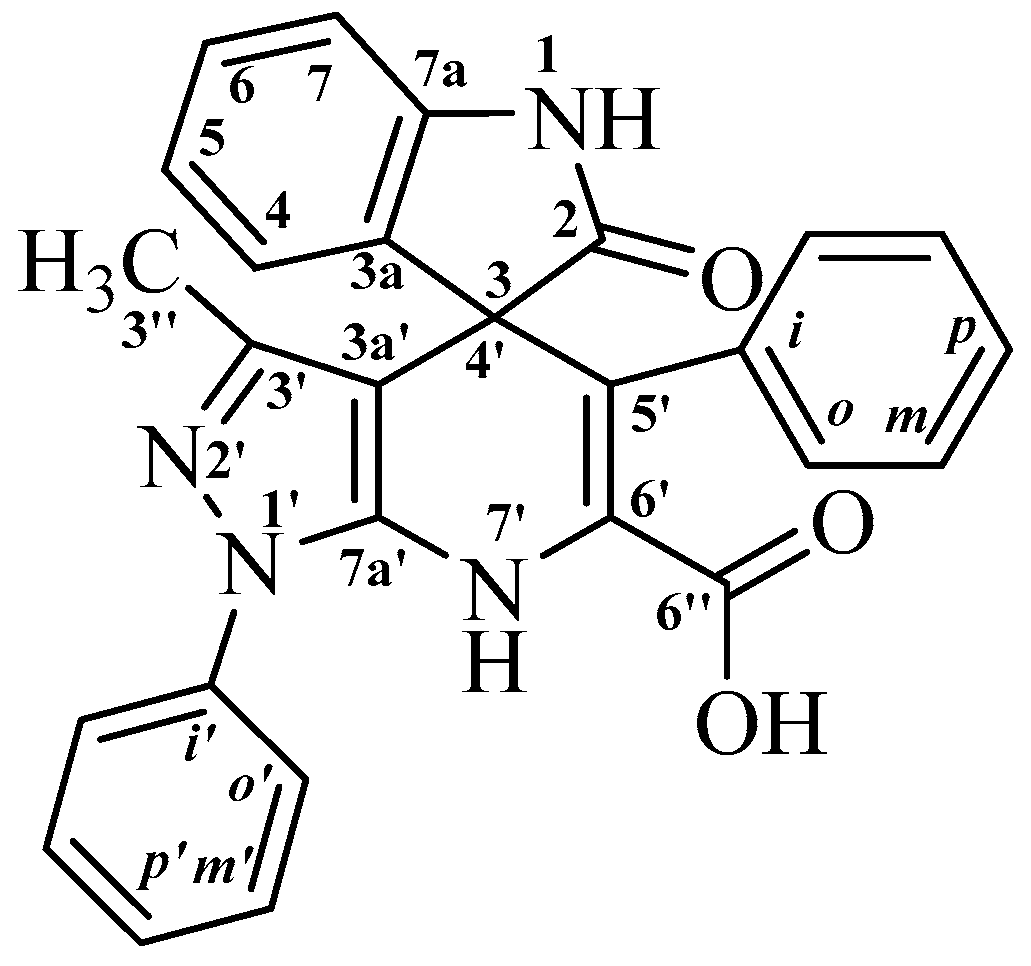
3′-Methyl-2-oxo-1′,5′-diphenyl-1′,7′-dihydrospiro[indoline-3,4′-pyrazolo[3,4-b]pyridine]-6′-carboxylic Acid
Abstract
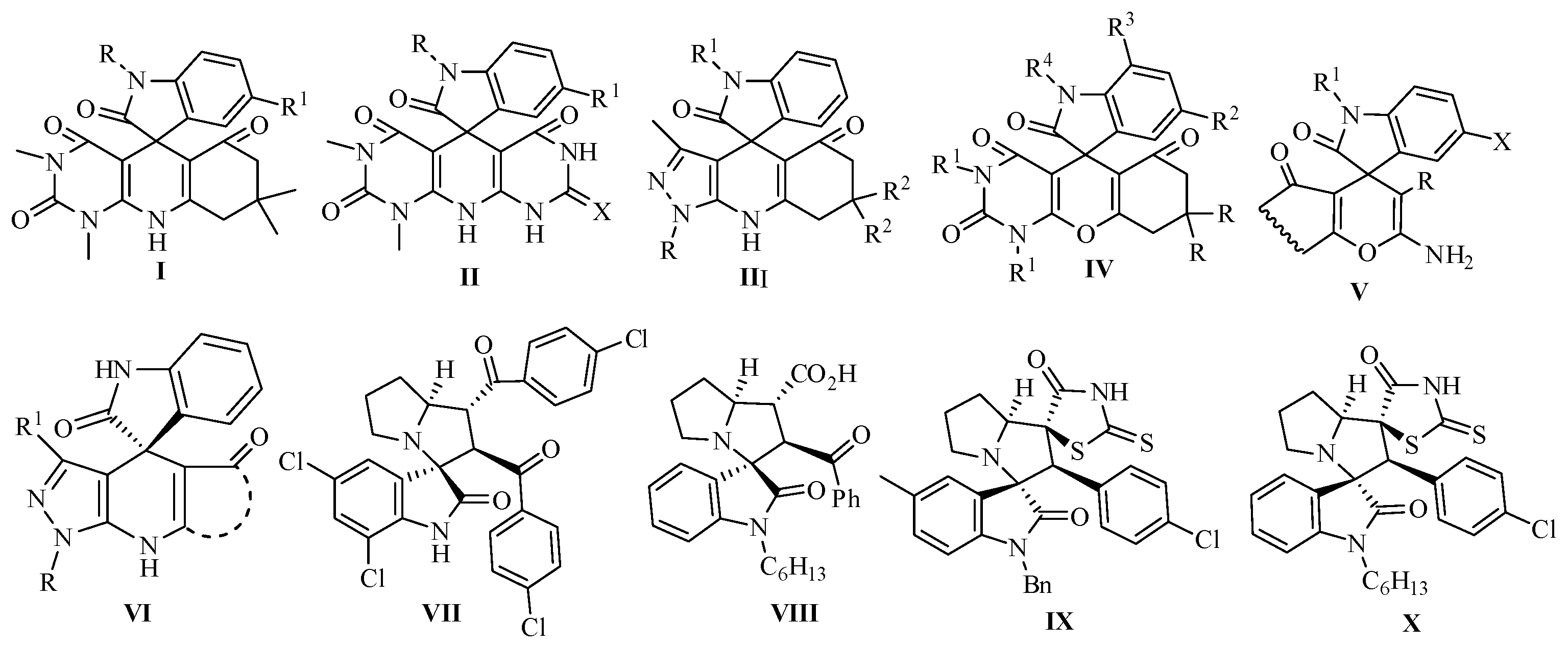
:1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. General Information
3.2. Synthesis of (±)-3′-Methyl-2-oxo-1′,5′-diphenyl-1′,7′-dihydrospiro[indoline-3,4′-pyrazolo[3,4-b]pyridine]-6′-carboxylic Acid
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sriram, D.; Bal, T.R.; Yogeeswari, P. Synthesis, Antiviral and Antibacterial Activities of Isatin Mannich Bases. Med. Chem. Res. 2005, 14, 211–228. [Google Scholar] [CrossRef]
- Murali, K.; Avinash, R.; Kirthiga, R.; Franzblau, S.G. Synthesis, antibacterial, and antitubercular studies of some novel isatin derivatives. Med. Chem. Res. 2012, 21, 4335–4340. [Google Scholar] [CrossRef]
- Karalı, N.; Güzel, Ö.; Özsoy, N.; Özbey, S.; Salman, A. Synthesis of new spiroindolinones incorporating a benzothiazole moiety as antioxidant agents. Eur. J. Med. Chem. 2010, 45, 1068–1077. [Google Scholar] [CrossRef] [PubMed]
- Thangamani, A. Regiospecific synthesis and biological evaluation of spirooxindolopyrrolizidines via [3+2] cycloaddition of azomethine ylide. Eur. J. Med. Chem. 2010, 45, 6120–6126. [Google Scholar] [CrossRef] [PubMed]
