4-(((4-Methoxyphenyl)amino)methyl)-N,N-dimethylaniline and 2-Methoxy-5-((phenylamino)methyl)phenol
Abstract
:1. Introduction
2. Results and Discussion
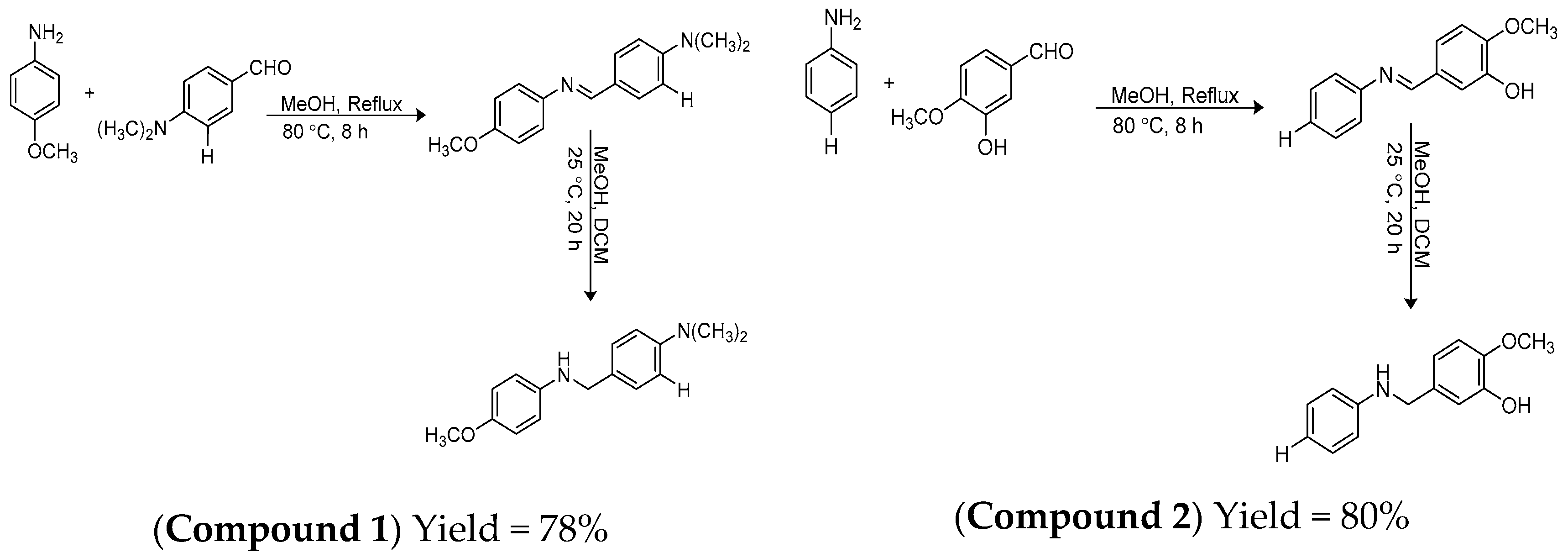
2.1. Synthesis of the Compounds
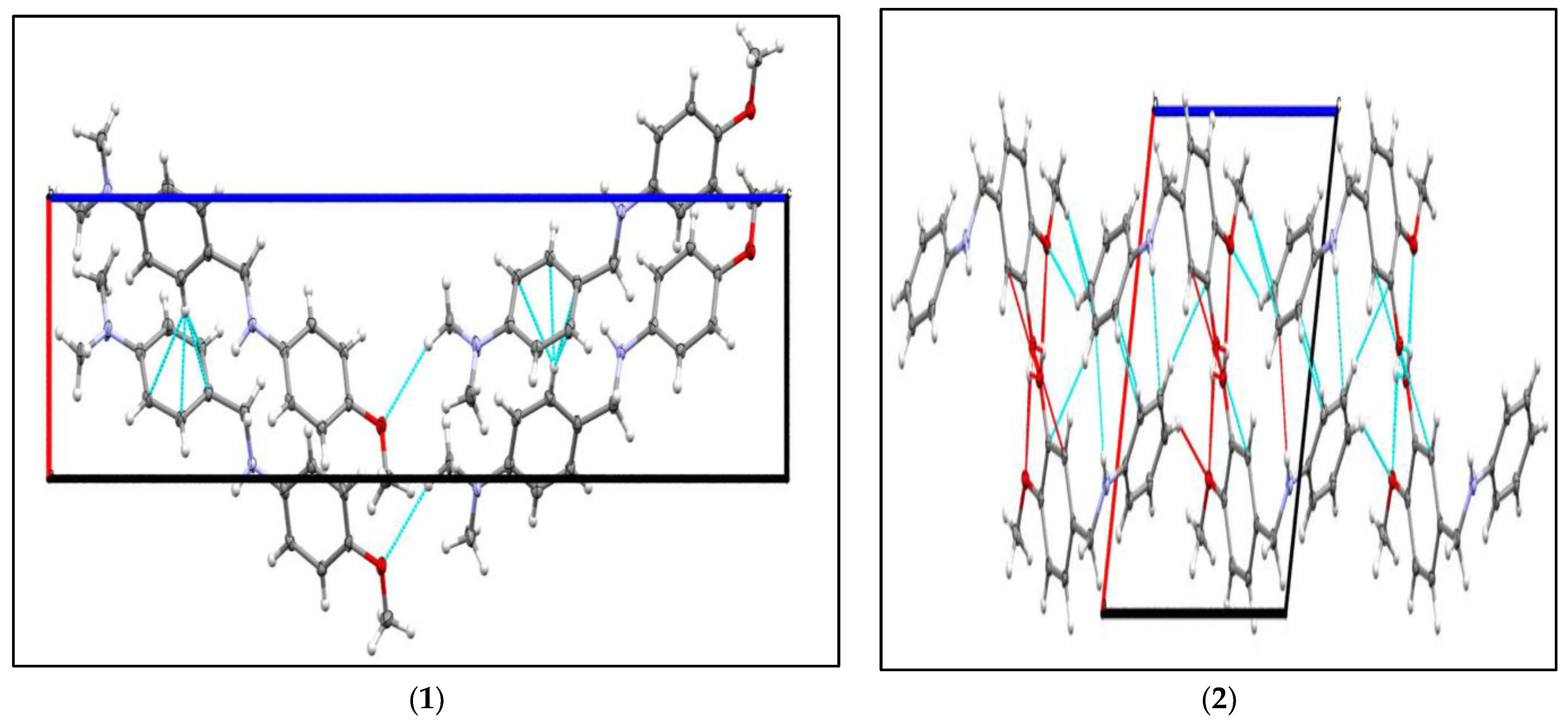
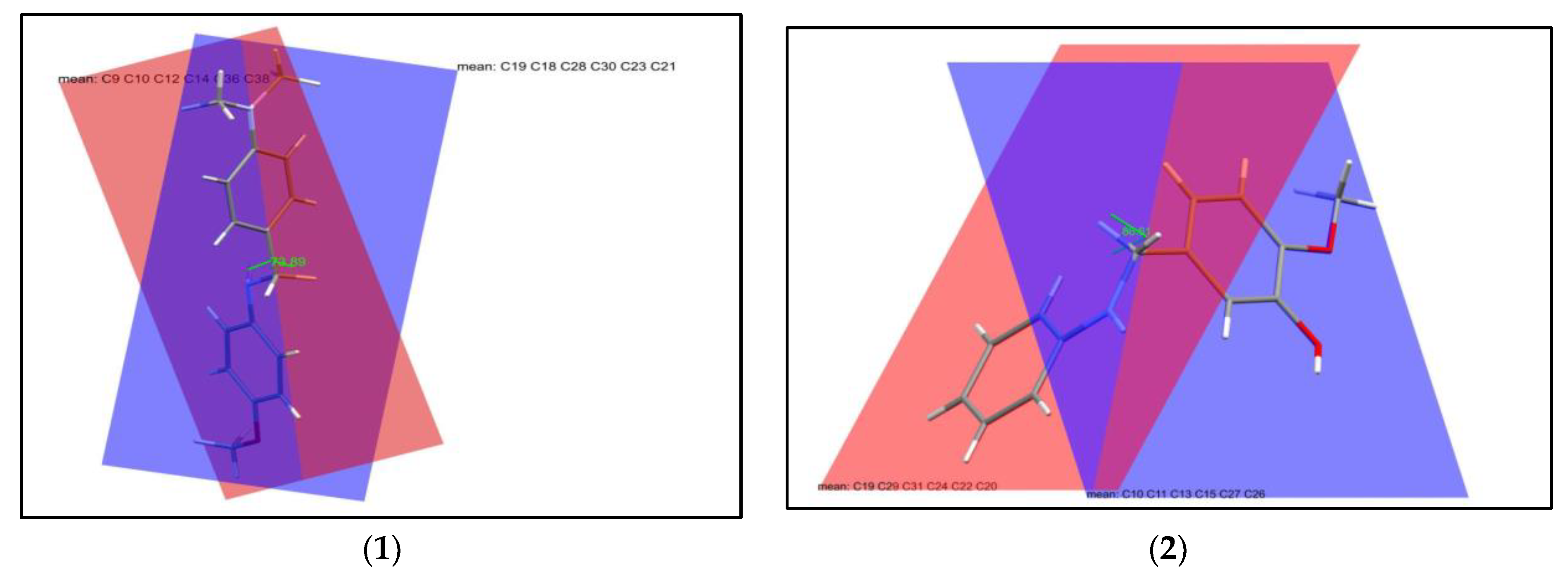
2.2. Molecular Structures of the Compounds
3. Materials and Methods
3.1. Synthesis of 4-(((4-Methoxyphenyl)amino)methyl)-N,N-dimethylaniline (1)
3.2. Synthesis of 2-Methoxy-5-((phenylamino)methyl)phenol (2)
3.3. Single Crystal X-ray Crystallography
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roose, P.; Eller, K.; Henkes, E.; Rossbacher, R.; Höke, H. Ullmann’s Encyclopedia of Industrial Chemistry; Weinheim Wiley-VCH-Verl: Weinheim, Germany, 2015; pp. 1–55. [Google Scholar]
- Wakamatsu, T.; Inaki, H.; Ogawa, A.; Watanabe, M.; Ban, Y. Reduction of nitriles and amides to amines with tetrabutylammonium borohydride in dichloromethane. Heterocycles 1980, 14, 1437–1440. [Google Scholar] [CrossRef]
- Severin, R.; Doye, S. The catalytic hydroamination of alkynes. Chem. Soc. Rev. 2007, 36, 1407–1420. [Google Scholar] [CrossRef]
