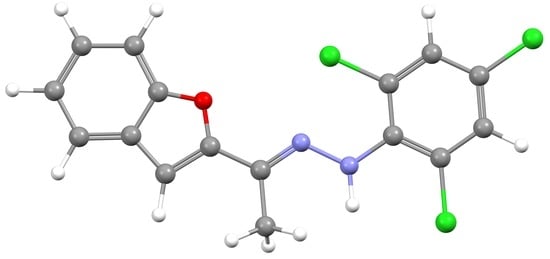
(E)-1-(1-(Benzofuran-2-yl)ethylidene)-2-(2,4,6-trichlorophenyl)hydrazine
Abstract
1. Introduction
2. Results and Discussion
2.1. Synthesis of 3
2.2. IR and NMR Spectroscopy of 3
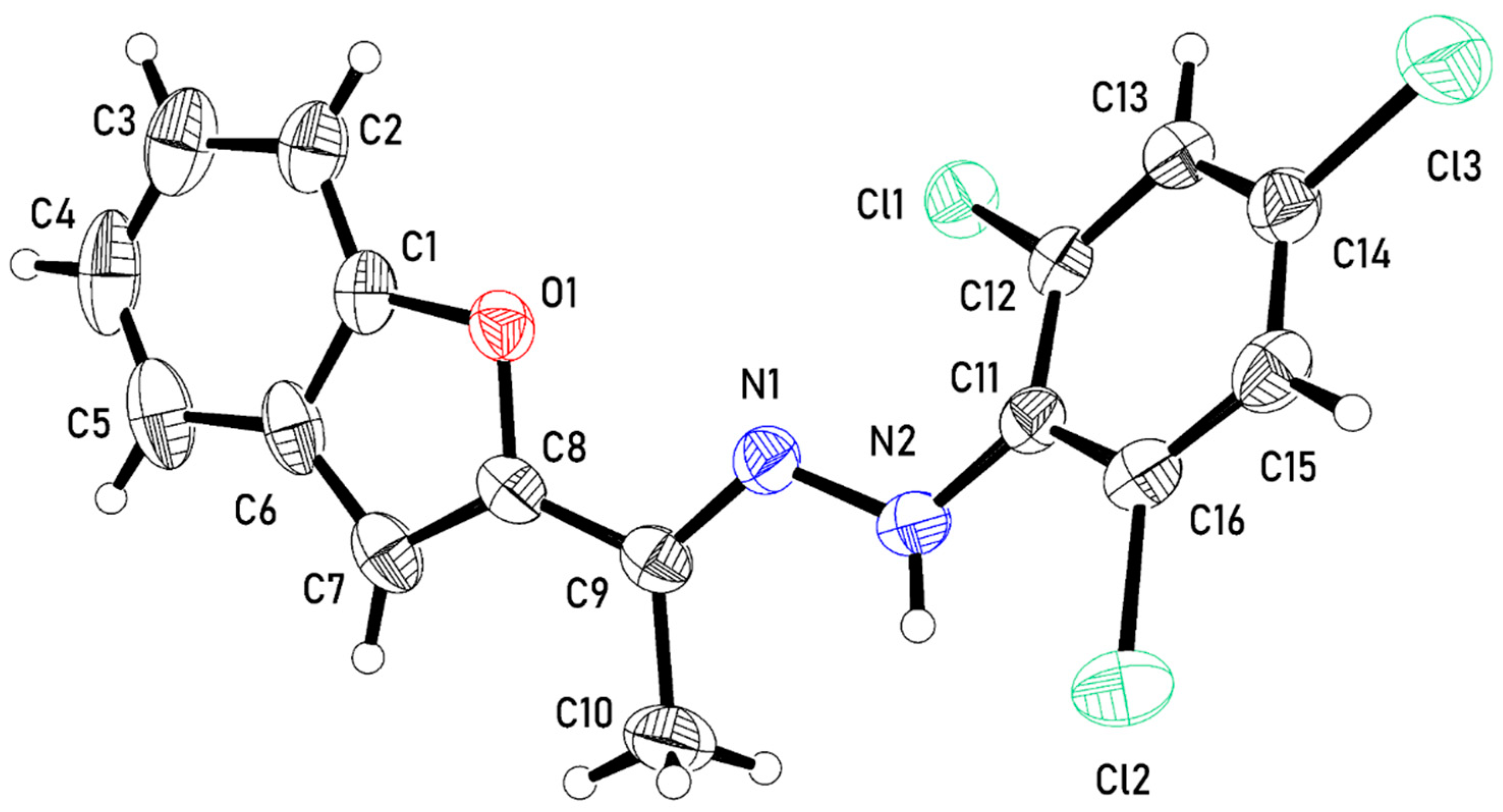
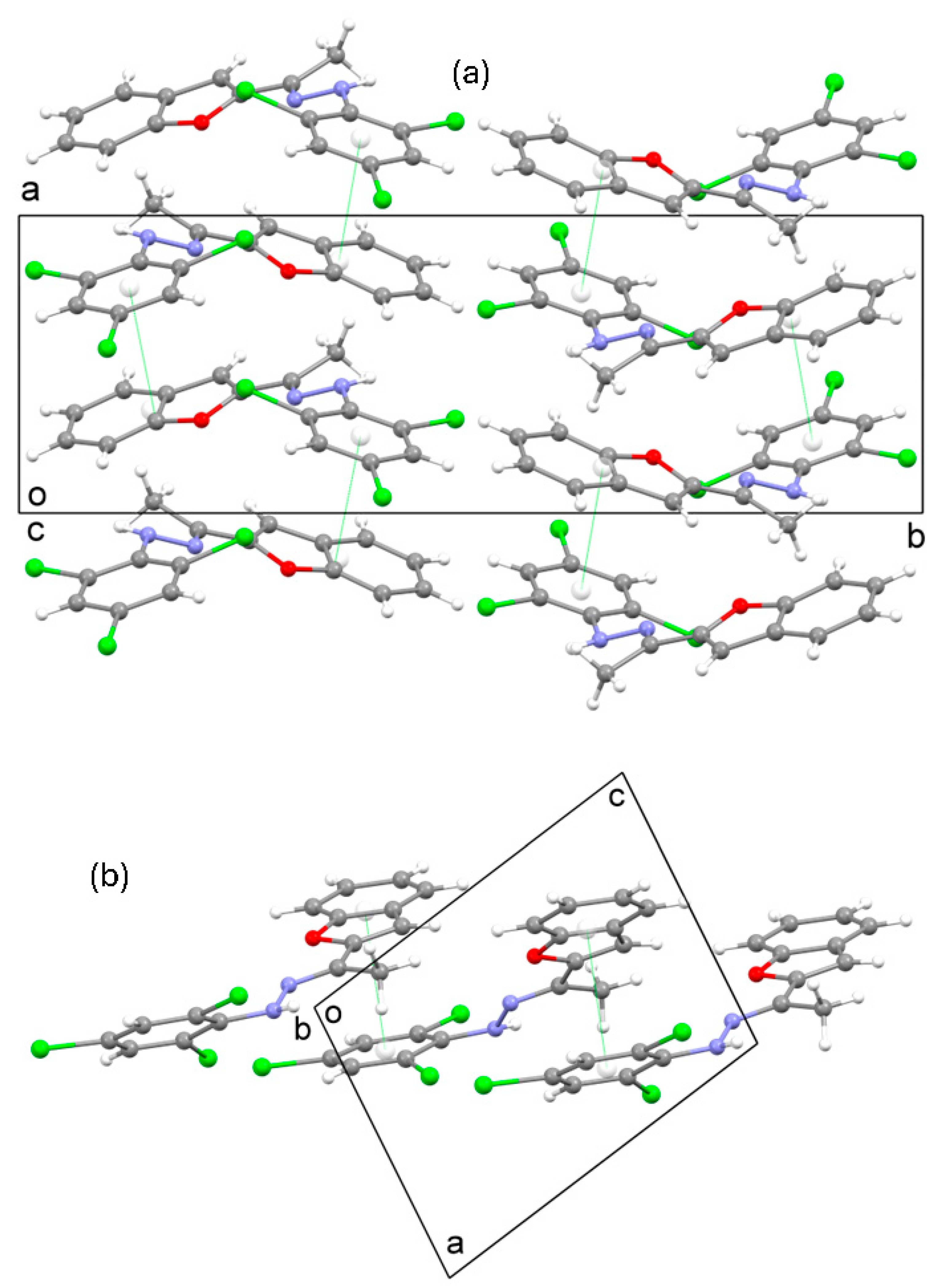
2.3. Crystal Structure of 3
3. Materials and Methods
3.1. General
3.2. Synthesis of 3
3.3. Crystal Structure Determination
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Khodarahmi, G.; Asadi, P.; Hassanzadeh, F.; Khodarahmi, E. Benzofuran as a promising scaffold for the synthesis of antimicrobial and antibreast cancer agents: A review. J. Res. Med. Sci. 2015, 20, 1094–1104. [Google Scholar] [CrossRef]
- Dwarakanath, D.; Gaonkar, S.L. Advances in synthetic strategies and medicinal importance of benzofurans: A review. Asian J. Org. Chem. 2022, 11, e202200282. [Google Scholar] [CrossRef]
- Miao, Y.; Hu, Y.; Yang, J.; Liu, T.; Sun, J.; Wang, X. Natural source, bioactivity and synthesis of benzofuran derivatives. RSC Adv. 2019, 9, 27510–27540. [Google Scholar] [CrossRef] [PubMed]
- Khanam, H.; Shamsuzzaman. Bioactive benzofuran derivatives: A review. Eur. J. Med. Chem. 2015, 97, 483–504. [Google Scholar] [CrossRef] [PubMed]
- Heravi, M.M.; Zadsirjan, V. Chapter Five-Recent Advances in the Synthesis of Benzo[b]furans. Adv. Heterocycl. Chem. 2015, 117, 261–376. [Google Scholar] [CrossRef]
