(E)-2-(6-(4′-(Diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5(4H)-ylidene)-2-(6-(4′-(diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5-yl)acetonitrile
Abstract
:1. Introduction
2. Results and Discussion
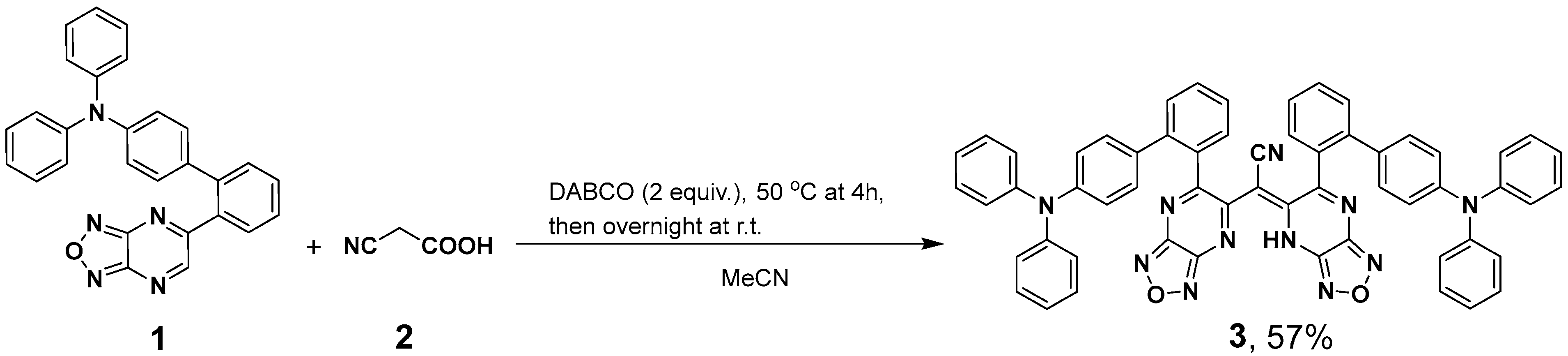
2.1. Synthesis of (E)-2-(6-(4′-(Diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5(4H)-ylidene)-2-(6-(4′-(diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5-yl)acetonitrile (3)
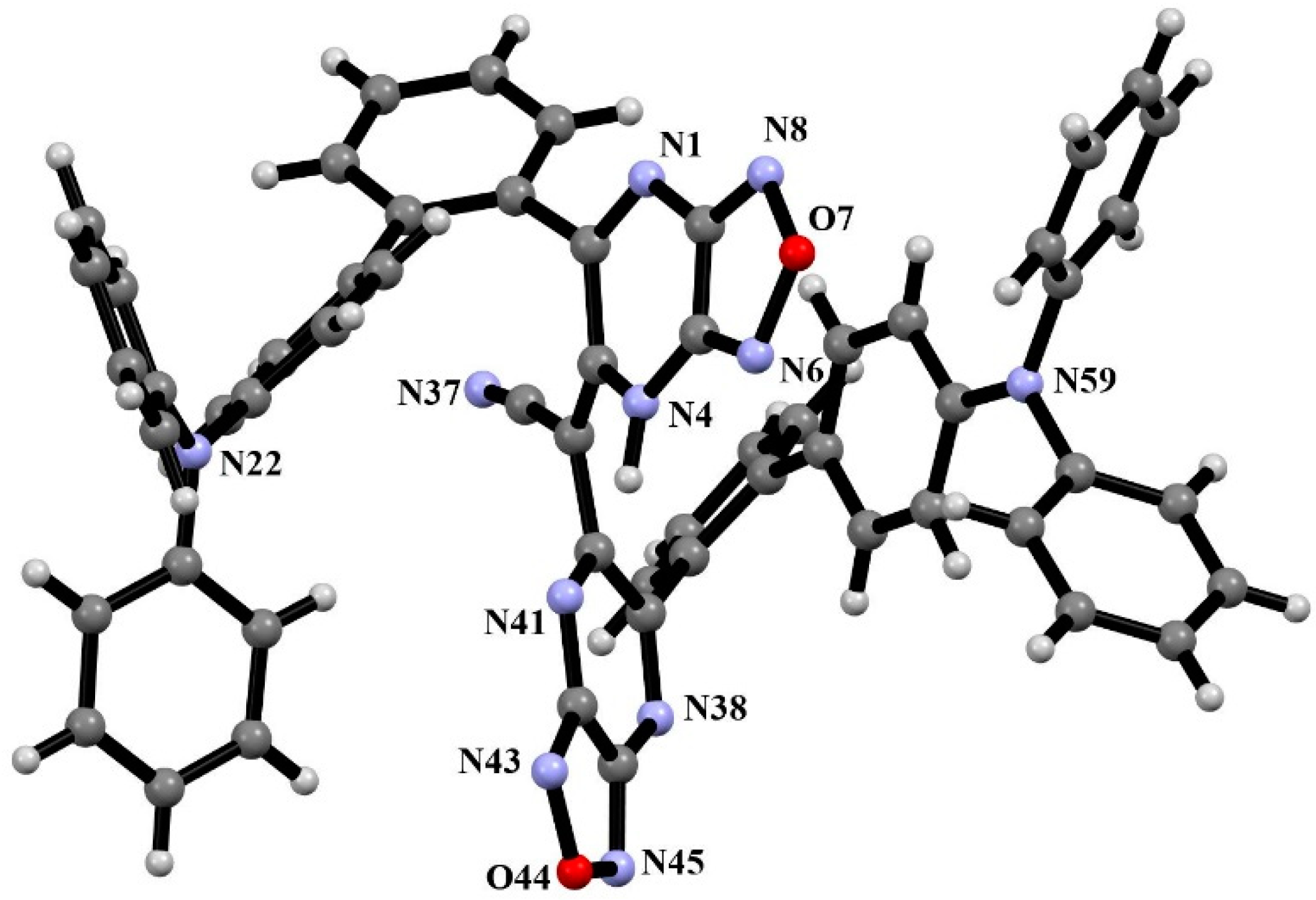
2.2. Crystal Structure of Compound (3)
2.3. Photophysical and Electrochemical Properties of the Compound (3)
3. Materials and Methods
Synthesis of (E)-2-(6-(4′-(Diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5(4H)-ylidene)-2-(6-(4′-(diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5-yl)acetonitrile (3) (Supplementary Materials)
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mancini, R.S.; Barden, C.J.; Weaver, D.F.; Reed, M.A. Furazans in Medicinal Chemistry. J. Med. Chem. 2021, 64, 1786–1815. [Google Scholar] [CrossRef] [PubMed]
- Fershtat, L.L. Recent advances in the synthesis and performance of 1,2,4,5-tetrazine-based energetic materials. FirePhysChem 2023, 3, 78–87. [Google Scholar] [CrossRef]
- Ovcharenko, V.I.; Sheremetev, A.B.; Strizhenko, K.V.; Fokin, S.V.; Romanenko, G.V.; Bogomyakov, A.S.; Morozov, V.A.; Syroeshkin, M.A.; Kozmenkova, A.Y.; Lalov, A.V.; et al. Novel organic magnet derived from pyrazine-fused furazans. Mendeleev Commun. 2021, 31, 784–788. [Google Scholar] [CrossRef]
- Verbitskiy, E.V.; Achelle, S.; Bureš, F.; le Poul, P.; Barsella, A.; Kvashnin, Y.A.; Rusinov, G.L.; Guen, F.R.-L.; Chupakhin, O.N.; Charushin, V.N. Synthesis, photophysical and nonlinear optical properties of [1,2,5]oxadiazolo[3,4-b]pyrazine-based linear push-pull systems. J. Photochem. Photobiol. A 2021, 404, 112900. [Google Scholar] [CrossRef]
- Verbitskiy, E.V.; Kvashnin, Y.A.; Baranova, A.A.; Khokhlov, K.O.; Chuvashov, R.D.; Yakovleva, Y.A.; Makarova, N.I.; Vetrova, E.V.; Metelitsa, A.V.; Rusinov, G.L.; et al. Novel fluorophores based on imidazopyrazine derivatives: Synthesis and photophysical characterization focusing on solvatochromism and sensitivity towards nitroaromatic compounds. Dye. Pigment. 2019, 168, 248–256. [Google Scholar] [CrossRef]
