[6-(Thiophen-2-yl)-2,2′-bipyridine]bis(triphenylphosphine) Copper(I) Tetrafluoroborate
Abstract
1. Introduction
2. Results and Discussion
2.1. Characterization with NMR Spectroscopy
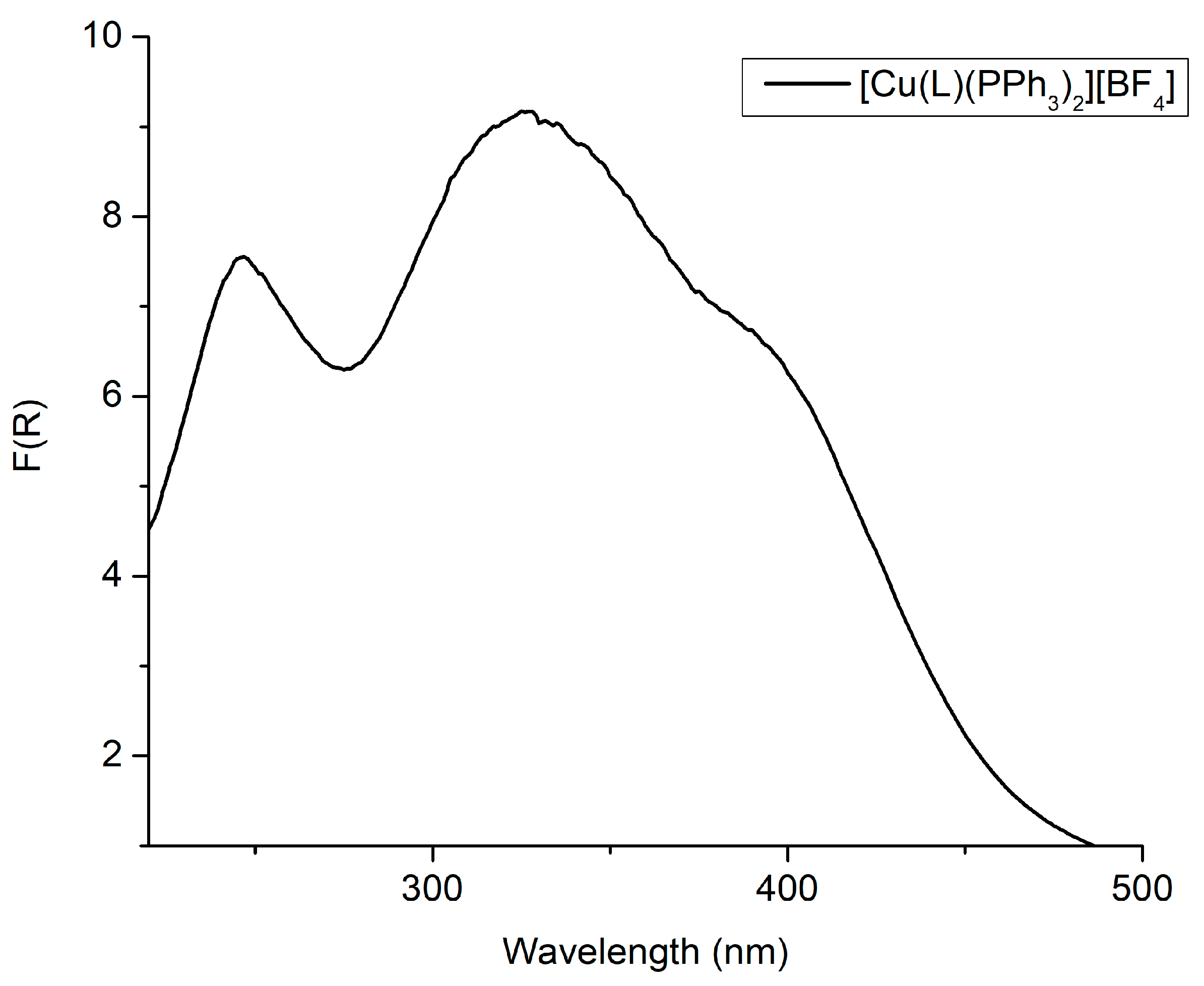
2.2. Absorption Spectrum
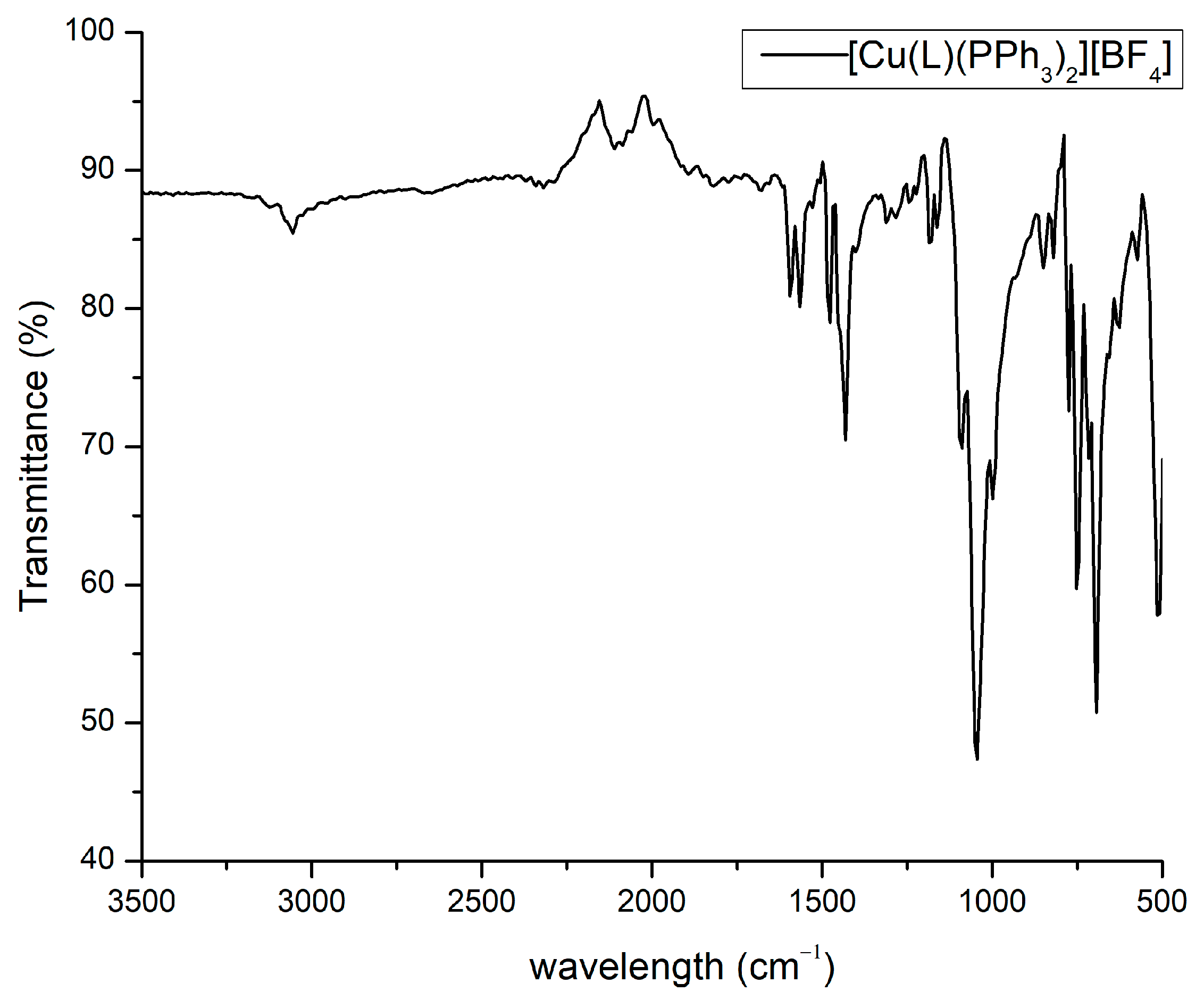
2.3. IR Spectroscopy
2.4. Emission Spectrum–Quantum Yield Calculation
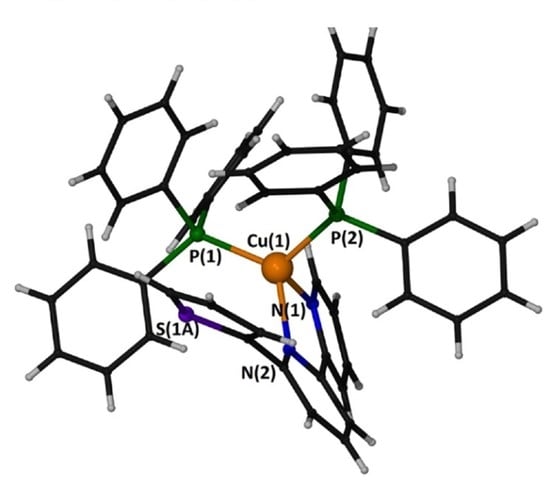
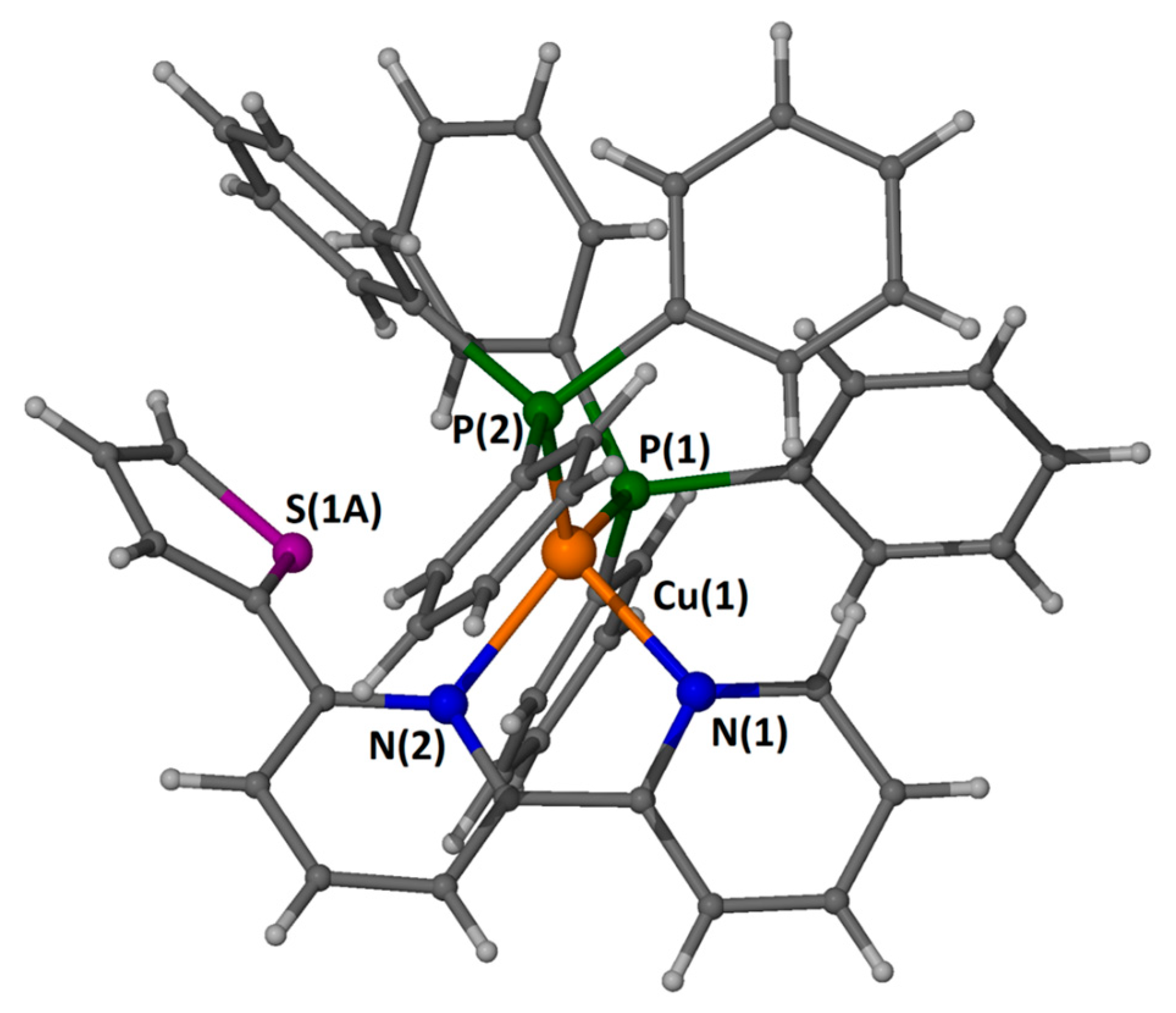
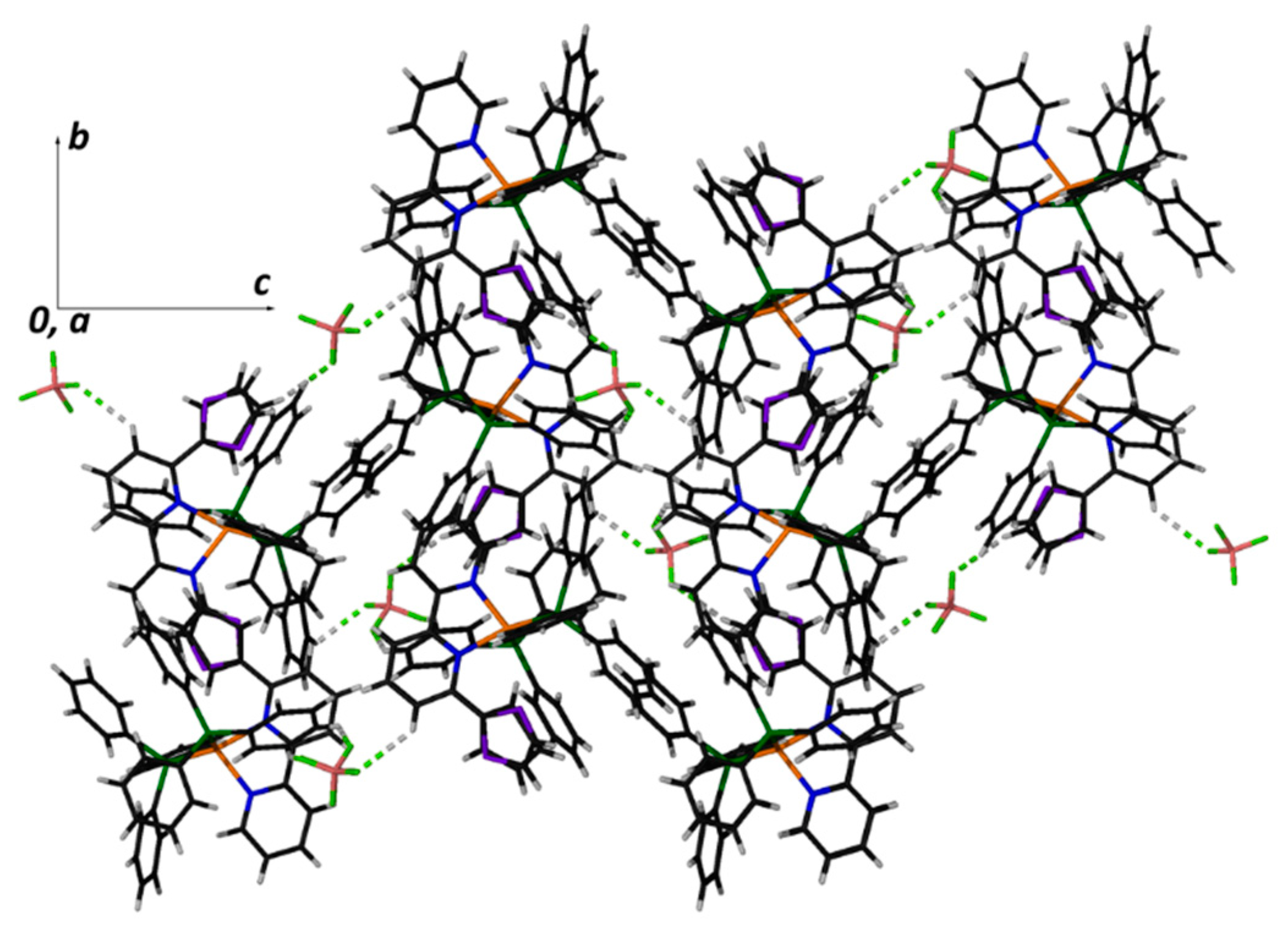
2.5. Description of the Structure
3. Materials and Methods
3.1. Materials
3.2. Methods
3.3. Crystal Structure Determination
3.4. Synthesis
Synthesis of the Complex [Cu(L)(PPh3)2][BF4] (1)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Min, J.; Zhang, Q.; Sun, W.; Cheng, Y.; Wang, L. Neutral copper(i) phosphorescent complexes from their ionic counterparts with 2-(2′-quinolyl)benzimidazole and phosphine mixed ligands. Dalton Trans. 2010, 40, 686–693. [Google Scholar] [CrossRef]
