Cis-Bis(2,2′-Azopyridinido)dicarbonylruthenium(II)
Abstract
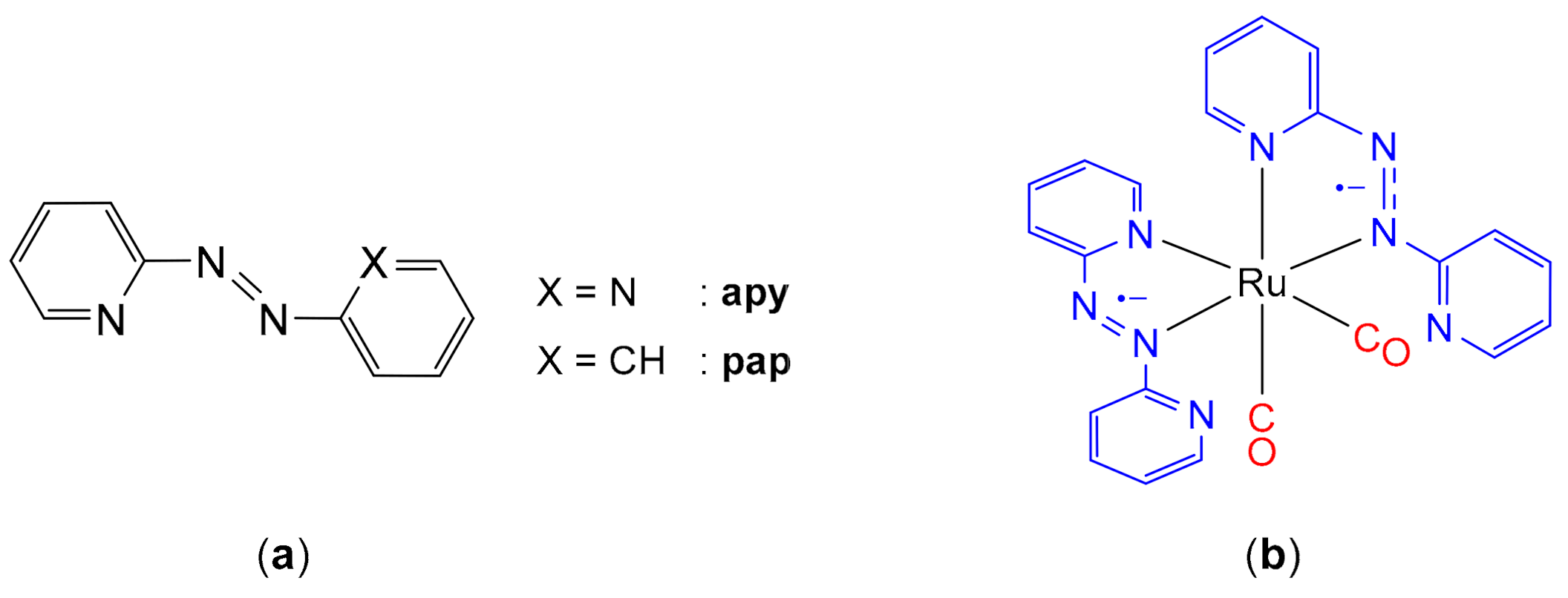
1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterization of the Diradical Complex
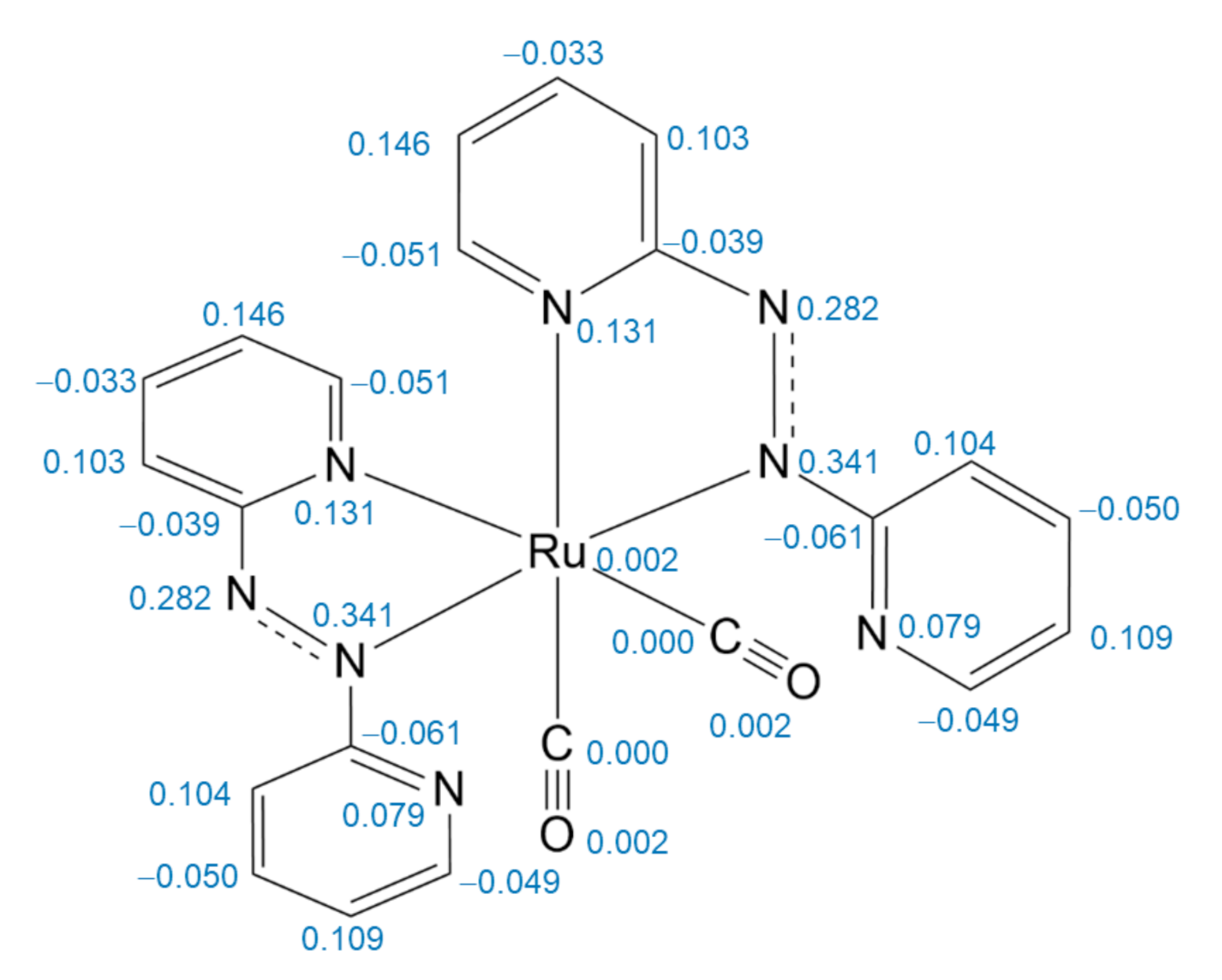
2.2. Electronic Structure Analysis
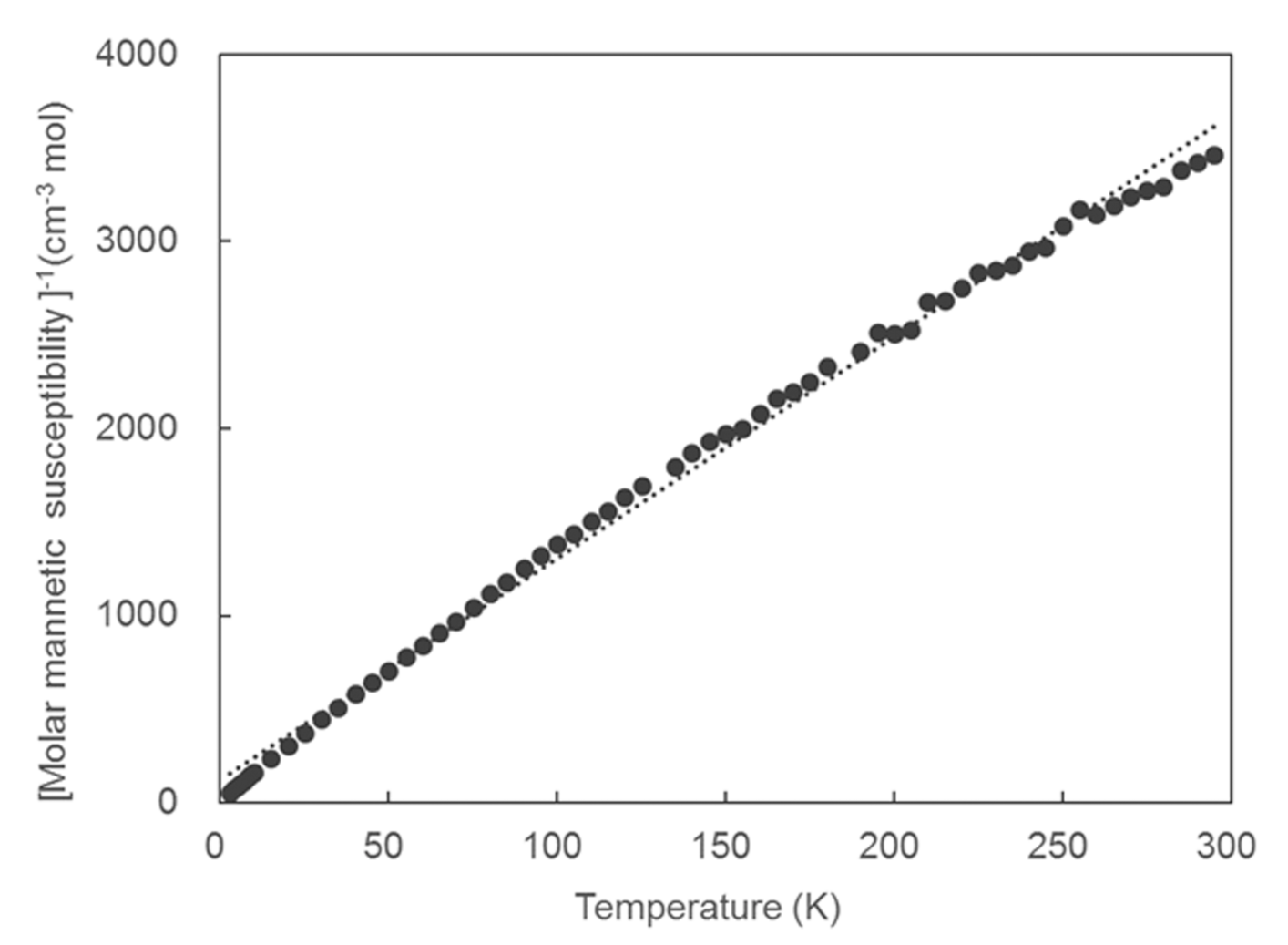
2.3. Magnetic Properties
3. Materials and Methods
3.1. Measurements
3.2. Synthesis of the Complex
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Kaim, W. Complexes with 2,2′-azobispyridine and related ‘S-frame’ bridging ligands containing the azo function. Coord. Chem. Rev. 2001, 219–221, 463–488. [Google Scholar] [CrossRef]
- Shivakumar, M.; Pramanik, K.; Bhattacharyya, I.; Chakravorty, A. Chemistry of metal-bound anion radicals. A family of mono- and bis(azopyridine) chelates of bivalent ruthenium. Inorg. Chem. 2000, 39, 4332–4338. [Google Scholar] [CrossRef] [PubMed]
- Das, D.; Mondal, T.K.; Mobin, S.M.; Lahiri, G.K. Sensitive valence structures of [(pap)2Ru(Q)]n (n = +2, +1, 0, –1, –2) with two different redox noninnocent ligands, Q = 3,5-di-tert-butyl-N-aryl-1,2-benzoquinonemonoimine and pap = 2-phenylazopyridine. Inorg. Chem. 2009, 48, 9800–9810. [Google Scholar] [CrossRef] [PubMed]
- Oyama, D.; Asuma, A.; Hamada, T.; Takase, T. Novel [Ru(polypyridine)(CO)2Cl2] and [Ru(polypyridine)2(CO)Cl]+-type complexes: Characterizing the effects of introducing azopyridyl ligands by electrochemical, spectroscopic and crystallographic measurements. Inorg. Chim. Acta 2009, 362, 2581–2588. [Google Scholar] [CrossRef]
