(2S,3R,6R)-2-[(R)-1-Hydroxyallyl]-4,4-dimethoxy-6-methyltetrahydro-2H-pyran-3-ol
Abstract
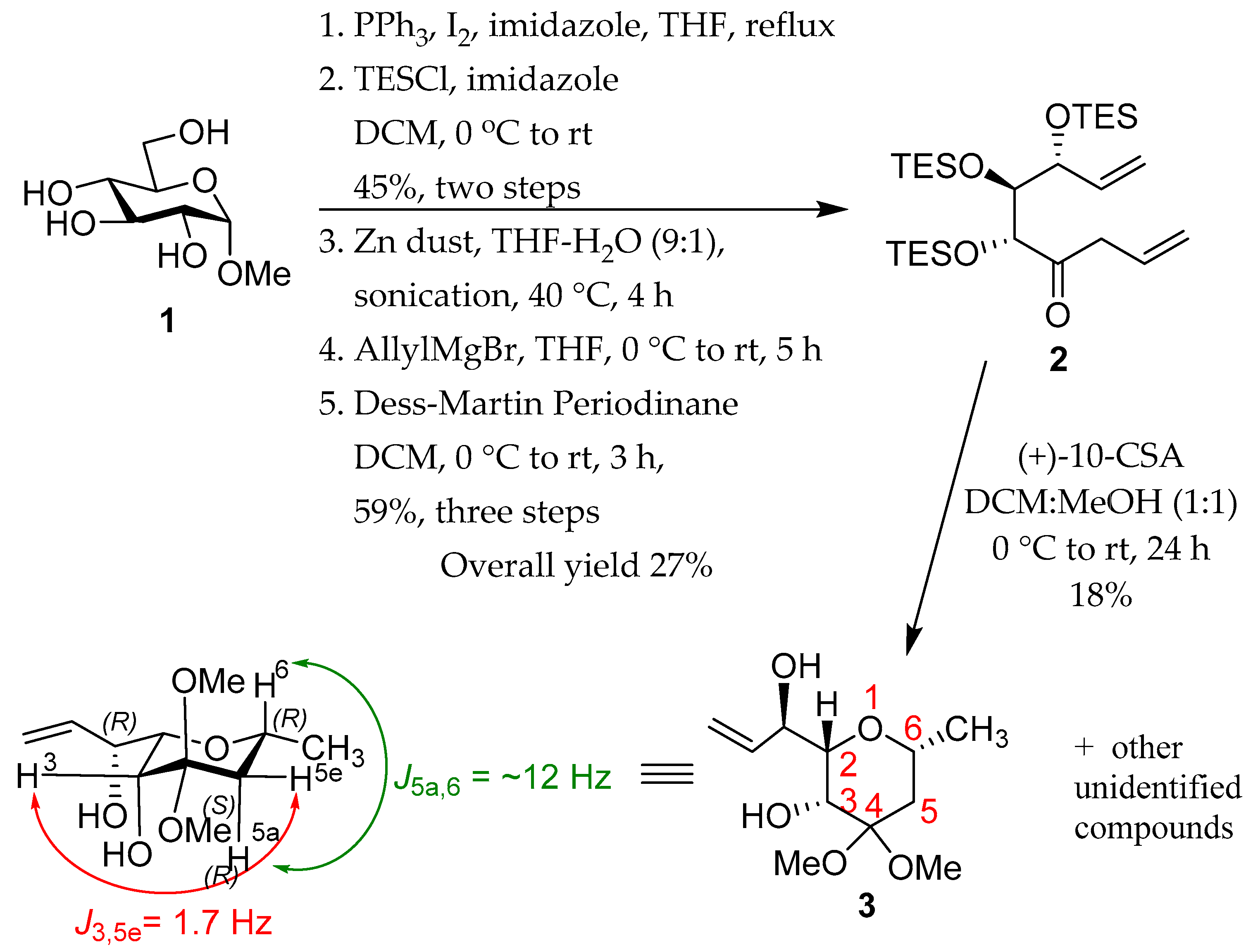
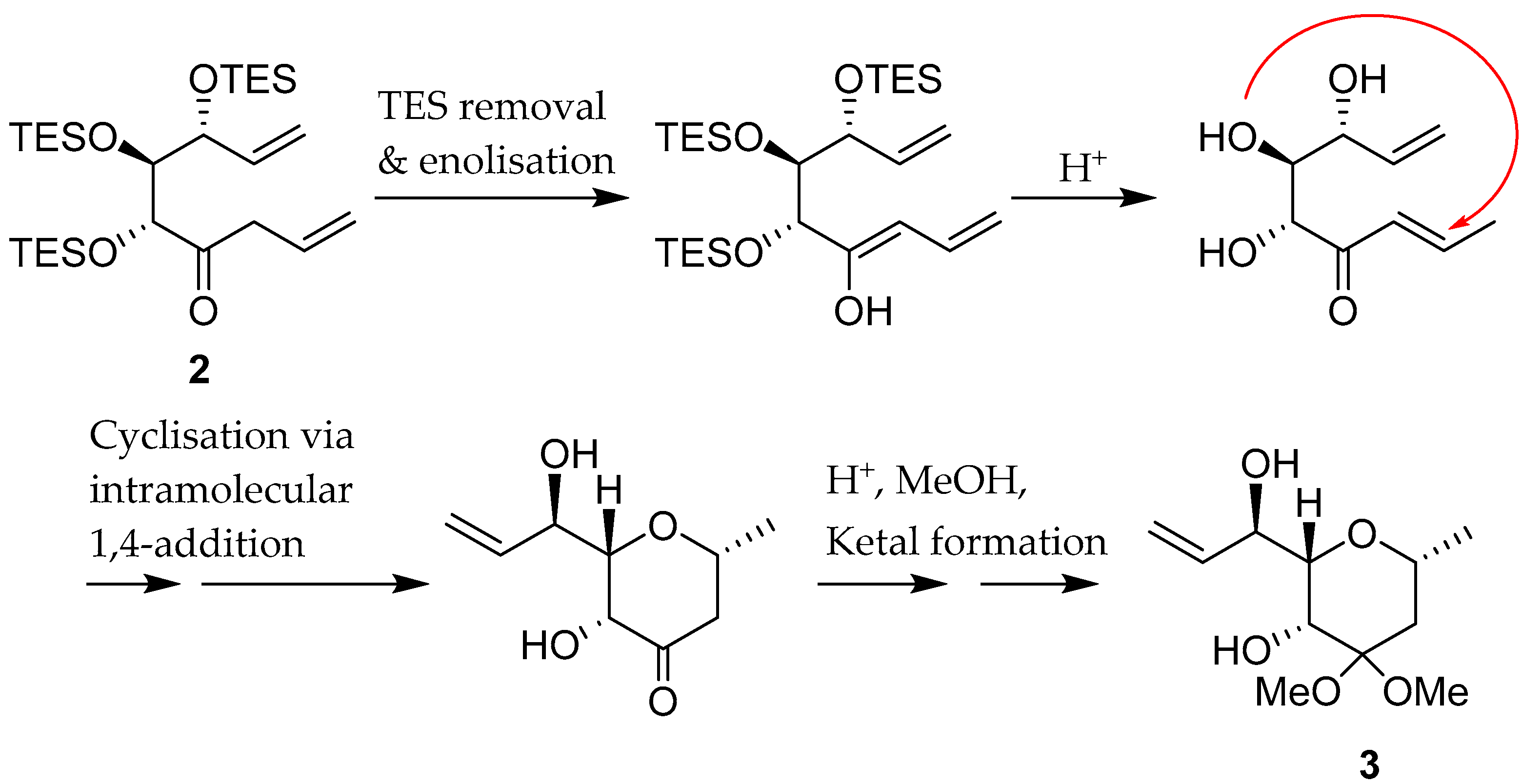
1. Introduction
2. Results and Discussion
3. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hevey, R. Strategies for the development of glycomimetic drug candidates. Pharmaceuticals 2019, 12, 55. [Google Scholar] [CrossRef] [PubMed]
- Ernst, B.; Magnani, J.L. From carbohydrate leads to glycomimetic drugs. Nat. Rev. Drug Discov. 2009, 8, 661–677. [Google Scholar] [CrossRef]
- Smith, D.C.; Lord, J.M.; Roberts, L.M.; Johannes, L. Glycosphingolipids as toxin receptors. Semin. Cell Dev. Biol. 2004, 15, 397–408. [Google Scholar] [CrossRef]
- Gamblin, S.J.; Skehel, J.J. Influenza hemagglutinin and neuraminidase membrane glycoproteins. J. Biol. Chem. 2010, 285, 28403–28409. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.; Murphy, P.V.; Gabius, H.J. Multivalent carbohydrate-lectin interactions: How synthetic chemistry enables insights into nanometric recognition. Molecules 2016, 21, 629. [Google Scholar] [CrossRef] [PubMed]
- Tamburrini, A.; Colombo, C.; Bernardi, A. Design and synthesis of glycomimetics: Recent advances. Med. Res. Rev. 2020, 40, 495–531. [Google Scholar] [CrossRef] [PubMed]
- Negi, A.; O’Reilly, C.; Jarikote, D.V.; Zhou, J.; Murphy, P.V. Multi-targeting protein-protein interaction inhibitors: Evolution of macrocyclic ligands with embedded carbohydrates (MECs) to improve selectivity. Eur. J. Med. Chem. 2019, 176, 292–309. [Google Scholar] [CrossRef] [PubMed]
