Improved Synthesis and Determination of the Biologically Active Diastereomer of YK11
Abstract
1. Introduction
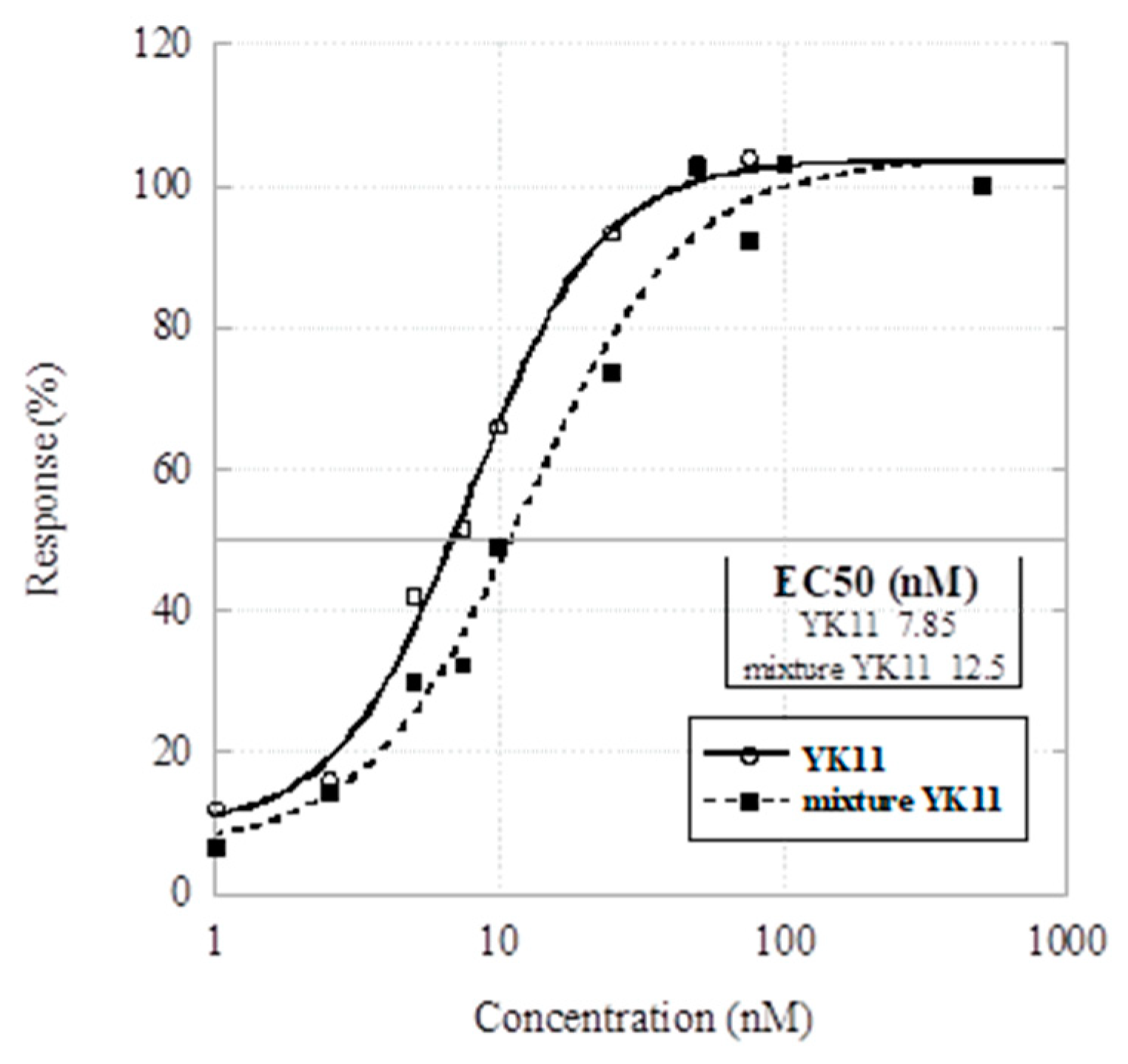
2. Results and Discussion
3. Materials and Methods
3.1. General Information
3.2. Synthesis of L1–L3
3.3. Synthesis of YK11 (2a)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Bhasin, S.; Jasuja, R. Selective androgen receptor modulators as function promoting therapies. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, R.; Mohler, M.L.; Bohl, C.E.; Miller, D.D.; Dalton, J.T. Selective Androgen Receptor Modulators (SARMs) as Function Promoting Therapies. Nucl. Recept. Signal. 2008, 6, e010. [Google Scholar]
- Kanno, Y.; Hikosaka, R.; Zhang, S.; Inoue, Y.; Nakahama, T.; Kato, K.; Yamaguchi, A.; Tominaga, N.; Kohra, S.; Arizono, K.; et al. (17a,20E)-17,20-[(1-Methoxyethylidene)bis(oxy)]-3-oxo-19-norpregna-4,20-diene-21-carboxylic Acid Methyl Ester (YK11) Is a Partial Agonist of the Androgen Receptor. Biol. Pharm. Bull. 2011, 34, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Yatsu, T.; Kusakabe, T.; Kato, K.; Inouye, Y.; Nemoto, K.; Kanno, Y. Selective Androgen Receptor Modulator, YK11, Up-Regulates Osteoblastic Proliferation and Differentiation in MC3T3-E1 Cells. Biol. Pharm. Bull. 2018, 41, 394–398. [Google Scholar] [CrossRef] [PubMed]
- Kato, K.; Nouchi, H.; Ishikura, K.; Takaishi, S.; Motodate, S.; Tanaka, H.; Okudaira, K.; TMochida, R.; Nishigaki, K.; Shigenobu, H.; et al. Facile Access to Spiro Furanone Skeleton Based on Pd(II)-Mediated Cyclization-Carbonylation of Propargylic Ester. Tetrahedron 2006, 62, 2545–2554. [Google Scholar] [CrossRef]