- Rana, S.; Blowers, E.C.; Tebbe, C.; Contreras, J.I.; Radhakrishnan, P.; Kizhake, S.; Zhou, T.; Rajule, R.N.; Arnst, J.L.; Munkarah, A.R.; et al. Isatin Derived Spirocyclic Analogues with α-Methylene-γ-butyrolactone as Anticancer Agents: A Structure–Activity Relationship Study. J. Med. Chem. 2016, 59, 5121–5127. [Google Scholar] [CrossRef] [PubMed]
- Khalafi-Nezhad, A.; Mohammadi, S. Magnetic, Acidic, Ionic Liquid-Catalyzed One-Pot Synthesis of Spirooxindoles. ACS Comb. Sci. 2013, 15, 512–518. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Shi, D. Efficient One-Pot Synthesis of Novel Spirooxindole Derivatives via Three-Component Reaction in Aqueous Medium. J. Comb. Chem. 2010, 12, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Mo, L.-P.; Zhao, F.-Y.; Zhang, Z.-H.; Liu, S.-X. One-Pot, Three-Component Synthesis of a Library of Spirooxindole-Pyrimidines Catalyzed by Magnetic Nanoparticle Supported Dodecyl Benzenesulfonic Acid in Aqueous Media. ACS Comb. Sci. 2012, 14, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Bazgir, A.; Hosseini, G.; Ghahremanzadeh, R. Copper Ferrite Nanoparticles: An Efficient and Reusable Nanocatalyst for a Green One-Pot, Three-component Synthesis of Spirooxindoles in Water. ACS Comb. Sci. 2013, 15, 530–534. [Google Scholar] [CrossRef] [PubMed]