- Zhang, Q.; Ding, J.; Cheng, Y.; Wang, L.; Xie, Z.; Jing, X.; Wang, F. Novel Heteroleptic CuI Complexes with Tunable Emission Color for Efficient Phosphorescent Light-Emitting Diodes. Adv. Funct. Mater. 2007, 17, 2983–2990. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhou, Q.; Cheng, Y.; Wang, L.; Ma, D.; Jing, X.; Wang, F. Highly Efficient Electroluminescence from Green-Light-Emitting Electrochemical Cells Based on CuI Complexes. Adv. Funct. Mater. 2006, 16, 1203–1208. [Google Scholar] [CrossRef]
- Armaroli, N.; Accorsi, G.; Holler, M.; Moudam, O.; Nierengarten, J.-F.; Zhou, Z.; Wegh, R.T.; Welter, R. Highly Luminescent CuI Complexes for Light-Emitting Electrochemical Cells. Adv. Mater. 2006, 18, 1313–1316. [Google Scholar] [CrossRef]
- Safin, D.A.; Mitoraj, M.P.; Robeyns, K.; Filinchuk, Y.; Velde, C.M.L.V. Luminescent mononuclear mixed ligand complexes of copper(i) with 5-phenyl-2,2′-bipyridine and triphenylphosphine. Dalton Trans. 2015, 44, 16824–16832. [Google Scholar] [CrossRef]
- Li, G.F.; Zhang, X.Y.; Li, R.F.; Liu, X.F. Synthesis and properties of two new Cu(I) complexes based on 5,6-substituted imidazole-2,9-dimethyl-1,10-phenanthroline and triphenylphosphine. Russ. J. Gen. Chem. 2016, 86, 387–390. [Google Scholar] [CrossRef]
