Diethyl 2-Cyano-3-oxosuccinate
Abstract
1. Introduction
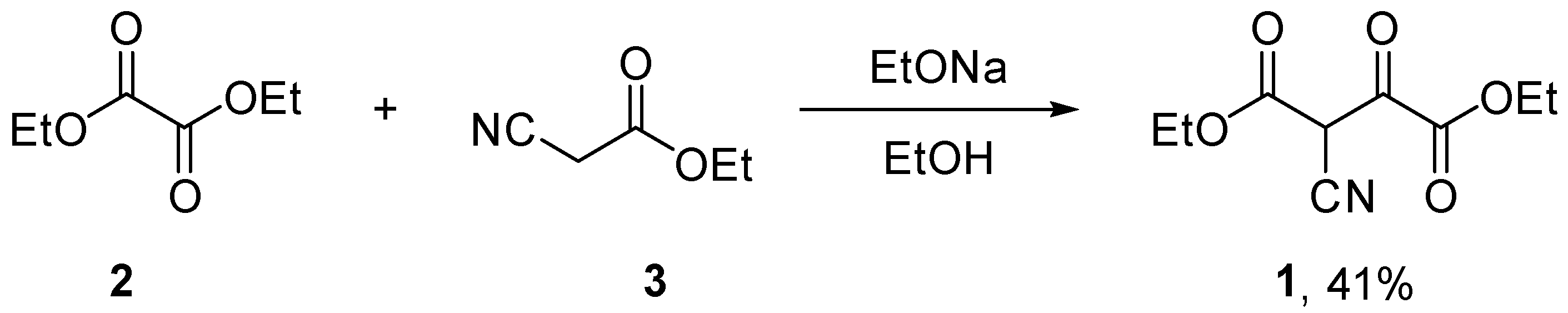
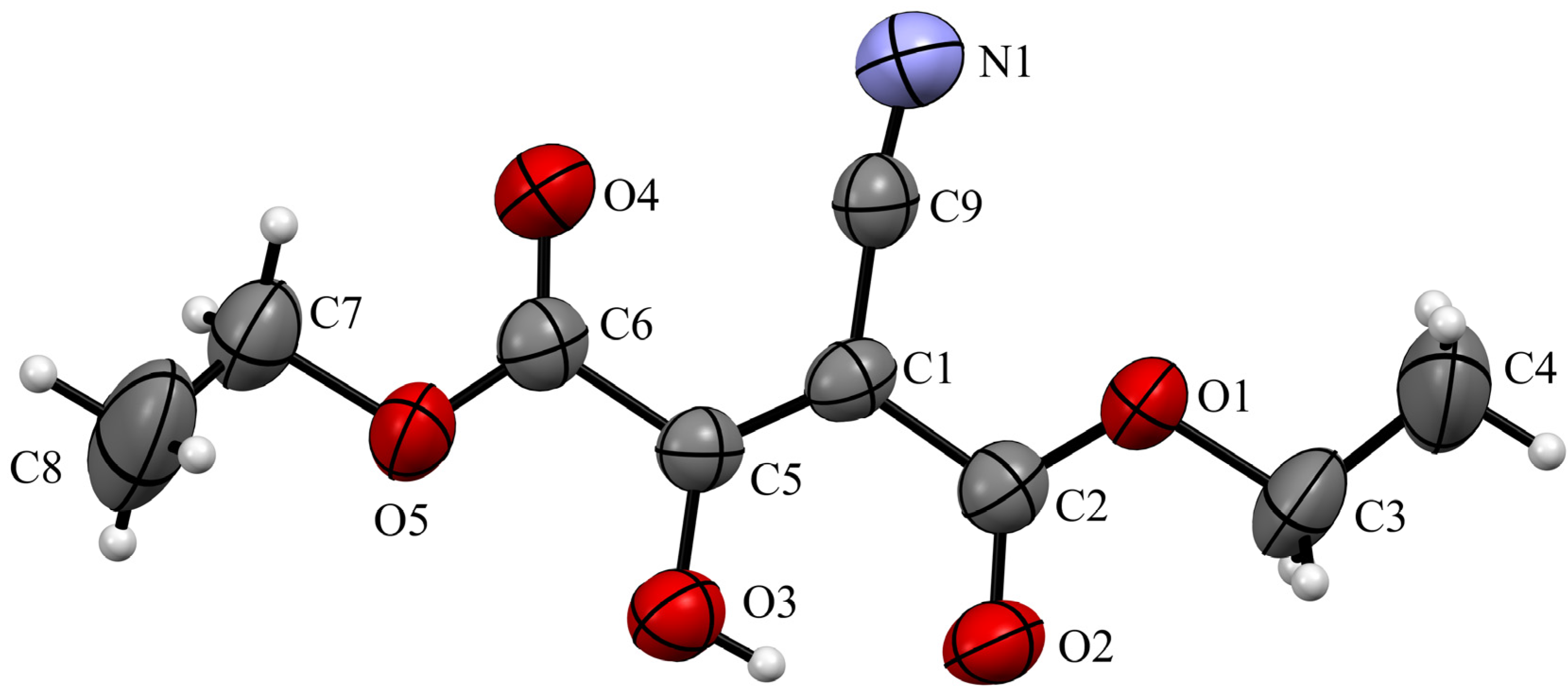
2. Results
3. Experimental Section
Diethyl 2-cyano-3-oxosuccinate (1)
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bertini, C. Azione del sodio e dell’idrossilammina sopra i fenilidrazoni dell’etere cianacetico e sintesi dell’etere cianossalacetico. Gazz. Chim. Ital. 1901, 23, 578–588. [Google Scholar]
- Trimbach, M. Action des chloroxalates d’ethyle et de methyle sur les ethers cyanacetiques. Bull. Soc. Chim. Fr. 1905, 33, 372–375. [Google Scholar]
- Schenone, P.; Fossa, P.; Menozzi, G. Reaction of 2-2-Dimethylaminomethylene-1,3-diones with Dinucleophiles. X. Synthesis of 5-Substituted Ethyl or Methyl 4-Isoxazolecarboxylates and Methyl 4-(2,2-dimethyl-1-propyl)-5-isoxazolescarboxylate. J. Heterocycl. Chem. 1991, 28, 453. [Google Scholar] [CrossRef]
- Borodkin, S.; Jonsson, S.; Cocolas, G.H.; McKee, R.L. Orotic acid analogs. 2,5-Disubstituted 6-hydroxy-4-carboxypyrimidines. J. Med. Chem. 1967, 10, 290–291. [Google Scholar] [CrossRef] [PubMed]
- Schinz, H.; Hinder, M. Preparation and properties of various α-keto β-lactones. Helv. Chim. Acta. 1947, 30, 1349–1373. [Google Scholar] [CrossRef] [PubMed]
- Kolind-Andersen, H.; Dyrnesli, R.; Lawesson, S.-O. Alkylation of some potential 2-oxoesters. Bull. Soc. Chim. Belg. 1975, 84, 341–360. [Google Scholar] [CrossRef]
- Jefford, C.W.; Kubota, T.; Zaslona, A. Intramolecular Carbenoid Reactions of Pyrrole Derivatives. A Total Synthesis of (±)-Ipalbidine. Helv. Chim. Acta. 1986, 69, 2048–2061. [Google Scholar] [CrossRef]
- Lienhard, G.E.; Jencks, W.P. The Reaction of Carbanions with N,S-Diacetylcysteamine. A Model for Enzymatic Carbon-Carbon Condensation. J. Am. Chem. Soc. 1965, 87, 3863–3874. [Google Scholar] [CrossRef] [PubMed]
- Curreli, F.; Belov, D.S.; Do Kwon, Y.; Ramesh, R.; Furimsky, A.M.; O’Loughlin, K.; Byrge, P.C.; Iyer, L.V.; Mirsalis, J.C.; Kurkin, A.V.; et al. Structure-based lead optimization to improve antiviral potency and ADMET properties of phenyl-1H-pyrrole-carboxamide entry inhibitors targeted to HIV-1 gp120. Eur. J. Med. Chem. 2018, 154, 367–391. [Google Scholar] [CrossRef]
- Shahak, I.; Bergman, E.D. Organic Fluorine Compounds. XVI. The Preparation and Reactions of Ethyl Ethoxalylfluoroacetate. J. Chem. Soc. 1960, 3225–3229. [Google Scholar] [CrossRef]