- Hultzsch, K.C.; Gribkov, D.V.; Hampel, F. Non-metallocene rare earth metal catalysts for the diastereoselective and enantioselective hydroamination of aminoalkenes. J. Org. Chem. 2005, 690, 4441–4452. [Google Scholar] [CrossRef]
- Seayad, J.; Tillack, A.; Hartung, C.G.; Beller, M. Base-Catalyzed Hydroamination of Olefins: An Environmentally Friendly Route to Amines. Adv. Synth. Catal. 2002, 344, 795–813. [Google Scholar] [CrossRef]
- Aghera, A.K.; Parsania, P.H. A cleaner approach for reduction of some symmetric diimines using NaBH4. Ind. J. Chem. 2009, 48B, 438–442. [Google Scholar]
- Billman, J.H.; Diesing, A.C. Reduction of Schiff bases with sodium borohydride. J. Org. Chem. 1957, 22, 1068–1070. [Google Scholar] [CrossRef]
- Ajibade, P.A.; Andrew, F.P.; Fatokun, A.A.; Oluwalana, A.E. Synthesis, characterization and in vitro screening for anticancer potential of Mn(II), Co(II), Cu(II), Zn(II), and Pt(II) methoxyphenyl dithiocarbamato complexes. J. Mol. Struct. 2021, 1230, 129894. [Google Scholar] [CrossRef]
- Paca, A.M.; Ajibade, P.A. Bis-(N-ethylphenyldithiocarbamato)palladium(II) as molecular precursor for palladium sulfide nanoparticles. J. Mol. Struct. 2021, 12435, 130777. [Google Scholar] [CrossRef]