- Kvashnin, Y.A.; Verbitskiy, E.V.; Eltsov, O.S.; Slepukhin, P.A.; Tameev, A.R.; Nekrasova, N.V.; Rusinov, G.L.; Nunzi, J.-M.; Chupakhin, O.N.; Charushin, V.N. Dibenzo[f,h]furazano[3,4-b]quinoxalines: Synthesis by intramolecular cyclization through direct transition metal-free C–H functionalization and electrochemical, photophysical, and charge mobility characterization. ACS Omega 2020, 5, 8200–8210. [Google Scholar] [CrossRef] [PubMed]
- Steparuk, A.S.; Kvashnin, Y.A.; Rusinov, G.L.; Verbitskiy, E.V.; Aleksandrov, A.E.; Lypenko, D.A.; Tameev, A.R.; Charushin, V.N. The first application of push-pull systems based on 1,2,5-oxadiazolo[3,4-b]pyrazine in organic light-emitting diodes and perovskite solar cells. Russ. Chem. Bull. 2023, 72, 527–533. [Google Scholar] [CrossRef]
- Kvashnin, Y.A.; Verbitskiy, E.V.; Rusinov, G.L.; Charushin, V.N. Modification and application of 1,2,5-oxadiazolo[3,4-b]pyrazine derivatives: Highlights and perspectives. Russ. Chem. Bull. 2022, 71, 1342–1362. [Google Scholar] [CrossRef]
- Grabowski, Z.R.; Rotkiewicz, K.; Siemiarczuk, A. Dual fluorescence of donor-acceptor molecules and the Twisted Intramolecular Charge Transfer (TICT) states. J. Lumin. 1979, 18–19, 420–424. [Google Scholar] [CrossRef]
- Grabowski, Z.R.; Rotkiewicz, K.; Rettig, W. Structural Changes Accompanying Intramolecular Electron Transfer: Focus on Twisted Intramolecular Charge-Transfer States and Structures. Chem. Rev. 2003, 103, 3899–4032. [Google Scholar] [CrossRef] [PubMed]
- Purushotham, U.; Narahari Sastry, G. Conjugate acene fused buckybowls: Evaluating their suitability for p-type, ambipolar and n-type air stable organic semiconductors. Phys. Chem. Chem. Phys. 2013, 15, 5039–5048. [Google Scholar] [CrossRef] [PubMed]
- Eakins, G.L.; Cooper, M.W.; Gerasimchuk, N.N.; Phillips, T.J.; Breyfogle, B.E.; Stearman, C.J. Structural influences impacting the role of the 9-ylidene bond in the electronic tuning of structures built upon 9-fluorenylidene scaffolds. Can. J. Chem. 2013, 91, 1059–1071. [Google Scholar] [CrossRef]
