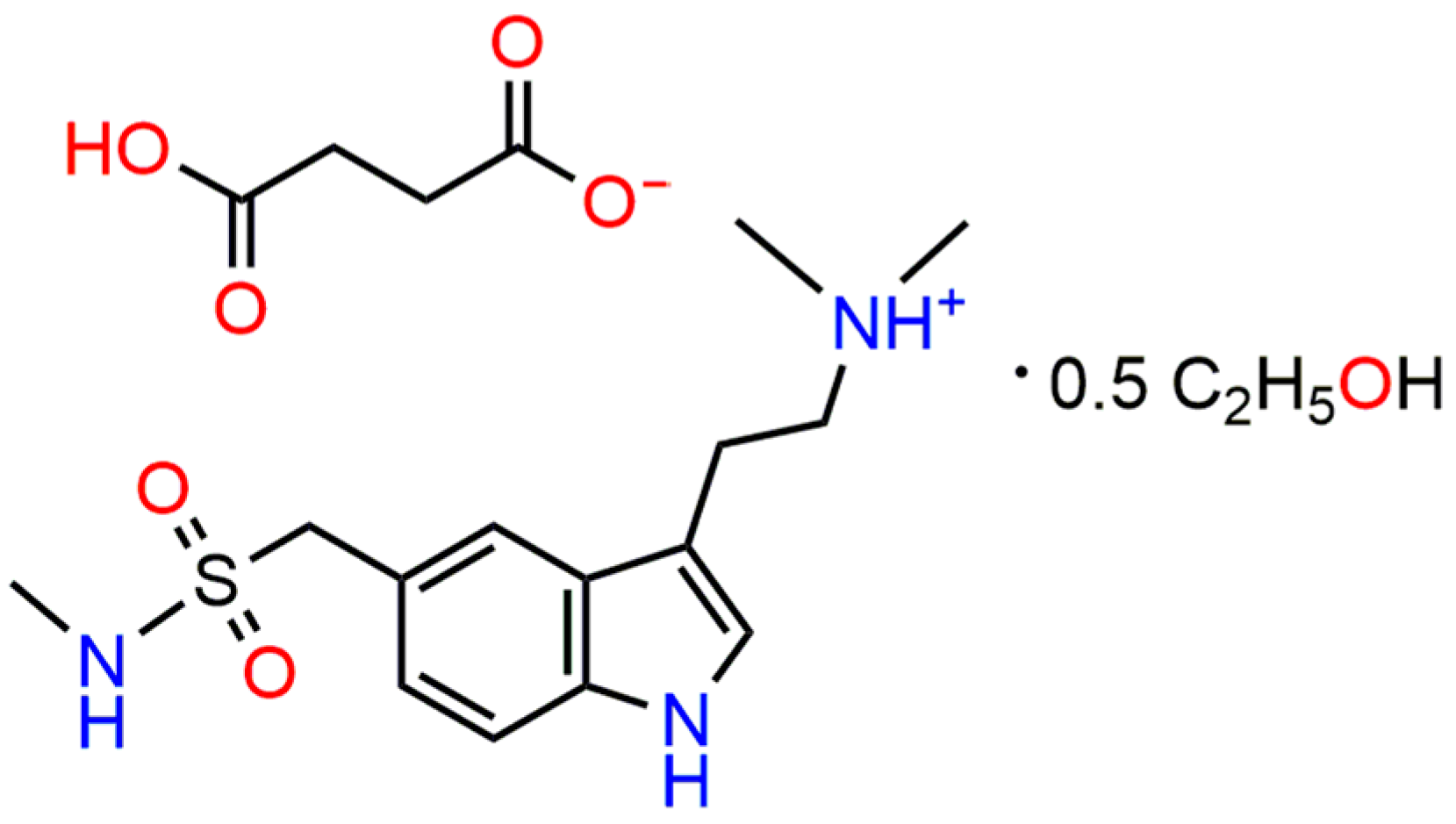
Sumatriptan Succinate Hemi(Ethanol Solvate)
Abstract
1. Introduction
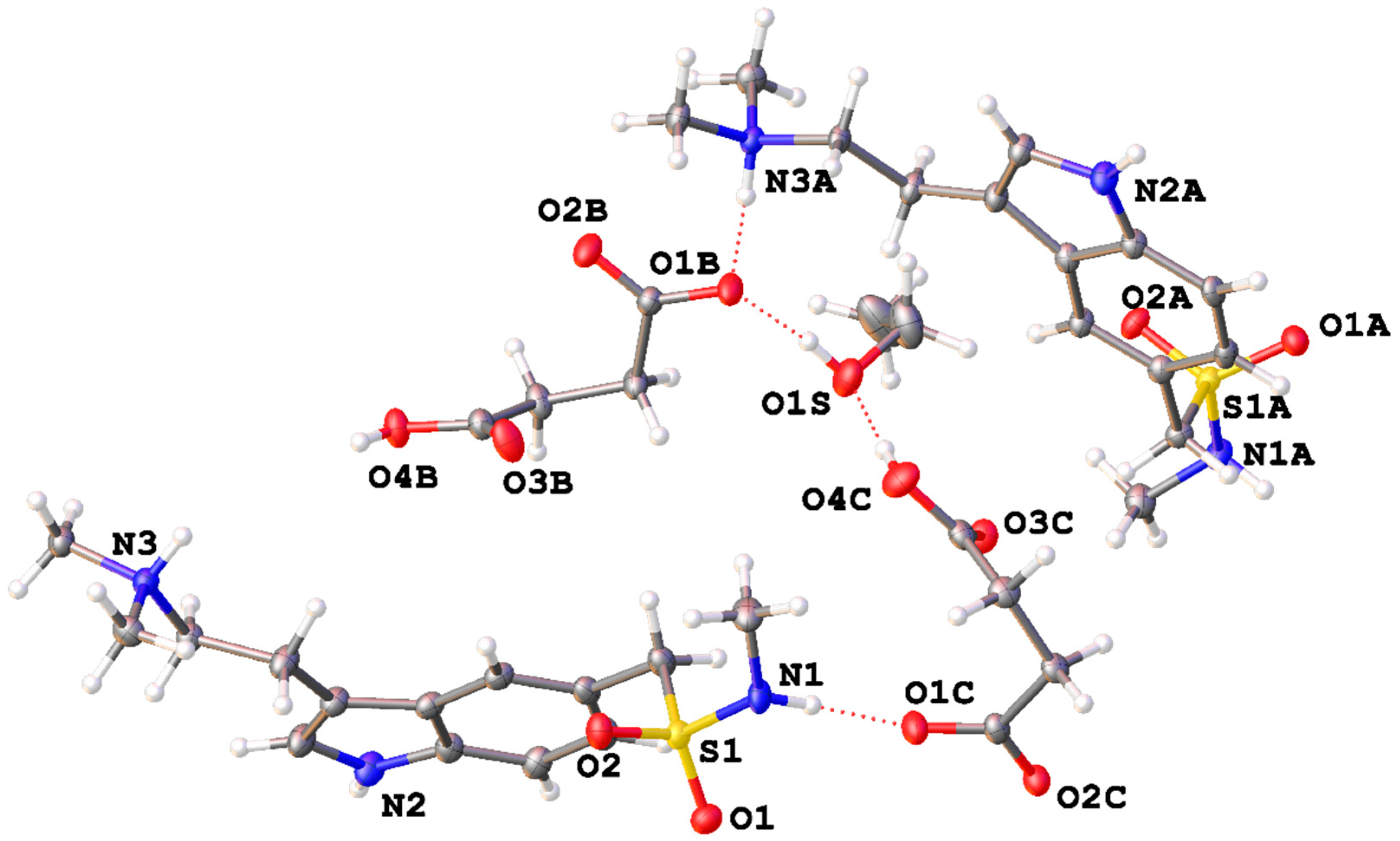
2. Results and Discussion
3. Materials and Methods
X-ray Diffraction
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ala, M.; Ghasemi, M.; Mohammad Jafari, R.; Dehpour, A.R. Beyond Its Anti-Migraine Properties, Sumatriptan Is an Anti-Inflammatory Agent: A Systematic Review. Drug Devel. Res. 2021, 82, 896–906. [Google Scholar] [CrossRef] [PubMed]
- Syed, Y.Y. Sumatriptan/Naproxen Sodium: A Review in Migraine. Drugs 2016, 76, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Tfelt-Hansen, P.; De Vries, P.; Saxena, P.R. Triptans in Migraine. Drugs 2000, 60, 1259–1287. [Google Scholar] [CrossRef] [PubMed]
- Pöstges, T.; Lehr, M. Metabolism of Sumatriptan Revisited. Pharmacol. Res. Perspect. 2023, 11, e01051. [Google Scholar] [CrossRef]
- Ravikumar, K.; Sridhar, B.; Krishnan, H. Sumatriptan, an Anti migraine Drug. Acta Cryst. Sect. E 2006, 62, o1086–o1088. [Google Scholar] [CrossRef]
- Ravikumar, K.; Swamy, G.Y.S.K.; Krishnan, H. α-{3-[2-(Di methyl ammonio) ethyl]-1H-Indol-5-Yl}-N-Methyl methanesulfon amide Succinate (Sumatriptan Succinate). Acta Cryst. Sect. E 2004, 60, o618–o620. [Google Scholar] [CrossRef]
- Korlyukov, A.A.; Dorovatovskii, P.V.; Vologzhanina, A.V. N-(4-Methyl-3-((4-(Pyridin-3-Yl)Pyrimidin-2-Yl)Amino)Phenyl)-4-((4-Methylpiperazin-1-Yl)Methyl)Benzamide. Molbank 2022, 2022, M1461. [Google Scholar] [CrossRef]
- Korlyukov, A.A.; Buikin, P.A.; Dorovatovskii, P.V.; Vologzhanina, A.V. Synthesis, NoSpherA2 Refinement, and Noncovalent Bonding of Abiraterone Bromide Monohydrate. Struct. Chem. 2023, 34, 1927–1934. [Google Scholar] [CrossRef]