- Steen, R.O.; Nurkkala, L.J.; Angus-Dunne, S.J.; Schmitt, C.X.; Constable, E.C.; Riley, M.J.; Bernhardt, P.V.; Dunne, S.J. The Role of Isomeric Effects on the Luminescence Lifetimes and Electrochemistry of Oligothienyl-Bridged Dinuclear Tris(2,2′-bipyridine)ruthenium(II) Complexes. Eur. J. Inorg. Chem. 2008, 2008, 1784–1794. [Google Scholar] [CrossRef]
- Leoni, E.; Mohanraj, J.; Holler, M.; Mohankumar, M.; Nierengarten, I.; Monti, F.; Sournia-Saquet, A.; Delavaux-Nicot, B.; Nierengarten, J.-F.; Armaroli, N. Heteroleptic Copper(I) Complexes Prepared from Phenanthroline and Bis-Phosphine Ligands: Rationalization of the Photophysical and Electrochemical Properties. Inorg. Chem. 2018, 57, 15537–15549. [Google Scholar] [CrossRef]
- Beaudelot, J.; Oger, S.; Peruško, S.; Phan, T.-A.; Teunens, T.; Moucheron, C.; Evano, G. Photoactive Copper Complexes: Properties and Applications. Chem. Rev. 2022, 122, 16365–16609. [Google Scholar] [CrossRef] [PubMed]