- Chand, K.; Rajeshwari; Hiremathad, A.; Singh, M.; Santos, M.A.; Keri, R.S. A review on antioxidant potential of bioactive heterocycle benzofuran: Natural and synthetic derivatives. Pharmacol. Rep. 2017, 69, 281–295. [Google Scholar] [CrossRef]
- Kadieva, M.G.; Oganesyan, É.T. Methods for the synthesis of benzofuran derivatives (review). Chem. Heterocycl. Compd. 1997, 33, 1245–1258. [Google Scholar] [CrossRef]
- Dawood, K.M. An update on benzofuran inhibitors: A patent review. Expert Opin. Ther. Pat. 2019, 29, 841–870. [Google Scholar] [CrossRef]
- Arce-Ramos, L.; Castillo, J.-C.; Becerra, D. Synthesis and biological studies of benzo[b]furan derivatives: A review from 2011 to 2022. Pharmaceuticals 2023, 16, 1265. [Google Scholar] [CrossRef]
- Jiang, S.; Wang, W.; Mou, C.; Zou, J.; Jin, Z.; Hao, G.; Chi, Y.R. Facile access to benzofuran derivatives through radical reactions with heteroatom-centered super-electron-donors. Nat. Commun. 2023, 14, 7381. [Google Scholar] [CrossRef]
- Verma, G.; Marella, A.; Shaquiquzzaman, M.; Akhtar, M.; Ali, M.R.; Alam, M.M. A review exploring biological activities of hydrazones. J. Pharm. Bioallied. Sci. 2014, 6, 69–80. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira Carneiro Brum, J.; França, T.C.C.; LaPlante, S.R.; Villar, J.D.F. Synthesis and biological activity of hydrazones and derivatives: A review. Mini Rev. Med. Chem. 2020, 20, 342–368. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Jamwal, P.; Vaid, H.; Gurubrahamam, R. Synthesis of alkynyl hydrazones from unprotected hydrazine and their reactivity as diazo precursors. Org. Lett. 2023, 11, 1889–1894. [Google Scholar] [CrossRef]