- Quiroga, J.; Portillo, S.; Pérez, A.; Gálvez, J.; Abonia, R.; Insuasty, B. An efficient synthesis of pyrazolo[3,4-b]pyridine-4-spiroindolinones by a three-component reaction of 5-aminopyrazoles, isatin, and cyclic β-diketones. Tetrahedron Lett. 2011, 52, 2664–2666. [Google Scholar] [CrossRef]
- Quiroga, J.; Romo, P.; Cobo, J.; Glidewell, C. Synthesis of spiro[indoline-3,3′-pyrrolizines] by 1,3-dipolar reactions between isatins, L-proline and electron-deficient alkenes. Acta Crystallogr. Sect. C Struct. Chem. 2017, 73, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Romo, P.; Quiroga, J.; Cobo, J.; Glidewell, C. Regio- and stereospecific assembly of dispiro[indoline-3,3′-pyrrolizine-1′,5′′-thiazolidines] from simple precursors using a one-pot procedure: Synthesis, spectroscopic and structural characterization, and a proposed mechanism of formation. Acta Crystallogr. Sect. C Struct. Chem. 2020, 76, 779–785. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-Y.; Shi, D.-Q. Three-component one-pot synthesis of pyrazolo[3,4-b]quinolin-5(6H)-one derivatives in aqueous media. J. Heterocycl. Chem. 2011, 49, 212–216. [Google Scholar] [CrossRef]
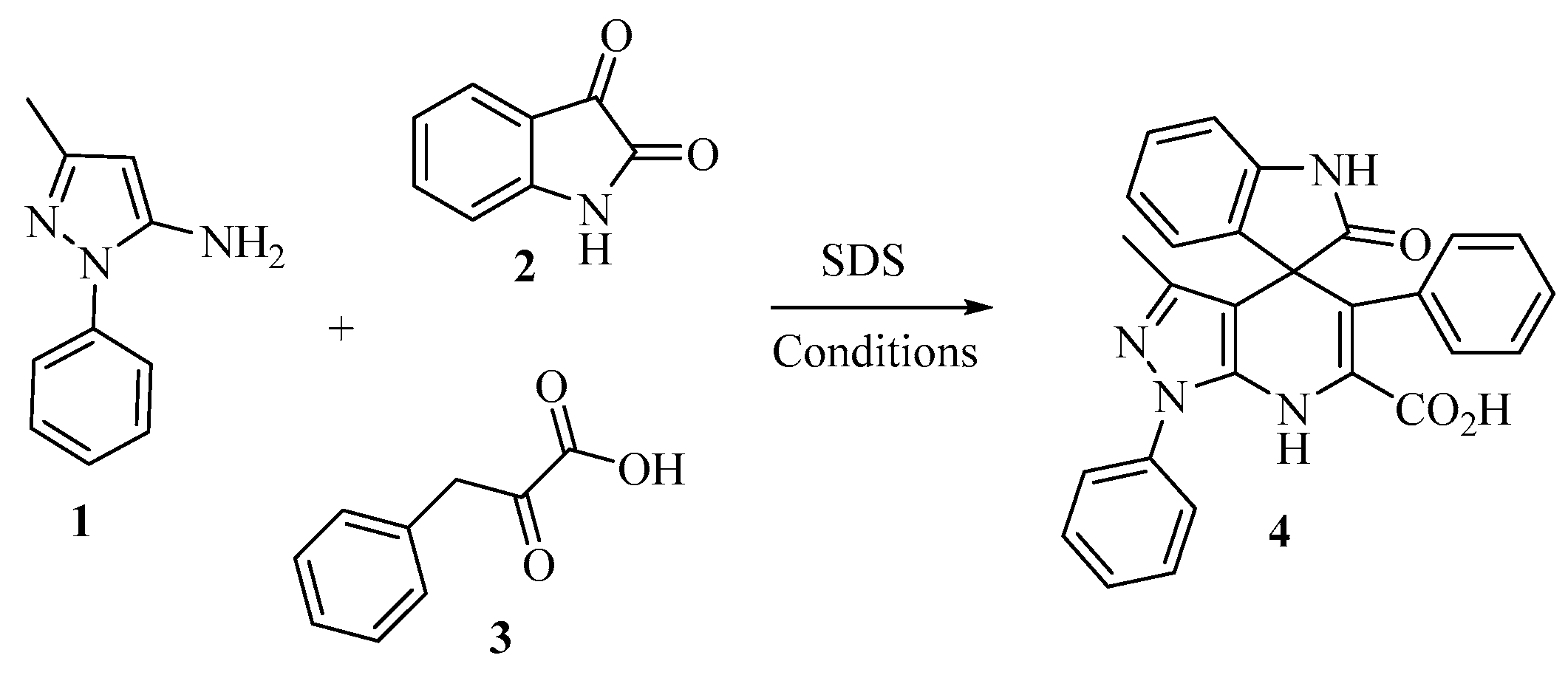
| Entry | Stoichiometry | Conditions | Yield (%) |
|---|---|---|---|
| 1 | H2O, reflux, 4 h | 21 | |
| 2 | 1 (0.6 mmol), 2 (0.6 mmol) 3 (0.6 mmol), SDS (0.1 g/mmol substrate) | H2O, MW, T: 90 °C, 100 W, 5 min | 76 |
