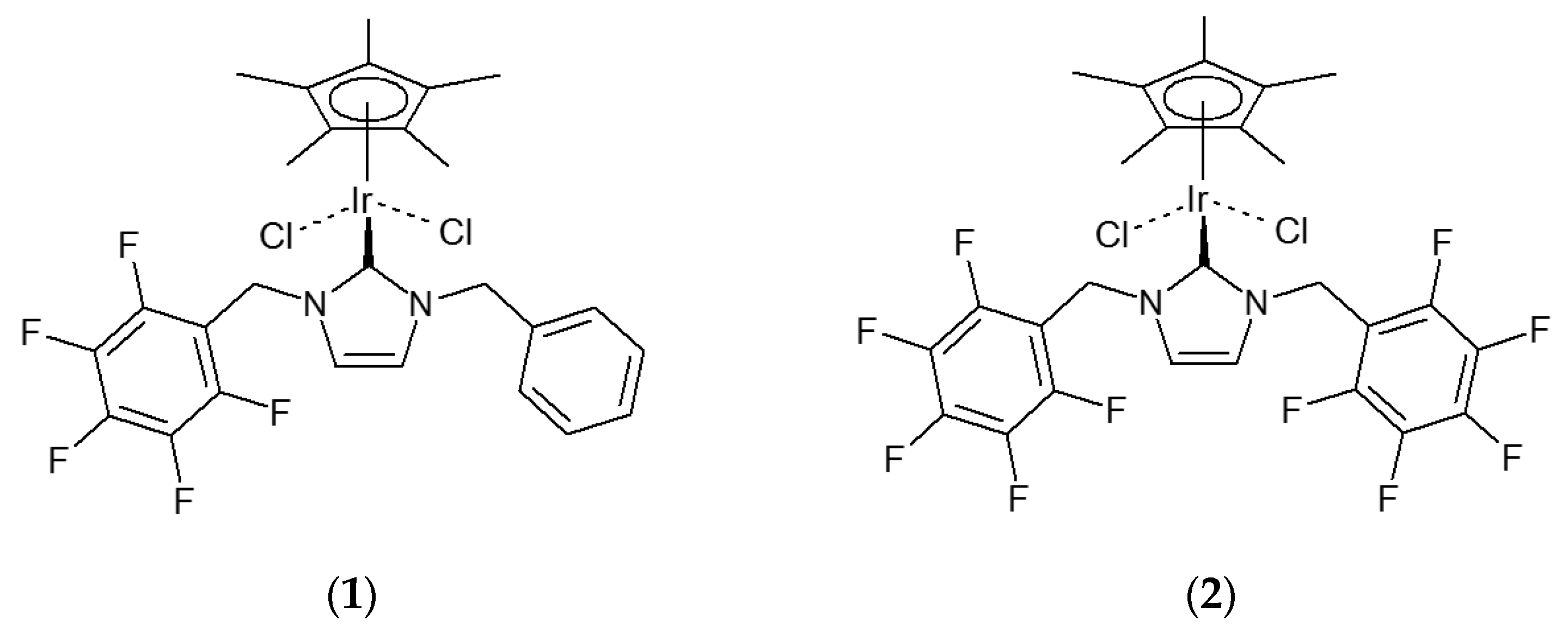
(η5-Pentamethylcyclopentadienyl)iridium{1,3-bis(2,3,4,5,6-pentafluorobenzyl)imidazolin-2-ylidene}dichloride
Abstract
1. Introduction
2. Results
3. Materials and Methods
3.1. Synthesis of (η5-Pentamethylcyclopentadienyl)iridium{1,3-bis(2,3,4,5,6-pentafluorobenzyl)imidazolin-2-ylidene}dichloride (2)
3.2. Single-Crystal XRD Determination
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Rufino-Felipe, E.; Valdés, H.; Germán-Acacio, J.M.; Reyes-Márquez, V.; Morales-Morales, D. Fluorinated N-Heterocyclic carbene complexes. Applications in catalysis. J. Organomet. Chem. 2020, 921, 121364. [Google Scholar] [CrossRef]
- Marr, A.C.; Pollock, C.L.; Saunders, G.C. Base free dynamic kinetic resolution of secondary alcohols using ‘piano stool’ complexes of N-heterocyclic carbenes. Organometallics 2007, 26, 3283–3285. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, Y.; Morgan, P.J.; Jackson, R.E.; Liu, X.; Saunders, G.C.; Lorenzini, F.; Marr, A.C. Designing effective homogeneous catalysis for glycerol valorisation: Selective synthesis of a value-added aldehyde from 1,3-propanediol via hydrogen transfer catalysed by a highly recyclable, fluorinated Cp*Ir(NHC) catalyst. Catal. Today 2018, 307, 248–259. [Google Scholar] [CrossRef]
- Pachal, S.R.; Saunders, G.C.; Weston, J.K. Complex Dependent Regioselectivity in the Nucleophilic Substitution of Fluorine of a Coordinated Tetrafluoropyridyl N-Heterocycle Carbene. Inorg. Chim. Acta 2013, 394, 558–562. [Google Scholar] [CrossRef]
- Thomas, H.P.; Marr, A.C.; Morgan, P.J.; Saunders, G.C. Tethering of Pentamethylcyclopentadienyl and N-Heterocycle Stabilized Carbene Ligands by Intramolecular 1,4-Addition to a Polyfluorophenyl Substituent. Organometallics 2018, 37, 1339–1341. [Google Scholar] [CrossRef]
- Marr, A.C.; Morgan, P.J.; Saunders, G.C.; Thomas, H.P. Conversion of haloform to carbonate by iridium N-heterocyclic carbene complexes and silver(I) oxide. Dalton Trans. 2019, 48, 1947–1949. [Google Scholar] [CrossRef] [PubMed]
- Morgan, P.J.; Hanson-Heine, M.W.D.; Thomas, H.P.; Saunders, G.C.; Marr, A.C.; Licence, P. C–F Bond Activation of a Perfluorinated Ligand Leading to Nucleophilic Fluorination of an Organic Electrophile. Organometallics 2020, 39, 2116–2124. [Google Scholar] [CrossRef]
- Thomas, H.P.; Wang, Y.; Lorenzini, F.; Coles, S.J.; Horton, P.N.; Marr, A.C.; Saunders, G.C. Cyclometallation via Carbon-Fluorine Bond Activation induced by Silver Particles. Organometallics 2017, 36, 960–963. [Google Scholar] [CrossRef]
- Marr, A.C.; Saunders, G.C.; Thomas, H.P.; Wang, Y. Orthometallation in Iridium N-Heterocycle Stabilized Carbene Complexes via Carbon—Halogen and Carbon—Hydrogen Bond Activation induced by Silver Nanoparticles. Inorg. Chim. Acta 2019, 486, 1–7. [Google Scholar] [CrossRef]
- Aznarez, F.; Gao, W.-X.; Lin, Y.-J.; Hahn, F.E.; Jin, G.-X. Preparation of polynuclear NHC complexes by post-synthetic modification of half-sandwich rhodium and iridium complexes bearing C-azolato ligands. Dalton Trans. 2018, 47, 9442–9452. [Google Scholar] [CrossRef] [PubMed]
