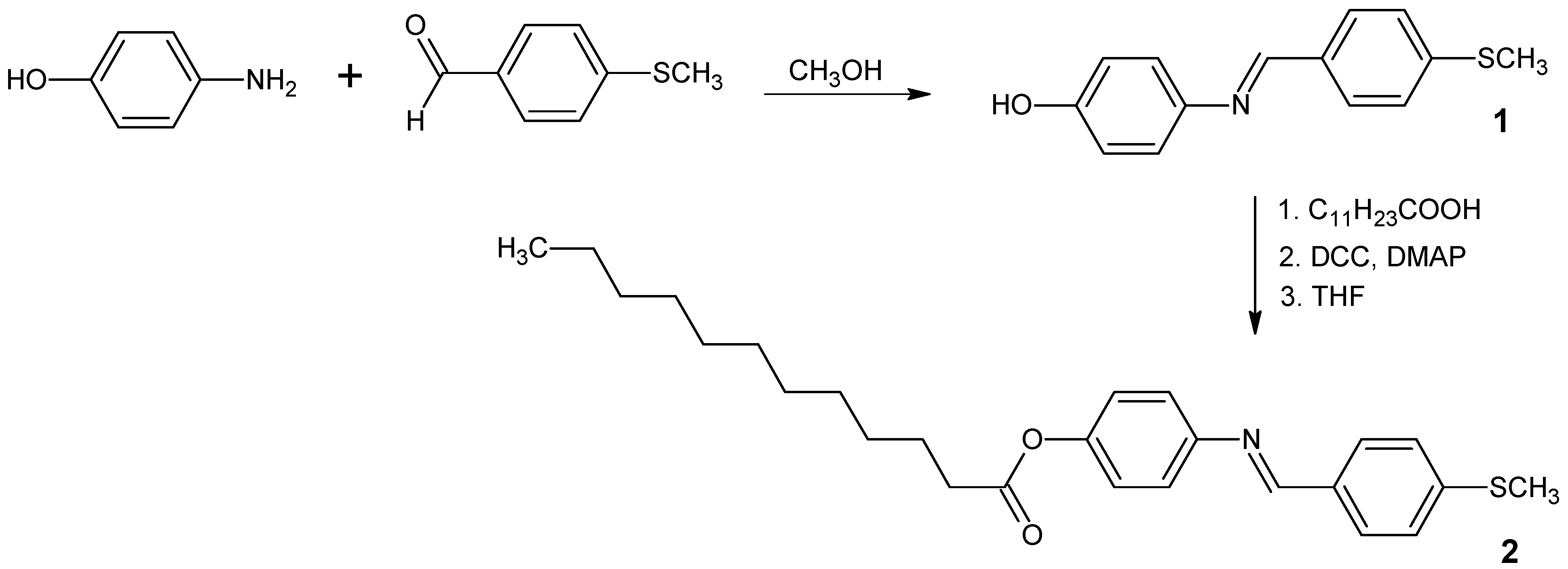
4-({[4-(Methylthio)phenyl]methylene}amino)phenyl Dodecanoate
Abstract
:Experimental
Supplementary Materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgements
References
- Petti, L.; Rippa, M.; Fiore, A.; Manna, L.; Mormile, P. Optically induced light modulation in an hybrid nanocomposite system of inorganic CdSe/CdS nanorods and nematic liquid crystals. Opt. Mater. 2010, 32, 1011–1016. [Google Scholar] [CrossRef]
- Shurpo, N.A.; Vakshtein, M.S.; Kamanina, N.V. Effect of CdSe/ZnS semiconductor quantum dots on the dynamic properties of nematic liquid-crystalline medium. Tech. Phys. Lett. 2010, 36, 319–321. [Google Scholar] [CrossRef]
- Hoang, M.H.; Cho, M.J.; Kim, K.H.; Lee, T.W.; Jin, J.I.; Choi, D.H. Semiconducting 2,3,6,7,10,11-hexakis{4-(5-dodecylthiophen-2-yl)phenyl]ethynyl}triphenylene and its discotic liquid crystalline properties. Chem. Lett. 2010, 39, 396–397. [Google Scholar] [CrossRef]
- Yuksel, F.; Atilla, D.; Ahsen, V. Synthesis and characterization of liquid crystalline unsymmetrically substituted phthalocyanines. Polyhedron 2007, 26, 4551–4556. [Google Scholar] [CrossRef]
- Zhang, B.Y.; Meng, F.B.; Tian, M.; Xiao, W.Q. Side-chain liquid-crystalline polysiloxanes containing ionic mesogens and cholesterol ester groups. React. Funct. Polym. 2005, 66, 551–558. [Google Scholar] [CrossRef]
- Yeap, G.Y.; Ha, S.T.; Lim, P.L.; Boey, P.L.; Ito, M.M.; Sanehisa, S.; Youhei, Y. Synthesis, physical and mesomorphic properties of Schiff’s base esters containing ortho-, meta- and para-substituents in benzylidene-4’-alkanoyloxyanilines. Liq. Cryst. 2006, 33, 205–211. [Google Scholar] [CrossRef]
- Ha, S.T.; Ong, L.K.; Ong, S.T.; Yeap, G.Y.; Wong, J.P.W.; Koh, T.M.; Lin, H.C. Synthesis and mesomorphic properties of new Schiff base esters with different alkyl chains. Chin. Chem. Lett. 2009, 20, 767–770. [Google Scholar] [CrossRef]
