Synthesis of a Novel 2-((4,5-Diphenyl-4H-1,2,4-triazol-3-yl)thio)acetaldehyde as a Bisulfite Adduct
Abstract
:1. Introduction
2. Results and Discussion
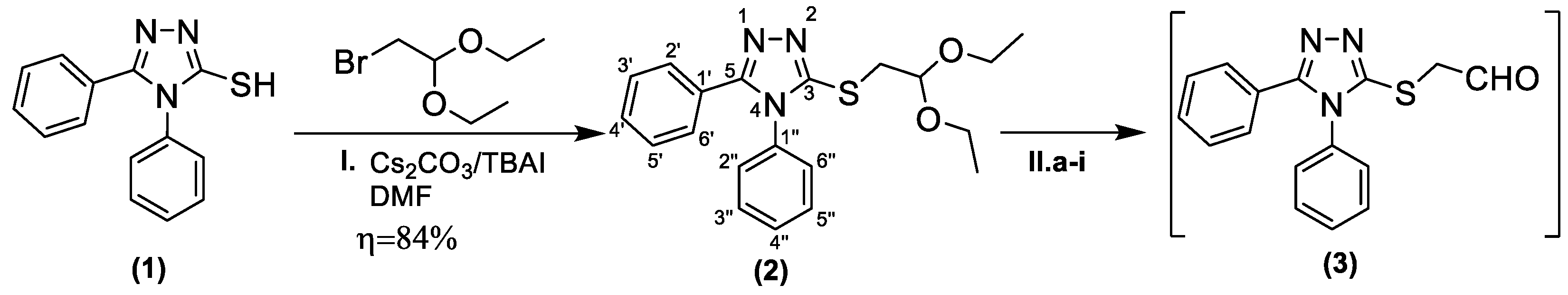
- In the first step, 1,2,4-triazole-3-thiol (1) was alkylated using 2-bromo-1,1-diethoxyethane (bromoacetaldehyde diethyl acetal) in basic medium provided by cesium carbonate, in 84% yield. The reaction follows a nucleophilic substitution mechanism via the sulfur atom. The resulting S-alkylated intermediary (2) was isolated and characterized using spectroscopic methods.
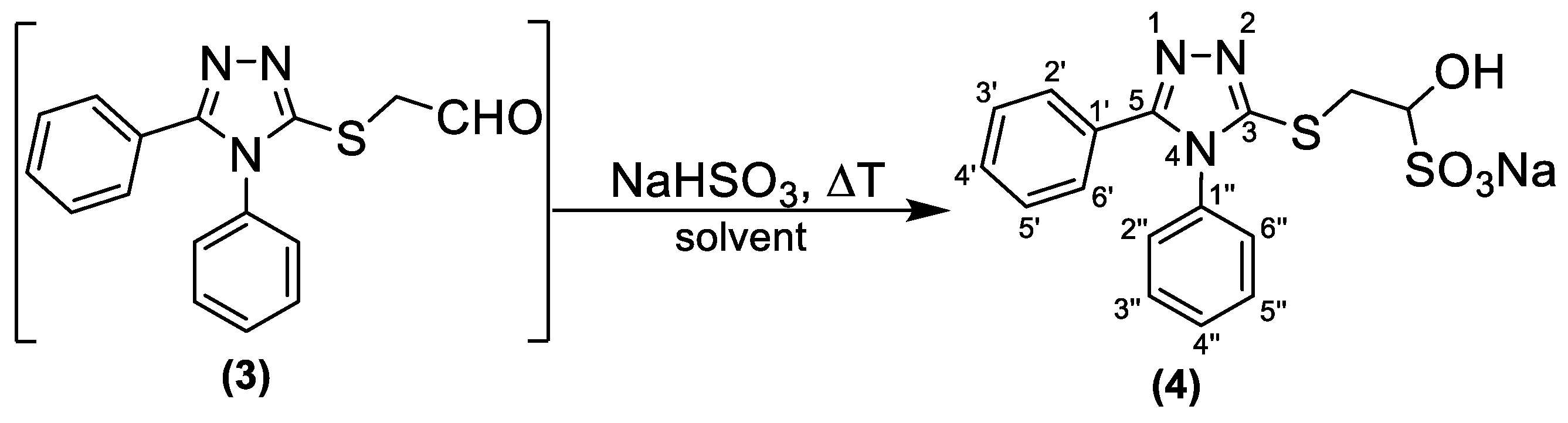
- The obtained acetal (2) was deprotected by using various reagents and conditions. For the preliminary analysis, we used TLC or 1H-NMR. TLC indicated whether the starting material was consumed, while the 1H-NMR analysis showed the absence or presence of the aldehydic proton. The aldehyde was separated from the reaction mixture by conversion into its bisulfite adduct. The resulting α-hydroxy sulfonic acid sodium salt (4) was isolated and characterized.

