(Z)-4-[2-(3,4-Difluorophenyl)hydrazono]-3-methyl-1H-pyrazol-5(4H)-one
Abstract
1. Introduction
2. Results and Discussion
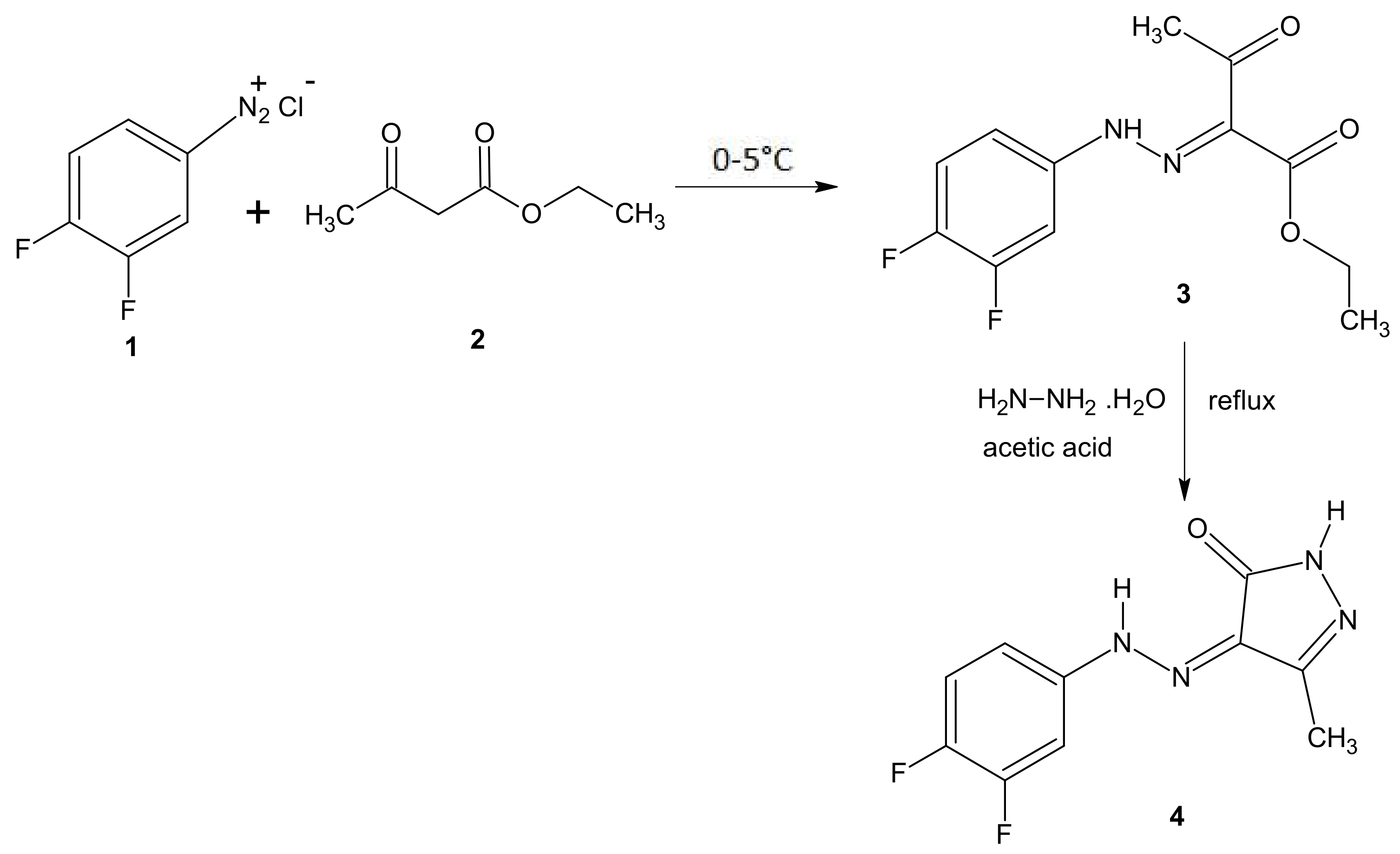
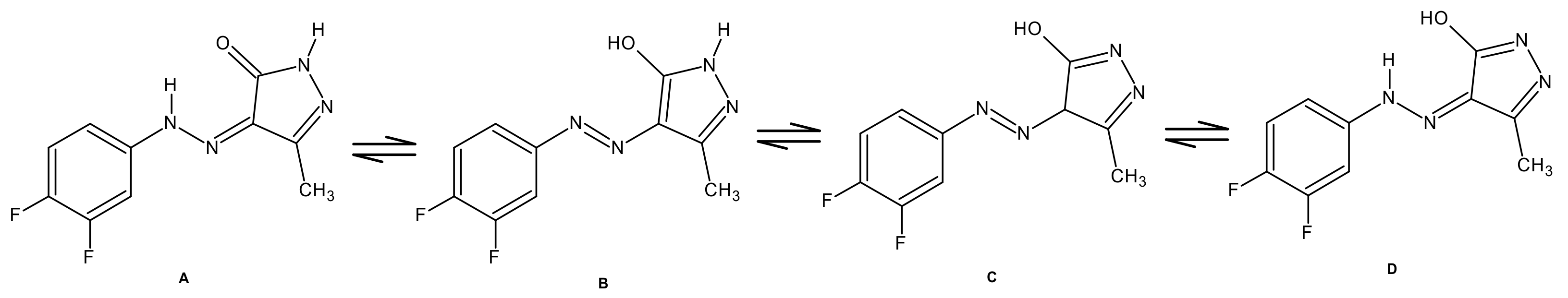
2.1. Chemistry
2.2. Anti-Inflammatory Studies
2.3. Antiproliferative Studies
2.4. Antibacterial Activity
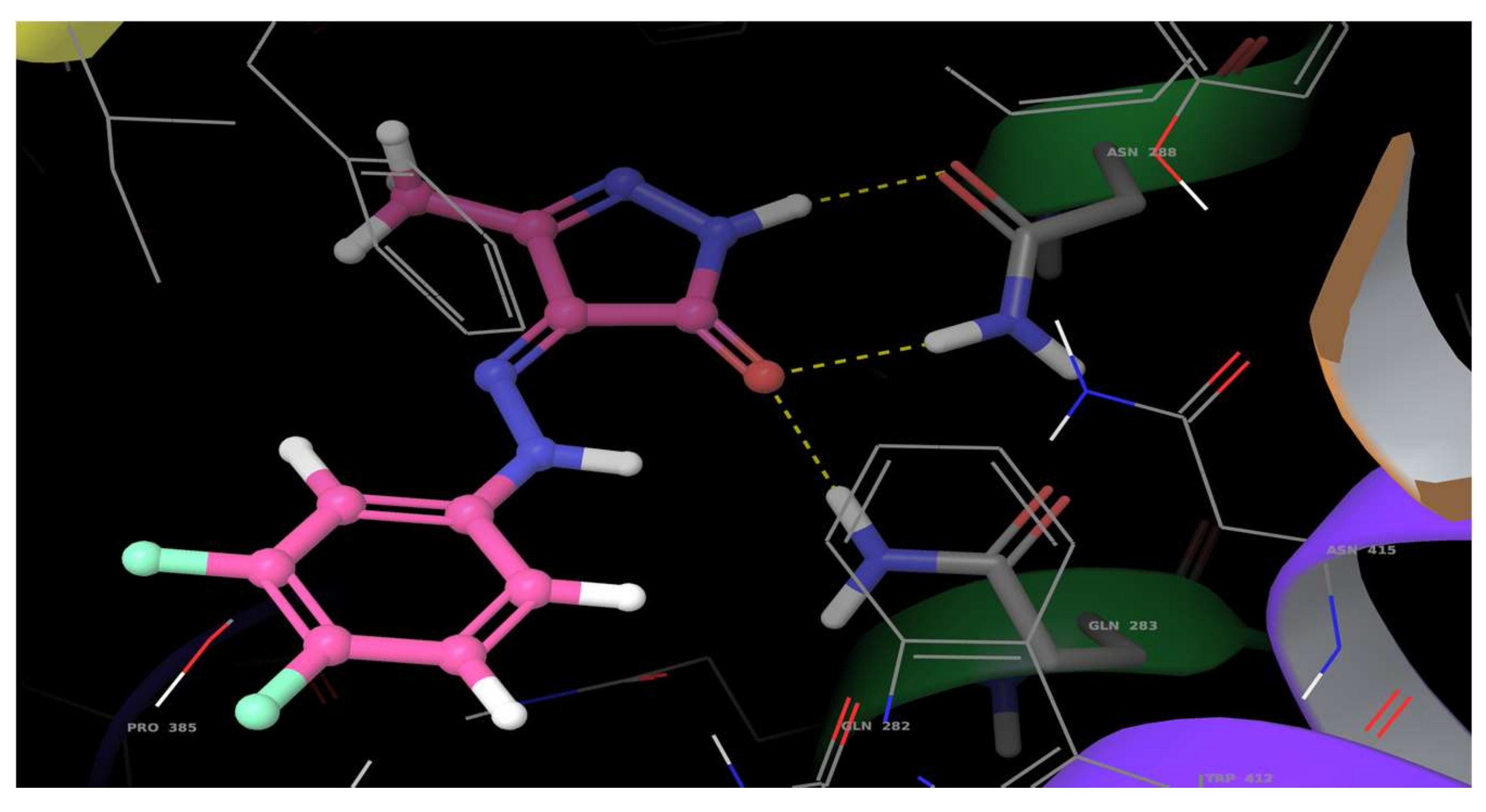
2.5. Molecular Docking Studies
3. Materials and Methods
3.1. Chemistry
3.1.1. General Information
Experimental
3.1.2. Synthesis of (Z)-4-[2-(3,4-Difluorophenyl)hydrazono]-3-methyl-1H-pyrazol-5(4H)-one (4)
3.2. Anti-Inflammatory Activity
3.3. Antiproliferative Studies
3.4. Antibacterial Activity
3.5. Molecular Docking Studies
4. Conclusions
Supplementary Materials
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Ei Ashry, E.S.H.; Awad, L.F.; Ibrahim, E.I.; Bdeewy, O.K. Synthesis of antipyrine derivatives derived from dimedone. Chin. J. Chem. 2007, 25, 570. [Google Scholar] [CrossRef]
- Uramaru, N.; Shigematsu, H.; Toda, A.; Eyanagi, R.; Kitamura, S.; Ohta, S. Design, synthesis, and pharmacological activity of nonallergenic pyrazolone-type antipyretic analgesics. J. Med. Chem. 2010, 53, 8727–8733. [Google Scholar] [CrossRef] [PubMed]
- Samshuddin, S.; Narayana, B.; Sarojini, B.K.; Khan, M.T.H.; Yathirajan, H.S.; Darshan Raj, C.G.; Raghavendra, R. Antimicrobial, analgesic, DPPH scavenging activities and molecular docking study of some 1,3,5-triaryl-2-pyrazolines. Med. Chem. Res. 2012, 21, 2012–2022. [Google Scholar] [CrossRef]
- Rao, B.S.; Akberali, P.M.; Holla, B.S.; Sarojini, B.K. Synthesis and studies on some new fluorine containing hydroxypyrazolines and 1H pyrazoles-as possible antiproliferative agents. J. Pharmacol. Toxicol. 2008, 3, 102–110. [Google Scholar]
- Bansal, E.; Srivastava, V.K.; Kumar, A. Synthesis and anti-inflammatory activity of 1-acetyl-5-substituted aryl-3-(β-aminonaphthyl)-2-pyrazolines and β-(substituted aminoethyl) amidonaphthalenes. Eur. J. Med. Chem. 2001, 36, 81–92. [Google Scholar] [CrossRef]
