Bicyclo[4.2.0]octa-1,3,5-trien-3-yl-dimethyl((E)-styryl)-silane
Abstract
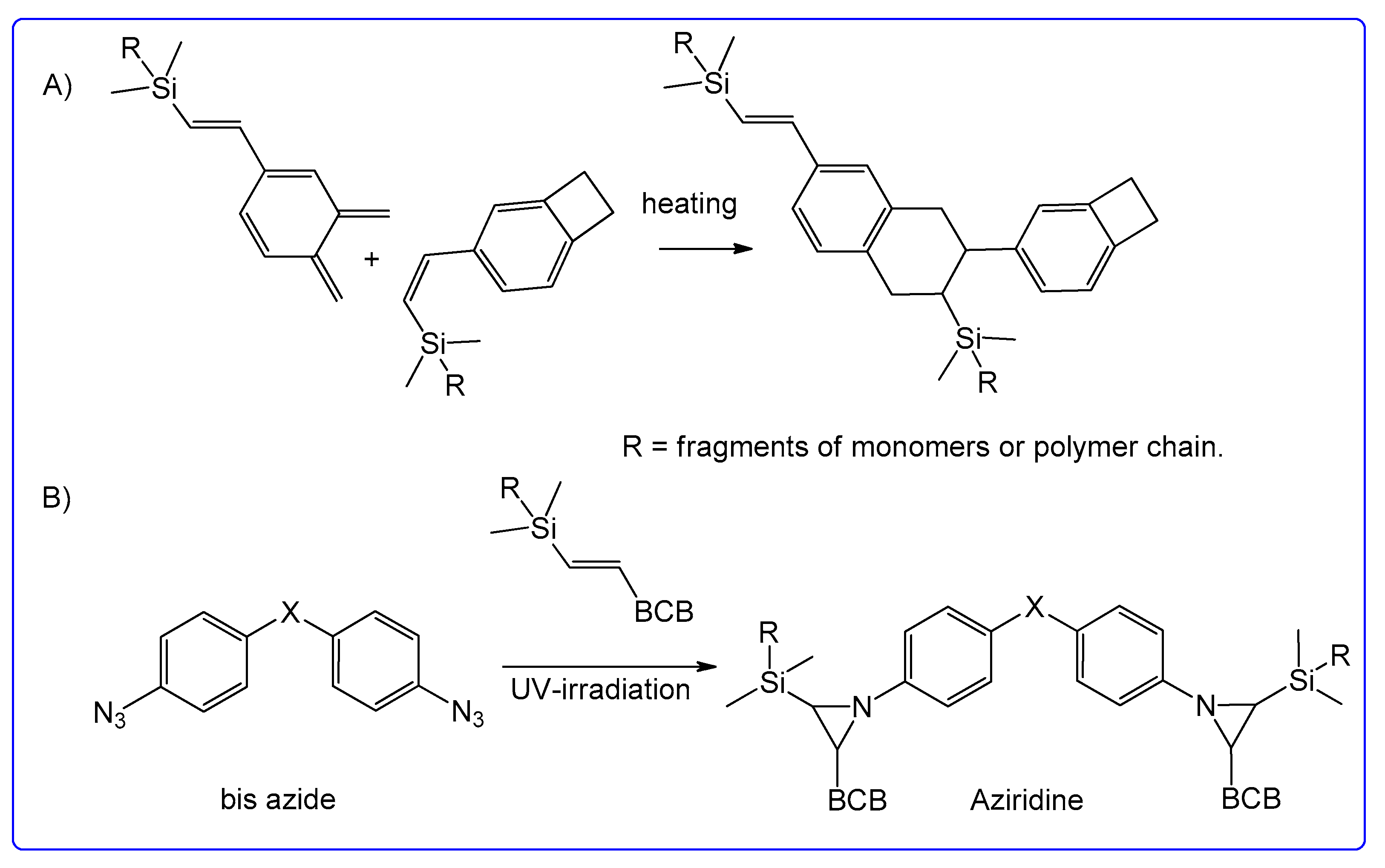
:1. Introduction
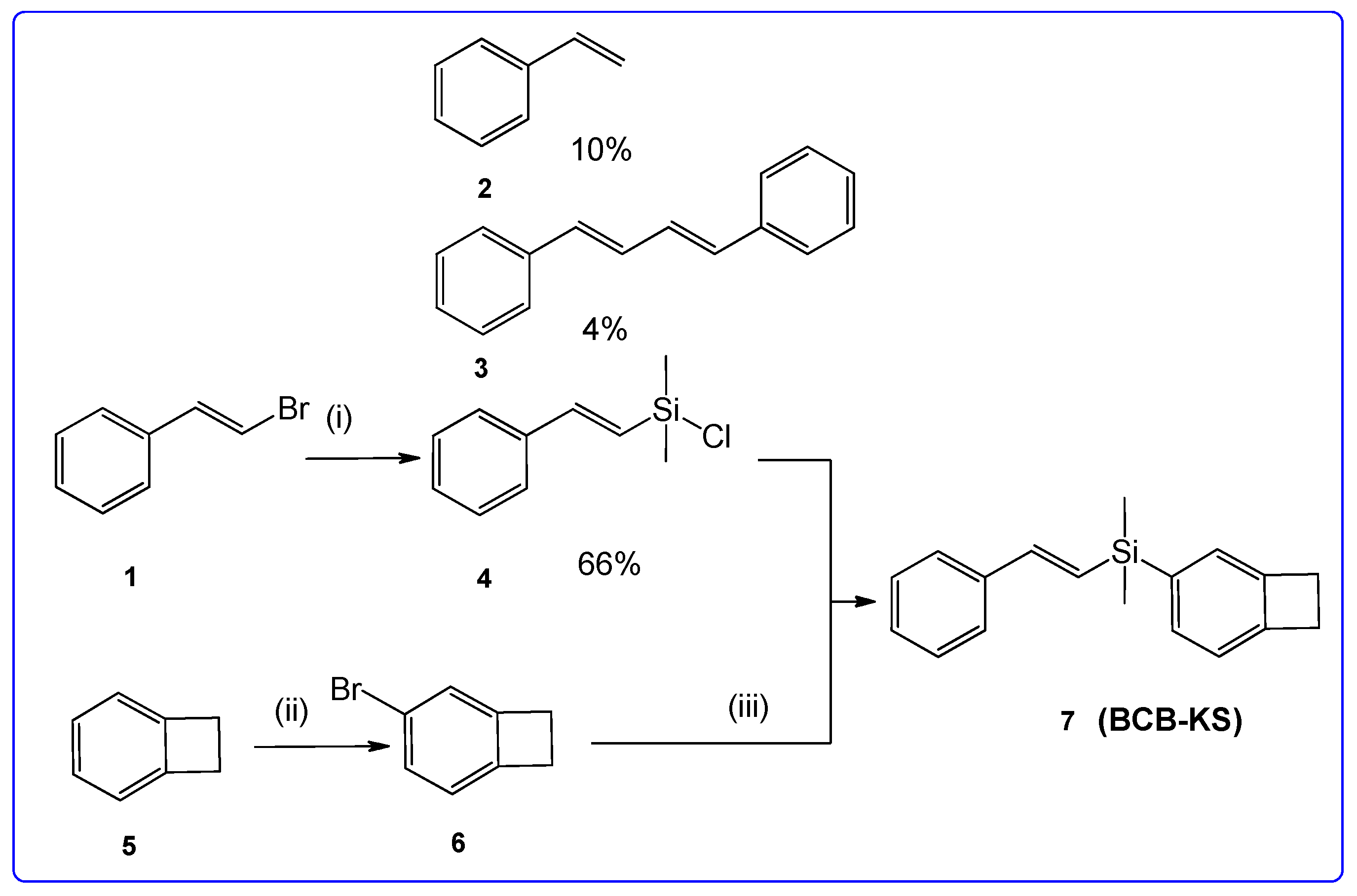
2. Results and Discussion
3. Materials and Methods
3.1. 4-Bromobenzocyclobutene (6)
3.2. Dimethylchloro((E)-styryl)silane (4)
3.3. Benzocyclobuten-3-yldimethyl((E)-styryl)silane (7) (BCB-KS)
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Pantoja, M.; Abenojar, J.; Martínez, M.A.; Velasco, F. Silane pretreatment of electrogalvanized steels: Effect on adhesive properties. Int. J. Adhes. Adhes. 2016, 65, 54–62. [Google Scholar] [CrossRef]
- Chen, B.; Lu, Z.; Meng, H.; Chen, Y.; Yang, L.; Zhang, H.; Xie, H.; Chen, C. Effectiveness of pre-silanization in improving bond performance of universal adhesives or self-adhesive resin cements to silica-based ceramics: Chemical and in vitro evidences. Dent. Mater. 2019, 35, 543–553. [Google Scholar] [CrossRef] [PubMed]
- Babanzadeh, S.; Mahjoub, A.R.; Mehdipour-Ataei, S. Novel soluble thermally stable silane-containing aromatic polyimides with reduced dielectric constant. Polym. Degrad. Stab. 2010, 95, 2492–2498. [Google Scholar] [CrossRef]
- Gelest. Industrial Silanes for Adhesives & Sealants, Coatings and Composites. Available online: https://www.gelest.com/wp-content/uploads/Industrial_Silanes.pdf (accessed on 20 December 2019).
