4-Aminoalkyl Quinolin-2-one Derivatives via Knorr Cyclisation of ω-Amino-β-Keto Anilides
Abstract
:1. Introduction
2. Results
3. Materials and Methods
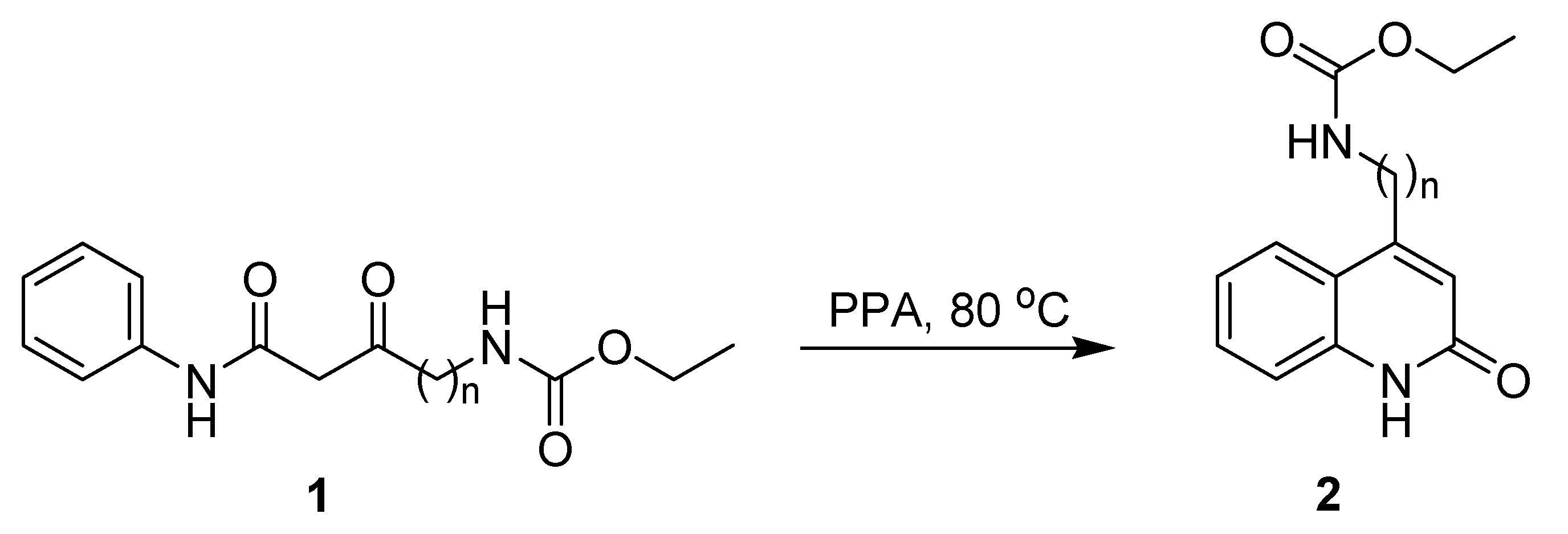
Synthetic Procedure
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shang, X.-F.; Natschke, S.L.M.; Liu, Y.-Q.; Guo, X.; Xu, X.-S.; Goto, M.; Li, J.-C.; Yang, G.-Z.; Lee, K.-H. Biologically active quinoline and quinazoline alkaloids part I. Med. Res. Rev. 2018, 38, 775–828. [Google Scholar] [CrossRef] [PubMed]
- Shang, X.-F.; Natschke, S.L.M.; Yang, G.-Z.; Liu, Y.-Q.; Guo, X.; Xu, X.-S.; Goto, M.; Li, J.-C.; Zhang, J.-Y.; Lee, K.-H. Biologically active quinoline and quinazoline alkaloids part II. Med. Res. Rev. 2018, 38, 1614–1660. [Google Scholar] [CrossRef] [PubMed]
- Nainwal, N.M.; Tasneem, S.; Akhtar, W.; Verma, G.; Khan, M.F.; Parvez, S.; Shaquiquzzaman, M.; Akhter, M.; Alam, M.M. Green recipes to quinoline: A review. Eur. J. Med. Chem. 2019, 164, 121–170. [Google Scholar] [CrossRef] [PubMed]
- Harry, N.A.; Ujwaldev, S.M.; Anilkumar, G. Recent advances and prospects in the metal-free synthesis of quinolines. Org. Biomol. Chem. 2020, 18, 9775–9790. [Google Scholar] [CrossRef]
- Tashima, T. The structural use of carbostyril in physiologically active substances. Bioorg. Med. Chem. Lett. 2015, 25, 3415–3419. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Harayama, T. A Concise and Versatile Synthesis of Viridicatin Alkaloids from Cyanoacetanilides. Org. Lett. 2009, 11, 1603–1606. [Google Scholar] [CrossRef]
- Tangella, Y.; Manasa, K.L.; Krishna, N.H.; Sridhar, B.; Kamal, A.; Babu, B.N. Regioselective Ring Expansion of Isatins with In Situ Generated α-Aryldiazomethanes: Direct Access to Viridicatin Alkaloids. Org. Lett. 2018, 20, 3639–3642. [Google Scholar] [CrossRef]
- Liang, P.; Zhang, Y.Y.; Yang, P.; Grond, S.; Zhang, Y.; Qian, Z.-J. Viridicatol and viridicatin isolated from a shark-gill-derived fungus Penicilliumpolonicum AP2T1 as MMP-2 and MMP-9 inhibitors in HT1080 cells by MAPKs signaling pathway and docking studies. Med. Chem. Res. 2019, 28, 1039–1048. [Google Scholar] [CrossRef]
- Einsiedler, M.; Jamieson, C.S.; Maskeri, M.A.; Houk, K.N.; Gulder, T.A.M. Fungal Dioxygenase AsqJ is Promiscuous and Bimodal: Substrate-Directed Formation of Quinolones versus Quinazolinones. Angew. Chem. Int. Ed. 2021, 60, 8297–8302. [Google Scholar] [CrossRef] [PubMed]