- Aznarez, F.; Iglesias, M.; Hepp, A.; Veit, B.; Sanz Miguel, P.J.; Oro, L.A.; Jin, G.-X.; Hahn, F.E. Iridium(III) Complexes Bearing Chelating Bis-NHC Ligands and Their Application in the Catalytic Reduction of Imines. Eur. J. Inorg. Chem. 2016, 2016, 4598–4603. [Google Scholar] [CrossRef]
- Aznarez, F.; Sanz Miguel, P.J.; Tan, T.T.Y.; Hahn, F.E. Preparation of Rhodium(III) Di-NHC Chelate Complexes Featuring Two Different NHC Donors via a Mild NaOAc-Assisted C–H Activation. Organometallics 2016, 35, 410–419. [Google Scholar] [CrossRef]
- Lockley, A.J.; Marr, A.C.; Saunders, G.C.; Thomas, H.P. Arene–polyfluoroarene π-π stacking between N-heterocyclic carbene ligands of pentamethylcyclopentadienyl group 9 metal complexes. J. Fluor. Chem. 2019, 220, 1–8. [Google Scholar] [CrossRef]
- Lampl, M.; Schlapp-Hackl, I.; Wurst, K.; Gelbrich, T.; Kopacka, H.; Muller, T.; Kreutz, C.; Naier, B.; Partl, G.J.; Kahlenberg, V.; et al. Synthetic and structural studies on pentafluorobenzylated imidazole systems. J. Fluor. Chem. 2019, 218, 51–62. [Google Scholar] [CrossRef]
- Bondi, A. van der Waals volumes and Radii. J. Phys. Chem. 1964, 68, 441–451. [Google Scholar] [CrossRef]