3. Materials and Methods
4. Experimental
4.1. Synthesis of 3-((2,2-Diethoxyethyl)thio)-4,5-diphenyl-4H-1,2,4-triazole (2)
4.2. Synthesis of 2-((4,5-Diphenyl-4H-1,2,4-triazol-3-yl)thio)acetaldehyde (3)
4.2.1. Method A
4.2.2. Method B
4.3. Synthesis of Sodium 2-((4,5-Diphenyl-4H-1,2,4-triazol-3-yl)thio)-1-hydroxyethane-1-sulfonate (Bisulfite Adduct) (4)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Sathyanarayana, R.; Poojary, B. Exploring Recent Developments on 1,2,4-Triazole: Synthesis and Biological Applications. J. Chin. Chem. Soc. 2020, 67, 459–477. [Google Scholar] [CrossRef]
- Küçükgüzel, Ş.G.; Çıkla-Süzgün, P. Recent Advances Bioactive 1,2,4-Triazole-3-Thiones. Eur. J. Med. Chem. 2015, 97, 830–870. [Google Scholar] [CrossRef] [PubMed]
- Aljaafari, M.N.; Alkhoori, M.A.; Hag-Ali, M.; Cheng, W.-H.; Lim, S.-H.-E.; Loh, J.-Y.; Lai, K.-S. Contribution of Aldehydes and Their Derivatives to Antimicrobial and Immunomodulatory Activities. Molecules 2022, 27, 3589. [Google Scholar] [CrossRef]
- Hoggarth, E. 251. Compounds Related to Thiosemicarbazide. Part II. 1-Benzoylthiosemicarbazides. J. Chem. Soc. 1949, 1163–1167. [Google Scholar] [CrossRef]
- Reid, J.R.; Heindel, N.D. Improved Syntheses of 5-Substituted-4-Amino-3-Mercapto-(4H)-1,2,4-Triazoles. J. Heterocycl. Chem. 1976, 13, 925–926. [Google Scholar] [CrossRef]
- Eweiss, N.F.; Bahajaj, A.A.; Elsherbini, E.A. Synthesis of Heterocycles. Part VI. Synthesis and Antimicrobial Activity of Some 4-Amino-5-Aryl-1,2,4-Triazole-3-Thiones and Their Derivatives. J. Heterocycl. Chem. 1986, 23, 1451–1458. [Google Scholar] [CrossRef]
- Siegfried, A.; Ernst, S. Ger. (East). Chem. Abstr. 1985, 106, 5048. [Google Scholar]
- Malbec, F.; Milcent, R.; Barbier, G. Dérivés de la dihydro-2,4 triazole-1,2,4 thione-3 et de l’amino-2 thiadiazole-1,3,4 à partir de nouvelles thiosemicarbazones d’esters. J. Heterocycl. Chem. 1984, 21, 1689–1698. [Google Scholar] [CrossRef]
- Altowyan, M.S.; Haukka, M.; Soliman, S.M.; Barakat, A.; Boraei, A.T.A.; Aboelmagd, A. Stereoselective Synthesis of New 4-Aryl-5-Indolyl-1,2,4-Triazole S- and N-β-Galactosides: Characterizations, X-ray Crystal Structure and Hirshfeld Surface Analysis. Crystals 2023, 13, 797. [Google Scholar] [CrossRef]
- Il’inykh, E.S.; Kim, D.G.; Valova, M.S.; Fedorova, O.V. Synthesis and Optical Properties of New S-Derivatives of 5,5′-(1,4-Phenylene)Bis(4H-1,2,4-Triazole-3-Thiol) and 5,5′,5″-(Benzene-1,3,5-Triyl)Tris(4H-1,2,4-Triazole-3-Thiol). Russ. J. Gen. Chem. 2019, 89, 2571–2576. [Google Scholar] [CrossRef]
