Abstract
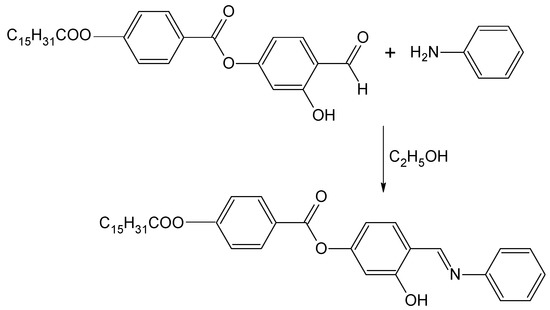
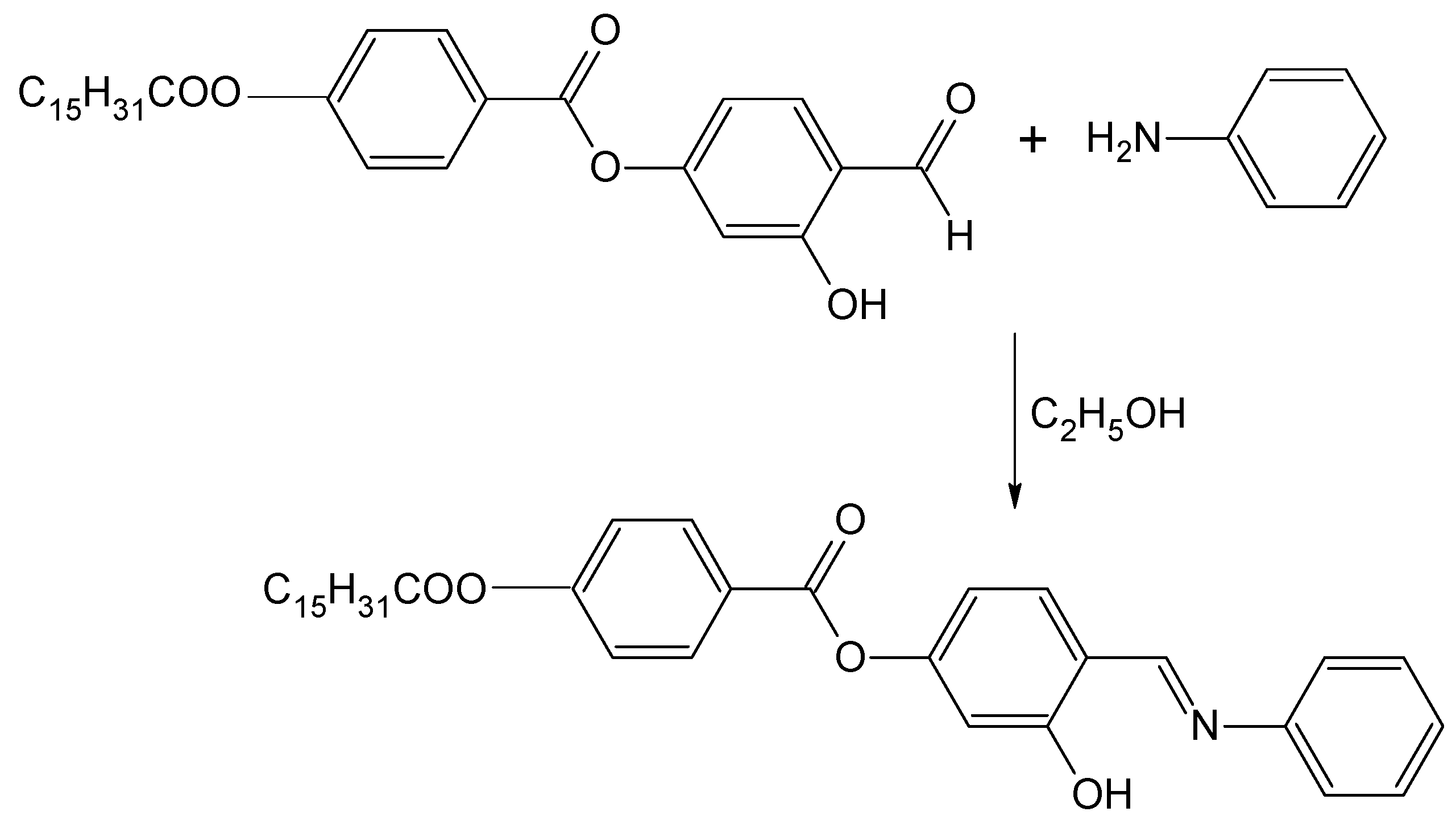
A new Schiff base ester, 3-hydroxy-4-[(phenylimino)methyl]phenyl 4-(hexadecanoyloxy)benzoate was synthesized and its IR, 1H-NMR, 13C-NMR and MS spectroscopic data are presented.