- White, C.; Yates, A.; Maitlis, P.M. (η5-Pentamethylcyclopentadienyl)rhodium and -iridium compounds. Inorg. Synth. 1992, 29, 228–234. [Google Scholar] [CrossRef]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Bourhis, L.J.; Dolomanov, O.V.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. The anatomy of a comprehensive constrained, restrained refinement program for the modern computing environment—Olex2 dissected. Acta Crystallogr. 2015, A71, 59–75. [Google Scholar] [CrossRef]

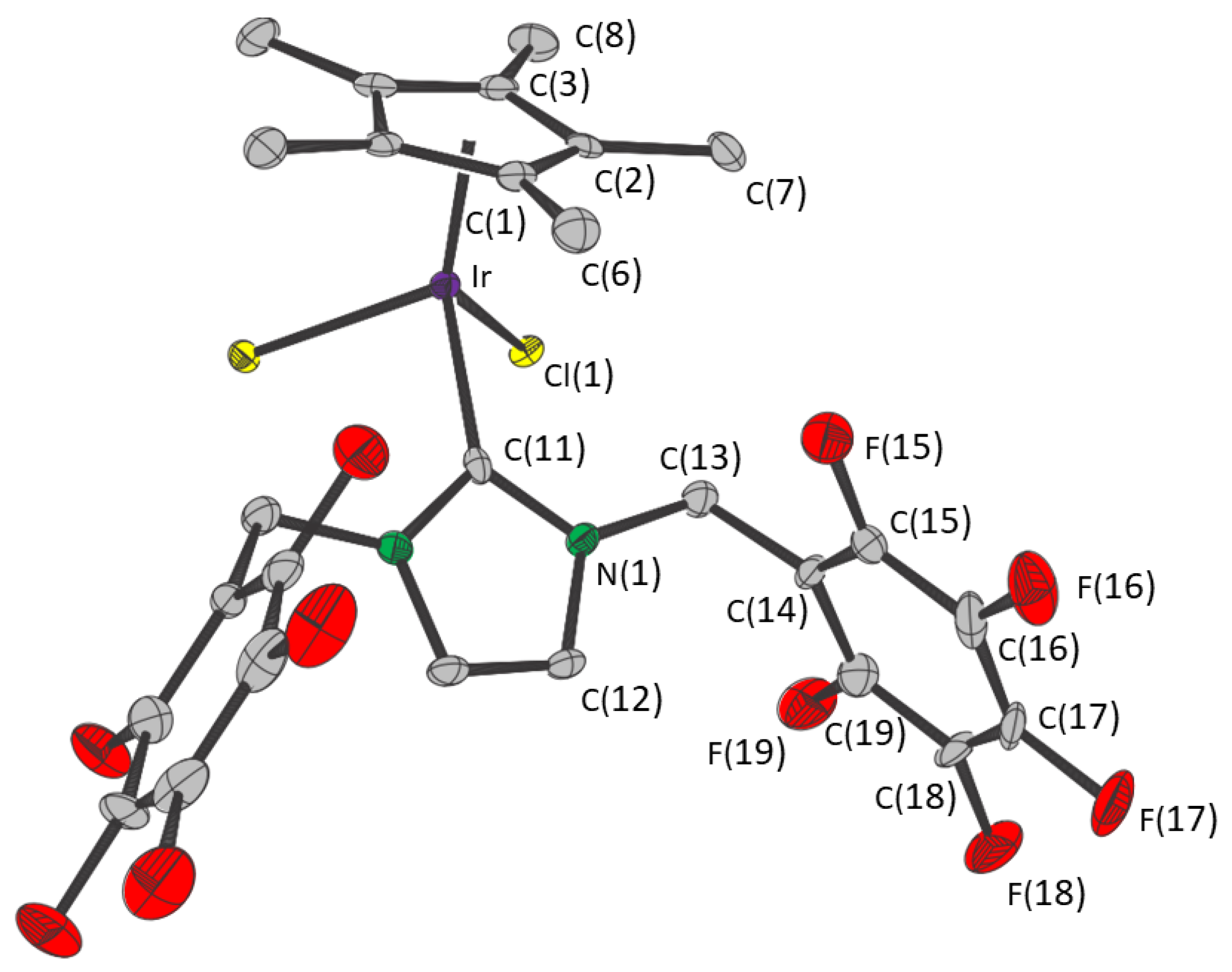
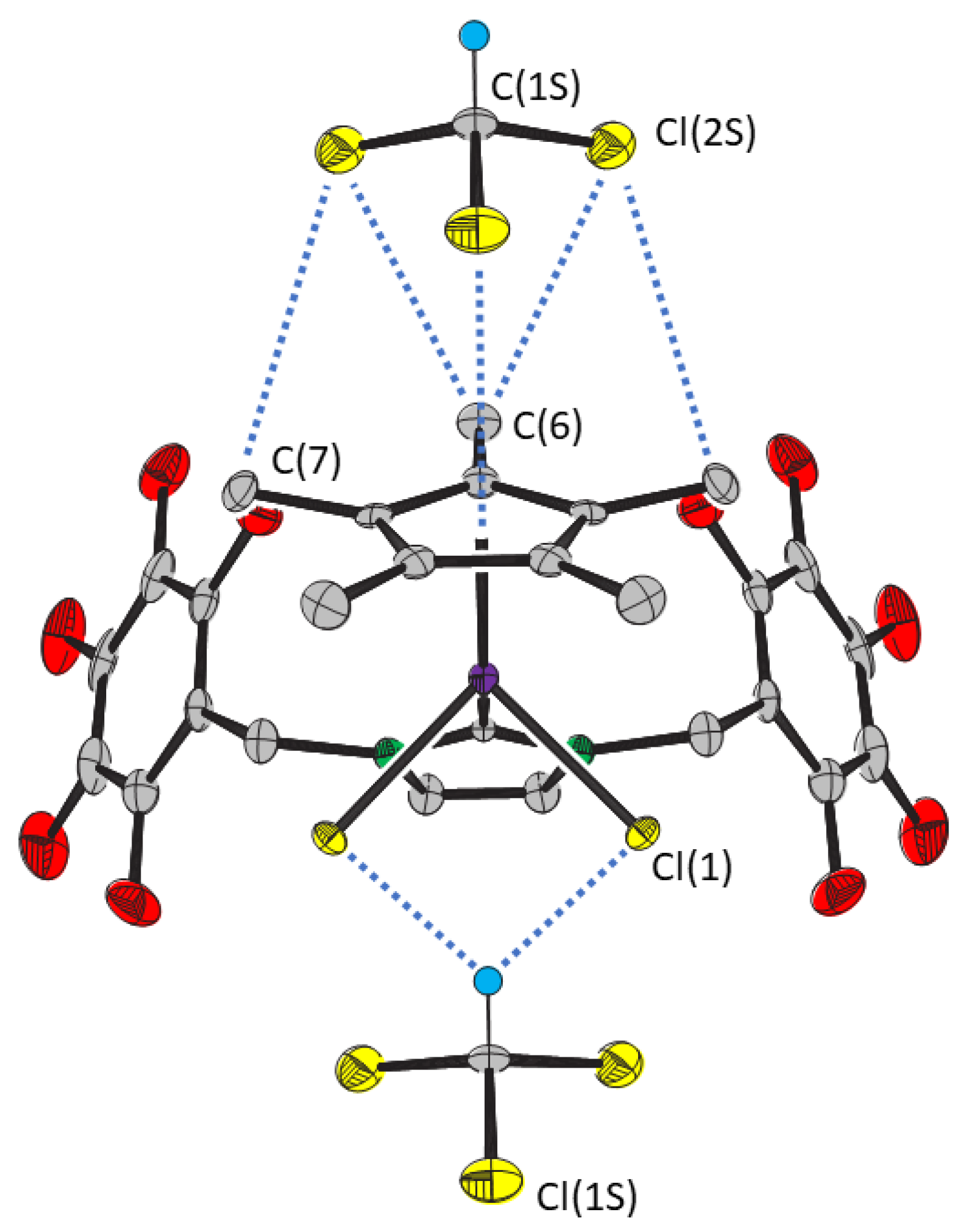
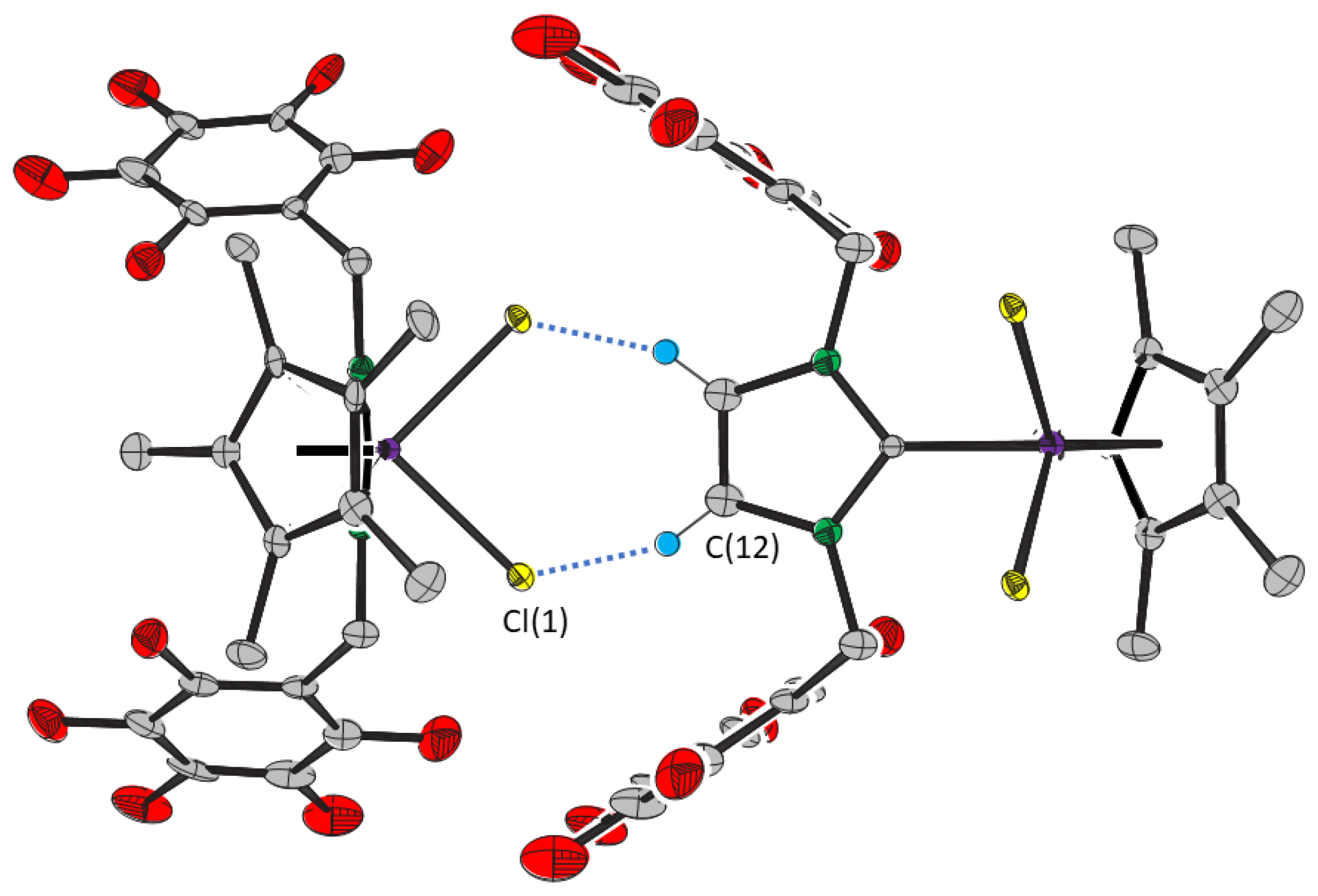
| Distance/Å | Angle/° | ||
|---|---|---|---|
| Cp*—Ir | 1.809(5) | Cp*—Ir—Cl(1) | 123.2(2) |
| Ir—Cl(1) | 2.4263(10) | Cp*—Ir—C(11) | 132.9(2) |
| Ir—C(11) | 2.049(6) | Cl(1)—Ir—Cl(1) | 87.71(5) |
| Cl(1)—Ir—C(11) | 88.35(12) | ||
| Cp*···Cl(1S) | 3.547(5) | Ir—Cp*···Cl(1S) | 174.8(2) |
| C(6)···Cl(2S) | 3.795(5) | ||
| C(7)···Cl(2S) | 3.830(5) | Ir—Cl(1)···C(1S) | 95.1(3) |
| Cl(1)···C(1S) | 3.458(8) | Ir—Cl(1)···C(12) | 103.9(1) |