- Ito, Y.; Kusakabe, T.; Dhage, Y.D.; Takahashi, K.; Sakata, K.; Sasai, H.; Kato, K. Total Synthesis of (−)-Graminin A Based on Asymmetric Cyclization Carbonylation of Propargyl Acetate. J. Org. Chem. 2019, 84, 16268–16277. [Google Scholar] [CrossRef] [PubMed]
- Kusakabe, T.; Mochida, T.; Ariyama, T.; Lee, D.; Ohkubo, S.; Takahashi, K.; Kato, K. PdII catalyzed ligand controlled synthesis of bis(3-furanyl)methanones and methyl 3-furancarboxylates. Org. Biomol. Chem. 2019, 17, 6860–6865. [Google Scholar] [CrossRef] [PubMed]
- The Crystallographic Data (CCDC-1974030) can be Obtained Free of Charge from the Cambridge Crystallographic Data Centre. Available online: www.ccdc.cam.ac.uk/data_request/cif (accessed on 7 April 2020).
- Ammann, S.E.; Liu, W.; White, M.C. Enantioselective Allylic C-H Oxidation of Terminal Olefins to Isochromans by Palladium(II)/Chiral Sulfoxide Catalysis. Angew. Chem. Int. Ed. 2016, 55, 9571–9575. [Google Scholar] [CrossRef] [PubMed]
- Piper, T.; Dib, J.; Putz, M.; Fusshöller, G.; Pop, V.; Lagojda, A.; Kuehne, D.; Geyer, H.; Schänzer, W.; Thevis, M. Studies on the in vivo metabolism of the SARM YK11: Identification and characterization of metabolites potentially useful for doping controls. Drug Test Anal. 2018, 10, 1646–1656. [Google Scholar] [CrossRef] [PubMed]
- Thevis, M.; Piper, T.; Dib, J.; Lagojda, A.; Kühne, D.; Lars, P.; Geyer, H.; Schänzer, W. Mass spectrometric characterization of the selective androgen receptor modulator (SARM) YK-11 for doping control purposes. Rapid Commun. Mass Spectrom. 2017, 31, 1175–1183. [Google Scholar] [CrossRef] [PubMed]