- Neff, S.A.; Lee, S.U.; Asami, Y.; Ahn, J.S.; Oh, H.; Baltrusaitis, J.; Gloer, J.B.; Wicklow, D.T. Aflaquinolones A−G: Secondary Metabolites from Marine and Fungicolous Isolates of Aspergillus spp. J. Nat. Prod. 2012, 75, 464–472. [Google Scholar] [CrossRef] [Green Version]
- Jia, W.-L.; Ces, S.V.; Fernández-Ibánez, M.A. Divergent Total Syntheses of Yaequinolone-Related Natural Products by Late-Stage C−H Olefination. J. Org. Chem. 2021, 86, 6259–6277. [Google Scholar] [CrossRef]
- Kalkhambkar, R.G.; Kulkarni, G.M.; Kamanavalli, C.M.; Premkumar, N.; Asdaq, S.M.B.; Sun, C.M. Synthesis and biological activities of some new fluorinated coumarins and 1-aza coumarins. Eur. J. Med. Chem. 2008, 43, 2178–2188. [Google Scholar] [CrossRef] [PubMed]
- Bonnefous, C.; Payne, J.E.; Roppe, J.; Zhuang, H.; Chen, X.; Symons, K.T.; Nguyen, P.M.; Sablad, M.; Rozenkrants, N.; Zhang, Y.; et al. Discovery of Inducible Nitric Oxide Synthase (iNOS) Inhibitor Development Candidate KD7332, Part 1: Identification of a Novel, Potent, and Selective Series of Quinolinone iNOS Dimerization Inhibitors that are Orally Active in Rodent Pain Models. J. Med. Chem. 2009, 52, 3047–3062. [Google Scholar] [CrossRef] [PubMed]
- Skepper, C.K.; Armstrong, D.; Balibar, C.J.; Bauer, D.; Bellamacina, C.; Benton, B.M.; Bussiere, D.; De Pascale, G.; De Vicente, J.; Dean, C.R.; et al. Topoisomerase Inhibitors Addressing Fluoroquinolone Resistance in Gram-Negative Bacteria. J. Med. Chem. 2020, 63, 7773–7816. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Yang, R.; Zhang-Negrerie, D.; Wang, J.; Du, Y.; Zhao, K. One-Pot Synthesis of 3-Hydroxyquinolin-2(1H)-ones from NPhenylacetoacetamide via PhI(OCOCF3)2-Mediated α-Hydroxylation and H2SO4-Promoted Intramolecular Cyclization. J. Org. Chem. 2013, 78, 5385–5392. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, Q.; Zhang, D.; Xin, X.; Zhang, R.; Zhou, F.; Dong, D. PPA-Mediated C-C Bond Formation: A Synthetic Route to Substituted Indeno[2,1-c]quinolin-6(7H)-ones. Org. Lett. 2013, 15, 776–779. [Google Scholar] [CrossRef]
- Sai, K.K.S.; Gilbert, T.M.; Klumpp, D.A. Knorr Cyclizations and Distonic Superelectrophiles. J. Org. Chem. 2007, 72, 9761–9764. [Google Scholar] [CrossRef] [PubMed]
