1,1,4,7-Tetramethyldecahydro-1H-cyclopropa[e]azulen-7-ol from the Stembark Chisocheton pentandrus
Abstract
:1. Introduction
2. Results
Extraction and Isolation
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Plant Material
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Supratman, U.; Naibaho, W.; Salam, S.; Maharani, R.; Hidayat, A.T.; Harneti, D.; Nurlelasari; Shiono, Y. Cytotoxic triterpenoid from the bark of Chisocheton patens Blume (Meliaceae). Phytochem. Lett. 2019, 30, 81–87. [Google Scholar] [CrossRef]
- Mohamad, K.; Hirasawa, Y.; Litaudon, M.; Awang, K.; Hamid, A.; Hadi, A.; Takeya, K.; Ekasari, W.; Widyawaruyanti, A.; Zaini, N.C.; et al. Ceramicines B–D, new antiplasmodial limonoids from Chisocheton ceramicus. Bioorg. Med. Chem. 2009, 17, 727–730. [Google Scholar] [CrossRef] [PubMed]
- Joshi, M.N.; Chowdhury, B.L.; Vishnoi, S.P.; Shoeb, A.; Kapil, R.S. Antiviral activity of (+)-odorinol. Planta Medica 1987, 53, 254–255. [Google Scholar] [CrossRef] [PubMed]
- Agbedahunsi, J.M.; Fakoya, F.A.; Adesanya, S.A. Studies on the anti-inflammatory and toxic effects of the stem bark of Khaya ivorensis (Meliaceae) on rats. Phytomedicine 2004, 11, 504–508. [Google Scholar] [CrossRef] [PubMed]
- Nurlelasari; Katja, D.G.; Harneti, D.; Wardayo, M.M.; Supratman, U.; Awang, K. Limonoids from the seeds of Chisocheton macrophyllus. Chem. Nat. Compd. 2017, 53, 83–87. [Google Scholar] [CrossRef]