- Paul, N.; Samanta, S.; Goswami, S. Redox induced electron transfer in doublet azo-anion diradical rhenium(II) complexes. Characterization of complete electron transfer series. Inorg. Chem. 2010, 49, 2649–2655. [Google Scholar] [CrossRef]
- Ghosh, P.; Samanta, S.; Roy, S.K.; Demeshko, S.; Meyer, F.; Goswami, S. Introducing a new azoaromatic pincer ligand. Isolation and characterization of redox events in its ferrous complexes. Inorg. Chem. 2014, 53, 4678–4686. [Google Scholar] [CrossRef]
- Oyama, D.; Mun, B.; Takase, T. Redox-induced reversible intramolecular carbon-nitrogen bond formation of an azopyridylruthenium complex: Control of carbonyl ligand photoreactivity caused by structural change of the complex. J. Organomet. Chem. 2015, 799–800, 173–178. [Google Scholar] [CrossRef]
- Zapata-Rivera, J.; Maynau, D.; Calzado, C.J. Evaluation of the magnetic interactions in salts containing [Ni(dmit)2]− radical anions. Chem. Mater. 2017, 29, 4317–4329. [Google Scholar] [CrossRef]
- Farcaş, A.-A.; Beu, T.A.; Bende, A. Light-induced spin transitions in Ni(II)-based macrocyclic-ligand complexes: A DFT study. J. Photochem. Photobiol. 2019, 376, 316–323. [Google Scholar] [CrossRef]
- Oyama, D.; Takatsuki, Y.; Fujita, R. Azopyridylruthenium(II) complexes containing Ru-C bonds: Synthesis, characterization and reactivity. Trends Inorg. Chem. 2010, 12, 31–40. [Google Scholar]