- Sarojini, B.K.; Vidyagayatri, M.; Darshanraj, C.G.; Bharath, B.R.; Manjunatha, H. DPPH Scavenging Assay of Novel 1, 3-disubstituted-1H-pyrazol-5-ols and their in silico Studies on Some Proteins Involved in Alzheimers Disease Signalling Cascade. Lett. Drug Des. Discov. 2010, 7, 214–224. [Google Scholar] [CrossRef]
- Higashi, Y.; Jitsuikia, D.; Chayama, K.; Yoshizumia, M. Edaravone (3-methyl-1-Phenyl-2-pyrazolin-5-one), anovel free radical scavenger, for treatment of cardiovascular diseases. Recent Patents Cardiovasc. Drug Discov. 2006, 1, 85–93. [Google Scholar] [CrossRef]
- Karcı, F.; Karcı, F. The synthesis and solvatochromic properties of some novel heterocyclic disazo dyes derived from barbituric acid. Dyes Pigment. 2008, 77, 451–456. [Google Scholar] [CrossRef]
- Desai, K.C.; Indorwala, N.S. Environmentally sustainable analytical reagent like 1-(2’-chloro-5’-sulphophenyl)-3-methyl-4-azo-(2′′-carboxy-5′′-sulphonic acid)-5-pyrazolone as a spectrophotometric reagent for Mn (II). J. Environ. Res. Dev. 2012, 6, 1024–1028. [Google Scholar]
- Byun, J.W.; Lee, D.H.; Lee, Y.S. Preparation of polymer-bound pyrazolone active esters for combinatorial chemistry. Tetrahed. Lett. 2003, 44, 8063–8067. [Google Scholar] [CrossRef]
- Clària, J. Cyclooxygenase-2 biology. Curr. Pharma. Design 2003, 9, 2177–2190. [Google Scholar] [CrossRef]
- Allavena, P.; Signorelli, M.; Chieppa, M.; Erba, E.; Bianchi, G.; Marchesi, F.; Olimpio, C.O.; Bonardi, C.; Garbi, A.; Lissoni, A.; et al. Anti-inflammatory Properties of the Novel Antitumour Agent Yondelis (Trabectedin): Inhibition of Macrophage Differentiation and Cytokine Production. Cancer Res. 2005, 65, 2964–2971. [Google Scholar] [CrossRef] [PubMed]