- Andrés-Tomé, I.; Fyson, J.; Dias, F.B.; Monkman, A.P.; Iacobellis, G.; Coppo, P. Copper(i) complexes with bipyridyl and phosphine ligands: A systematic study. Dalton Trans. 2012, 41, 8669–8674. [Google Scholar] [CrossRef]
- Bozic-Weber, B.; Constable, E.C.; Housecroft, C.E.; Kopecky, P.; Neuburger, M.; Zampese, J.A. The intramolecular aryl embrace: From light emission to light absorption. Dalton Trans. 2011, 40, 12584–12594. [Google Scholar] [CrossRef] [PubMed]
- Son, J.-K.; Zhao, L.-X.; Basnet, A.; Thapa, P.; Karki, R.; Na, Y.; Jahng, Y.; Jeong, T.C.; Jeong, B.-S.; Lee, C.-S.; et al. Synthesis of 2,6-diaryl-substituted pyridines and their antitumor activities. Eur. J. Med. Chem. 2008, 43, 675–682. [Google Scholar] [CrossRef] [PubMed]
- Constable, E.C.; Henney, R.P.G.; Leese, T.A.; Tocher, D.A. Cyclometallation reactions of 6-phenyl-2,2′-bipyridine; a potential C,N,N-donor analogue of 2,2′: 6′,2″-terpyridine. Crystal and molecular structure of dichlorobis(6-phenyl-2,2′-bipyridine)ruthenium(II). J. Chem. Soc. Dalton Trans. 1990, 2, 443–449. [Google Scholar] [CrossRef]