- Ajibade, P.A.; Fatokun, A.A.; Andrew, F.P. Synthesis, characterization and anticancer studies of Mn(II), Cu(II), Zn(II) and Pt(II) dithiocarbamate complexes-crystal structures of the Cu(II) and Pt(II) complexes. Inorg. Chim. Acta 2020, 504, 119431. [Google Scholar] [CrossRef]
- Jones, R.S.G. Tryptamine: A neuromodulator or neurotransmitter in mammalian brain? Prog. Neurobiol. 1982, 19, 117–139. [Google Scholar] [CrossRef]
- Knight, A.W.; Greenway, G.M. Relationship between structural attributes and observed electrogenerated chemiluminescence (ECL) activity of tertiary amines as potential analytes for the tris (2, 2-bipyridine) ruthenium(II) ECL reaction. A review. Analyst 1996, 121, 101R–106R. [Google Scholar] [CrossRef]
- Svejstrup, T.D.; Ruffoni, A.; Juliá, F.; Aubert, V.M.; Leonori, D. Synthesis of Arylamines via Aminium Radicals. Angew. Chem. 2017, 129, 15144–15148. [Google Scholar] [CrossRef] [Green Version]
- Kumar, C.T.K.; Keshavayya, J.; Rajesh, T.N.; Peethambar, S.K.; Ali, A.R.S. Synthesis, characterization, and biological activity of 5-phenyl-1, 3, 4-thiadiazole-2-amine incorporated azo dye derivatives. Org. Chem. Int. 2013, 2013, 370626. [Google Scholar]