- Ahmed, F.; Xiong, H. Recent developments in 1,2,3-triazole-based chemosensors. Dyes Pigm. 2021, 185, 108905. [Google Scholar] [CrossRef]
- Saini, N.; Wannasiri, C.; Chanmungkalakul, S.; Prigyai, N.; Ervithayasuporn, V.; Kiatkamjornwong, S. Furan/thiophene-based fluorescent hydrazones as fluoride and cyanide sensors. J. Photochem. Photobiol. A Chem. 2019, 385, 112038. [Google Scholar] [CrossRef]
- Aysha, T.S.; Mohamed, M.B.I.; El-Sedik, M.S.; Youssef, Y.A. Multi-functional colorimetric chemosensor for naked eye recognition of Cu2+, Zn2+ and Co2+ using new hybrid azo-pyrazole/pyrrolinone ester hydrazone dye. Dyes Pigm. 2021, 196, 109795. [Google Scholar] [CrossRef]
- Govindasamy, V.; Perumal, S.; Sekar, I.; Madheswaran, B.; Karuppannan, S.; Kuppannan, S.B. Phenothia-zine-thiophene hydrazide dyad: An efficient “on-off” chemosensor for highly selective and sensitive de-tection of Hg2+ ions. J. Fluoresc. 2021, 31, 667–674. [Google Scholar] [CrossRef]
- Zhao, S.; Chen, L.; Liu, F.; Fan, Y.; Liu, Y.; Han, Y.; Hu, Y.; Su, J.; Song, C. Rapid and selective detection of aluminum ion using 1,2,3-triazole-4,5-dicarboxylic acid-functionalized gold nanoparticle-based colorimetric sensor. RSC Adv. 2021, 11, 30635–30645. [Google Scholar] [CrossRef]
- Abdel-Wahab, B.F.; Mohamed, H.A.; Kariuki, B.M.; El-Hiti, G.A. (2Z,5Z)-5-((3-(Benzofuran-2-yl)-1-phenyl-1H-pyrazol-4-yl)methylene)-2-((4-methoxyphenyl)imino)-3-phenylthiazolidin-4-one. Molbank 2023, 2023, M1665. [Google Scholar] [CrossRef]