- Kobayashi, K.; Tanaka, K. Reactivity of CO2 activated on transition metals and sulfur ligands. Inorg. Chem. 2015, 54, 5085–5095. [Google Scholar] [CrossRef] [PubMed]
- Oyama, D.; Abe, R.; Takase, T. CO-ligand photodissociation in two Ru(II) complexes affected by different polypyridyl supporting ligands. Chem. Lett. 2017, 46, 1412–1414. [Google Scholar] [CrossRef]
- Akatsuka, K.; Abe, R.; Takase, T.; Oyama, D. Coordination chemistry of Ru(II) complexes of an asymmetric bipyridine analogue: Synergistic effects of supporting ligand and coordination geometry on reactivities. Molecules 2020, 25, 27. [Google Scholar] [CrossRef]
- Luckhaus, D.; Yamamoto, Y.-I.; Suzuki, T.; Signorell, R. Genuine binding energy of the hydrated electron. Sci. Adv. 2017, 3, e1603224. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16 (Revision, A.03); Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Lee, C.; Yang, W.; Parr, R.G. Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density. Phys. Rev. 1988, B37, 785–789. [Google Scholar] [CrossRef]
- Hehre, W.J.; Ditchfield, R.; Pople, J.A. Self-consistent molecular orbital methods. XII. Further extensions of Gaussian-type basis sets for use in molecular orbital studies of organic molecules. J. Chem. Phys. 1972, 56, 2257–2261. [Google Scholar] [CrossRef]
- Francl, M.M.; Pietro, W.J.; Hehre, W.J.; Binkley, J.S.; Gordon, M.S.; DeFrees, D.J.; Pople, J.A. Self-consistent molecular orbital methods. XXIII. A polarization-type basis set for second-row elements. J. Chem. Phys. 1982, 77, 3654–3665. [Google Scholar] [CrossRef]