| Cl(1)···C(12) | 3.449(5) | Cl(1)···C(12)—N(1) | 138.1(3) |
| Cl(1)···C(12)—C(12) | 107.1(3) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bandaru, S.K.; Marr, A.C.; Saunders, G.C. (η5-Pentamethylcyclopentadienyl)iridium{1,3-bis(2,3,4,5,6-pentafluorobenzyl)imidazolin-2-ylidene}dichloride. Molbank 2023, 2023, M1641. https://doi.org/10.3390/M1641
Bandaru SK, Marr AC, Saunders GC. (η5-Pentamethylcyclopentadienyl)iridium{1,3-bis(2,3,4,5,6-pentafluorobenzyl)imidazolin-2-ylidene}dichloride. Molbank. 2023; 2023(2):M1641. https://doi.org/10.3390/M1641
Chicago/Turabian StyleBandaru, Sravan K., Andrew C. Marr, and Graham C. Saunders. 2023. "(η5-Pentamethylcyclopentadienyl)iridium{1,3-bis(2,3,4,5,6-pentafluorobenzyl)imidazolin-2-ylidene}dichloride" Molbank 2023, no. 2: M1641. https://doi.org/10.3390/M1641
APA StyleBandaru, S. K., Marr, A. C., & Saunders, G. C. (2023). (η5-Pentamethylcyclopentadienyl)iridium{1,3-bis(2,3,4,5,6-pentafluorobenzyl)imidazolin-2-ylidene}dichloride. Molbank, 2023(2), M1641. https://doi.org/10.3390/M1641