- Greenhough, A.; Smartt, H.J.M.; Moore, A.E.; Roberts, H.R.; Williams, A.C.; Paraskeva, C.; Kaidi, A. The COX-2/PGE2pathway: Key roles in the hallmarks of cancer and adaptation to the tumour microenvironment. Carcinogenesis 2009, 30, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Dias, L.R.S.; Salvador, R.R.S. Pyrazole Carbohydrazide Derivatives of Pharmaceutical Interest. Pharmaceuticals 2012, 5, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.A.; Azam, F.; Rghigh, A.M.; Gbaj, A.; Zetrini, A.E. Structure-based design, synthesis, molecular docking, and biological activities of 2-(3-benzoyl phenyl) propanoic acid derivatives as dual mechanism drugs. J. Pharm. Bioallied Sci. 2012, 4, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Cha, Y.I.; DuBois, R.N. NSAIDs and cancer prevention: Targets downstream of COX-2. Annul. Rev. Med. 2007, 58, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Thun, M.J.; Henley, S.J.; Patrono, C. Nonsteroidal anti-inflammatory drugs as anticancer agents: mechanistic, pharmacologic, and clinical issues. J. Natl. Cancer. Inst. 2002, 94, 252–266. [Google Scholar] [CrossRef] [PubMed]
- Lanas, A. Nonsteroidal anti-inflammatory drugs and cyclooxygenase inhibition in the gastrointestinal tract: a trip from peptic ulcer to colon cancer. Am. J. Med. Sci. 2009, 338, 96–106. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, J.F.; Conalty, M.L. Proceedings of the Royal Irish Academy Section B. Biol. Geol. Chem. Sci. 1980, 80B, 385–394. [Google Scholar]
- Filler, R.; Saha, R. Fluorine in medicinal chemistry: a century of progress and a 60-year retrospective of selected highlights. Future Med. Chem. 2009, 1, 777–791. [Google Scholar] [CrossRef] [PubMed]