- Socea, L.; Barbuceanu, S.; Socea, B.; Draghici, C.; Apostol, T.-V.; Pahontu, E.; Olaru, O. New Heterocyclic Compounds from 1,2,4-Triazoles Class with Potential Cytotoxic Activity. Rev. Chim. 2017, 68, 2503–2508. [Google Scholar] [CrossRef]
- Wuts, P.G.M. Greene’s Protective Groups in Organic Synthesis, 5th ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; pp. 559–615. [Google Scholar]
- Salvatore, R.N.; Smith, R.A.; Nischwitz, A.K.; Gavin, T. A Mild and Highly Convenient Chemoselective Alkylation of Thiols Using Cs2CO3–TBAI. Tetrahedron Lett. 2005, 46, 8931–8935. [Google Scholar] [CrossRef]
- Taylor, H.M.; Hauser, C.R. α-(N,N-dimethylamino)phenylacetonitrile. Org. Synth. 1963, 43, 25. [Google Scholar] [CrossRef]
- Young, S.D.; Buse, C.T.; Heathcock, C.H. 2-methyl-2-(trimethylsiloxy)pentan-3-one. Org. Synth. 1985, 63, 79. [Google Scholar] [CrossRef]
- Sun, J.; Dong, Y.; Cao, L.; Wang, X.; Wang, S.; Hu, Y. Highly Efficient Chemoselective Deprotection of O,O-Acetals and O,O-Ketals Catalyzed by Molecular Iodine in Acetone. J. Org. Chem. 2004, 69, 8932–8934. [Google Scholar] [CrossRef] [PubMed]
- Tufariello, J.J.; Winzenberg, K. A nitrone-based synthesis of the pyrrolizidine alkaloid croalbinecine. Tetrahedron Lett. 1986, 27, 1645–1648. [Google Scholar] [CrossRef]
- Lacoske, M.H.; Xu, J.; Mansour, N.; Gao, C.; Theodorakis, E.A. Synthetic strategies toward the decalin motif of maklamicin and related spirotetronates. Org. Chem. Front. 2015, 2, 388–393. [Google Scholar] [CrossRef]
- Parker, K.A.; Su, D.S. Synthesis of C-aryl furanosides by the “reverse polarity” strategy. J. Org. Chem. 1996, 61, 2191–2194. [Google Scholar] [CrossRef]
- Nishimata, T.; Sato, Y.; Mori, M. Palladium-Catalyzed Asymmetric Allylic Substitution of 2-Arylcyclohexenol Derivatives: Asymmetric Total Syntheses of (+)-Crinamine, (−)-Haemanthidine, and (+)-Pretazettine. J. Org. Chem. 2004, 69, 1837–1843. [Google Scholar] [CrossRef]
- Kumar, R.; Kumar, D.; Chakraborti, A.K. Perchloric Acid Adsorbed on Silica Gel (HClO4–SiO2) as an Inexpensive, Extremely Efficient, and Reusable Dual Catalyst System for Acetal/Ketal Formation and Their Deprotection to Aldehydes/Ketones. Synthesis 2007, 299–303. [Google Scholar] [CrossRef]
- Stipanovic, R.D.; Turner, R.B. Cyclohexenone-4-acetic Acid Derivatives from the Addition of Diazoacetic Ester to β,γ-Unsaturated Ketals. J. Org. Chem. 1968, 33, 3261–3263. [Google Scholar] [CrossRef]
