Pyridinium Pentachloridooxomolybdate(V), (PyH)2[MoOCl5], Revisited: Single-Crystal X-ray Structure Determination
Abstract
:1. Introduction
2. Results
3. Materials and Methods
3.1. General
3.2. Synthesis
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schwarz, G.; Mendel, R.R.; Ribbe, M.W. Molybdenum cofactors, enzymes and pathways. Nature 2009, 460, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Burgmayer, S.J.N.; Stiefel, E.I. Molybdenum enzymes, cofactors, and systems: The chemical uniqueness of molybdenum. J. Chem. Educ. 1985, 62, 943. [Google Scholar] [CrossRef] [Green Version]
- Modec, B.; Dolenc, D. Molybdenum complexes with citrate revisited. A mononuclear [MoVOCl4(H2O)]− ion as a new synthetic entry. Inorg. Chim. Acta 2019, 495, 119006. [Google Scholar] [CrossRef]
- Modec, B.; Dolenc, D.; Kasunič, M. Complexation of molybdenum(V) with glycolic acid: An unusual orientation of glycolato ligand in {Mo2O4}2+ complexes. Inorg. Chem. 2008, 47, 3625–3633. [Google Scholar] [CrossRef]
- James, R.G.; Wardlaw, W. Co-ordination compounds of quinquevalent molybdenum. J. Chem. Soc. 1927, 2145–2156. [Google Scholar] [CrossRef]
- Hanson, G.R.; Brunette, A.A.; McDonell, A.C.; Murray, K.S.; Wedd, A.G. Electronic properties of thiolate compounds of oxomolybdenum(V) and their tungsten and selenium analogs. Effects of oxygen-17, molybdenum-98, and molybdenum-95 isotope substitution upon ESR spectra. J. Am. Chem. Soc. 1981, 103, 1953–1959. [Google Scholar] [CrossRef]