- Bohm, H.J.; Banner, D.; Bendels, S.; Kansy, M.; Kuhn, B.; Muller, K.; Sander, U.O.; Stahl, M. Fluorine in Medicinal Chemistry. Chem. Biol. Chem. 2004, 5, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Penning, T.D.; Talley, J.J.; Bertenshaw, S.R.; Carter, J.S.; Collins, P.W.; Docter, S.; Graneto, M.J.; Lee, L.F.; Malecha, J.W.; Miyashiro, J.M.; et al. Synthesis and Biological Evaluation of the 1.5 Diarylpyrazole Class of Cyclooxygenase-2 Inhibitors: Identification of 4-[5-(4-Methylphenyl)-3-(trifluoromethyl)-1H-pyrazole-1-yl] benzenesulfonamide (SC-58634, Celecoxib). J. Med. Chem. 1997, 40, 1347–1365. [Google Scholar] [CrossRef] [PubMed]
- Vidyagayatri, M.; Darshanraj, C.G.; Sarojini, B.K.; Sreenivasa, S.; Jayaramu, M. Synthesis, characterization, evaluation of antiproliferative and antimicrobial properties of new pyrazole derivatives. J. Pharm. Res. 2011, 4, 2787–2790. [Google Scholar]
- Sarojini, B.K.; Mohan, B.J.; Narayana, B.; Yathirajan, H.S.; Jasinski, J.P.; Butcher, R.J. (Z)-4-[2-(2,4-Dimethylphenyl)hydrazinylidene]-3-methylpyrazol-5(1H)-one. Acta Cryst. E 2013, E69, o532. [Google Scholar] [CrossRef] [PubMed]
- Fun, H.K.; Quah, C.K.; Kalluraya, B. 4-[2-(4-Chlorophenyl)hydrazinylidene]-3-methyl-1H-pyrazol-5(4H)-one. Acta Cryst. E 2011, E67, o2670. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.K.; Hassan, M.A.; Mohamed, M.M.; El-Sayed, A.M. Metal salt-catalyzed diazocoupling of 3-substituted-1H-pyrazol-2-in-5-ones in aqueous medium. Dyes Pigment. 2005, 66, 241–245. [Google Scholar] [CrossRef]
- Hassan, A.E.A.; Moustafa, A.H.; Tolbah, M.M.; Zohdy, H.F.; Haikal, A.Z. Synthesis and Antimicrobial Evaluationof Novel Pyrazolones and Pyrazolone Nucleosides. Nucleos. Nucleot. Nucl. 2012, 31, 783–800. [Google Scholar] [CrossRef] [PubMed]
- Warburg, O. On the origin of cancer cells. Science. 1956, 123, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, K.; Finer-Moore, J.S.; Pedersen, B.P.; Caboni, L.; Waight, A.; Hillig, R.C.; Bringmann, P.; Heisler, I.; Muller, T.; Siebeneicher, H.; et al. Mechanism of inhibition of human glucose transporter GLUT1 is conserved between cytochalasin B and phenylalanine amides. Proc. Natl. Acad. Sci. USA 2016, 113, 4711–4716. [Google Scholar] [CrossRef] [PubMed]
