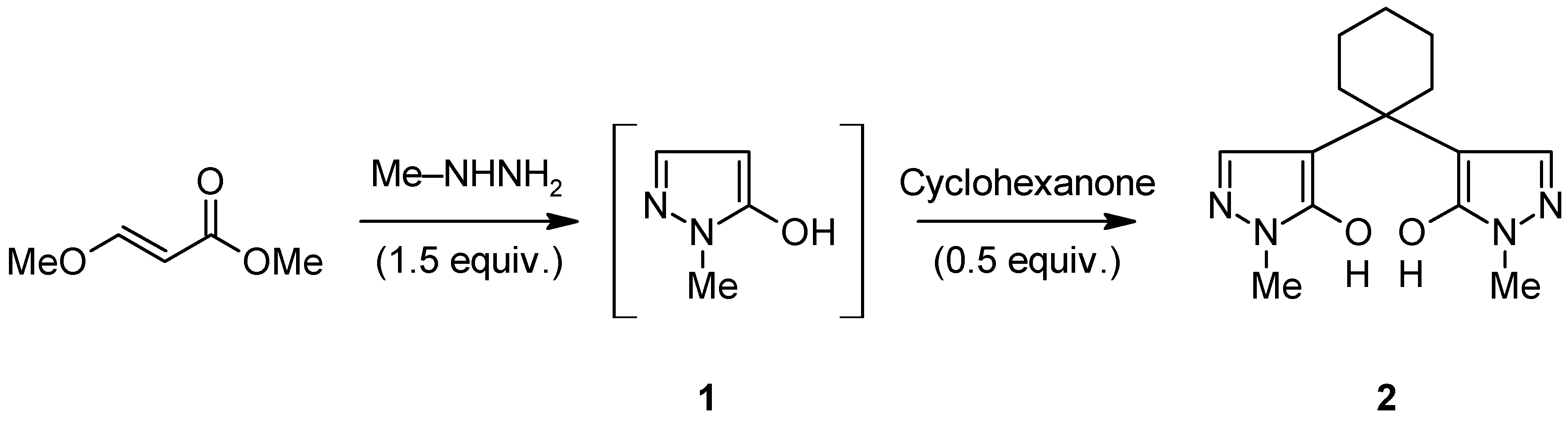
Synthesis of 4,4'-(Cyclohexane-1,1-diyl)bis(1-methyl- 1H-pyrazol-5-ol)
Abstract
:1. Introduction
2. Experimental
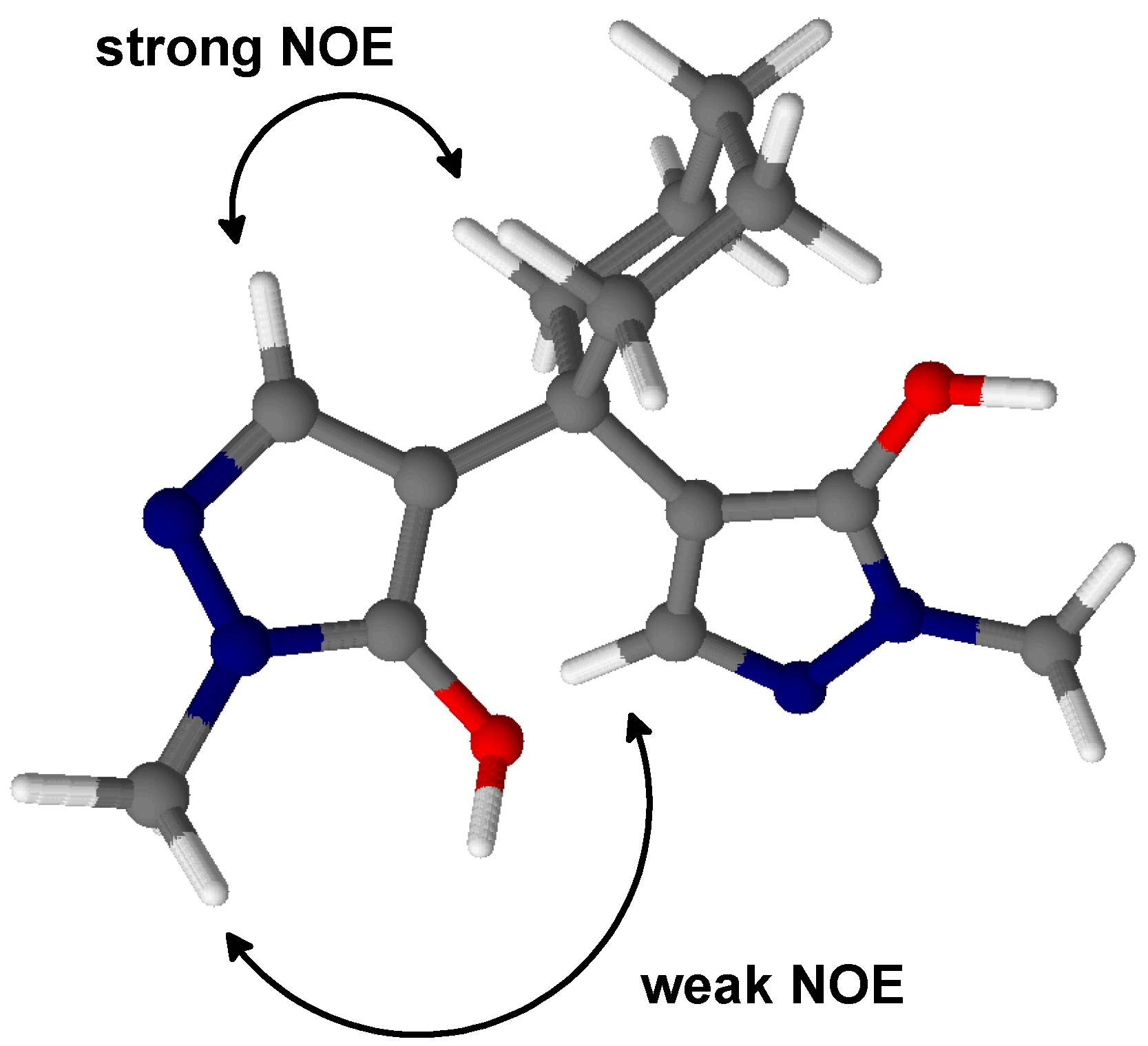
2.1. 4,4'-(Cyclohexane-1,1-diyl)bis(1-methyl-1H-pyrazol-5-ol) (2)
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Supplementary File 4Supplementary File 5Supplementary File 6References and Notes
- Eller, G.A.; Wimmer, V.; Haring, A.W.; Holzer, W. An efficient approach to heterocyclic analogues of xanthone: a short synthesis of all possible pyrido[5,6]pyrano[2,3-c]pyrazol-4(1H)-ones. Synthesis 2006, 24, 4219. [Google Scholar] [CrossRef]
- Eller, G.A.; Haring, A.W.; Datterl, B.; Zwettler, M.; Holzer, W. Tri- and tetracyclic heteroaromatic systems: synthesis of novel benzo-, benzothieno- and thieno-fused pyrano[2,3-c]pyrazol-4(1H)-ones. Heterocycles 2007, 71, 87. [Google Scholar] [CrossRef]
- Eller, G.A.; Holzer, W. A convenient approach to heterocyclic building blocks: synthesis of novel ring systems containing a [5,6]pyrano[2,3-c]pyrazol-4(1H)-one moiety. Molecules 2007, 12, 60. [Google Scholar] [CrossRef] [PubMed]
- Eller, G.A.; Datterl, B.; Holzer, W. Pyrazolo[4',3':5,6]pyrano[2,3-b]quinoxalin-4(1H)-one: synthesis and characterization of a novel tetracyclic ring system. J. Heterocycl. Chem. 2007, 44, 1139. [Google Scholar] [CrossRef]
- Eller, G.A.; Wimmer, V.; Holzer, W. Synthesis of novel polycyclic ring systems containing two pyrano[2,3-c]pyrazol-4(1H)-one moieties. Khim. Geterotsikl. Soedin. 2007, 1251. [Google Scholar]Chem. Heterocycl. Comp. 2007, 43, 1060. [CrossRef]
- Maywald, V.; Steinmetz, A.; Rack, M.; Gotz, N.; Gotz, R.; Henkelmann, J.; Becker, H.; Aiscar Bayeto, J.J. Preparation of 1-substituted 5- or 3-hydroxypyrazoles from alkoxyacrylates and hydrazines. PCT Int. Appl. WO 0031042, 2000. [Chem. Abstr., 2000, 133, 4655]. [Google Scholar]