- Thevis, M.; Walpurgis, K.; Thomas, A. Analytical Approaches in Human Sports Drug Testing: Recent Advances, Challenges, and Solutions. Anal. Chem. 2020, 92, 506–523. [Google Scholar] [CrossRef] [PubMed]
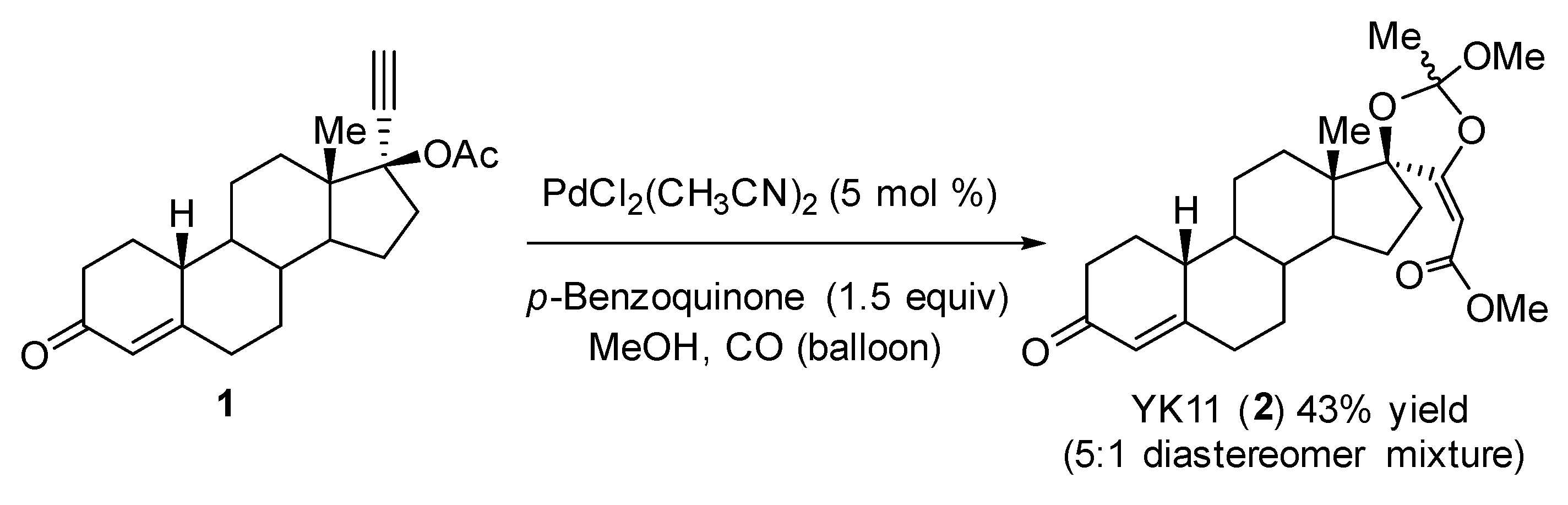


| Entry | Ligand | Temp. | Time | Yield (%) of 2 | dr |
|---|---|---|---|---|---|
| 1 | L1 | 0 | 36 | 63 | 4:1 |
| 2 | L2 | 0 | 108 | Complexmixture | - |
| 3 | L3 | 25 | 36 | 25 | 4:1 |
| 4 a | L1 | 0 | 18 | 73 | 7:1 |
| 5 a | L1 | −10 | 48 | 74 | 12:1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kanno, Y.; Kusakabe, T.; Saito, N.; Kikkawa, S.; Takahashi, K.; Azumaya, I.; Nemoto, K.; Kato, K. Improved Synthesis and Determination of the Biologically Active Diastereomer of YK11. Molbank 2020, 2020, M1125. https://doi.org/10.3390/M1125
Kanno Y, Kusakabe T, Saito N, Kikkawa S, Takahashi K, Azumaya I, Nemoto K, Kato K. Improved Synthesis and Determination of the Biologically Active Diastereomer of YK11. Molbank. 2020; 2020(2):M1125. https://doi.org/10.3390/M1125
Chicago/Turabian StyleKanno, Yuichiro, Taichi Kusakabe, Nao Saito, Shoko Kikkawa, Keisuke Takahashi, Isao Azumaya, Kiyomitsu Nemoto, and Keisuke Kato. 2020. "Improved Synthesis and Determination of the Biologically Active Diastereomer of YK11" Molbank 2020, no. 2: M1125. https://doi.org/10.3390/M1125
APA StyleKanno, Y., Kusakabe, T., Saito, N., Kikkawa, S., Takahashi, K., Azumaya, I., Nemoto, K., & Kato, K. (2020). Improved Synthesis and Determination of the Biologically Active Diastereomer of YK11. Molbank, 2020(2), M1125. https://doi.org/10.3390/M1125






