N,N'-1,2-Phenylenebis[4-(chloromethyl)benzamide]
Abstract
:Experimental Section
General
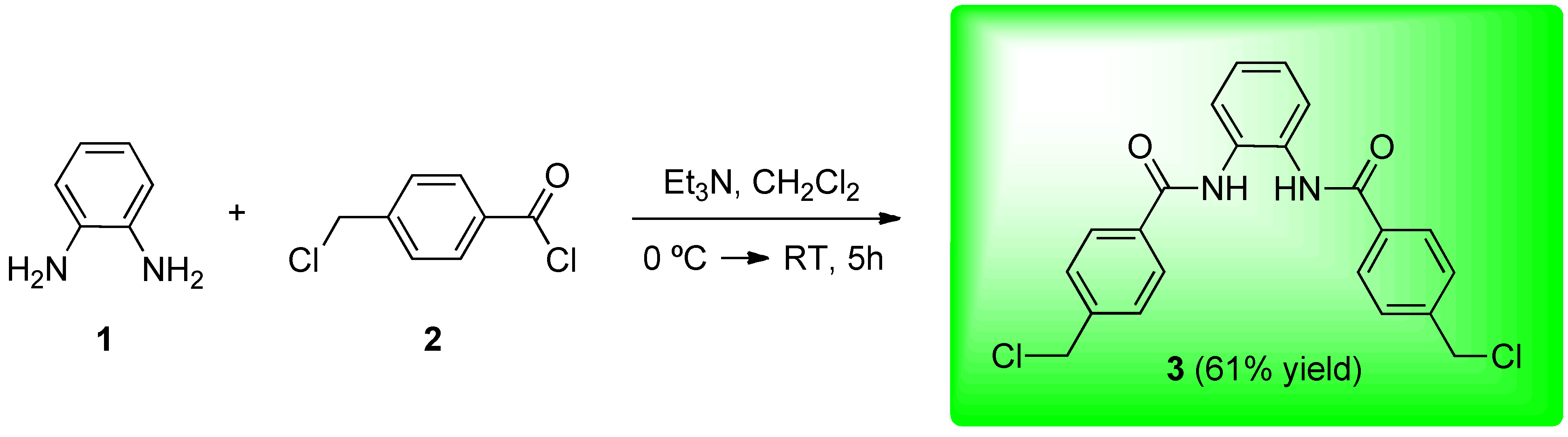
Synthesis of N,N'-1,2-phenylenebis[4-(chloromethyl)benzamide] (3)
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Supplementary File 4Acknowledgements
References and Notes
- Norman, M.H.; Rigdon, G.C.; Hall, W.R.; Navas, F. Structure-activity relationships of a series of substituted benzamides: Potent D2/5-HT2 antagonists and 5-HT1a agonists as neuroleptic agents. J. Med. Chem. 1996, 39, 1172–1188. [Google Scholar] [CrossRef] [PubMed]
- Liberg, D.; Lazarevic, B.; Pero, R.W.; Leanderson, T. N-substituted benzamides inhibit NFκB activation and induce apoptosis by separate mechanisms. Br. J. Cancer 1999, 81, 981–988. [Google Scholar] [CrossRef] [PubMed]
- Reich, S.H.; Johnson, T.; Wallace, M.B.; Kephart, S.E.; Fuhrman, S.A.; Worland, S.T.; Matthews, D.A.; Hendrickson, T.F.; Chan, F.; Meador, J., III; Ferre, R.A.; Brown, E.L.; DeLisle, D.M.; Patick, A.K.; Binford, S.L.; Ford, C.E. Substituted Benzamide Inhibitors of Human Rhinovirus 3C Protease: Structure-Based Design, Synthesis, and Biological Evaluation. J. Med. Chem. 2000, 43, 1670–1683. [Google Scholar] [CrossRef] [PubMed]
- Zollinger, H. Color Chemistry: Syntheses, Properties and Applications of Organic Dyes and Pigments, 3rd ed.; Wiley-VCH: Weinheim, Germany, 2003. [Google Scholar]
- Pero, R.W.; Olsson, A.; Amiri, A.; Chaplin, D. Multiple mechanisms of action of the benzamides and nicotinamides as sensitizers of radiotherapy: Opportunities for drug design. Cancer Detect. Prev. 1998, 22, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Rossberg, M.; Lendle, W.; Pfleiderer, G.; Tögel, A.; Dreher, E.-L.; Langer, E.; Rassaerts, H.; Kleinschmidt, P.; Strack, H.; Cook, R.; Beck, U.; Lipper, K.-A.; Torkelson, T.R.; Löser, E.; Beutel, K.K.; Mann, T. Chlorinated Hydrocarbons. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2006. [Google Scholar]
- For the synthesis of the linear analogues using p-phenylenediamine, see: Misawa, Y.; Koumura, N.; Matsumoto, H.; Tamaoki, N.; Yoshida, M. Hydrogels Based on Surfactant-Free Ionene Polymers with N,Nʹ′-(p-Phenylene)dibenzamide Linkages. Macromolecules 2008, 41, 8841–8846. [Google Scholar]
- See Supplementary Files.
- Armarego, W.L.F.; Perrin, D.D. Purification of Laboratory Chemicals, 4th ed.; Butterworth-Heinemann: Oxford, UK, 1996. [Google Scholar]
- The qualitative TLC analysis of the reaction crude showed the presence of additional minor products. Unfortunately, we were unable to isolate and identify such products by column chromatography. Nevertheless, the major loss in the yield comes most likely from the recrystal¬lization, filtration and washing procedures used to purify the title compound.
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bachl, J.; Díaz, D.D. N,N'-1,2-Phenylenebis[4-(chloromethyl)benzamide]. Molbank 2010, 2010, M705. https://doi.org/10.3390/M705
Bachl J, Díaz DD. N,N'-1,2-Phenylenebis[4-(chloromethyl)benzamide]. Molbank. 2010; 2010(4):M705. https://doi.org/10.3390/M705
Chicago/Turabian StyleBachl, Jürgen, and David D. Díaz. 2010. "N,N'-1,2-Phenylenebis[4-(chloromethyl)benzamide]" Molbank 2010, no. 4: M705. https://doi.org/10.3390/M705
APA StyleBachl, J., & Díaz, D. D. (2010). N,N'-1,2-Phenylenebis[4-(chloromethyl)benzamide]. Molbank, 2010(4), M705. https://doi.org/10.3390/M705