- Eller, G.A.; Holzer, W. A one-step synthesis of pyrazolone. Molbank 2006, M464. [Google Scholar] [CrossRef]
- ACD/3D Viewer, version 10.00. Advanced Chemistry Development, Inc.: Toronto, ON, Canada, 2006; www.acdlabs.com.
- Upon trituration and subsequent recrystallization of the oily residue with/from i.e. diethyl ether, pure methylpyrazolone 1 can be obtained as colourless crystals. Mp 108.5–111 °C. 1H NMR (500 MHz, DMSO-d6): δ (ppm) 10.90 (br s, 1H, OH), 7.09 (d, 3J(H-3,H-4) = 1.9 Hz, 1H, H-3), 5.30 (d, 3J(H-4,H-3) = 1.9 Hz, 1H, H-4), 3.47 (s, 3H, NMe). 13C NMR (125 MHz, DMSO-d6): δ (ppm) 152.6 (C-5, 2J(C-5,H-4) = 5.7 Hz, 3J(C-5,H-3) = 10.5 Hz, 3J(C-5,NMe) = 1.9 Hz), 137.1 (C-3, 1J = 182.7 Hz, 2J(C-4,H-3) = 5.1 Hz), 86.1 (C-4, 1J = 176.1 Hz), 32.9 (NMe, 1J = 139.3 Hz). 15N NMR (50 MHz, DMSO-d6): δ (ppm) −201.9 (N-1); N-2 was not found.
- Sample Availability: Compounds 1 and 2 are available from MDPI.
© 2008 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Eller, G.A.; Holzer, W. Synthesis of 4,4'-(Cyclohexane-1,1-diyl)bis(1-methyl- 1H-pyrazol-5-ol). Molbank 2008, 2008, M569. https://doi.org/10.3390/M569
Eller GA, Holzer W. Synthesis of 4,4'-(Cyclohexane-1,1-diyl)bis(1-methyl- 1H-pyrazol-5-ol). Molbank. 2008; 2008(2):M569. https://doi.org/10.3390/M569
Chicago/Turabian StyleEller, Gernot A., and Wolfgang Holzer. 2008. "Synthesis of 4,4'-(Cyclohexane-1,1-diyl)bis(1-methyl- 1H-pyrazol-5-ol)" Molbank 2008, no. 2: M569. https://doi.org/10.3390/M569
APA StyleEller, G. A., & Holzer, W. (2008). Synthesis of 4,4'-(Cyclohexane-1,1-diyl)bis(1-methyl- 1H-pyrazol-5-ol). Molbank, 2008(2), M569. https://doi.org/10.3390/M569