- Buikin, P.; Vologzhanina, A.; Novikov, R.; Dorovatovskii, P.; Korlyukov, A. Abiraterone Acetate Complexes with Biometals: Synthesis, Characterization in Solid and Solution, and the Nature of Chemical Bonding. Pharmaceutics 2023, 15, 2180. [Google Scholar] [CrossRef]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge Structural Database. Acta Cryst. Sect. B 2016, 72, 171–179. [Google Scholar] [CrossRef]
- Taylor, R.; Wood, P.A. A Million Crystal Structures: The Whole Is Greater than the Sum of Its Parts. Chem. Rev. 2019, 119, 9427–9477. [Google Scholar] [CrossRef] [PubMed]
- Delori, A.; Galek, P.T.A.; Pidcock, E.; Jones, W. Quantifying Homo- and Heteromolecular Hydrogen Bonds as a Guide for Adduct Formation. Chem. Eur. J. 2012, 18, 6835–6846. [Google Scholar] [CrossRef] [PubMed]
- Macrae, C.F.; Sovago, I.; Cottrell, S.J.; Galek, P.T.A.; McCabe, P.; Pidcock, E.; Platings, M.; Shields, G.P.; Stevens, J.S.; Towler, M.; et al. Mercury 4.0: From Visualization to Analysis, Design and Prediction. J. Appl. Cryst. 2020, 53, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Galek, P.T.A.; Allen, F.H.; Fábián, L.; Feeder, N. Knowledge-Based H-Bond Prediction to Aid Experimental Polymorph Screening. CrystEngComm 2009, 11, 2634–2639. [Google Scholar] [CrossRef]
- Sarkar, N.; Sinha, A.S.; Aakeröy, C.B. Systematic Investigation of Hydrogen-Bond Propensities for Informing Co-Crystal Design and Assembly. CrystEngComm 2019, 21, 6048–6055. [Google Scholar] [CrossRef]
