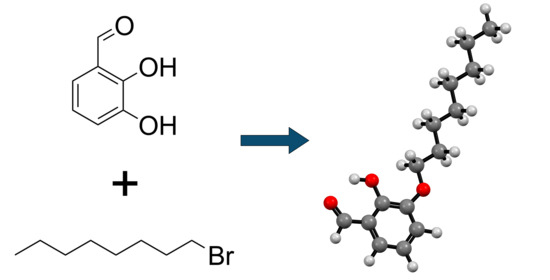
2-Hydroxy-3-octyloxybenzaldehyde
Abstract
:1. Introduction
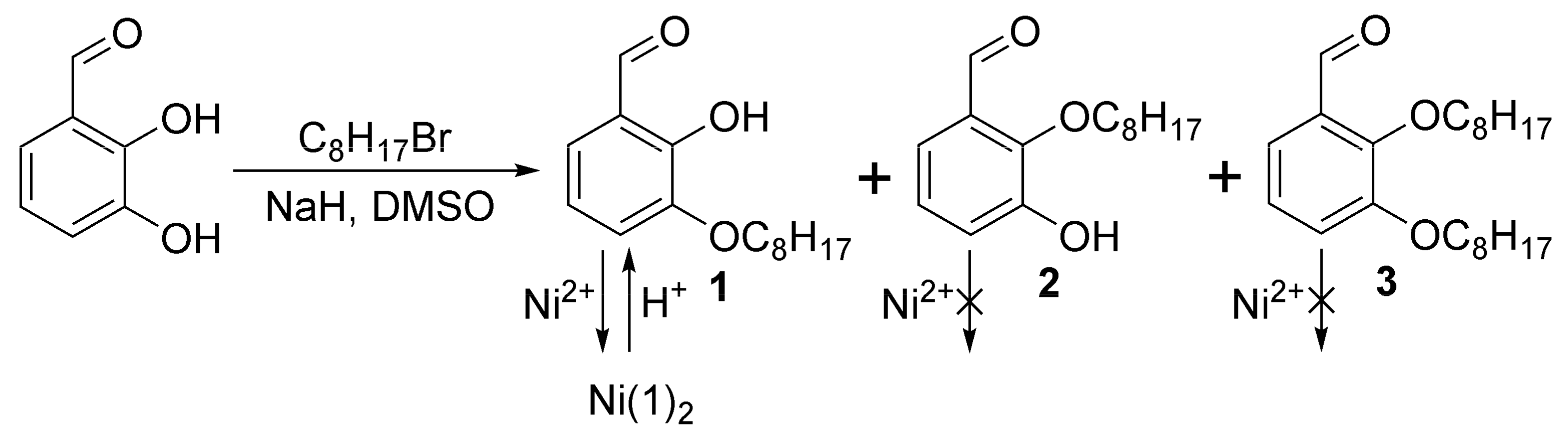
2. Results
3. Materials and Methods
3.1. General Considerations
3.2. Synthesis of 2-Hydroxy-3-octyloxybenzaldehyde
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vereshchagin, A.A.; Lukyanov, D.A.; Kulikov, I.R.; Panjwani, N.A.; Alekseeva, E.A.; Behrends, J.; Levin, O.V. The Fast and the Capacious: A [Ni(Salen)]-TEMPO Redox-Conducting Polymer for Organic Batteries. Batter. Supercaps 2021, 4, 336–346. [Google Scholar] [CrossRef]
- Chepurnaya, I.A.; Karushev, M.P.; Alekseeva, E.V.; Lukyanov, D.A.; Levin, O.V. Redox-conducting polymers based on metal- salen complexes for energy storage applications. Pure Appl. Chem. 2020, 92, 1239–1258. [Google Scholar] [CrossRef]
- Alekseeva, E.V.; Chepurnaya, I.A.; Malev, V.V.; Timonov, A.M.; Levin, O.V. Polymeric nickel complexes with salen-type ligands for modification of supercapacitor electrodes: Impedance studies of charge transfer and storage properties. Electrochim. Acta 2017, 225, 378–391. [Google Scholar] [CrossRef]
- Eliseeva, S.N.; Alekseeva, E.V.; Vereshchagin, A.A.; Volkov, A.I.; Vlasov, P.S.; Konev, A.S.; Levin, O.V. Nickel-Salen Type Polymers as Cathode Materials for Rechargeable Lithium Batteries. Macromol. Chem. Phys. 2017, 218, 1700361. [Google Scholar] [CrossRef]
- Chen, J.; Wagner, P.; Tong, L.; Wallace, G.G.; Officer, D.L.; Swiegers, G.F. A Porphyrin-Doped Polymer Catalyzes Selective, Light-Assisted Water Oxidation in Seawater. Angew. Chem. Int. Ed. 2012, 51, 1907–1910. [Google Scholar] [CrossRef] [PubMed]
- De Castro, B.; Ferreira, R.; Freire, C.; García, H.; Palomares, E.J.; Sabater, M.J. Photochemistry of nickel salen based complexes and relevance to catalysis. New J. Chem. 2002, 26, 405–410. [Google Scholar] [CrossRef]
- Wesley Jeevadason, A.; Kalidasa Murugavel, K.; Neelakantan, M.A. Review on Schiff bases and their metal complexes as organic photovoltaic materials. Renew. Sustain. Energy Rev. 2014, 36, 220–227. [Google Scholar] [CrossRef]
- Konev, A.S.; Kayumov, M.Y.; Karushev, M.P.; Novoselova, Y.V.; Lukyanov, D.A.; Alekseeva, E.V.; Levin, O.V. Polymeric Metal Salen-Type Complexes as Catalysts for Photoelectrocatalytic Hydrogen Peroxide Production. ChemElectroChem 2018, 5, 3138–3142. [Google Scholar] [CrossRef]
- Novozhilova, M.V.; Smirnova, E.A.; Karushev, M.P.; Timonov, A.M.; Malev, V.V.; Levin, O.V. Synthesis and study of catalysts of electrochemical oxygen reduction reaction based on polymer complexes of nickel and cobalt with Schiff bases. Russ. J. Electrochem. 2016, 52, 1183–1190. [Google Scholar] [CrossRef]
- Sukwattanasinitt, M.; Nantalaksakul, A.; Potisatityuenyong, A.; Tuntulani, T.; Chailapakul, O.; Praphairakait, N. An Electrochemical Sensor from a Soluble Polymeric Ni−salen Complex. Chem. Mater. 2003, 15, 4337–4339. [Google Scholar] [CrossRef]