- Yamazaki, S.; Sugiura, H.; Ohashi, S.; Ishizuka, K.; Saimu, R.; Mikata, Y.; Ogawa, A. Intramolecular [2 + 2] and [4 + 2] Cycloaddition Reactions of Cinnamylamides of Ethenetricarboxylate in Sequential Processes. J. Org. Chem. 2016, 81, 10863–10886. [Google Scholar] [CrossRef] [PubMed]
- Pandey, G.; Adate, P.A.; Puranik, V.G. Organocatalytic dynamic kinetic resolution via conjugate addition: Synthesis of chiral trans-2,5-dialkylcyclohexanones. Org. Biomol. Chem. 2012, 10, 8260–8267. [Google Scholar] [CrossRef] [PubMed]
- Bailley, N.A.; Coates, E.; Robertson, G.B.; Bonati, F.; Ugo, R. The crystal and molecular structures of acetylacetonatobiscarbonyl-rhodium(I) and of some fluoro-substituted complexes. Chem. Commun. 1967, 1041–1042. [Google Scholar] [CrossRef]
- Kashiwabara, T.; Tanaka, M. Arylglyoxylrhodium Complexes, Their Thermolysis, and Attempted Generation by Carbonylation of an Aroylrhodium Complex. Organometallics 2011, 30, 239–244. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. A 2008, A64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Brandenburg, K.; Brendt, M. DIAMOND, Release 2.1 d.; Crystal Impact GbR: Bonn, Germany, 2000; pp. 302–303. [Google Scholar]
- Albert, A.; Serjeant, E.P. The Determination of Ionization Constants. A Laboratory Manual, 3rd ed.; Chapman and Hall: New York, NY, USA, 1984. [Google Scholar]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Markov, O.N.; Moiseev, A.E.; Tarasevich, B.N.; Tafeenko, V.A.; Beloglazkina, E.K.; Shtil, A.A.; Finko, A.V. Diethyl 2-Cyano-3-oxosuccinate. Molbank 2023, 2023, M1634. https://doi.org/10.3390/M1634
Markov ON, Moiseev AE, Tarasevich BN, Tafeenko VA, Beloglazkina EK, Shtil AA, Finko AV. Diethyl 2-Cyano-3-oxosuccinate. Molbank. 2023; 2023(2):M1634. https://doi.org/10.3390/M1634
Chicago/Turabian StyleMarkov, Oleg N., Alexander E. Moiseev, Boris N. Tarasevich, Victor A. Tafeenko, Elena K. Beloglazkina, Alexander A. Shtil, and Alexander V. Finko. 2023. "Diethyl 2-Cyano-3-oxosuccinate" Molbank 2023, no. 2: M1634. https://doi.org/10.3390/M1634
APA StyleMarkov, O. N., Moiseev, A. E., Tarasevich, B. N., Tafeenko, V. A., Beloglazkina, E. K., Shtil, A. A., & Finko, A. V. (2023). Diethyl 2-Cyano-3-oxosuccinate. Molbank, 2023(2), M1634. https://doi.org/10.3390/M1634