- Modec, B.; Brenčič, J.V. Novel methanol-containing oxomolybdate(V) complexes: Synthesis and structural characterisation of intermediates in the formation of {Mo2O4}2+ clusters from [MoOCl4(H2O)]− and [MoOBr4]− precursors. Eur. J. Inorg. Chem. 2005, 2005, 1698–1709. [Google Scholar] [CrossRef]
- Gray, H.B.; Hare, C.R. The electronic structures and spectra of chromyl and molybdenyl ions. Inorg. Chem. 1962, 1, 363–368. [Google Scholar] [CrossRef]
- Blake, A.B.; Cotton, F.A.; Wood, J.S. The crystal, molecular, and electronic structures of a binuclear oxomolybdenum(V) xanthate complex. J. Am. Chem. Soc. 1964, 86, 3024–3031. [Google Scholar] [CrossRef]
- Bondi, A. van der Waals volumes and radii. J. Phys. Chem. 1964, 68, 441–451. [Google Scholar] [CrossRef]
- Janiak, C. A critical account on π–π stacking in metal complexes with aromatic nitrogen-containing ligands. J. Chem. Soc. Dalton Trans. 2000, 3885–3896. [Google Scholar] [CrossRef]
- Junk, P.C.; Atwood, J.L. Hydrogen-bonded tetramethylethylenediammonium and triphenylphosphonium complexes derived from liquid clathrate media. J. Coord. Chem. 1999, 46, 505–518. [Google Scholar] [CrossRef]
- Seyedsadjadi, S.A.; Ghammamy, S.; Rezaeibehbahani, G. The crystal and molecular structure of bis(tetramethylammonium) pentachlorooxomolybdate(V)-acetonitrile(1:1). Cryst. Res. Technol. 2005, 40, 727–730. [Google Scholar] [CrossRef]