- Supriatno; Nurlelasari; Herlina, T.; Harneti, D.; Maharani, R.; Hidayat, A.T.; Mayanti, M.; Supratman, U.; Azmi, M.N.; Shiono, Y. A new limonoid from stembark of Chisocheton pentandrus (Meliaceae). Nat. Prod. Res. 2018, 32, 1–7. [Google Scholar] [CrossRef]
- Katja, D.G.; Farabi, K.; Nurlelasari; Harneti, D.; Mayanti, T.; Supratman, U.; Awang, K.; Hayashi, H. Cytotoxic constituents from the bark of Chisocheton cumingianus (Meliaceae). J. Asian Nat. Prod. Res. 2016, 19, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Katja, D.G.; Farabi, K.; Harneti, D.; Mayanti, T.; Supratma, U. Cytotoxic triterpenoid from the stembark of Chisocheton celebicus (Meliaceae). Makara J. Sci. 2017, 21, 8–12. [Google Scholar] [CrossRef]
- Kaplan, M.A.C.; Pugialli, H.R.L.; Lopes, D.; Gottlieb, H.E. The stereochemistry of ledol from Renealmia chrysotrycha. Phytochemistry 2000, 55, 749–753. [Google Scholar] [CrossRef]
- Phongmaykin, J.; Kumamoto, T.; Ishikawa, T.; Suttisri, R.; Saifah, E. A new sesquiterpene and other terpenoid constituents of Chisocheton penduliflorus. Arch. Pharm. Res. 2008, 31, 21–27. [Google Scholar] [CrossRef] [PubMed]
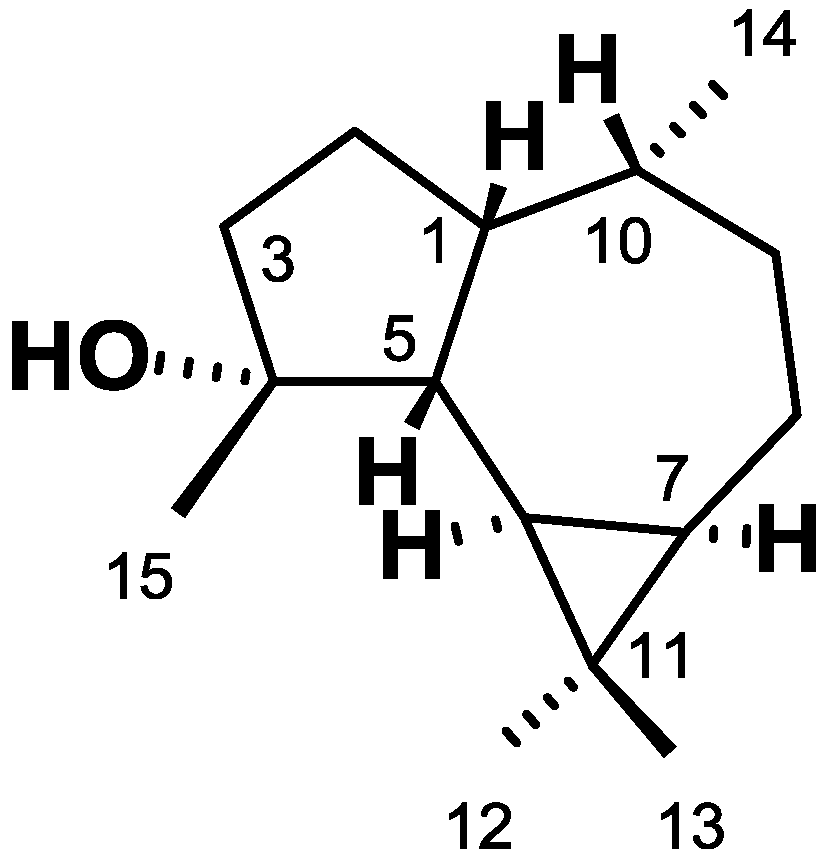
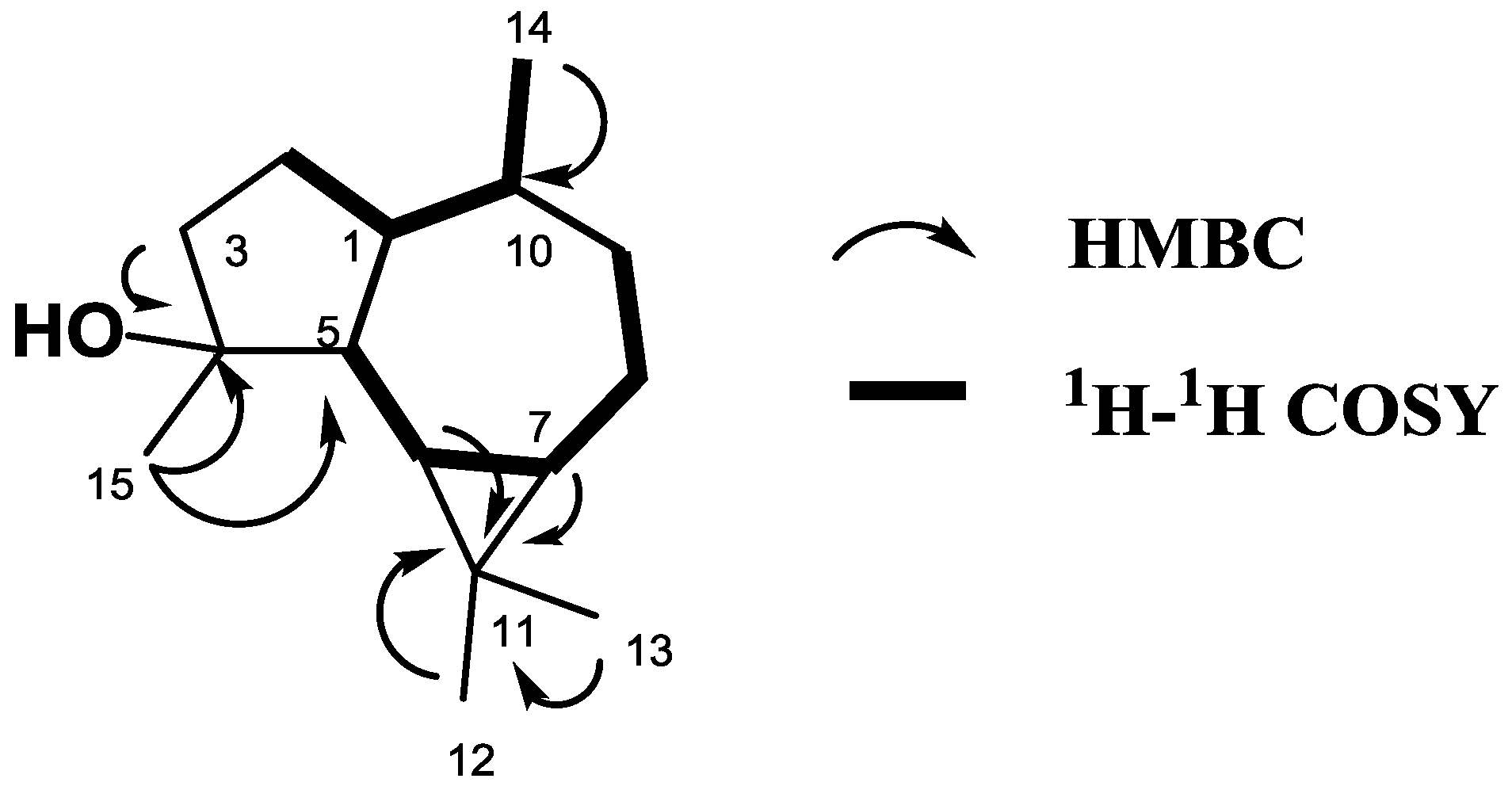
| C | δC | δH (ΣH., mult., J = Hz) |
|---|---|---|
| 1 | 39.7 | 1.72 (1H, m) |
| 2 | 29.1 | 1.15 (1H, m) 1.68 (1H, m) |
| 3 | 37.8 | 1.45 (1H, m) 1.59 (1H, m) |
| 4 | 76.6 | - |