- Kariuki, B.M.; Abdel-Wahab, B.F.; Mohamed, H.A.; Bekheit, M.S.; El-Hiti, G.A. Synthesis and characterization of novel 2-(1,2,3-triazol-4-yl)-4,5-dihydro-1H-pyrazol-1-yl)thiazoles and 2-(4,5-dihydro-1H-pyrazol-1-yl)-4-(1H-1,2,3-triazol-4-yl)thiazoles. Molecules 2022, 27, 8904. [Google Scholar] [CrossRef]
- Abdel-Wahab, B.F.; Bekheit, M.S.; Kariuki, B.M.; El-Hiti, G.A. (E)-N’-(1-(Benzofuran-2-yl)ethylidene)-1-(4-methoxyphenyl)-5-methyl-1H-1,2,3-triazole-4-carbohydrazide. Molbank 2023, 2023, M1657. [Google Scholar] [CrossRef]
- Liman, W.; Ait Lahcen, N.; Oubahmane, M.; Hdoufane, I.; Cherqaoui, D.; Daoud, R.; El Allali, A. Hybrid molecules as potential drugs for the treatment of HIV: Design and applications. Pharmaceuticals 2022, 15, 1092. [Google Scholar] [CrossRef] [PubMed]
- Khalid, M.; Arshad, M.N.; Tahir, M.N.; Asiri, A.M.; Naseer, M.M.; Ishaq, M.; Khan, M.U.; Shafiq, Z. An efficient synthesis, structural (SC-XRD) and spectroscopic (FTIR, 1HNMR, MS spectroscopic) characterization of novel benzofuran-based hydrazones: An experimental and theoretical studies. J. Mol. Struct. 2000, 1216, 128318. [Google Scholar] [CrossRef]
- Alotaibi, M.H.; Mohamed, H.A.; Abdel-Wahab, B.F.; Hegazy, A.S.; Kariuki, B.M.; El-Hiti, G.A. Crystal structure of N’-(1-(benzofuran-2-yl)ethylidene)-2-cyanoacetohydrazide, C13H11N3O2. Z. Kristallogr.-New Cryst. Struct. 2019, 234, 361–362. [Google Scholar] [CrossRef]