- Shevchenko, A.P.; Blatov, V.A. Simplify to Understand: How to Elucidate Crystal Structures? Struct. Chem. 2021, 32, 507–519. [Google Scholar] [CrossRef]
- Blatov, V.A.; O’Keeffe, M.; Proserpio, D.M. Vertex-, Face-, Point-, Schläfli-, and Delaney-Symbols in Nets, Polyhedra and Tilings: Recommended Terminology. CrystEngComm 2010, 12, 44–48. [Google Scholar] [CrossRef]
- Shevchenko, A.P.; Shabalin, A.A.; Karpukhin, I.Y.; Blatov, V.A. Topological Representations of Crystal Structures: Generation, Analysis and Implementation in the TopCryst System. Sci. Technol. Adv. Mat. Methods 2022, 2, 250–265. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT—Integrated Space-Group and Crystal-Structure Determination. Acta Cryst. Sect. A 2015, A71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Cryst. Sect. C 2015, C71, 3–8. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A Complete Structure Solution, Refinement and Analysis Program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]


| HSum+·HSucc− (ETITEG [6]) | 1 | ||||||
|---|---|---|---|---|---|---|---|
| Donor | Acceptor | Propensity | Observed | Donor | Acceptor | Propensity | Observed |
| R-COOH | CO2 | 0.85 | Yes | R-COOH | CO2 | 0.84 | Yes |
| SO2 | 0.51 | SO2 | 0.46 | ||||
| COOH | 0.33 | COOH | 0.26 | ||||
| R-OH | 0.34 | Yes | |||||
| Ammonium R3NH+ | CO2 | 0.90 | Yes | Ammonium R3NH+ | CO2 | 0.81 | Yes |
| SO2 | 0.63 | SO2 | 0.43 | ||||
| COOH | 0.43 | COOH | 0.23 | ||||
| R-OH | 0.31 | ||||||
| Indole NH | CO2 | 0.97 | Yes | Indole NH | CO2 | 0.87 | |
| SO2 | 0.88 | SO2 | 0.52 | ||||
| COOH | 0.79 | COOH | 0.31 | Yes | |||
| R-OH | 0.40 | ||||||