- Levchenko, K.S.; Chudov, K.A.; Adamov, G.E.; Poroshin, N.O.; Shmelin, P.S.; Grebennikov, E.P.; Parshikov, Y.G. Photocurable and Thermosetting Polymer Materials on the Basis of Benzocyclobutene and Its Derivatives for Electronics. Russ. J. Gen. Chem. 2018, 88, 2793–2812. [Google Scholar] [CrossRef]
- Levchenko, K.S.; Chudov, K.A.; Demin, D.Y.; Adamov, G.E.; Poroshin, N.O.; Shmelin, P.S.; Grebennikov, E.P.; Chvalun, S.N.; Zubov, V.P. Synthesis of photo and thermosetting monomers and polymers based on benzocyclobutene. Russ. Chem. Bull. 2019, 68, 1321–1342. [Google Scholar] [CrossRef]
- Hu, H.; Liu, L.; Li, Z.; Zhao, C.; Huang, Y.; Chang, G.; Yang, J. Benzocyclobutene/vinylphenyl-introduced polycarbosilanes with low dielectric constant, high temperature performance and photopatternability. Polymer 2015, 66, 58–66. [Google Scholar] [CrossRef]
- Huanga, Y.; Zhanga, S.; Hua, H.; Weia, X.; Yub, H.; Yanga, J. Synthesis of poly(silmethylene)s via ring-opening polymerization of benzocyclobutene functionalized disilacyclobutene and their low-dielectric and thermal properties. Polym. Adv. Technol. 2017, 28, 1480–1488. [Google Scholar] [CrossRef]
- Yang, J.; Liu, S.; Zhu, F.; Huang, Y.; Li, B.; Zhang, L. New Polymers Derived from 4-Vinylsilylbenzocyclobutene Monomer with Good Thermal Stability, Excellent Film-Forming Property, and Low-Dielectric Constant. J. Polym. Sci. Part A Polym. Chem. 2011, 49, 381–391. [Google Scholar] [CrossRef]
- Yang, L.; Cao, K.; Huang, Y.; Chang, G.; Zhu, F.; Yang, J. Synthesis and properties of cross-linkable polysiloxane via incorporating benzocyclobutene. High Perform. Polym. 2014, 26, 463–469. [Google Scholar] [CrossRef]
- So, Y.-H.; Hahn, S.F.; Li, Y.; Reinhard, M.T. Styrene 4-Vinylbenzocyclobutene Copolymer for Microelectronic Applications. J. Polym. Sci. Part A Polym. Chem. 2008, 46, 2799–2806. [Google Scholar] [CrossRef]
- Ohba, K. Overview of Photo-definable Benzocyelobutene. Polym. Photopolym. Sci. Technol. 2002, 15, 177–182. [Google Scholar] [CrossRef]
- Kirchhoff, R.A.; Bruza, K.J. Polymers from Benzocyclobutenes. In High Performance Polymers; Hergenrother, P.M., Ed.; Advances in Polymer Science; Springer: Berlin/Heidelberg, Germany, 1994; Volume 117, pp. 1–66. [Google Scholar] [CrossRef]
- Pandarus, V.; Ciriminna, R.; Gingras, G.; Béland, F.; Kaliaguine, S.; Pagliaro, M. Waste-free and efficient hydrosilylation of olefins. Green Chem. 2019, 21, 129–140. [Google Scholar] [CrossRef]
- Brook, M.A.; Neuy, A. The beta-effect: Changing the ligands on silicon. J. Org. Chem. 1990, 55, 3609–3616. [Google Scholar] [CrossRef]
- Roth, M.; Ahles, M.; Gawrisch, C.; Schwalm, T.; Schmechel, R.; Melzer, C.; von Seggern, H.; Rehahn, M. Rodlike Tetracene Derivatives. Chem. Eur. J. 2017, 23, 13445–13454. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Z.; Wang, Z.-Y.; Ni, S.-F.; Dang, L.; Lee, H.K.; Peng, X.-S.; Wong, H.N.C. Ligand-Free Iron-Catalyzed Carbon(sp2)−Carbon(sp2) Oxidative Homo-Coupling of Alkenyllithiums. Org. Lett. 2019, 21, 700–704. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, B.P.S.; Sarkar, A. Functionalized vinylsilanes via highly efficient and recyclable Pt-nanoparticle catalysed hydrosilylation of alkynes. Dalton Trans. 2017, 46, 8709–8715. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Levchenko, K.S.; Chudov, K.A.; Demin, D.Y.; Shmelin, P.S.; Grebennikov, E.P. Bicyclo[4.2.0]octa-1,3,5-trien-3-yl-dimethyl((E)-styryl)-silane. Molbank 2020, 2020, M1102. https://doi.org/10.3390/M1102
Levchenko KS, Chudov KA, Demin DY, Shmelin PS, Grebennikov EP. Bicyclo[4.2.0]octa-1,3,5-trien-3-yl-dimethyl((E)-styryl)-silane. Molbank. 2020; 2020(1):M1102. https://doi.org/10.3390/M1102
Chicago/Turabian StyleLevchenko, Konstantin S., Konstantin A. Chudov, Dmitri Yu. Demin, Pavel S. Shmelin, and Evgeny P. Grebennikov. 2020. "Bicyclo[4.2.0]octa-1,3,5-trien-3-yl-dimethyl((E)-styryl)-silane" Molbank 2020, no. 1: M1102. https://doi.org/10.3390/M1102
APA StyleLevchenko, K. S., Chudov, K. A., Demin, D. Y., Shmelin, P. S., & Grebennikov, E. P. (2020). Bicyclo[4.2.0]octa-1,3,5-trien-3-yl-dimethyl((E)-styryl)-silane. Molbank, 2020(1), M1102. https://doi.org/10.3390/M1102