- Constable, E.C.; Henney, R.P.G.; Tocher, D.A. Co-ordination chemistry of 2-phenyl-6-(2-thienyl)pyridine and 2,6-bis(2-thienyl)pyridine; new ambidentate ligands. J. Chem. Soc. Dalton Trans. 1992, 16, 2467–2474. [Google Scholar] [CrossRef]
- Zhang, L.; Zuo, Q. A series of blue-green-yellow-red emitting Cu(I) complexes: Molecular structure and photophysical performance. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 223, 117280. [Google Scholar] [CrossRef]
- Shi, L.; Li, B.; Lu, S.; Zhu, D.; Li, W. Synthesis, characterization and oxygen-sensing properties of a novel luminescent Cu(I) complex. Appl. Organomet. Chem. 2009, 23, 379–384. [Google Scholar] [CrossRef]
- Yang, W.; Yang, W.; Liu, W.; Qin, W. Study on the synthesis, characterization, photophysical performance and oxygen-sensing behavior of a luminescent Cu(I) complex with large conjugation plane. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 104, 56–63. [Google Scholar] [CrossRef]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge Structural Database. Acta Crystallogr. Sect. B Struct. Sci. Cryst. Eng. Mater. 2016, 72, 171–179. [Google Scholar] [CrossRef]