- Voigt, T.; Gerding-Reimers, C.; Ngoc Tran, T.T.; Bergmann, S.; Lachance, H.; Schölermann, B.; Brockmeyer, A.; Janning, P.; Ziegler, S.; Waldmann, H. A natural product inspired tetrahydropyran collection yields mitosis modulators that synergistically target CSE1L and tubulin. Angew. Chem. Int. Ed. 2013, 52, 410–414. [Google Scholar] [CrossRef] [PubMed]
- Kohli, K.; Prajapati, R.; Sharma, B.K. Bio-based chemicals from renewable biomass for integrated biorefineries. Energies 2019, 12, 233. [Google Scholar] [CrossRef]
- Bhatt, U.; Christmann, M.; Quitschalle, M.; Claus, E.; Kalesse, M. The first total synthesis of (+)-ratjadone. J. Org. Chem. 2001, 66, 1885–1893. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.R.; Ihle, D.C.; Plummer, S.V. Total synthesis of (−)-Ratjadone. Org. Lett. 2001, 3, 1383–1386. [Google Scholar] [CrossRef] [PubMed]
- Moynihan, L. Synthesis of Iminosugar C-Glycosides via Tandem Allylic-Azide Rearrangement—Huisgen Cycloaddition. Ph.D. Thesis, National University of Ireland, Galway, Ireland, 2012. [Google Scholar]
- Skaanderup, P.R.; Poulsen, C.S.; Hyldtoft, L.; Jørgensen, M.R.; Madsen, R. Regioselective conversion of primary alcohols into iodides in unprotected Methyl furanosides and pyranosides. Synthesis (Stuttg) 2002, 2002, 1721–1727. [Google Scholar] [CrossRef]
- Moynihan, L.; Chadda, R.; McArdle, P.; Murphy, P.V. Allylic azide rearrangement in tandem with huisgen cycloaddition for stereoselective annulation: Synthesis of C-Glycosyl iminosugars. Org. Lett. 2015, 17, 6226–6229. [Google Scholar] [CrossRef] [PubMed]