- Barbot, F.; Miginiac, P. Preparation of 3-alkenals and 3-alkynals by hydrolysis of the corresponding acetals. Synthesis 1983, 1983, 651–654. [Google Scholar] [CrossRef]
- Molla, E.; Abser, N.; Islam, M. Synthesis and Characterization of Some 4-Aryl Substituted Thiosemicarbazides, N-Alkyloxybenzaldehydes Containing Long Alkyl Chains and Their Corresponding Thiosemicarbazones. Jahangirnagar Univ. J. Sci. 2018, 41, 31–42. [Google Scholar]
- Siwek, A.; Wujec, M.; Dobosz, M.; Wawrzycka-Gorczyca, I. Study of Direction of Cyclization of 1-Azolil-4-Aryl/Alkyl-Thiosemicarbazides. Heteroat. Chem. 2010, 21, 521–532. [Google Scholar] [CrossRef]
- Pareek, A.K.; Joseph, P.E.; Seth, D.S. Convenient Synthesis, Characterization of Some Novel Substituted 3-Methyl-2-Pyrazoline-5-Ones and Substituted 3,5-Dimethyl Pyrazoles. Orient. J. Chem. 2010, 26, 1467–1471. [Google Scholar]
- Pintea, B.N.V. A Simple and Inexpensive Capillary Holder for Thin-Layer Chromatography. J. Chem. Educ. 2011, 88, 673–674. [Google Scholar] [CrossRef]
| No. | Method | Conversion, % | Ref. |
|---|---|---|---|
| 1. | I2, acetone, r.t., and reflux | n.o. a | [16] |
| 2. | CF3COOH, DCM, r.t., and reflux | 100 | [17] |
| 3. | concd. HCl, THF-H2O, reflux | 100 | [18] |
| 4. | Na2S2O4, THF-H2O, reflux | n.o. | [19] |
| 5. | FeCl3/SiO2, acetone, r.t. | n.o. | [20] |
| 6. | HClO4/SiO2, EtOH 96%-H2O, r.t. | n.o. | [21] |
| 7. | HClO4/SiO2, THF-H2O, reflux | n.o. | - |
| 8. | TsOH, acetone, r.t. | n.o. | [22] |
| 9. | HCOOH 80%, r.t. | 27 | - |
| 10 | HCOOH 98%, r.t. | 47 | [23] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pintea, B.-N.; Burcă, I.; Badea, V.; Peter, F. Synthesis of a Novel 2-((4,5-Diphenyl-4H-1,2,4-triazol-3-yl)thio)acetaldehyde as a Bisulfite Adduct. Molbank 2023, 2023, M1715. https://doi.org/10.3390/M1715
Pintea B-N, Burcă I, Badea V, Peter F. Synthesis of a Novel 2-((4,5-Diphenyl-4H-1,2,4-triazol-3-yl)thio)acetaldehyde as a Bisulfite Adduct. Molbank. 2023; 2023(3):M1715. https://doi.org/10.3390/M1715
Chicago/Turabian StylePintea, Beniamin-Nicolae, Ion Burcă, Valentin Badea, and Francisc Peter. 2023. "Synthesis of a Novel 2-((4,5-Diphenyl-4H-1,2,4-triazol-3-yl)thio)acetaldehyde as a Bisulfite Adduct" Molbank 2023, no. 3: M1715. https://doi.org/10.3390/M1715
APA StylePintea, B.-N., Burcă, I., Badea, V., & Peter, F. (2023). Synthesis of a Novel 2-((4,5-Diphenyl-4H-1,2,4-triazol-3-yl)thio)acetaldehyde as a Bisulfite Adduct. Molbank, 2023(3), M1715. https://doi.org/10.3390/M1715