Mesomorphic materials have many practical applications, in particular as display devices, organic light emitting diodes, anisotropic networks, photoconductors and semiconductor materials [1,2,3]. Strong demand of new mesomorphic compounds for applications has encouraged the synthesis and study of numerous mesogens in particular, thermotropic liquid crystals [4,5]. Wide-ranging research on Schiff base core systems has been carried out since the discovery of MBBA, which exhibited room temperature nematic phase [6]. Several studies have been conducted on ester type of Schiff bases due to their interesting properties and substantial temperature range [7,8,9,10,11,12,13]. Thus, we report here another new derivative containing an hexadecanoyloxy chain, 3-hydroxy-4-[(phenylimino)methyl]phenyl 4-(hexadecanoyloxy)benzoate.
Experimental
4-(4-n-Hexadecanoyloxybenzoyloxy)-2-hydroxybenzaldehye was prepared according to a method that was described in our previous work [13]. In a round-bottom flask, a mixture of the aldehyde (2.48 g, 5.0 mmol), aniline (0.47 g, 5.0 mmol) and absolute ethanol (40 mL) was refluxed with stirring for 3 h. The reaction mixture was filtered and the solvent was removed from the filtrate by evaporation. Recrystallization from absolute ethanol gave the title compound as a yellow solid (1.14 g, 40%).
Melting point: 153–154 °C
MS (EI): m/z (rel. int. %) = 572 (1) (M+).
IR (KBr): νmax/ cm−1 2953, 2916, 2848 (C-H aliphatic), 1752 (C=O of C15H31COO- fragment), 1743 (C=O of benzoate), 1628 (C=N), 1605 (C=C aromatic), 1283 (C-O).
1H-NMR (400 MHz, CDCl3): δ/ppm 0.90 (t, 3H, J = 6.5 Hz, CH3-), 1.22–1.42 (m, 24H, CH3-(CH2)12-), 1.80 (quint, 2H, J = 7.2 Hz, -CH2-CH2COO-), 2.63 (t, 2H, J = 7.4 Hz, -CH2-COO-), 6.86 (dd, 1H, J = 2.2, 8.4 Hz, Ar-H), 6.92 (d, 1H, J = 2.2 Hz, Ar-H), 7.26–7.34 (m, 5H, Ar-H), 7.40–7.48 (m, 3H, Ar-H), 8.26 (dd, 2H, J = 2.3, 8.7 Hz, Ar-H), 8.66 (s, 1H, CH=N), 13.67 (s, 1H, O-H).
13C-NMR (100 MHz, CDCl3): δ/ppm 172.09 (C=O of C15H31COO-), 164.25 (C=O of benzoate), 162.17 (C=N), 163.0, 155.5, 155.0, 148.6, 133.6, 132.3, 129.9, 127.0, 126.8, 122.3, 121.6, 117.7, 113.3 and 111.0 for aromatic carbons, 34.8 (-CH2COO-), 25.3 (-CH2CH2COO-), 32.3, 30.1, 30.0, 29.9, 29.8, 29.7, 29.5, 23.1 (CH3(CH2)12), 14.5 (CH3(CH2)12).
Elemental analysis: Calculated for C36H45NO5, 75.63%, H, 7.93%, N, 2.45%; Found: C, 75.66%, H, 7.91%, N, 2.44%.
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgments
Authors would like to thank Universiti Tunku Abdul Rahman and Universitit Sains Malaysia for the financial supports and research facilities.