- Wadt, W.R.; Hay, P.J. Ab initio effective core potentials for molecular calculations. Potentials for main group elements Na to Bi. J. Chem. Phys. 1985, 82, 284–298. [Google Scholar] [CrossRef]
- Baldwin, D.A.; Lever, A.B.P.; Parish, R.V. Complexes of 2,2′-azopyridine with iron(II), cobalt(II), nickel(II), copper(I), and copper(II). Infrared study. Inorg. Chem. 1969, 8, 107–115. [Google Scholar] [CrossRef]
- Hotze, A.C.G.; Caspers, S.E.; DeVos, D.; Kooijman, H.; Spek, A.L.; Flamigni, A.; Bacac, M.; Sava, G.; Haasnoot, J.G.; Reedijk, J. Structure-dependent in vitro cytotoxicity of the isomeric complexes [Ru(L)2Cl2] (L=o-tolylazopyridine and 4-methyl-2-phenylazopyridine) in comparison to [Ru(azpy)2Cl2]. J. Biol. Inorg. Chem. 2004, 9, 354–364. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takase, T.; Kainuma, S.; Kanno, T.; Oyama, D. Cis-Bis(2,2′-Azopyridinido)dicarbonylruthenium(II). Molbank 2021, 2021, M1182. https://doi.org/10.3390/M1182
Takase T, Kainuma S, Kanno T, Oyama D. Cis-Bis(2,2′-Azopyridinido)dicarbonylruthenium(II). Molbank. 2021; 2021(1):M1182. https://doi.org/10.3390/M1182
Chicago/Turabian StyleTakase, Tsugiko, Shuya Kainuma, Takatoshi Kanno, and Dai Oyama. 2021. "Cis-Bis(2,2′-Azopyridinido)dicarbonylruthenium(II)" Molbank 2021, no. 1: M1182. https://doi.org/10.3390/M1182
APA StyleTakase, T., Kainuma, S., Kanno, T., & Oyama, D. (2021). Cis-Bis(2,2′-Azopyridinido)dicarbonylruthenium(II). Molbank, 2021(1), M1182. https://doi.org/10.3390/M1182