| Sulfonamide SO2NH | CO2 | 0.92 | Yes | Sulfonamide SO2NH | CO2 | 0.91 | Yes |
| SO2 | 0.68 | SO2 | 0.62 | ||||
| COOH | 0.50 | COOH | 0.40 | ||||
| ROH | CO2 | 0.84 | Yes | ||||
| SO2 | 0.47 | ||||||
| COOH | 0.26 | ||||||
| R-OH | 0.34 | ||||||
| D–H···A | D–H | H···A | D···A | D–H···A | |
|---|---|---|---|---|---|
| 1 | N(1)–H(1)···O(1C) | 0.90(3) | 1.95(3) | 2.844(4) | 173(4) |
| 2 | N(2)–H(2)···O(3B i) | 0.89(3) | 2.07(3) | 2.877(5) | 151(3) |
| 3 | N(3)–H(3)···O(2C) | 0.89(3) | 1.81(3) | 2.669(4) | 162(3) |
| 4 | N(1A)–H(1AA)···O(2B ii) | 0.87(3) | 1.95(3) | 2.794(4) | 163(4) |
| 5 | N(2A)–H(2AA)···O(3C iii) | 0.89(3) | 2.06(3) | 2.866(4) | 161(1) |
| 6 | N(3A)–H(3A)···O(1B) | 0.88(4) | 1.80(4) | 2.636(4) | 158(5) |
| 7 | O(4C)–H(4C)···O(1S) | 0.86(3) | 1.68(3) | 2.532(4) | 170(4) |
| 8 | O(1S)–H(1S)···O(1B) | 0.85(3) | 1.78(3) | 2.603(4) | 162(5) |
| 9 | O(4B)–H(4B)···O(2C iii) | 0.84(4) | 1.70(4) | 2.526(4) | 165(4) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Buikin, P.A.; Vologzhanina, A.V.; Korlyukov, A.A. Sumatriptan Succinate Hemi(Ethanol Solvate). Molbank 2024, 2024, M1766. https://doi.org/10.3390/M1766
Buikin PA, Vologzhanina AV, Korlyukov AA. Sumatriptan Succinate Hemi(Ethanol Solvate). Molbank. 2024; 2024(1):M1766. https://doi.org/10.3390/M1766
Chicago/Turabian StyleBuikin, Petr A., Anna V. Vologzhanina, and Alexander A. Korlyukov. 2024. "Sumatriptan Succinate Hemi(Ethanol Solvate)" Molbank 2024, no. 1: M1766. https://doi.org/10.3390/M1766
APA StyleBuikin, P. A., Vologzhanina, A. V., & Korlyukov, A. A. (2024). Sumatriptan Succinate Hemi(Ethanol Solvate). Molbank, 2024(1), M1766. https://doi.org/10.3390/M1766