- Marchetti, F.; Pampaloni, G.; Zacchini, S. Lactam/MoCl5 interaction in CH2Cl2: Synthesis and X-ray characterization of protonated δ-valerolactam salts. RSC Adv. 2013, 3, 10007–10013. [Google Scholar] [CrossRef]
- Modec, B.; Brenčič, J.V. Anions of 1,3,5-benzenetricarboxylic and heptanedioic acids serving as bridges between dimolybdenum(V) metal–metal bonded units: Preparation and structural characterization of dinuclear and tetranuclear complexes. Eur. J. Inorg. Chem. 2005, 2005, 4325–4334. [Google Scholar] [CrossRef]
- Vrdoljak, V.; Milić, D.; Cindrić, M.; Matković-Čalogović, D.; Cinčić, D. Synthesis of novel molybdenum(V) complexes: Structural characterization of two thiosemicarbazonato complexes [MoOCl2{C6H4(O)CH:NNHC:SNHC6H5}] and [MoOCl2{C10H6(O)CH:NNHC:SNHC6H5}]·CH3CN, and two oxohalomolybdates NH4[MoOCl4(CH3CN)] and [C5H5NH]2[MoOCl5]·CH2Cl2. Polyhedron 2007, 26, 3363–3372. [Google Scholar] [CrossRef]
- Isovitsch, R.A.; May, J.G.; Fronczek, F.R.; Maverick, A.W. Photoredox reactions of oxomolybdenum(V) with phosphines. Polyhedron 2000, 19, 1437–1446. [Google Scholar] [CrossRef]
- Agilent. CrysAlis PRO; Agilent Technologies Ltd.: Yarnton, UK, 2014. [Google Scholar]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. Olex2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A 2015, 71, 3–8. [Google Scholar] [CrossRef] [Green Version]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Spek, A.L. Structure