- Gao, X.; Hu, Y. Development of n-type organic semiconductors for thin film transistors: A viewpoint of molecular design. J. Mater. Chem. C 2014, 2, 3099–3117. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Cryst. 2015, A71, 3–8. [Google Scholar] [CrossRef] [PubMed]




| Compound | UV–Vis Absorption | Fluorescence | Φ, % | ||
|---|---|---|---|---|---|
| λabsmax, nm | εmax, M−1 cm−1 | Excitation λex, nm | Emission λem, nm | ||
| 3 | 307 468 680 | 43,200 12,500 1500 | 260, 405 | 523 | <1 |
| Ehred a, eV | Ehox a, eV | EelLUMO, eV | EelHOMO, eV | EDFTLUMO, a eV | EDFTHOMO, a eV | Egopt, b eV | Egel, eV | EgDFT, eV |
|---|---|---|---|---|---|---|---|---|
| −0.63 | 0.58 | −4.17 | −5.38 | −3.65 | −5.28 | 1.46 | 1.21 | 1.63 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kvashnin, Y.A.; Slepukhin, P.A.; Gazizov, D.A.; Zhilina, E.F.; Rusinov, G.L.; Verbitskiy, E.V.; Charushin, V.N. (E)-2-(6-(4′-(Diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5(4H)-ylidene)-2-(6-(4′-(diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5-yl)acetonitrile. Molbank 2023, 2023, M1714. https://doi.org/10.3390/M1714
Kvashnin YA, Slepukhin PA, Gazizov DA, Zhilina EF, Rusinov GL, Verbitskiy EV, Charushin VN. (E)-2-(6-(4′-(Diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5(4H)-ylidene)-2-(6-(4′-(diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5-yl)acetonitrile. Molbank. 2023; 2023(3):M1714. https://doi.org/10.3390/M1714
Chicago/Turabian StyleKvashnin, Yuriy A., Pavel A. Slepukhin, Denis A. Gazizov, Ekaterina F. Zhilina, Gennady L. Rusinov, Egor V. Verbitskiy, and Valery N. Charushin. 2023. "(E)-2-(6-(4′-(Diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5(4H)-ylidene)-2-(6-(4′-(diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5-yl)acetonitrile" Molbank 2023, no. 3: M1714. https://doi.org/10.3390/M1714
APA StyleKvashnin, Y. A., Slepukhin, P. A., Gazizov, D. A., Zhilina, E. F., Rusinov, G. L., Verbitskiy, E. V., & Charushin, V. N. (2023). (E)-2-(6-(4′-(Diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5(4H)-ylidene)-2-(6-(4′-(diphenylamino)-[1,1′-biphenyl]-2-yl)-[1,2,5]oxadiazolo[3,4-b]pyrazin-5-yl)acetonitrile. Molbank, 2023(3), M1714. https://doi.org/10.3390/M1714






