Benzyl (R)-2-(Acetylthio)Propanoate: A Promising Sulfur Isoster of (R)-Lactic Acid and Ester Precursors
Abstract
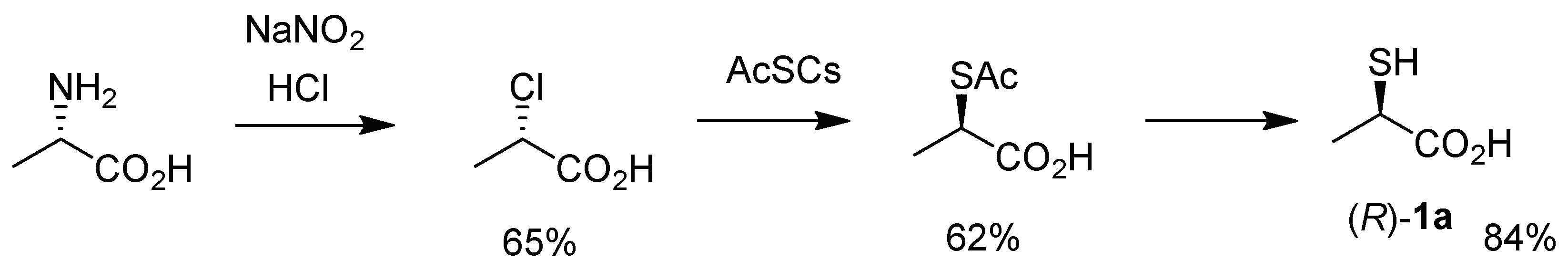
:1. Introduction
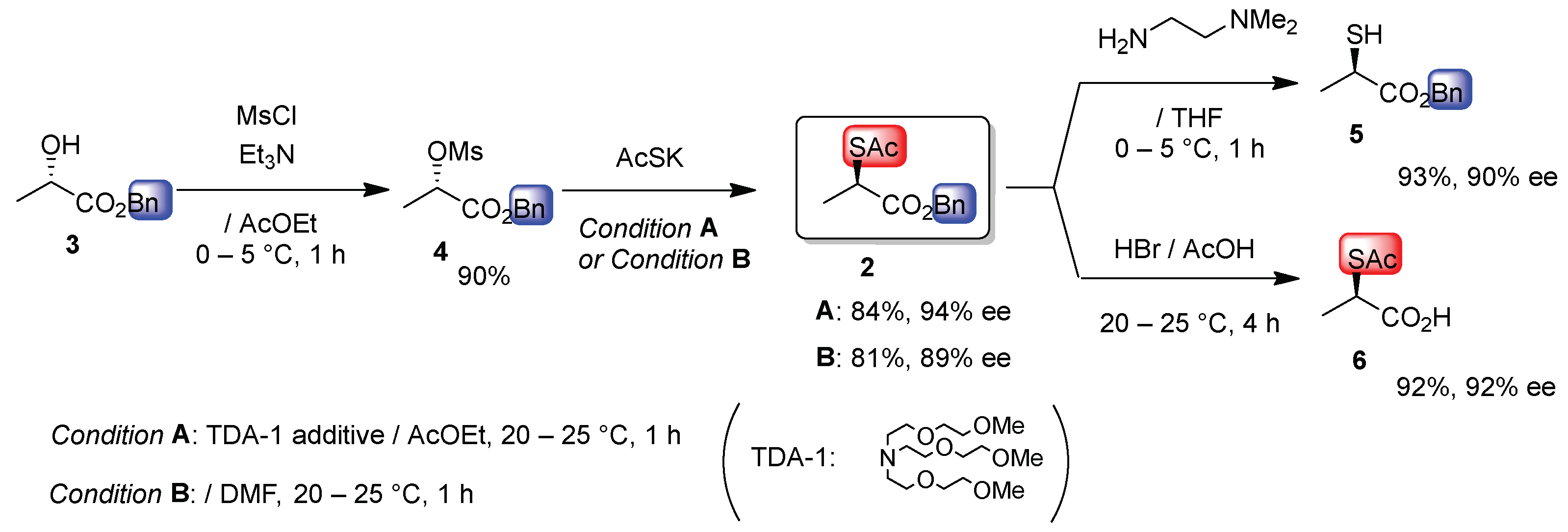
2. Results
3. Experimental Section

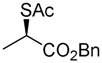
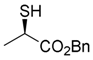

4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Toru, T.; Bolm, C. Organosulfur Chemistry in Asymmetric Synthesis; Wiley-VCH: Weinheim, Germany, 2008. [Google Scholar]
- Clayden, J.; MacLellan, P. Asymmetric Synthesis of Tertiary Thiols and Thioesters. Beilstein. J. Org. Chem. 2011, 7, 582–595. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, R.; Tanabe, Y. Chiral Syntheses of Methyl (R)-2-Sulfanylcarboxylic Esters and Acids with Optical Purity Determination using HPLC. Chirality 2018, 30, 816–827. [Google Scholar] [CrossRef] [PubMed]
- Labadarios, D.; Davis, M.; Portmann, B.; Williams, R. Paracetamol-induced Hepatic Necrosis in the Mouse: Relationship between Covalent Binding, Hepatic Glutathione Depletion, and Protective Effect of α-Mercaptopropionylglycine. Biochem. Pharmacol. 1977, 26, 31–35. [Google Scholar] [CrossRef]
- Wang, H.; Ma, C.; Zhou, J.; Liu, X.Q. Stereoselective Analysis of Tiopronin Enantiomers in Rat Plasma Using High-Performance Liquid Chromatography-Electrospray Ionization Mass Spectrometry After Chiral Derivatization. Chirality 2009, 21, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Oishi, H.; Noto, T.; Sasaki, M.; Suzuki, K.; Hayashi, T.; Okazaki, H.; Ando, K.; Sawada, M. Thiolactomycin, a New Antibiotic. I. Taxonomy of the Producing Organism, Fermentation and Biological Properties. J. Antibiot. 1982, 35, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Tsuzuki, K.; Omura, S. Synthesis and Biological Activities of Thiotetromycin Analogs. J. Antibiot. 1983, 36, 1589–1591. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.L.; Salvono, J.M. Total Synthesis of (±) thiolactomycin. Tetrahedron Lett. 1984, 25, 5243–5246. [Google Scholar] [CrossRef]