- Bernet, B.; Vasella, A. Carbocyclische verbindungen aus monosacchariden. II. Umsetzungen in der Mannosereihe. Helv. Chim. Acta 1979, 62, 2400–2410. [Google Scholar] [CrossRef]
- Skaanderup, P.R.; Hyldtoft, L.; Madsen, R. Zinc-mediated fragmentation of methyl 6-deoxy-6-iodo-hexopyranosides. Monatsh. Chem. 2002, 133, 467–472. [Google Scholar] [CrossRef]
- Sasaki, M.; Kawashima, Y.; Fuwa, H. Studies toward the total synthesis of amphidinoliden: Stereocontrolled synthesis of the c13-c29 segment. Heterocycles 2015, 90, 579–599. [Google Scholar] [CrossRef]
- Constantino, M.G.; Lacerda, V.; Tasic, L.; da Silva, G.; Rittner, R. Principal component analysis of long-range ‘W’ coupling constants of some cyclic compounds. J. Mol. Struct. 2001, 597, 129–136. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bennett, J.; Murphy, P.V. (2S,3R,6R)-2-[(R)-1-Hydroxyallyl]-4,4-dimethoxy-6-methyltetrahydro-2H-pyran-3-ol. Molbank 2020, 2020, M1140. https://doi.org/10.3390/M1140
Bennett J, Murphy PV. (2S,3R,6R)-2-[(R)-1-Hydroxyallyl]-4,4-dimethoxy-6-methyltetrahydro-2H-pyran-3-ol. Molbank. 2020; 2020(2):M1140. https://doi.org/10.3390/M1140
Chicago/Turabian StyleBennett, Jack, and Paul V. Murphy. 2020. "(2S,3R,6R)-2-[(R)-1-Hydroxyallyl]-4,4-dimethoxy-6-methyltetrahydro-2H-pyran-3-ol" Molbank 2020, no. 2: M1140. https://doi.org/10.3390/M1140
APA StyleBennett, J., & Murphy, P. V. (2020). (2S,3R,6R)-2-[(R)-1-Hydroxyallyl]-4,4-dimethoxy-6-methyltetrahydro-2H-pyran-3-ol. Molbank, 2020(2), M1140. https://doi.org/10.3390/M1140