- Rajput, A.P.; Rajput, S.S. Synthesis and Microbial Screening of Seven Membered Heterocyclic ring Compounds from 1,2-diaminobenzene. Int. J. Pharm. Tech. Res. 2009, 1, 900–904. [Google Scholar]
- Arunkumar, S.; Ilango, K.; Manikandan, R.S.; Ramalakshmi, N. Synthesis and anti-inflammatory activity of some novel pyrazole derivatives of gallic acid. Euro. J. Chem. 2009, 6, S123–S128. [Google Scholar] [CrossRef][Green Version]
- Cetin, Y.; Bullerman, L.B. Cytotoxicity of Fusarium mycotoxins to mammalian cell cultures as determined by the MTT bioassay. Food. Chem. Toxicol. 2005, 43, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Paris, C.; Bertoglio, J.; Breard, J. Lysosomal and mitochondrial pathways in miltefosine-induced apoptosis in U937 cells. Apoptosis 2007, 12, 1257–1267. [Google Scholar] [CrossRef] [PubMed]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in vitro evaluating antimicrobial activity: A review. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef] [PubMed]
| Compound | % Inhibition | |
|---|---|---|
| 2 h | 4 h | |
| 4 | 68.29 ± 3.52 * | 71.16 ± 1.94 ** |
| PYZ-F | 61.32 ± 1.16 * | 63.13 ± 3.82 * |
| PYZ-di-CH3 | 59.36 ± 3.62 | 64.23 ± 2.44 * |
| Diclofenac sodium | 70.12 ± 1.82 | 79.76 ± 2.57 |
| Control (Tween-80, 1%) | NA | NA |
| Compound | IC50 (µM) | ||
|---|---|---|---|
| Cell Lines | |||
| HeLa | HepG2 | EAT | |
| 4 | 18 | 27 | 36 |
| PYZ-F | 124 | 112 | 46 |
| PYZ-di-CH3 | 92 | 89 | 113 |
| Compound | Minimum Inhibitory Concentration (μM) | |||
|---|---|---|---|---|
| Bacterial Strains | ||||
| Staphylococcus Aureus | Escherichia coli | Pseudomonas aeruginosa | Klebsiellapneumoniae | |
| 4 | 26.2 | 26.2 | 26.2 | 26.2 |
| PYZ-F | 56.8 | 56.8 | 113.6 | 113.6 |
