163, E88–E111. [Google Scholar] [CrossRef] [PubMed]
- Holopainen, I.J.; Aho, J.; Vornanen, M.; Huuskonen, H. Phenotypic plasticity and predator effects on morphology and physiology of crucian carp in nature and in the laboratory. J. Fish Biol. 1997, 50, 781–798. [Google Scholar] [CrossRef]
- Brönmark, C.; Miner, J.G. Predator-induced phenotypical change in body morphology in crucian carp. Science 1992, 258, 1348–1350. [Google Scholar] [CrossRef] [PubMed]
- Repka, S.; Walls, M.; Kettle, M. Neck spine protects Daphnia Pulex from predation by Chaoborus, but individuals with longer tail spine are at a greater risk. J. Plankton Res. 1995, 17, 393–403. [Google Scholar] [CrossRef]
- Hoverman, J.T.; Auld, J.R.; Relyea, R.A. Putting prey back together again: Integrating predator-induced behavior, morphology, and life history. Oecologia 2005, 144, 481–491. [Google Scholar] [CrossRef] [PubMed]
- Haskell, A.; Graham, T.E.; Griffin, C.R.; Hestbeck, J.B. Size related survival of headstarted redbelly turtles (Pseudemys rubriventris) in Massachusetts. J. Herpetol. 1996, 30, 524–527. [Google Scholar] [CrossRef]
- Kats, L.B.; Dill, L.M. The scent of death: Chemosensory assessment of predation risk by prey animals. Écoscience 1998, 5, 361–394. [Google Scholar] [CrossRef]
- Epp, K.J.; Gabor, C.R. Innate and learned predator recognition mediated by chemical signals in Eurycea nana. Ethology 2008, 114, 607–615. [Google Scholar] [CrossRef]
- Schoeppner, N.M.; Relyea, R.A. Damage, digestion, and defence: The roles of alarm cues and kairomones for inducing prey defences. Ecol. Lett. 2005, 8, 505–512. [Google Scholar] [CrossRef] [PubMed]
- DeWitt, T.J.; Sih, A.; Wilson, D.S. Cost and limits of phenotypic plasticity. Trends Ecol. Evol. 1998, 13, 77–81. [Google Scholar] [CrossRef]
- Van Buskirk, J.; Relyea, R.A. Selection for phenotypic plasticity in Rana sylvatica tadpoles. Biol. J. Linn. Soc. 1998, 65, 301–328. [Google Scholar] [CrossRef]
- Schoeppner, N.M.; Relyea, R.A. Interpreting the smells of predation: How alarm cues and kairomones induce different prey defences. Funct. Ecol. 2009, 23, 1114–1121. [Google Scholar] [CrossRef]
- Schoeppner, N.M.; Relyea, R.A. Detecting small environmental differences: Risk-response curves for predator-induced behavior and morphology. Oecologia 2008, 154, 743–754. [Google Scholar] [CrossRef] [PubMed]
- Van Buskirk, J.; Muller, C.; Portmann, A.; Surbeck, M. A test of the risk allocation hypothesis: Tadpole responses to temporal change in predation risk. Behav. Ecol. 2002, 13, 526–530. [Google Scholar] [CrossRef]
- Lima, S.L.; Bednekoff, P.A. Temporal variation in danger drives antipredator behavior: The predation risk allocation hypothesis. Am. Nat. 1999, 153, 649–659. [Google Scholar] [CrossRef]
- Sih, A.; McCarthy, T.M. Prey responses to pulses of risk and safety: Testing the risk allocation hypothesis. Anim. Behav. 2002, 63, 437–443. [Google Scholar] [CrossRef]
- Barry, M.J.; Syal, S. Metabolic responses of tadpoles to chemical predation cues. Hydrobiologia 2013, 700, 267–276. [Google Scholar] [CrossRef]
- Reed, D.H.; Lowe, E.H.; Briscoe, D.A.; Frankham, R. Fitness and adaptation in a novel environment: Effect of inbreeding, prior environment, and lineage. Evolution (N. Y.) 2003, 57, 1822–1828. [Google Scholar] [CrossRef]
- Handelsman, C.A.; Broder, E.D.; Dalton, C.M.; Ruell, E.W.; Myrick, C.A.; Reznick, D.N.; Ghalambor, C.K. Predator-induced phenotypic plasticity in metabolism and rate of growth: Rapid adaptation to a novel environment. Integr. Comp. Biol. 2013, 53, 975–988. [Google Scholar] [CrossRef] [PubMed]
- D’Anna, G.; Giacalone, V.M.; Vega Fernández, T.; Vaccaro, A.M.; Pipitone, C.; Mirto, S.; Mazzola, S.; Badalamenti, F. Effects of predator and shelter conditioning on hatchery-reared white seabream Diplodus sargus (L., 1758) released at sea. Aquaculture 2012, 356–357, 91–97. [Google Scholar] [CrossRef]
- Mirza, R.S.; Chivers, D.P. Predator-recognition training enhances survival of brook trout: Evidence from laboratory and field- enclosure studies. Can. J. Zool. 2000, 2208, 2198–2208. [Google Scholar] [CrossRef]
- Griffin, A.S.; Blumstein, D.T.; Evans, C.S. Review: Training captive-bred or translocated animals to avoid predators. Conserv. Biol. 2000, 14, 1317–1326. [Google Scholar] [CrossRef]
| MANOVA | Df | Pillai | f Value | p Value |
|---|---|---|---|---|
| Treatment | 1, 122 | 0.128 | 5.88 | <0.001 |
| Date | 1, 122 | 0.318 | 18.61 | <0.001 |
| Treatment*Date | 1, 122 | 0.087 | 3.81 | 0.012 |
| REPEATED MEASURES ANOVA | Active | Stationary | Floating | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Df | f value | p value | Df | f value | p value | Df | f value | p value | |
| Treatment | 1 | 9.43 | 0.003 | 1 | 3.70 | 0.057 | 1 | 11.21 | 0.001 |
| Treatment*Date | 1 | 5.83 | 0.017 | 1 | 0.80 | 0.372 | 1 | 5.41 | 0.022 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kenison, E.K.; Williams, R.N. Training for Translocation: Predator Conditioning Induces Behavioral Plasticity and Physiological Changes in Captive Eastern Hellbenders (Cryptobranchus alleganiensis alleganiensis) (Cryptobranchidae, Amphibia). Diversity 2018, 10, 13. https://doi.org/10.3390/d10010013
Kenison EK, Williams RN. Training for Translocation: Predator Conditioning Induces Behavioral Plasticity and Physiological Changes in Captive Eastern Hellbenders (Cryptobranchus alleganiensis alleganiensis) (Cryptobranchidae, Amphibia). Diversity. 2018; 10(1):13. https://doi.org/10.3390/d10010013
Chicago/Turabian StyleKenison, Erin K., and Rod N. Williams. 2018. "Training for Translocation: Predator Conditioning Induces Behavioral Plasticity and Physiological Changes in Captive Eastern Hellbenders (Cryptobranchus alleganiensis alleganiensis) (Cryptobranchidae, Amphibia)" Diversity 10, no. 1: 13. https://doi.org/10.3390/d10010013
APA StyleKenison, E. K., & Williams, R. N. (2018). Training for Translocation: Predator Conditioning Induces Behavioral Plasticity and Physiological Changes in Captive Eastern Hellbenders (Cryptobranchus alleganiensis alleganiensis) (Cryptobranchidae, Amphibia). Diversity, 10(1), 13. https://doi.org/10.3390/d10010013