- Lin, Y.-R.; Huang, J.-S.; Zhong, M.-H. [6-(4-Bromophenyl)-2,2′-bipyridine-κ2N,N′]bis(triphenylphosphine-κP)copper(I) tetrafluoridoborate. Acta Crystallogr. Sect. E Struct. Rep. Online 2011, 67, m1187. [Google Scholar] [CrossRef]
- Tikkanen, W.R.; Kueger, C.; Bomben, K.D.; Jolly, W.L.; Kaska, W.; Ford, P.C. Synthesis, characterization, and x-ray molecular structures of mono- and dinuclear copper complexes with 2,7-bis(2-pyridyl)-1,8-naphthyridine. Inorg. Chem. 1984, 23, 3633. [Google Scholar] [CrossRef]
- Sequeira, D.; Baptista, P.V.; Valente, R.; Piedade, M.F.M.; Garcia, M.H.; Morais, T.S.; Fernandes, A.R. Cu(I) complexes as new antiproliferative agents against sensitive and doxorubicin-resistant colorectal cancer cells: Synthesis, characterization, and mechanisms of action. Dalton Trans. 2021, 50, 1845. [Google Scholar] [CrossRef]
- Bruker, APEX 3. In SAINT, SHELXT; Bruker AXS Inc.: Fitchburg, WI, USA, 2016.