validation in chemical crystallography. Acta Crystallogr. Sect. D 2009, 65, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Macrae, C.F.; Bruno, I.J.; Chisholm, J.A.; Edgington, P.R.; McCabe, P.; Pidcock, E.; Rodriguez-Monge, L.; Taylor, R.; van de Streek, J.; Wood, P.A. Mercury CSD 2.0—New features for the visualization and investigation of crystal structures. J. Appl. Crystallogr. 2008, 41, 466–470. [Google Scholar] [CrossRef]
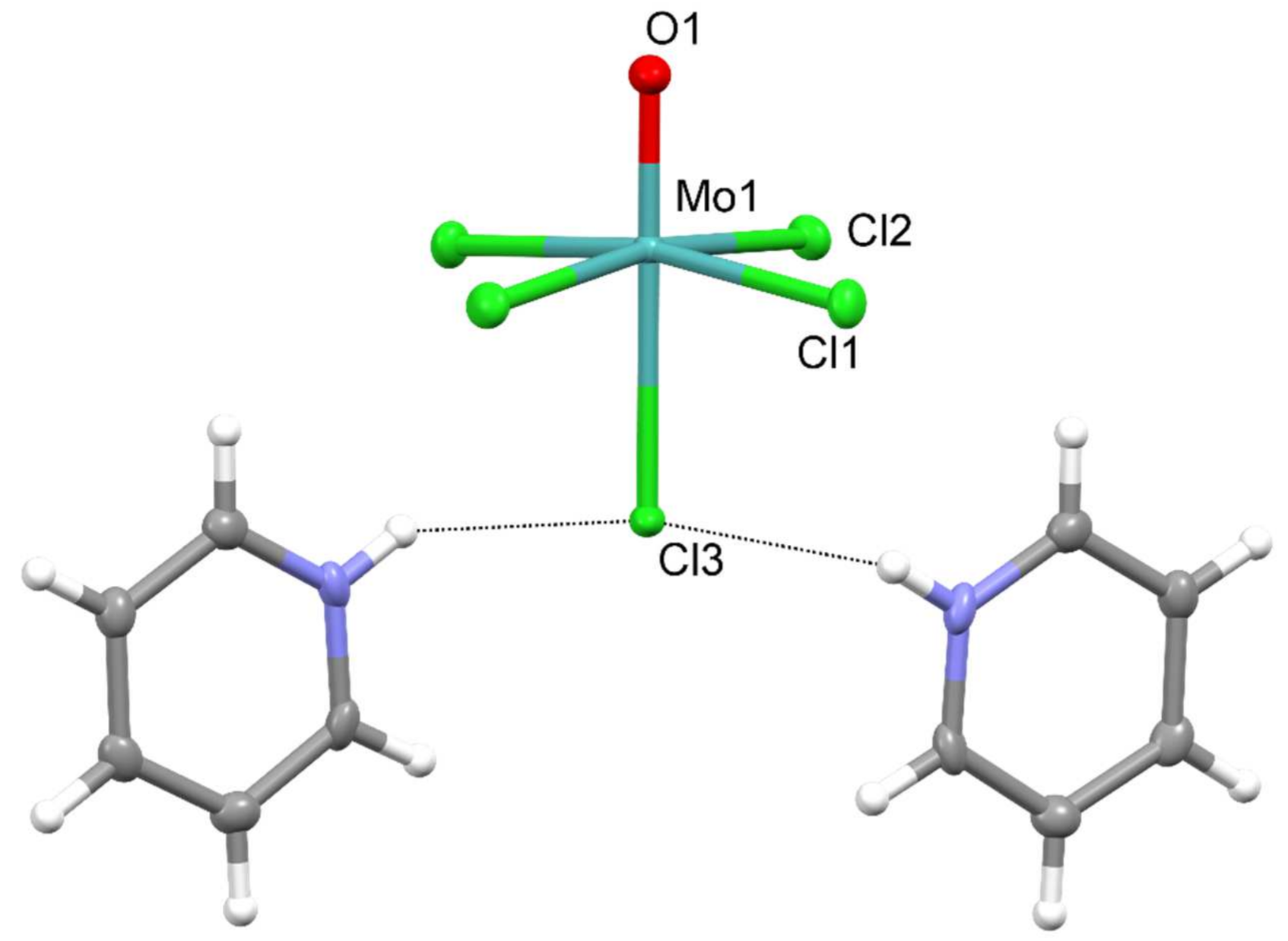
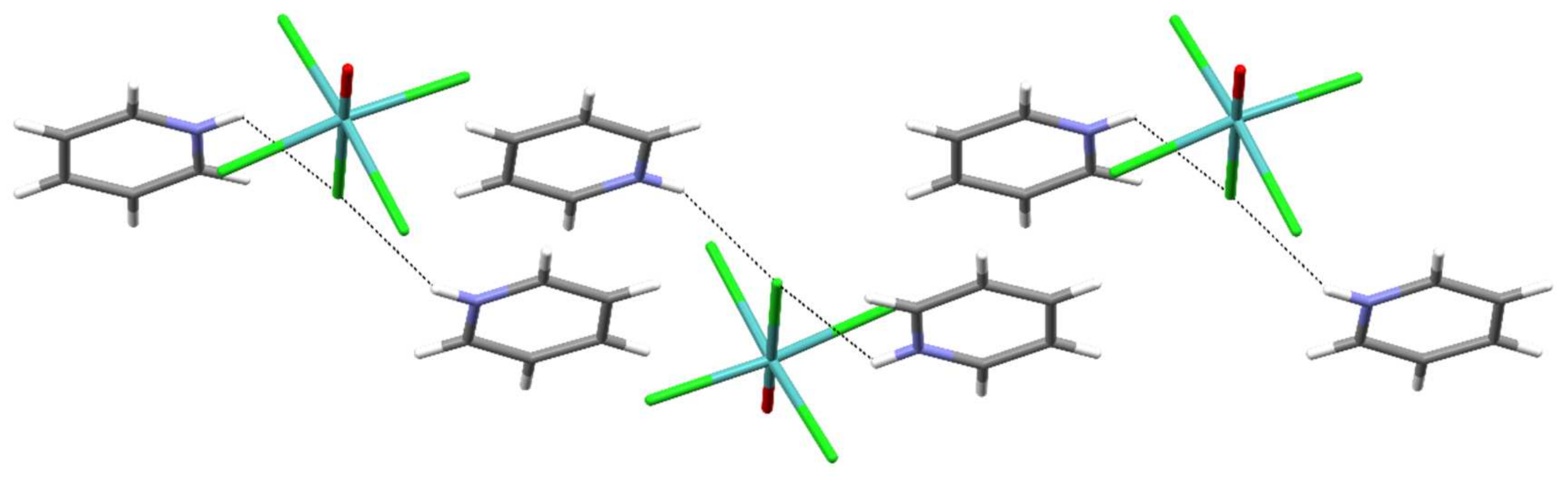
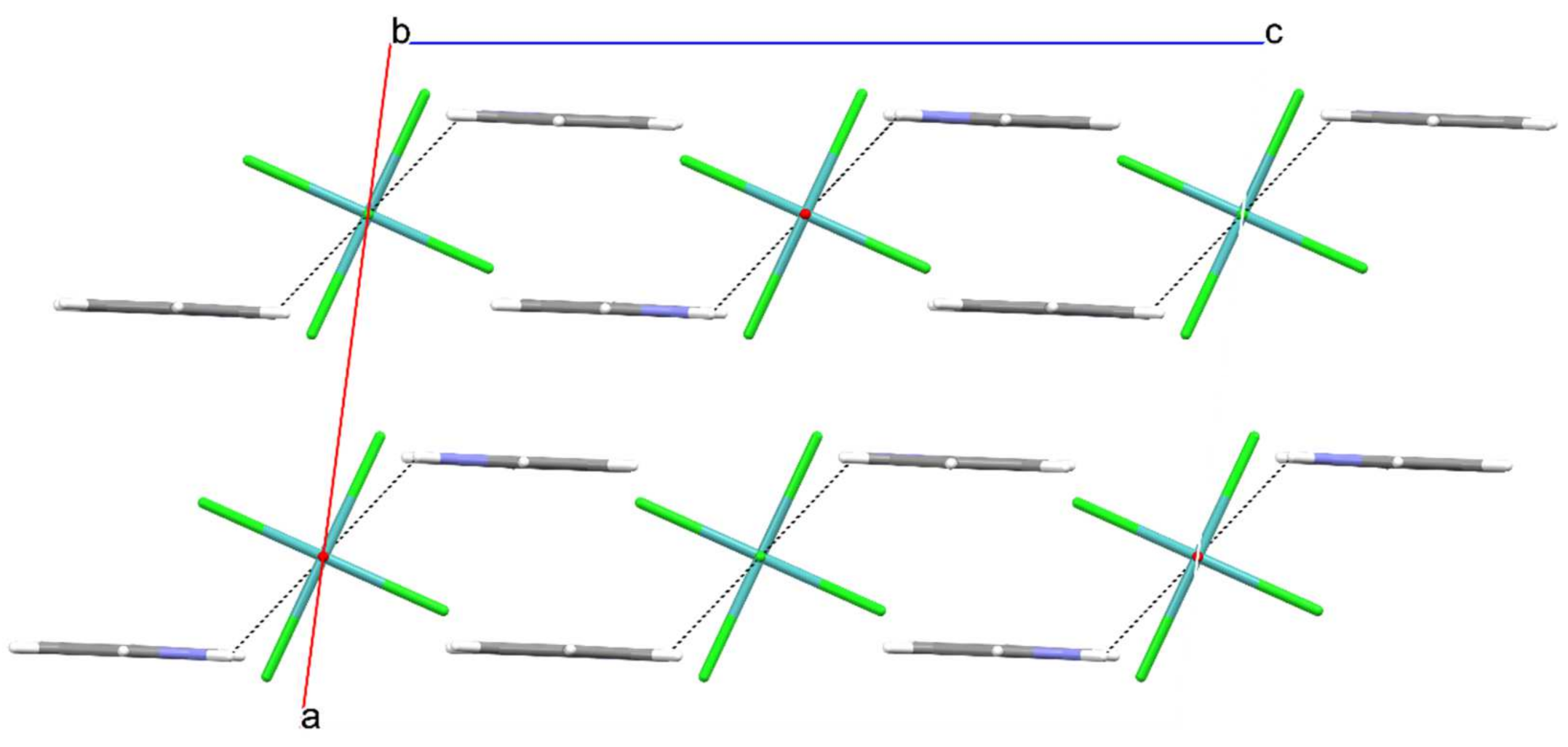
| Parameter | |
|---|---|
| Mo(1)–O(1) | 1.665(3) |
| Mo(1)–Cl(1) | 2.4072(8) |
| Mo(1)–Cl(2) | 2.3845(8) |
| Mo(1)–Cl(3) | 2.6038(10) |
| O(1)–Mo(1)–Cl(1) | 95.88(2) |
| O(1)–Mo(1)–Cl(2) | 95.295(19) |
| O(1)–Mo(1)–Cl(3) | 180.0 |
| Cl(1)–Mo(1)–Cl(2) | 90.44(3) |
| Cl(2)–Mo(1)–Cl(1) 1 | 88.47(3) |
| Cl(3)–Mo(1)–Cl(1) | 84.12(2) |
| Cl(3)–Mo(1)–Cl(2) | 84.705(19) |
| Hydrogen Bond | |
| Type | Parameters |
| N–H∙∙∙Cl | N(1)∙∙∙Cl(3)[2–x, –1–y, 2–z] = 3.092(3) |
| N(1)–H(1) = 0.83(3) | |
| H(1)∙∙∙Cl(3)[2–x, –1–y, 2–z] = 2.51(3) | |
| N(1)–H(1)∙∙∙Cl(3)[2–x, –1–y, 2–z] = 129(2) | |
| π∙∙∙π Stacking | |
| Type | Parameters 1 |
| PyH+∙∙∙PyH+ | Cg∙∙∙Cg = 3.5058(2) |
| α = 0.0 | |
| slippage = 0.944 |
| Data | (PyH)2[MoOCl5] |
|---|---|
| Empirical formula | C10H12Cl5MoN2O |
| Formula weight | 449.41 |
| Crystal system | monoclinic |