- McFadden, J.M.; Frehywot, G.L.; Townsend, C.A. A Flexible Route to (5R)-Thiolactomycin, a Naturally Occurring Inhibitor of Fatty Acid Synthesis. Org. Lett. 2002, 4, 3859–3862. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, Y.; Suzukamo, G.; Komuro, Y.; Imanishi, N.; Morooka, S.; Enomoto, M.; Kojima, A.; Sanemitsu, Y.; Mizutani, M. Structure-Activity Relationship of Optically Active 2-(3-Pyridyl)thiazolidin-4-Ones as an Anti-PAF Antagonist. Tetrahedron Lett. 1991, 32, 379–382. [Google Scholar] [CrossRef]
- Tanabe, Y.; Yamamoto, H.; Murakami, M.; Yanagi, K.; Kubota, Y.; Okumura, H.; Sanemitsu, Y.; Suzukamo, G. Synthetic Study of the Highly Potent and Selective Anti-Platelet Activating Factor Thiazolidin-4-one Agents and Related Compounds. J. Chem. Soc. Perkin Trans. 1 1995, 935–947. [Google Scholar] [CrossRef]
- Greenlee, M.L.; Laub, J.B.; Balkovec, J.M.; Hammond, M.L.; Hammond, G.G.; Pompliano, D.L. Epstein-Toney, J.H. Synthesis and SAR of thioester and Thiol Inhibitors of IMP-1 Metallo-β-Lactamase. Bioorg. Med. Chem. Lett. 1999, 9, 2549–2554. [Google Scholar] [CrossRef]
- Singh, J.; Kronenthal, D.R.; Schwinden, M.; Godfrey, J.D.; Fox, R.; Vawter, E.J.; Zhang, B.; Kissick, T.P.; Patel, B.; Mneimne, O.; et al. Efficient Asymmetric Synthesis of the Vasopeptidase Inhibitor BMS-189921. Org. Lett. 2003, 5, 3155–3158. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.M.; Li, J.Y.; Chen, L.L.; Li, J.; Ye, Q.Z.; Nan, F.J. Design and Synthesis of Chromogenic Thiopeptolide Substrates as MetAPs Active Site Probes. Bioorg. Med. Chem. 2004, 12, 2853–2861. [Google Scholar] [CrossRef] [PubMed]
- Kumar, I.; Jolly, R.S. Transpeptidation Reactions of a Specific Substrate Catalyzed by the Streptomyces R61 DD-Peptidase: Characterization of a Chromogenic Substrate and Acyl Acceptor Design. Biochemistry 2005, 44, 9971–9979. [Google Scholar] [CrossRef] [PubMed]
