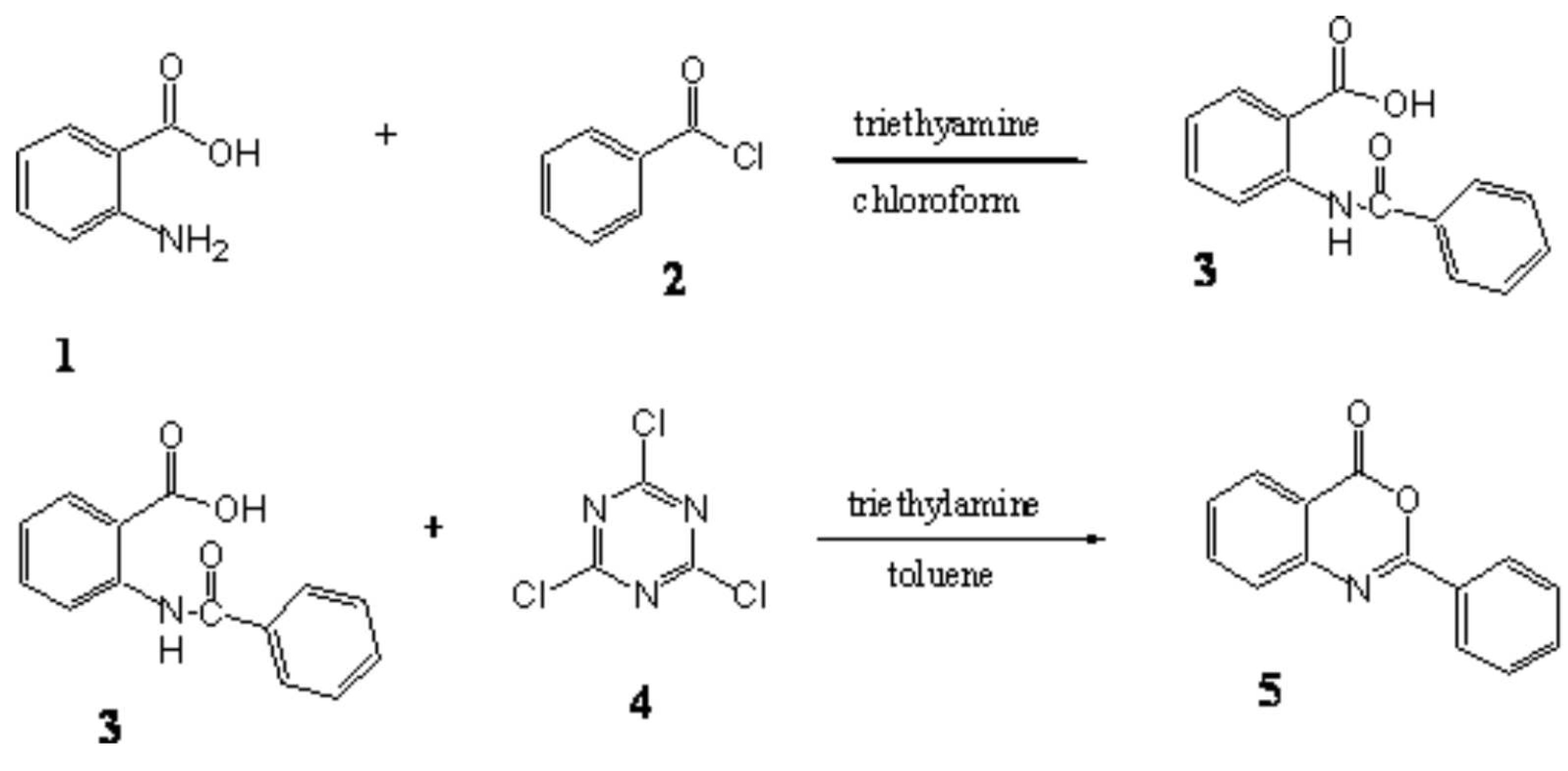
Synthesis of 4H-3,1-benzoxazin-4-one 2-phenyl Using Cyanuric Chloride as a Cyclization Agent
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgement
References
- Bulluci, C.; Gualtieri, F.; Chiarini, A. Eur. J. Med. Chem. 1987, 22, 473.
- Wiker, P.; Wilson, A. J. Am. Chem. Soc. 1955, 77, 5598.
- Bouillant, M. L.; Farre-Bonvin, J.; Ricci, P. Tetrahedron Letters 1983, 24, 51.
- Donchet, M.; Martin-Tuguy, J.; Marais, A.; Pupet, A. Phytochem 1981, 23, 1901.
- Shariat, M.; Abdollahi, S. Molecules 2004, 9, 705–712. [PubMed]
- Bain, D. I.; Smalley, R. K. J. Chem. Soc. C 1968, 13, 1593–7. [CrossRef]
- Sample Availability: Available from the authors.
© 2005 MDPI. All rights reserved.
Share and Cite
Abdollahi, S.; Shariat, M. Synthesis of 4H-3,1-benzoxazin-4-one 2-phenyl Using Cyanuric Chloride as a Cyclization Agent. Molbank 2005, 2005, M448. https://doi.org/10.3390/M448
Abdollahi S, Shariat M. Synthesis of 4H-3,1-benzoxazin-4-one 2-phenyl Using Cyanuric Chloride as a Cyclization Agent. Molbank. 2005; 2005(6):M448. https://doi.org/10.3390/M448
Chicago/Turabian StyleAbdollahi, Sohrab, and Mehdi Shariat. 2005. "Synthesis of 4H-3,1-benzoxazin-4-one 2-phenyl Using Cyanuric Chloride as a Cyclization Agent" Molbank 2005, no. 6: M448. https://doi.org/10.3390/M448
APA StyleAbdollahi, S., & Shariat, M. (2005). Synthesis of 4H-3,1-benzoxazin-4-one 2-phenyl Using Cyanuric Chloride as a Cyclization Agent. Molbank, 2005(6), M448. https://doi.org/10.3390/M448