- Ryabukhin, D.S.; Gurskaya, L.Y.; Fukin, G.K.; Vasilyev, A.V. Superelectrophilic activation of N-aryl amides of 3-arylpropynoic acids: Synthesis of quinolin-2(1H)-one derivatives. Tetrahedron 2014, 70, 6428–6443. [Google Scholar] [CrossRef]
- Guan, M.; Pang, Y.; Zhang, J.; Zhao, Y. Pd-Catalyzed sequential β-C(sp3)–H arylation and intramolecular amination of δ-C(sp2)–H bonds for synthesis of quinolinones via an N,O-bidentate directing group. Chem. Commun. 2016, 52, 7043–7046. [Google Scholar] [CrossRef]
- Han, J.; Wu, X.; Zhang, Z.; Wang, L. Palladium-catalyzed arylation/cyclization/desulfonation cascades toward 4-aryl quinolin-2(1H)-ones with diaryliodonium salts. Tetrahedron Lett. 2017, 58, 3433–3436. [Google Scholar] [CrossRef]
- Silva, V.L.M.; Silva, A.M.S. Palladium-Catalysed Synthesis and Transformation of Quinolones. Molecules 2019, 24, 228. [Google Scholar] [CrossRef] [Green Version]
- Peng, J.-B.; Chen, B.; Qi, X.; Ying, J.; Wu, X.-F. Palladium-catalyzed synthesis of quinolin-2(1H)-ones: The unexpected reactivity of azodicarboxylate. Org. Biomol. Chem. 2018, 16, 1632–1635. [Google Scholar] [CrossRef]
- Cheng, Y.-C.; Chen, Y.-Y.; Chuang, C.-P. Cobalt salt-catalyzed carbocyclization reactions of α-bromo-N-phenylacetamide derivatives. Org. Biomol. Chem. 2017, 15, 2020–2032. [Google Scholar] [CrossRef]
- Petersen, W.F.; Taylor, R.J.K.; Donald, J.R. Photoredox-catalyzed procedure for carbamoyl radical generation: 3,4-dihydroquinolin-2-one and quinolin-2-one synthesis. Org. Biomol. Chem. 2017, 15, 5831–5845. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, C.; He, J.-Y.; Bai, Q.-F.; Feng, G. Silver-Catalyzed Decarboxylative Radical Addition/Cyclization of Oxamic Acids with Alkenes towards quinolin-2-ones. Synlett 2020, 31, 1517–1522. [Google Scholar] [CrossRef]
- Fan, H.; Pan, P.; Zhang, Y.; Wang, W. Synthesis of 2-Quinolinones via a Hypervalent Iodine(III)-Mediated Intramolecular Decarboxylative Heck-Type Reaction at Room Temperature. Org. Lett. 2018, 20, 7929–7932. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, S.; Hara, K.; Hashimoto, H.; Hirayama, Y.; Champagne, P.A.; Houk, K.N.; Tang, Y.; Watanabe, K. Enzymatic one-step ring contraction for quinolone biosynthesis. Nat. Commun. 2018, 9, 2826. [Google Scholar] [CrossRef]