- Sheldrick, G.M. SADABS; University of Göttingen: Göttingen, Germany, 1996. [Google Scholar]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Hübschle, C.B.; Sheldrick, G.M.; Dittrich, B. ShelXle: A Qt graphical user interface for SHELXL. J. Appl. Crystallogr. 2011, 44, 1281–1284. [Google Scholar] [CrossRef] [PubMed]
- Spek, A.L. Structure validation in chemical crystallography. Acta Crystallogr. Sect. D Biol. Crystallogr. 2009, 65, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Barbour, L.J. X-Seed—A Software Tool for Supramolecular Crystallography. J. Supramol. Chem. 2001, 1, 189–191. [Google Scholar] [CrossRef]
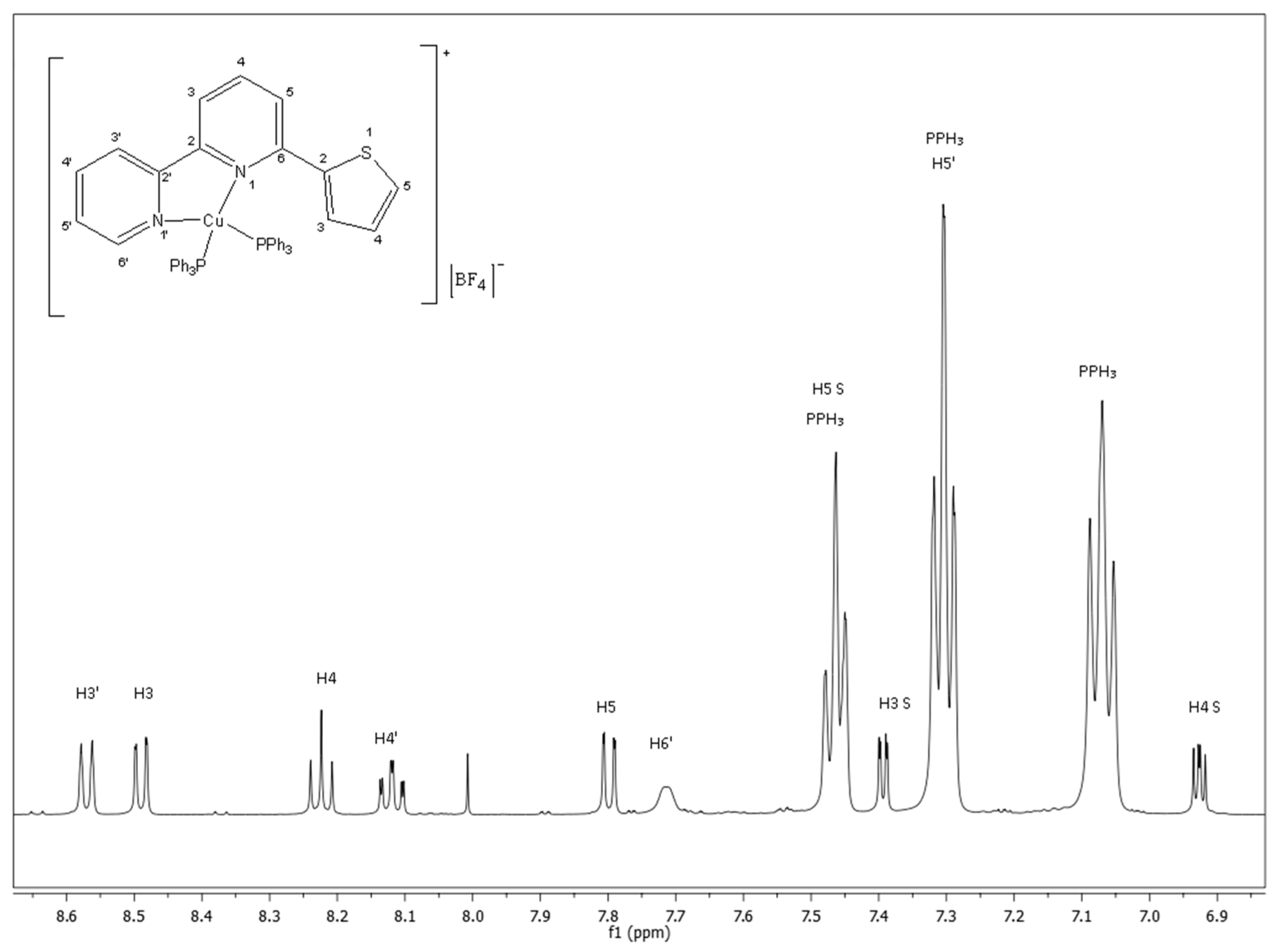

| H | L | Complex | Δδ = δcoml − δL |
|---|---|---|---|
| H6′ | 8.71 | 7.71 | −1.00 |
| H5′ | 7.45 | 7.31 | −0.14 |
| H4′ | 7.99 | 8.12 | 0.13 |
| H3′ | 8.57 | 8.57 | 0.00 |
| H3 | 8.38 | 8.49 | 0.11 |
| H4 | 7.97 | 8.23 | 0.26 |
| H5 | 7.90 | 7.80 | −0.10 |
| H3 (thiophene) | 7.59 | 7.40 | −0.19 |
| H4 (thiophene) | 7.20 | 6.93 | −0.27 |
| H5 (thiophene) | 7.83 | 7.47 | −0.36 |
| Bond Distances (Å) | Bond Angles (°) | ||
|---|---|---|---|
| Cu(1)-N(1) | 2.1127(19) | N(1)-Cu(1)-N(2) | 78.93(8) |