- Pinheiro, H.M.; Touraud, E.; Thomas, O. Aromatic amines from azo dye reduction: Status review with emphasis on direct UV spectrophotometric detection in textile industry wastewaters. Dyes Pigm. 2004, 61, 121–139. [Google Scholar] [CrossRef]
- Hogarth, G. Metal-dithiocarbamate complexes: Chemistry and biological activity. Mini Rev. Med. Chem. 2012, 12, 1202–1215. [Google Scholar] [CrossRef] [PubMed]
- Cozzi, P.G. Metal–Salen Schiff base complexes in catalysis: Practical aspects. Chem. Soc. Rev. 2004, 33, 410–421. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.F.; Xia, H.T.; Yang, S.P.; Wang, D.Q. 2-[(4-Chlorophenyl) aminomethyl]-6-methoxyphenol. Acta Cryst. 2007, E63, o3561. [Google Scholar] [CrossRef]
- Shu, H.; Yu, N.S.; Xie, G.L.; Chen, L.Z. 2-[(4-Methoxyanilino) methyl] phenol. Acta Cryst. 2011, E67, o2304. [Google Scholar] [CrossRef]
- Qu, Y.; Tian, L.J.; Dong, J. 2-(Anilinomethyl) phenol. Acta Cryst. 2007, E63, o4832. [Google Scholar] [CrossRef]
- Caracelli, I.; Maganhi, S.H.; Cardoso, J. D-O.; Cunha, R.L.; Vega-Teijido, M.A.; Zukerman-Schpector, J.; Tiekink, E.R. Crystallographic and docking (Cathepsins B, K, L and S) studies on bioactive halotelluroxetanes. Z. Kristallogr. 2018, 233, 113. [Google Scholar] [CrossRef] [Green Version]
- Bruker: APEX2; Bruker AXS Inc.: Madison, WI, USA, 2011.