- Marinozzi, M.; Carotti, A.; Sardella, R.; Buonerba, F.; Ianni, F.; Natalini, B.; Passeri, D.; Rizzo, G.; Pellicciari, R. Asymmetric Synthesis of the Four Diastereoisomers of a Novel Non-Steroidal Farnesoid X Receptor (FXR) Agonist: Role of the Chirality on the Biological Activity. Bioorg. Med. Chem. 2013, 21, 3780–3789. [Google Scholar] [CrossRef] [PubMed]
- Strijtveen, B.; Kellogg, R.M. Synthesis and Determination of Enantiomeric Excesses of Non-Racemic Tert-Thiols Derived from Chiral Secondary α-Mercaptocarboxylic Acids. Tetrahedron 1987, 43, 5039–5054. [Google Scholar] [CrossRef]
- Seebach, D.; Neaf, R.; Calderani. α-Alkylation of α-Heterosubstituted Carboxylic Acids without Racemization: EPC-Syntheses of Tertiary Alcohols and Thiols. Tetrahedron 1984, 40, 1313–1324. [Google Scholar] [CrossRef]
- Kapilashrami, K.; Bommineni, G.R.; Machutta, C.A.; Kim, P.; Lai, C.T.; Simmerling, C.; Picart, F.; Tonge, P.J. Thiolactomycin-Based β-Ketoacyl-AcpM Synthase A (KasA) Inhibitors. J. Biol. Chem. 2013, 288, 6045–6052. [Google Scholar] [CrossRef] [PubMed]
- De Vries, A.H.M.; Hof, R.P.; Staal, D.; Kellogg, R.M.; Feringa, B.L. Diastereoselective Synthesis of Pyridyl Substituted Thiazolidin-4-Ones. New Ligands for the Cu(I) Catalyzed Asymmetric Conjugate Addition of Diethylzinc to Enones. Tetrahedron Asymmetry 1997, 8, 539–1543. [Google Scholar] [CrossRef]
- Owen, L.N.; Rahman, M.B. The Synthesis and Reduction of Optically Active 2-Mercaptopropionic Acid and Some Derivatives. J. Chem. Soc. C 1971, 2432–2440. [Google Scholar] [CrossRef]
- Strijtveen, B.; Kellogg, R.M. Synthesis of (Racemization Prone) Optically Active Thiols by SN2 Substitution Using Cesium Thiocarboxylates. J. Org. Chem. 1986, 51, 3664–3671. [Google Scholar] [CrossRef]
- Zhang, Y.Z.; Zhu, S.F.; Cai, Y.; Mao, H.X.; Zhou, Q.L. Copper-Catalyzed Enantioselectivecarbenoidinsertion into S–H Bonds. Chem. Commun. 2009, 5362–5364. [Google Scholar] [CrossRef] [PubMed]
- Tanabe, Y.; Okabe, T.; Kakimizu, A.; Ohno, N.; Yoshioka, H. An Improved Method for Preparation of N-Alkyl-2(3H)-benzothiazolone. Bull. Chem. Soc. Jpn. 1983, 56, 1255–1256. [Google Scholar] [CrossRef]