| Space group | I 2/a |
| T [K] | 150.00(10) |
| λ [Å] | 1.54184 |
| a [Å] | 12.4835(5) |
| b [Å] | 8.0098(3) |
| c [Å] | 15.7823(8) |
| α [°] | 90 |
| β [°] | 97.459(4) |
| γ [°] | 90 |
| V [Å3] | 1564.72(12) |
| Z | 4 |
| Dcalc [g/cm3] | 1.908 |
| μ [mm−1] | 14.662 |
| Size [mm] | 0.08 × 0.09 × 0.10 |
| Collected reflections | 3368 |
| Unique reflections | 1518 |
| Observed reflections | 1367 |
| Rint | 0.0369 |
| R1 (I > 2σ(I)) | 0.0330 |
| wR2 (all data) | 0.0847 |
| Goodness of fit | 1.09 |
| Largest diff. peak/hole [e−/Å3] | 1.2/−1.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Šturm, J.; Modec, B. Pyridinium Pentachloridooxomolybdate(V), (PyH)2[MoOCl5], Revisited: Single-Crystal X-ray Structure Determination. Molbank 2021, 2021, M1249. https://doi.org/10.3390/M1249
Šturm J, Modec B. Pyridinium Pentachloridooxomolybdate(V), (PyH)2[MoOCl5], Revisited: Single-Crystal X-ray Structure Determination. Molbank. 2021; 2021(3):M1249. https://doi.org/10.3390/M1249
Chicago/Turabian StyleŠturm, Jaka, and Barbara Modec. 2021. "Pyridinium Pentachloridooxomolybdate(V), (PyH)2[MoOCl5], Revisited: Single-Crystal X-ray Structure Determination" Molbank 2021, no. 3: M1249. https://doi.org/10.3390/M1249
APA StyleŠturm, J., & Modec, B. (2021). Pyridinium Pentachloridooxomolybdate(V), (PyH)2[MoOCl5], Revisited: Single-Crystal X-ray Structure Determination. Molbank, 2021(3), M1249. https://doi.org/10.3390/M1249