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. A complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, A64, 339–341. [Google Scholar]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C27, 3–8. [Google Scholar]

| 1 | 2 | |
|---|---|---|
| Formula | C16H20N2O | C14H15NO2 |
| Dcalc/g cm−3 | 1.230 | 1.330 |
| μ(MoKα)/mm−1 | 0.077 | 0.089 |
| Formula Weight | 256.34 | 229.27 |
| Colour | colourless | colourless |
| Shape | block | Plank |
| Size/mm3 | 0.78 × 0.34 × 0.32 | 0.38 × 0.21 × 0.14 |
| Crystal System | orthorhombic | monoclinic |
| Space Group | Pca21 | P21 |
| a/Å | 6.590(1) | 9.9964(2) |
| b/Å | 7.278(1) | 5.65940(10) |
| c/Å | 28.870(4) | 10.6027(2) |
| α/° | 90 | 90 |
| β/° | 90 | 107.3100(10) |
| γ/° | 90 | 90 |
| V/Å3 | 1384.6(3) | 572.666(19) |
| Z/Z’ | 4/1 | 2/1 |
| Wavelength/Å | 0.71073 | 0.71073 |
| Θmin/° | 1.411 | 2.012 |
| Θmax/° | 25.969 | 28.390 |
| Measured Refl. | 8346 | 11733 |
| Independent Refl. | 2612 | 2860 |
| Reflections Used | 2521 | 2767 |
| Rint | 0.0268 | 0.0190 |
| Parameters | 176 | 156 |
| Largest Peak | 0.289 | 0.252 |
| Deepest Hole | −0.200 | −0.177 |
| GooF | 1.189 | 1.047 |
| wR2 (all data) | 0.1507 | 0.0766 |
| wR2 | 0.1483 | 0.0756 |
| R1 (all data) | 0.0433 | 0.0290 |
| R1 | 0.0418 | 0.0279 |
| D | H | A | d(D-H)/Å | d(H-A)/Å | d(D-A)/Å | D-H-A/Deg | |
|---|---|---|---|---|---|---|---|
| 1 | C11 | H11 | N21 | 0.95 | 2.63 | 3.463(4) | 146.4 |
| 2 | O2 | H2 | O11 | 0.84 | 2.14 | 2.8885(15) | 148.9 |
| O2 | H2 | O21 | 0.84 | 2.29 | 2.9277(5) | 133.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ajibade, P.A.; Andrew, F.P. 4-(((4-Methoxyphenyl)amino)methyl)-N,N-dimethylaniline and 2-Methoxy-5-((phenylamino)methyl)phenol. Molbank 2021, 2021, M1274. https://doi.org/10.3390/M1274
Ajibade PA, Andrew FP. 4-(((4-Methoxyphenyl)amino)methyl)-N,N-dimethylaniline and 2-Methoxy-5-((phenylamino)methyl)phenol. Molbank. 2021; 2021(3):M1274. https://doi.org/10.3390/M1274
Chicago/Turabian StyleAjibade, Peter A., and Fartisincha P. Andrew. 2021. "4-(((4-Methoxyphenyl)amino)methyl)-N,N-dimethylaniline and 2-Methoxy-5-((phenylamino)methyl)phenol" Molbank 2021, no. 3: M1274. https://doi.org/10.3390/M1274
APA StyleAjibade, P. A., & Andrew, F. P. (2021). 4-(((4-Methoxyphenyl)amino)methyl)-N,N-dimethylaniline and 2-Methoxy-5-((phenylamino)methyl)phenol. Molbank, 2021(3), M1274. https://doi.org/10.3390/M1274