References and Notes
- Petti, L.; Rippa, M.; Fiore, A.; Manna, L.; Mormile, P. Optically induced light modulation in an hybrid nanocomposite system of inorganic CdSe/CdS nanorods and nematic liquid crystals. Opt. Mater. 2010, 32, 1011–1016. [Google Scholar] [CrossRef]
- Shurpo, N.A.; Vakshtein, M.S.; Kamanina, N.V. Effect of CdSe/ZnS semiconductor quantum dots on the dynamic properties of nematic liquid-crystalline medium. Tech. Phys. Lett. 2010, 36, 319–321. [Google Scholar] [CrossRef]
- Hoang, M.H.; Cho, M.J.; Kim, K.H.; Lee, T.W.; Jin, J.I.; Choi, D.H. Semiconducting 2,3,6,7,10,11-hexakis{4-(5-dodecylthiophen-2-yl)phenyl]ethynyl}triphenylene and its discotic liquid crystalline properties. Chem. Lett. 2010, 39, 396–397. [Google Scholar] [CrossRef]
- Yuksel, F.; Atilla, D.; Ahsen, V. Synthesis and characterization of liquid-crystalline asymmetric phthalocyanines. Polyhedron 2007, 26, 4551–4556. [Google Scholar] [CrossRef]
- Zhang, B.Y.; Meng, F.B.; Tian, M.; Xiao, W.Q. Side-chain liquid-crystalline polysiloxanes containing ionic mesogens and cholesterol ester groups. React. Funct. Polym. 2007, 66, 551–558. [Google Scholar] [CrossRef]
- Kelker, H.; Scheurle, B. Liquid-crystalline (nematic) phase with a particularly loq solidification point. Angew. Chem. Int. Ed. Engl. 1969, 8, 884–885. [Google Scholar] [CrossRef]
- Eran, B.E.; Nesrullajev, A.; Canli, N.Y. Characterization and investigation of the mesogenic, thermo-morphological and thermotropic properties of new chiral (S)-5-octyloxy-2-[{4-(2-methylbuthoxy)phenylimino(methyl)phenol liquid crystalline compound. Mat. Chem. Phys. 2008, 111, 555–558. [Google Scholar] [CrossRef]
- Ha, S.T.; Ong, L.K.; Ong, S.T.; Yeap, G.Y.; Wong, J.P.W.; Koh, T.M.; Lin, H.C. Synthesis and mesomorphic properties of new Schiff base esters with different alkyl chains. Chin. Chem. Lett. 2009, 20, 767–770. [Google Scholar] [CrossRef]
- Parra, M.; Vergara, J.; Zuniga, C.; Soto, E.; Sierra, T.; Serrano, J.L. New chiral Schiff’s bases with a 1,3,4-thiadizole ring in the mesogenic core: Synthesis, mesomorphic. Liq. Cryst. 2004, 32, 457–462. [Google Scholar] [CrossRef]
- Prajapati, A.K.; Varia, C.C. Azomesogens with polar chloro, nitro and phenolic –OH substituents. Liq. Cryst. 2008, 35, 1271–1277. [Google Scholar] [CrossRef]
- Vora, R.; Prajapati, A.K.; Kevat, J. Effect of terminal branching on mesomorphism. Mol. Cryst. Liq. Cryst. 2001, 357, 229–237. [Google Scholar] [CrossRef]
- Yeap, G.Y.; Ha, S.T.; Lim, P.L.; Boey, P.L.; Ito, M.M.; Sanehisa, S.; Youhei, Y. Synthesis, physical and mesomorphic properties of Schiff’s base esters containing ortho-, meta- and para-substituents in benzylidene-4'-alkanoyloxyanilines. Liq. Cryst. 2006, 33, 205–211. [Google Scholar] [CrossRef]
- Yeap, G.Y.; Ha, S.T.; Boey, P.L.; Mahmood, W.A.K.; Ito, M.M.; Youhei, Y. Synthesis and characterization of some new mesogenic Schiff base esters N-[4-(4-n-hexadecanoyloxybenzoyloxy)benzylidene]-4-substituted anilines. Mol. Cryst. Liq. Cryst. 2006, 452, 73–90. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).