| 5 | 58.2 | 1.69 (1H, m) |
| 6 | 22.3 | 0.10 (1H, t, 9.3) |
| 7 | 28.6 | 0.51 (1H, ddd, 6.0, 9.6) |
| 8 | 18.8 | 1.29 (1H, m) 1.52 (1H, m) |
| 9 | 25.8 | 1.49 (1H, m) 1.54 (1H, m) |
| 10 | 38.5 | 1.85 (1H, m) |
| 11 | 18.4 | - |
| 12 | 16.3 | 0.88 (3H, s) |
| 13 | 28.7 | 0.92 (3H, s) |
| 14 | 16.1 | 0.84 (3H, d, 6.8) |
| 15 | 32.1 | 1.04 (3H, s) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fareza, M.S.; Nurlelasari; Supratman, U.; Katja, D.G.; Husna, M.H.; Awang, K. 1,1,4,7-Tetramethyldecahydro-1H-cyclopropa[e]azulen-7-ol from the Stembark Chisocheton pentandrus. Molbank 2019, 2019, M1092. https://doi.org/10.3390/M1092
Fareza MS, Nurlelasari, Supratman U, Katja DG, Husna MH, Awang K. 1,1,4,7-Tetramethyldecahydro-1H-cyclopropa[e]azulen-7-ol from the Stembark Chisocheton pentandrus. Molbank. 2019; 2019(4):M1092. https://doi.org/10.3390/M1092
Chicago/Turabian StyleFareza, Muhamad Salman, Nurlelasari, Unang Supratman, Dewa Gede Katja, Muhamad Hafiz Husna, and Khalijah Awang. 2019. "1,1,4,7-Tetramethyldecahydro-1H-cyclopropa[e]azulen-7-ol from the Stembark Chisocheton pentandrus" Molbank 2019, no. 4: M1092. https://doi.org/10.3390/M1092
APA StyleFareza, M. S., Nurlelasari, Supratman, U., Katja, D. G., Husna, M. H., & Awang, K. (2019). 1,1,4,7-Tetramethyldecahydro-1H-cyclopropa[e]azulen-7-ol from the Stembark Chisocheton pentandrus. Molbank, 2019(4), M1092. https://doi.org/10.3390/M1092