- Tanabe, Y.; Sanemitsu, Y. A Covenient Synthesis of 3-Chloromethyl-2(3H)-benzothiazolone. Synthesis 1988, 482–484. [Google Scholar] [CrossRef]
- Sanemitsu, Y.; Kawamura, S.; Tanabe, Y. Regioselective α-Methoxycarbonyl-sulfenylation of Ketones and Aldehydes: A Versatile Method for Preparation of Thiazolones, Thiadiazinones, and 3-Indolethiols. J. Org. Chem. 1992, 57, 1053–1056. [Google Scholar] [CrossRef]
- Shotaro, I.; Nakatsuji, H.; Tanabe, Y. Straightforward Synthesis of N-Methyl-4-(Pin)B-2(3H)-benzothiazol-2-one: A promising Cross-Coupling Reagent. Molbank 2018, 1, M976. [Google Scholar] [CrossRef]
- Strijtveen, B.; Feringa, B.L.; Kellogg, R.M. Methyl Phosphoric Dichloride as Reagent for the Determination of the Enantiomeric Excess of Chiral Thiols. Scope and Limitation. Tetrahedron 1987, 43, 123–130. [Google Scholar] [CrossRef]
- Robl, J.A.; Kronenthal, D.R.; Goderey, J.D., Jr. Bicyclic Carboxylic Acids as Inhibitors of Neutral Endopeptidase and Angiotensin-Converting Enzyme. Euro. Patent EP 629627, 21 December 1994. [Google Scholar]
Sample Availability: Samples of the compounds 2, 5 and 6 are available from the authors. |
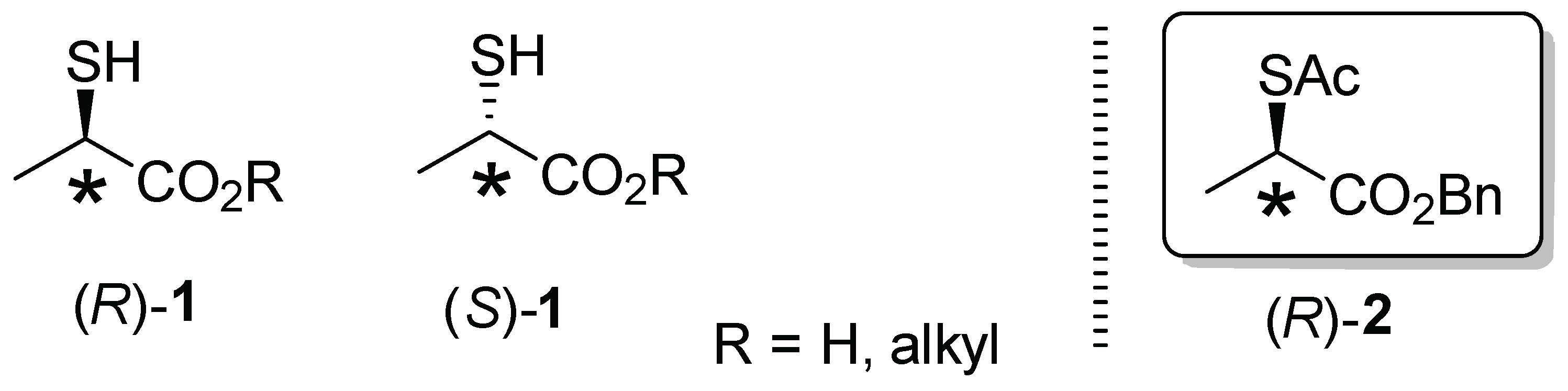
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sasaki, R.; Kawamoto, M.; Tanabe, Y. Benzyl (R)-2-(Acetylthio)Propanoate: A Promising Sulfur Isoster of (R)-Lactic Acid and Ester Precursors. Molbank 2018, 2018, M1010. https://doi.org/10.3390/M1010
Sasaki R, Kawamoto M, Tanabe Y. Benzyl (R)-2-(Acetylthio)Propanoate: A Promising Sulfur Isoster of (R)-Lactic Acid and Ester Precursors. Molbank. 2018; 2018(3):M1010. https://doi.org/10.3390/M1010
Chicago/Turabian StyleSasaki, Ryosuke, Momoyo Kawamoto, and Yoo Tanabe. 2018. "Benzyl (R)-2-(Acetylthio)Propanoate: A Promising Sulfur Isoster of (R)-Lactic Acid and Ester Precursors" Molbank 2018, no. 3: M1010. https://doi.org/10.3390/M1010
APA StyleSasaki, R., Kawamoto, M., & Tanabe, Y. (2018). Benzyl (R)-2-(Acetylthio)Propanoate: A Promising Sulfur Isoster of (R)-Lactic Acid and Ester Precursors. Molbank, 2018(3), M1010. https://doi.org/10.3390/M1010