| PYZ-di-CH3 | 108.6 | NI | 108.6 | 217.3 |
| Nitrofurazone | <31.56 | <31.56 | <31.56 | <31.56 |
| Control | 00 | 00 | 00 | 00 |
| Compounds | Interacting Residue | Docking Score | Type of Bond | Bond Length (Å) |
|---|---|---|---|---|
| Inhibitor (2S)-3-(4-Fluorophenyl)-2-[2-(3-hydroxyphenyl)ethanoylamino]-~{N}-[(1S)-1-phenylethyl]propanamide | Glu380 | −7.738 | H-bond | 1.98 |
| Gln282 | H-bond | 2.51 | ||
| Trp388 | H-bond | 1.99 | ||
| (Z)-4-[2-(3,4-Difluorophenyl)hydrazono]-3-methyl-1H-pyrazol-5(4H)-one (4) | Asn288 | −6.177 | H-bond | 1.92 |
| Asn288 | H-bond | 2.06 | ||
| Gln283 | H-bond | 1.96 | ||
| (Z)-4-[2-(4-Fluorophenyl)hydrazono]-3-methyl-1H-pyrazol-5(4H)-one (Pyz-F) | Gln282 | −5.347 | H-bond | 2.45 |
| Gln283 | H-bond | 1.92 | ||
| Asn288 | H-bond | 2.07 | ||
| Asn288 | H-bond | 1.88 | ||
| (Z)-4-[2-Phenyl hydrazono]-3-methyl-1H-pyrazol-5(4H)-one | Asn288 | −4.673 | H-bond | 2.08 |
| Gln283 | H-bond | 1.92 | ||
| Asn411 | H-bond | 2.06 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohan, B.J.; Sarojini, B.K.; Narayana, B.; Sindhu Priya, E.S. (Z)-4-[2-(3,4-Difluorophenyl)hydrazono]-3-methyl-1H-pyrazol-5(4H)-one. Molbank 2018, 2018, M989. https://doi.org/10.3390/M989
Mohan BJ, Sarojini BK, Narayana B, Sindhu Priya ES. (Z)-4-[2-(3,4-Difluorophenyl)hydrazono]-3-methyl-1H-pyrazol-5(4H)-one. Molbank. 2018; 2018(2):M989. https://doi.org/10.3390/M989
Chicago/Turabian StyleMohan, Billava Jayappa, Balladka Kunhanna Sarojini, Badiadka Narayana, and Enumadishetty Srinivasulu Sindhu Priya. 2018. "(Z)-4-[2-(3,4-Difluorophenyl)hydrazono]-3-methyl-1H-pyrazol-5(4H)-one" Molbank 2018, no. 2: M989. https://doi.org/10.3390/M989
APA StyleMohan, B. J., Sarojini, B. K., Narayana, B., & Sindhu Priya, E. S. (2018). (Z)-4-[2-(3,4-Difluorophenyl)hydrazono]-3-methyl-1H-pyrazol-5(4H)-one. Molbank, 2018(2), M989. https://doi.org/10.3390/M989