- Tang, H.; Tang, Y.; Kurnikov, I.V.; Liao, H.-J.; Chan, N.-L.; Kurnikova, M.G.; Guo, Y.; Chang, W.-C. Harnessing the Substrate Promiscuity of Dioxygenase AsqJ and Developing Efficient Chemoenzymatic Synthesis for Quinolones. ACS Catal. 2021, 11, 7186–7192. [Google Scholar] [CrossRef]
- Selig, P.; Bach, T. Enantioselective Total Synthesis of the Melodinus Alkaloid (+)-Meloscine. Angew. Chem. Int. Ed. 2008, 47, 5082–5084. [Google Scholar] [CrossRef]
- Selig, P.; Herdtweck, E.; Bach, T. Total Synthesis of Meloscine by a [2+2]-Photocycloaddition/Ring-Expansion Route. Chem. Eur. J. 2009, 15, 3509–3525. [Google Scholar] [CrossRef]
- Brandes, S.; Selig, P.; Bach, T. Stereoselective Intra-and Intermolecular [2+2] Photocycloaddition Reactions of 4-(2′-Aminoethyl)quinolones. Synlett 2004, 2588–2590. [Google Scholar] [CrossRef]
- Selig, P.; Bach, T. Photochemistry of 4-(2¢-Aminoethyl)quinolones: Enantioselective Synthesis of Tetracyclic Tetrahydro-1aH-pyrido[4′,3′:2,3]-cyclobuta[1,2-c] Quinoline-2,11(3H,8H)-diones by Intra-and Intermolecular [2+2]-Photocycloaddition Reactions in Solution. J. Org. Chem. 2006, 71, 5662–5673. [Google Scholar] [CrossRef] [PubMed]
- Yanev, P.; Angelov, P. Synthesis of functionalised β-keto amides by aminoacylation/domino fragmentation of β-enamino amides. Beilstein J. Org. Chem. 2018, 14, 2602–2606. [Google Scholar] [CrossRef] [PubMed]
| Product | n | Yield (%) |
|---|---|---|
| 2a | 1 | 90 |
| 2b | 2 | 80 |
| 2c | 3 | 85 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Angelov, P.; Velichkova, S.; Yanev, P. 4-Aminoalkyl Quinolin-2-one Derivatives via Knorr Cyclisation of ω-Amino-β-Keto Anilides. Molbank 2021, 2021, M1266. https://doi.org/10.3390/M1266
Angelov P, Velichkova S, Yanev P. 4-Aminoalkyl Quinolin-2-one Derivatives via Knorr Cyclisation of ω-Amino-β-Keto Anilides. Molbank. 2021; 2021(3):M1266. https://doi.org/10.3390/M1266
Chicago/Turabian StyleAngelov, Plamen, Stilyana Velichkova, and Pavel Yanev. 2021. "4-Aminoalkyl Quinolin-2-one Derivatives via Knorr Cyclisation of ω-Amino-β-Keto Anilides" Molbank 2021, no. 3: M1266. https://doi.org/10.3390/M1266
APA StyleAngelov, P., Velichkova, S., & Yanev, P. (2021). 4-Aminoalkyl Quinolin-2-one Derivatives via Knorr Cyclisation of ω-Amino-β-Keto Anilides. Molbank, 2021(3), M1266. https://doi.org/10.3390/M1266