- Politanskaya, L.; Bagryanskaya, I.; Tretyakov, E. Synthesis of polyfluorinated arylhydrazines, arylhydrazones and 3-methyl-1-aryl-1H-indazoles. J. Fluorine Chem. 2018, 214, 48–57. [Google Scholar] [CrossRef]
- Coşkun, D.; Tekin, S.; Sandal, S.; Coşkun, M.F. Synthesis, characterization, and anticancer activity of new benzofuran substituted chalcones. J. Chem. 2016, 2016, 7678486. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Cryst. 2015, A71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar] [CrossRef]
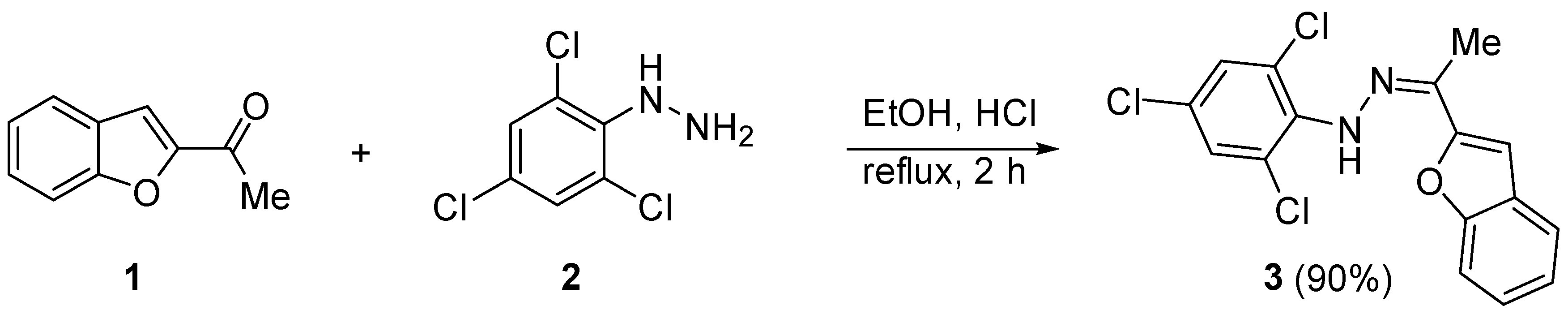
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdel-Wahab, B.F.; Mohamed, H.A.; Kariuki, B.M.; El-Hiti, G.A. (E)-1-(1-(Benzofuran-2-yl)ethylidene)-2-(2,4,6-trichlorophenyl)hydrazine. Molbank 2024, 2024, M1790. https://doi.org/10.3390/M1790
Abdel-Wahab BF, Mohamed HA, Kariuki BM, El-Hiti GA. (E)-1-(1-(Benzofuran-2-yl)ethylidene)-2-(2,4,6-trichlorophenyl)hydrazine. Molbank. 2024; 2024(1):M1790. https://doi.org/10.3390/M1790
Chicago/Turabian StyleAbdel-Wahab, Bakr F., Hanan A. Mohamed, Benson M. Kariuki, and Gamal A. El-Hiti. 2024. "(E)-1-(1-(Benzofuran-2-yl)ethylidene)-2-(2,4,6-trichlorophenyl)hydrazine" Molbank 2024, no. 1: M1790. https://doi.org/10.3390/M1790
APA StyleAbdel-Wahab, B. F., Mohamed, H. A., Kariuki, B. M., & El-Hiti, G. A. (2024). (E)-1-(1-(Benzofuran-2-yl)ethylidene)-2-(2,4,6-trichlorophenyl)hydrazine. Molbank, 2024(1), M1790. https://doi.org/10.3390/M1790