- Ha, S.T.; Ong, L.K.; Sivasothy, Y.; Yeap, G.Y.; Boey, P.L.; Lin, H.C. New mesogenic Schiff base esters with polar chloro substituent: synthesis, thermotropic properties and x-ray diffraction studies. Am. J. Appl. Sci. 2010, 7, 214–220. [Google Scholar] [CrossRef]
- Ha, S.T.; Ong, L.K.; Win, Y.F.; Koh, T.M.; Yeap, G.Y. Synthesis of New Schiff Base: 4-[(Pyridin-3-ylmethylene)amino]phenyldodecanoate. Molbank 2008, 2008, M582. [Google Scholar] [CrossRef]
- Ha, S.T.; Ong, L.K.; Win, Y.F.; Koh, T.M.; Yeap, G.Y. 4-[(Pyridin-3-ylmethylene)amino]phenylhexadecanoate. Molbank 2009, 2009, M584. [Google Scholar] [CrossRef]
- Ha, S.T.; Ong, L.K.; Win, Y.F.; Koh, T.M.; Yeap, G.Y. 4-[(Pyridin-3-ylmethylene)amino]phenyltetradecanoate. Molbank 2009, 2009, M585. [Google Scholar] [CrossRef]
- Ha, S.T.; Ong, L.K.; Win, Y.F.; Koh, T.M.; Yeap, G.Y. 4-[(Pyridin-3-ylmethylene)amino]phenyloctadecanoate. Molbank 2009, 2009, M591. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ha, S.-T.; Lee, T.-L.; Win, Y.-F.; Yeap, G.-Y. 4-({[4-(Methylthio)phenyl]methylene}amino)phenyl Dodecanoate. Molbank 2011, 2011, M735. https://doi.org/10.3390/M735
Ha S-T, Lee T-L, Win Y-F, Yeap G-Y. 4-({[4-(Methylthio)phenyl]methylene}amino)phenyl Dodecanoate. Molbank. 2011; 2011(3):M735. https://doi.org/10.3390/M735
Chicago/Turabian StyleHa, Sie-Tiong, Teck-Leong Lee, Yip-Foo Win, and Guan-Yeow Yeap. 2011. "4-({[4-(Methylthio)phenyl]methylene}amino)phenyl Dodecanoate" Molbank 2011, no. 3: M735. https://doi.org/10.3390/M735
APA StyleHa, S.-T., Lee, T.-L., Win, Y.-F., & Yeap, G.-Y. (2011). 4-({[4-(Methylthio)phenyl]methylene}amino)phenyl Dodecanoate. Molbank, 2011(3), M735. https://doi.org/10.3390/M735