| Cu(1)-N(2) | 2.1409(18) | N(1)-Cu(1)-P(1) | 104.06(5) |
| Cu(1)-P(1) | 2.2747(5) | N(2)-Cu(1)-P(1) | 124.90(5) |
| Cu(1)-P(2) | 2.2830(6) | N(1)-Cu(1)-P(2) | 112.20(5) |
| N(2)-Cu(1)-P(2) | 107.64(5) | ||
| P(1)-Cu(1)-P(2) | 120.46(2) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kouvatsis, P.; Glykos, D.; Plakatouras, J.C.; Malandrinos, G. [6-(Thiophen-2-yl)-2,2′-bipyridine]bis(triphenylphosphine) Copper(I) Tetrafluoroborate. Molbank 2023, 2023, M1605. https://doi.org/10.3390/M1605
Kouvatsis P, Glykos D, Plakatouras JC, Malandrinos G. [6-(Thiophen-2-yl)-2,2′-bipyridine]bis(triphenylphosphine) Copper(I) Tetrafluoroborate. Molbank. 2023; 2023(1):M1605. https://doi.org/10.3390/M1605
Chicago/Turabian StyleKouvatsis, Panagiotis, Dimitrios Glykos, John C. Plakatouras, and Gerasimos Malandrinos. 2023. "[6-(Thiophen-2-yl)-2,2′-bipyridine]bis(triphenylphosphine) Copper(I) Tetrafluoroborate" Molbank 2023, no. 1: M1605. https://doi.org/10.3390/M1605
APA StyleKouvatsis, P., Glykos, D., Plakatouras, J. C., & Malandrinos, G. (2023). [6-(Thiophen-2-yl)-2,2′-bipyridine]bis(triphenylphosphine) Copper(I) Tetrafluoroborate. Molbank, 2023(1), M1605. https://doi.org/10.3390/M1605