| 3 | EtOH, reflux, 4 h | 47 | |
| 4 | EtOH, MW, T: 80 °C, 100 W, 5 min | 29 |
| δ (ppm) | Carbon Atom | NH-1 | NH-7′ |
|---|---|---|---|
| 55.4 | C-3 (spiro) | 3J | |
| 135.3 | C-3a | 3J | |
| 141.4 | C-7a | 2J | |
| 178.8 | C-2 (CO) | 2J | |
| 99.0 | C-3a′ | 3J | |
| 116.7 | C-5′ | 3J | |
| 165.6 | C-6″ (-CO2H) | 3J |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romo, P.E.; Insuasty, B.; Quiroga, J.; Abonia, R. 3′-Methyl-2-oxo-1′,5′-diphenyl-1′,7′-dihydrospiro[indoline-3,4′-pyrazolo[3,4-b]pyridine]-6′-carboxylic Acid. Molbank 2021, 2021, M1214. https://doi.org/10.3390/M1214
Romo PE, Insuasty B, Quiroga J, Abonia R. 3′-Methyl-2-oxo-1′,5′-diphenyl-1′,7′-dihydrospiro[indoline-3,4′-pyrazolo[3,4-b]pyridine]-6′-carboxylic Acid. Molbank. 2021; 2021(2):M1214. https://doi.org/10.3390/M1214
Chicago/Turabian StyleRomo, Pablo E., Braulio Insuasty, Jairo Quiroga, and Rodrigo Abonia. 2021. "3′-Methyl-2-oxo-1′,5′-diphenyl-1′,7′-dihydrospiro[indoline-3,4′-pyrazolo[3,4-b]pyridine]-6′-carboxylic Acid" Molbank 2021, no. 2: M1214. https://doi.org/10.3390/M1214
APA StyleRomo, P. E., Insuasty, B., Quiroga, J., & Abonia, R. (2021). 3′-Methyl-2-oxo-1′,5′-diphenyl-1′,7′-dihydrospiro[indoline-3,4′-pyrazolo[3,4-b]pyridine]-6′-carboxylic Acid. Molbank, 2021(2), M1214. https://doi.org/10.3390/M1214