- Martin, C.S.; Dadamos, T.R.L.; Teixeira, M.F.S. Development of an electrochemical sensor for determination of dissolved oxygen by nickel–salen polymeric film modified electrode. Sens. Actuators B Chem. 2012, 175, 111–117. [Google Scholar] [CrossRef]
- Martins, T.S.; Bott-Neto, J.L.; Raymundo-Pereira, P.A.; Ticianelli, E.A.; Machado, S.A.S. An electrochemical furosemide sensor based on pencil graphite surface modified with polymer film Ni-salen and Ni(OH)2/C nanoparticles. Sens. Actuators B Chem. 2018, 276, 378–387. [Google Scholar] [CrossRef]
- Dahm, C.E.; Peters, D.G. Catalytic reduction of α, ω-dihaloalkanes with nickel(I) salen as a homogeneous-phase and polymer-bound mediator. J. Electroanal. Chem. 1996, 406, 119–129. [Google Scholar] [CrossRef]
- Vilas-Boas, M.; Santos, I.C.; Henderson, M.J.; Freire, C.; Hillman, A.R.; Vieil, E. Electrochemical Behavior of a New Precursor for the Design of Poly[Ni(salen)]-Based Modified Electrodes. Langmuir 2003, 19, 7460–7468. [Google Scholar] [CrossRef] [Green Version]
- Keller, F.; Rippert, A.J. New, Axially Chiral, Bimetallic Catalysts for Asymmetric Alkylation of Aldehydes with Diethylzinc. Helv. Chim. Acta 1999, 82, 125–137. [Google Scholar] [CrossRef]
- Malev, V.V.; Levin, O.V.; Timonov, A.M. Quasi-equilibrium voltammetric curves resulting from the existence of two immobile charge carriers within electroactive polymer films. Electrochim. Acta 2013, 108, 313–320. [Google Scholar] [CrossRef]
- Han, S.; Zhang, F.-F.; Qian, H.-Y.; Chen, L.-L.; Pu, J.-B.; Xie, X.; Chen, J.-Z. Design, syntheses, structure–activity relationships and docking studies of coumarin derivatives as novel selective ligands for the CB2 receptor. Eur. J. Med. Chem. 2015, 93, 16–32. [Google Scholar] [CrossRef] [PubMed]
- 3-Substituted Coumarin Derivative and Use Thereof; Shanghai Inst Materia Medica: Shanghai, China; University Zhejiang: Hangzhou, China, 2013; pp. 1–33.
- Liu, D.; Shen, J.; Xu, K.; Zhang, W. Salviandic Acid A Intermediate and Preparation Method Thereof. CN108358761B, 13 March 2018. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Novoselova, J.V.; Vereshchagin, A.A.; Kalnin, A.Y.; Lukyanov, D.A.; Levin, O.V. 2-Hydroxy-3-octyloxybenzaldehyde. Molbank 2021, 2021, M1264. https://doi.org/10.3390/M1264
Novoselova JV, Vereshchagin AA, Kalnin AY, Lukyanov DA, Levin OV. 2-Hydroxy-3-octyloxybenzaldehyde. Molbank. 2021; 2021(3):M1264. https://doi.org/10.3390/M1264
Chicago/Turabian StyleNovoselova, Julia V., Anatoliy A. Vereshchagin, Arseniy Y. Kalnin, Daniil A. Lukyanov, and Oleg V. Levin. 2021. "2-Hydroxy-3-octyloxybenzaldehyde" Molbank 2021, no. 3: M1264. https://doi.org/10.3390/M1264
APA StyleNovoselova, J. V., Vereshchagin, A. A., Kalnin, A. Y., Lukyanov, D. A., & Levin, O. V. (2021). 2-Hydroxy-3-octyloxybenzaldehyde. Molbank, 2021(3), M1264. https://doi.org/10.3390/M1264